Abstract
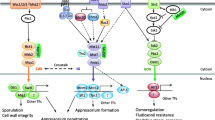
Mitogen-activated protein (MAP) kinases have the hallmark motif TXY and function in key signal transduction pathways in eukaryotic organisms. Most ascogenous plant pathogenic fungi have three MAPK pathways that regulate different developmental and infection processes. In the rice blast fungus Magnaporthe oryzae, the Pmk1 and Mps1 MAP kinases with the TEY motif are essential for appressorium formation, penetration, and invasive growth. Osm1 is the third MAP kinase that has the TGY motif and functions in osmoregulation. Although orthologs of Pmk1 and Mps1 are important for pathogenesis in all the plant pathogens studied, Osm1 orthologs have species-specific roles in stress responses and pathogenesis. Because of their functions in fungal development and pathogenesis, it is important to determine the expression and activation of MAP kinases under different growth conditions or infection stages. In this chapter, we describe methods for protein extraction and detection of the activation of the three MAP kinases in M. oryzae with the commercially available anti-TpEY or anti-TpGY phosphorylation-specific antibodies. Similar approaches can be used to monitor MAP kinase activation in other plant pathogenic fungi.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Zhao X, Mehrabi R, Xu JR (2007) Mitogen-activated protein kinase pathways and fungal pathogenesis. Eukaryot Cell 6(10):1701–1714
Hamel LP, Nicole MC, Duplessis S et al (2012) Mitogen-activated protein kinase signaling in plant-interacting fungi: distinct messages from conserved messengers. Plant Cell 24(4):1327–1351
Kondoh K, Nishida E (2007) Regulation of MAP kinases by MAP kinase phosphatases. Biochim Biophys Acta 1773(8):1227–1237
Pearson G, Robinson F, Gibson T et al (2001) Mitogen-activated protein (MAP) kinase pathways: regulation and physiological functions. Endocr Rev 22(2):153–183
Dean RA, Talbot NJ, Ebbole DJ et al (2005) The genome sequence of the rice blast fungus Magnaporthe grisea. Nature 434(7036):980–986
Zhao X, Kim Y, Park G et al (2005) A mitogen-activated protein kinase cascade regulating infection-related morphogenesis in Magnaporthe grisea. Plant Cell 17(4):1317–1329
Xu JR, Hamer JE (1996) MAP kinase and cAMP signaling regulate infection structure formation and pathogenic growth in the rice blast fungus Magnaporthe grisea. Genes Dev 10(21):2696–2706
Xu JR, Staiger CJ, Hamer JE (1998) Inactivation of the mitogen-activated protein kinase Mps1 from the rice blast fungus prevents penetration of host cells but allows activation of plant defense responses. Proc Natl Acad Sci U S A 95(21):12713–12718
Dixon KP, Xu JR, Smirnoff N et al (1999) Independent signaling pathways regulate cellular turgor during hyperosmotic stress and appressorium-mediated plant infection by Magnaporthe grisea. Plant Cell 11(10):2045–2058
Li G, Zhou X, Xu JR (2012) Genetic control of infection-related development in Magnaporthe oryzae. Curr Opin Microbiol 15(6):678–684
Liu W, Zhou X, Li G et al (2011) Multiple plant surface signals are sensed by different mechanisms in the rice blast fungus for appressorium formation. PLoS Pathog 7(1):e1001261
Zhang X, Liu W, Li Y et al (2017) Expression of HopAI interferes with MAP kinase signalling in Magnaporthe oryzae. Environ Microbiol 19(10):4190–4204
Li G, Zhang X, Tian H et al (2017) MST50 is involved in multiple MAP kinase signaling pathways in Magnaporthe oryzae. Environ Microbiol 19(5):1959–1974
Zheng D, Zhang S, Zhou X et al (2012) The FgHOG1 pathway regulates hyphal growth, stress responses, and plant infection in Fusarium graminearum. PLoS One 7(11):e49495
Ramamoorthy V, Zhao X, Snyder AK et al (2007) Two mitogen-activated protein kinase signalling cascades mediate basal resistance to antifungal plant defensins in Fusarium graminearum. Cell Microbiol 9(6):1491–1506
Leung H, Borromeo ES, Bernardo MA et al (1988) Genetic analysis of virulence in the rice blast fungus Magnaporthe grisea. Phytopathology 78(9):1227–1233
Jones CG, Hare DJ, Compton SJ (1989) Measuring plant protein with the Bradford assay: 1. Evaluation and standard method. J Chem Ecol 15(3):979–992
Gallagher SR (2012) One-dimensional SDS gel electrophoresis of proteins. Curr Protoc Protein Sci Chapter 10:Unit 10.1.1-44
Gilda JE, Gomes AV (2015) Western blotting using in-gel protein labeling as a normalization control: stain-free technology. Proteomic Profiling: Methods and Protocols 1295:381–391
Acknowledgments
This work was supported by a grant from US Wheat and Barley Scab Initiative and a grant from the National Research Initiative of the United States Department of Agriculture National Institute for Food and Agriculture (award number 2013-68004-20378).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Zhang, X., Bian, Z., Xu, JR. (2018). Assays for MAP Kinase Activation in Magnaporthe oryzae and Other Plant Pathogenic Fungi. In: Ma, W., Wolpert, T. (eds) Plant Pathogenic Fungi and Oomycetes. Methods in Molecular Biology, vol 1848. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-8724-5_8
Download citation
DOI: https://doi.org/10.1007/978-1-4939-8724-5_8
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8723-8
Online ISBN: 978-1-4939-8724-5
eBook Packages: Springer Protocols