Abstract
There is growing concern over the potential effects of high-intensity sonar on wild fish populations and commercial fisheries. Acoustic telemetry was employed to measure the movements of free-ranging reef fish and sea turtles in Port Canaveral, FL, in response to routine submarine sonar testing. Twenty-five sheepshead (Archosargus probatocephalus), 28 gray snapper (Lutjanus griseus), and 29 green sea turtles (Chelonia mydas) were tagged, with movements monitored for a period of up to 4 months using an array of passive acoustic receivers. Baseline residency was examined for fish and sea turtles before, during, and after the test event. No mortality of tagged fish or sea turtles was evident from the sonar test event. There was a significant increase in the daily residency index for both sheepshead and gray snapper at the testing wharf subsequent to the event. No broad-scale movement from the study site was observed during or immediately after the test.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Midfrequency sonar has been demonstrated to induce behavioral responses in marine mammals (Tyack et al. 2011; DeRuiter et al. 2013); however, the effect of sonar on other marine species is not well understood. Although it has been suggested that high-intensity midfrequency sonar is potentially capable of injury or mortality in fish species that are very close to the source, neither has been demonstrated in a field setting (Jørgensen et al. 2005; Popper 2008), and behavioral studies examining the effects of midfrequency sonar on unrestrained fish in their natural habitat are limited (Popper 2008; Slabbekoorn et al. 2010; Normandeau Associates, Inc. 2012). The most prominent studies thus far were conducted in enclosed, restrained environments from which behavior cannot be extrapolated to wild animals (Schwarz and Greer 1984; Jørgensen et al. 2005; Popper et al. 2007), have only investigated the immediate behavioral responses of a single species (Knudsen et al. 1992, 1994; Gearin et al. 2000), or did not include direct behavioral observations or assess long-term survival (Culik et al. 2001).
There are also very limited studies of sea turtle behavioral response to high-intensity sounds, with most efforts focused on measuring avoidance or changes in swimming activity in response to air gun sounds (O’Hara and Wilcox 1990; Moein et al. 1994; McCauley et al. 2000; Weir 2007). Although the range of turtle hearing is likely limited to an upper level of 2 kHz, to date there have been no behavioral response studies of turtles to midfrequency sonar.
The main objective of this study was to use fine-scale acoustic telemetry to assess the residency of fish and sea turtles before, during, and after a routine submarine sonar test event. Sheepshead (Archosargus probatocephalus) and gray snapper (Lutjanus griseus) were chosen as target fish species due to known site fidelity to hard-bottom habitats in the study area (Reyier et al. 2010), hearing ranges and sensitivities that are characteristic of fish without highly derived hearing specializations (Popper 2008), and the widespread occurrence of the Sparidae and Lutjanidae families in subtropical and tropical coastal marine waters throughout the world. The green sea turtle (Chelonia mydas) was also a target species because it possesses similar auditory structures and presumed hearing capabilities as the six sea turtle species that comprise the family Cheloniidae, and a previous mark-recapture study has indicated the importance of the Cape Canaveral region as a foraging and resting area for juveniles (Ehrhart et al. 2007).
2 Methods
2.1 Study Area
Port Canaveral is a man-made multiuse harbor on the central east coast of Florida that supports cruise ship terminals, recreational and commercial fishing interests, and military testing and training activities. The wharf structures within the Port create valuable habitats that have resulted in robust resident populations of tropical reef fish species (Reyier et al. 2010). A significant portion of the daily feeding and resting activity of the target species is centralized on the wharf, rock revetments, and associated floating dock spaces. Although fish and turtles are exposed to low levels of anthropogenic noise regularly from the activity at Port Canaveral, pier-side testing of navy sonar systems occurs on an intermittent basis and typically for short durations.
The sonar test examined in this study occurred at the northern end of Trident Wharf over a 2-day period in June 2012. The sonar signals, duty cycle, and source levels followed a typical protocol for a pier-side submarine sonar test event.
2.2 Acoustic Telemetry Arrays
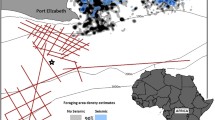
Two array configurations were utilized in this study to collect data at different temporal and spatial scales. Vemco VR2W (AMIRIX Systems, Inc.) autonomous telemetry receivers were deployed in the West, Middle, and Trident Basins to provide broad-scale coverage within the Port Canaveral area (Fig. 152.1). Regional tracking for those tagged fish that emigrated out of the Port Canaveral study area was accomplished via the Florida Atlantic coast telemetry (FACT) array, which at the time consisted of 200 acoustic receivers deployed across a 300-km section of the east Florida coastline. Vemco receivers within the immediate Canaveral Harbor study area were bottom mounted and secured via a guy anchor.
Lotek WHS3250 receivers (Lotek Wireless, Inc.) comprised the second fine-scale array and were secured via a guy anchor or bracket mounted directly offset from pilings on the interior of Trident Wharf in the Trident Basin only. Receivers mounted on wharf pilings were outfitted with acoustic scatterboards to help minimize acoustic reflection and localization errors. These custom-built boards were composed of closed cell foam and plywood mounted directly to pilings supporting a receiver.
2.3 Collection and Tagging
Lotek multimode transmitters (MM-MR-8-SO-TP) were used to gather movement data for both fish species and a subset of the turtles. These transmitters emit two discrete coded signals that are separately compatible with existing Vemco and Lotek receivers. The expected battery life for the transmitters ranged from 73 to 163 days depending on the pulse interval of signal emissions. Seventeen of the turtles were tagged with Vemco V9P-2L tags, which are only compatible with Vemco receivers.
A total of 25 sheepshead and 28 gray snapper were captured either by gill net or hook-line angling 1 week before the test event. Target fish had a minimum weight of 300 g in air to ensure that the tag accounted for no more than 2% of body weight (Winter 1983). Fish were anesthetized in a solution of 75 mg/L of tricaine methanosulfonate (MS-222, Western Chemical, Inc.) in seawater in a 10-gal holding tank. The fish were then placed ventral side up for surgical implantation of acoustic transmitters while the gills were irrigated with fresh seawater. The incisions were closed with two absorbable sutures followed by application of tissue adhesive and triple antibiotic ointment. Standard length, weight, method of capture, and release condition were recorded for all fish. Each fish was fitted with a small external dart tag (Hallprinta) in case of angler recapture. The fish were allowed to fully recover in aerated seawater for a minimum of 10 min and were observed swimming normally before release.
Twenty-nine green sea turtles were captured using tangle nets or dip nets. The tangle nets consisted of multifilament nylon twine mesh hung from a braided polypropylene top line and a braided polypropylene continuous lead-core bottom line. The turtles were also opportunistically captured with long-handled, large-hoop dip nets. After transport to the shore, standard morphometric measurements were recorded and the turtle was tagged externally using inconel alloy flipper tags and a passive internal transponder (PIT) tag (Destron-Fearing) inserted subcutaneously in the right front flipper proximal to the wrist joint. The Vemco or Lotek acoustic transmitters were attached externally with two-part marine epoxy to the 3rd and 4th right or left costal scutes depending on the condition of the scutes. Once the epoxy hardened, the turtle was released back into the basin.
2.4 Residency
Raw detection data from each receiver were filtered to minimize the probability of accepting false-positive detections (Pincock 2008). Residency indexes (RIs) were calculated on a daily basis for all fish to represent the proportion of a given time window that an individual was detected on the Trident Wharf Vemco receivers (TB1, TB2, and TB6). RIs were also calculated for combined detections on all three wharf receivers (a fish was considered present if it was detected on any of the three wharf receivers) to examine broader scale residency at the wharf. Before, during, and after sonar test time windows utilized in comparisons were structured by days 1–7, 8–9, and 10–16 posttagging, respectively. The Wilcoxon matched-pairs signed-rank test was used to test for significant differences between the pretest and posttest time windows at α ≤ 0.05.
RIs were also calculated for Lotek receivers along the Trident Wharf for fish only. Before, during, and after sonar test periods were structured in 36-h continuous-time windows. For this comparison, detections were combined into three receiver zones representing subsections of the wharf habitat. Receiver (REC) zones 1, 2, and 3 were composed of REC09/10/11, REC12/13/14, and REC15/16/17, respectively. Tests for significance between groups were conducted utilizing a nonparametric repeated-measures one-way ANOVA. Post hoc tests for significance between paired groups (before to during, during to after, and before to after) were conducted utilizing Wilcoxon matched-pairs signed-rank test. Significant differences were considered at α ≤ 0.017 after a Bonferroni correction for multiple comparisons.
3 Results
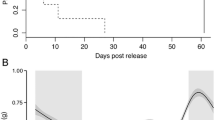
Fish and turtle data were monitored on the Canaveral Vemco and FACT arrays for 125 days after release of the animals. The tagged fish were detected on Vemco receivers for a median of 88 days (range 5–125 days, mode 125 days), while turtles were detected on Vemco receivers for a median of 63 days (range 6–125 days, mode 115 days). Figure 152.2 shows the number of detections of each species on Trident Wharf for the first 60 days after the start of tagging.
Table 152.1 shows a summary of the mean ± SE values for the RI on Trident Wharf. For sheepshead, there was a significant increase in the RI at TB6 (z = −2.06, n = 25, P = 0.04) and at the Trident Wharf receivers combined (z = −2.36, n = 25, P = 0.02) after the test event. Gray snapper showed a significant decrease in residency at TB2 posttest (z = −2.50, n = 28, P = 0.013), although there was a significant increase at the Trident Wharf receivers combined (z = −2.01, n = 287, P = 0.045). Green sea turtles overall had lower RI values on the wharf and also showed a significant decrease in residency at TB2 only (z = −2.72, n = 29, P = 0.007).
In the short term, significant differences between time periods were reported for the hourly RI for sheepshead at REC09/10/11 (H = 10.71, df = 2, P = 0.005) and for gray snapper at all Lotek receivers combined (H = 6.46, df = 2, P = 0.040). Post hoc comparisons showed a significant decrease in hourly residency for sheepshead at REC09/10/11 for the before test compared with the during test time period (z = −3.15, n = 25, P = 0.002). Post hoc comparisons for gray snapper were not significant. A summary of mean ± SE values for the hourly RIs in the Trident Basin are presented in Table 152.2.
4 Discussion
Sheepshead and gray snapper exposed to a typical pier-side submarine sonar test event showed some short-term declines in residency near particular individual receivers during and after the test, but no long term displacement from the wharf habitat overall was observed for any species. Sonar testing is infrequent in the Trident Basin, especially within the short-term residence time of 2–4 weeks typical of these species. Although the sonar signals may represent a novel stimulus, Port Canaveral is a very urban environment, and marine species here may be habituated to higher levels of anthropogenic sound, including vessel and construction noise. These individuals could therefore be less likely to respond than naïve populations. Nevertheless, the sheepshead, gray snapper, and green sea turtles present in the Trident Basin did not demonstrate significant displacement in response to a typical pier-side submarine sonar test event.
References
Culik BM, Koschinski S, Tregenza N, Ellis GM (2001) Reactions of harbor porpoises Phocoena phocoena and herring Clupea harengus to acoustic alarms. Mar Ecol Prog Ser 211:255–260
DeRuiter SL, Southall BL, Calambokidis J, Zimmer WMX, Sadykova D, Falcone EA, Friedlaender AS, Joseph JE, Moretti D, Schorr GS, Thomas L, Tyack PL (2013) First direct measurements of behavioural responses by Cuvier’s beaked whales to mid-frequency active sonar. Biol Lett 9, 20130223. doi:10.1098/rsbl.2013.0223
Ehrhart LM, Redfoot WE, Bagley DA (2007) Marine turtles of the central region of the Indian River Lagoon System, Florida. Fla Sci 70:415–434
Gearin PJ, Gosho ME, Lakke JL, Cooke L, Delong RL, Hughes KM (2000) Experimental testing of acoustic alarms (pingers) to reduce bycatch of harbor porpoise, Phoceoena phocoena, in the state of Washington. J Cetacean Res Manage 2:1–9
Jørgensen R, Olsen KK, Falk-Petersen IB, Kanapthippilai P (2005) Investigations of potential effects of low frequency sonar signals on survival, development and behaviour of fish larvae and juveniles. The Norwegian College of Fishery Science, University of Tromsø, Tromsø, Norway
Knudsen FR, Enger PS, Sand O (1992) Awareness reactions and avoidance responses to sound in juvenile Atlantic salmon, Salmo salar. J Fish Biol 40:523–534
Knudsen FR, Enger PS, Sand O (1994) Avoidance responses to low frequency sound in downstream migrating Atlantic salmon smolt, Salmo salar. J Fish Biol 45:227–233
McCauley RD, Fewtrell J, Duncan AJ, Jenner C, Jenner MN, Penrose JD, Prince RIT, Adhitya A, Murdoch J, McCabe K (2000) Marine seismic surveys: analysis and propagation of air-gun signals; and effects of air-gun exposure on humpback whales, sea turtles, fishes and squid. Report prepared for the Australian Petroleum Producers Exploration Association by the Centre for Marine Science and Technology, Curtin University, Perth, WA
Moein SE, Musick JA, Keinath JA, Barnard DE, Lenhardt M, George R (1994) Evaluation of seismic sources for repelling sea turtles from hopper dredges. Final report submitted to the US Army Corps of Engineers Waterways Experiment Station by the Virginia Institute of Marine Science, College of William and Mary, Gloucester Point, VA
Normandeau Associates, Inc. (2012) Effects of noise on fish, fisheries, and invertebrates in the U.S. Atlantic and Arctic from energy industry sound-generating activities. A workshop report prepared under Contract No. M11PC00031 for the Bureau of Ocean Energy Management, US Department of the Interior. A
O’Hara J, Wilcox JR (1990) Avoidance responses of loggerhead turtles, Caretta caretta, to low frequency sound. Copeia 2:564–567
Pincock DG (2008) False detections: what they are and how to remove them from detection data. Document DOC-004691, version 03, Vemco, Halifax, NS, Canada. Available at http://www.vemco.com/pdf/false_detections.pdf. Accessed Apr 2012
Popper AN (2008) Effects of mid- and high-frequency sonars on fish. Report prepared under Contract N66604-07 M-6056, Naval Undersea Warfare Center Division, Newport, RI
Popper AN, Halvorsen MB, Kane A, Miller DL, Smith ME, Song J, Stein P, Wysocki LE (2007) The effects of high-intensity, low-frequency active sonar on rainbow trout. J Acoust Soc Am 122:623–635
Reyier EA, Scheidt DM, Lowers RH et al (2010) A characterization of biological resources within the Cape Canaveral Air Force Station Trident Submarine Basin and adjacent marine waters of Port Canaveral, Florida (May 2008–Apr 2010). Final report submitted to the US Air Force 45th Space Wing Natural Assets Office, Sept 2010
Schwarz AL, Greer GL (1984) Responses of Pacific herring (Clupea harengus pallasi) to some underwater sounds. Can J Fish Aquat Sci 41:1183–1192
Slabbekoorn H, Bouton N, van Opzeeland I, ten Cate C, Popper AN (2010) A noisy spring: the impact of globally rising underwater sound levels on fish. Trends Ecol Evol 25:419–427
Tyack PL, Zimmer WMX, Moretti D, Southall BL, Claridge DE, Durban JW, Clark CW, D’Amico A, DiMarzio N, Jarvis S, McCarthy E, Morrissey R, Ward J, Boyd IL (2011) Beaked whales respond to simulated and actual navy sonar. PLoS ONE 6, e17009. doi:10.1371/journal.pone.0017009
Weir CR (2007) Observations of marine turtles in relation to seismic airgun sound off Angola. Mar Turtle News 116:17–20
Winter JD (1983) Underwater biotelemetry. In: Nielson LA, Johnson DL (eds) Fisheries techniques. American Fisheries Society, Bethesda, MD
Acknowledgments
We thank Jane Provancha, LCDR James Westermeyer and the Naval Ordnance Test Unit, and Don George and Angy Chambers at the Air Force 45th Space Wing Natural Assets Office for logistical support. We also thank Doug Scheidt, Carla Garreau, Russell Lowers, Karen Holloway-Adkins, Jennifer James, Carter Watterson, Colin Lazauski, David MacDuffee, Tony Ruffa, and the Florida Atlantic Coast Telemetry Array Community. This project was approved by the Kennedy Space Center Institutional Animal Care and Use Committee and the Naval Undersea Warfare Center Environmental Review Board. Funding was provided by the US Fleet Forces Command N46, the Canaveral Port Authority, and the Naval Undersea Warfare Center In-House Laboratory Independent Research Program.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media New York
About this paper
Cite this paper
Watwood, S.L., Iafrate, J.D., Reyier, E.A., Redfoot, W.E. (2016). Behavioral Response of Reef Fish and Green Sea Turtles to Midfrequency Sonar. In: Popper, A., Hawkins, A. (eds) The Effects of Noise on Aquatic Life II. Advances in Experimental Medicine and Biology, vol 875. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-2981-8_152
Download citation
DOI: https://doi.org/10.1007/978-1-4939-2981-8_152
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-2980-1
Online ISBN: 978-1-4939-2981-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)