Abstract
Heightened awareness of the relevance of early lung development on subsequent lung health and the need to identify lung disease before changes become irreversible, has resulted in increased efforts to monitor lung function from birth and throughout the preschool years. International collaborative efforts to adapt techniques and develop standardized protocols, together with increased availability of appropriate commercial devices mean that it is now possible to perform a wide range of pulmonary function tests (PFTs) in infants and preschool children less than 6 years of age. The aims of this chapter are to (a) briefly describe which PFTs can be performed in spontaneously breathing sleeping infants and awake preschool children using commercially available equipment, (b) discuss how to interpret PFTs in children under 5 years, and (c) consider the extent to which these tests might contribute to clinical management of infants and preschool children.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Respiratory function tests
- Infant
- Children
- Child
- preschool
- Normal values
- Lung function
- Spirometry
- Lung volumes
- Airways resistance
- Repeatability of results
Introduction
Pulmonary function tests (PFTs) are an integral component of clinical management in school-aged children and adults with lung disease. By contrast, the lack of suitable equipment and difficulties in undertaking such measurements in small, potentially uncooperative subjects meant that, until recently, assessments of pulmonary function in those less than 5 years of age was restricted to specialized research establishments. The realization that insults to the developing lung may have lifelong effects, with much of the burden of respiratory disease in later life having its origins prenatally or during the first years of life, has focused attention on the need to develop sensitive methods of assessing respiratory function in infants and preschool children. Assessment of respiratory function in the very young is relevant not only to our understanding of respiratory health and disease, during childhood, but also throughout later life. Such tests can provide objective outcome measures to identify early determinants of respiratory function, distinguish changes due to disease from those related to growth and development, and be used to evaluate the effects of new therapeutic advances as part of well-designed research studies. However, their role in clinical management or as a diagnostic tool remains limited, as discussed below.
The aims of this chapter are to:
-
Briefly describe which PFTs can be performed in spontaneously breathing sleeping infants (birth to ~2 years) and awake preschool children (3–5 years of age) using commercially available equipment.
-
Discuss how to interpret PFTs in children under 5 years.
-
Consider the extent to which these tests may contribute to clinical management of infants and preschool children.
This chapter focuses on commonly used tests for which commercially available equipment is available. It will not cover the use of forced oscillation techniques or the multiple breath washout technique which are covered elsewhere in this book (Chap. 10 ), nor the application of PFTs in ventilated infants or young children. Due to the enormous increase in literature in this field during recent years, references cited are generally limited to those published in the past 5 years, the bibliographies of which would inform the interested reader regarding prior relevant literature.
Methods of Assessing Pulmonary Function in Infants and Young Children
Although assessment of lung size, compliance, and gas-exchanging surface area may be valuable when assessing the impact of congenital cardiac defects or disruption of alveolar development in survivors of bronchopulmonary dysplasia [1–8], most respiratory problems beyond the neonatal period are characterized by some form of central or peripheral airway obstruction. Consequently, the most frequently used PFTs in early life are those designed to assess airway function by measuring either airways resistance or forced expiratory flows and volumes. There is, however, increasing awareness that airway obstruction may be determined not only by the caliber of the airways but also by the compliance of the airway wall and the elastic recoil of the surrounding parenchyma, leading to the search for suitable outcomes that will reflect these characteristics and hence improve the interpretation of results. Furthermore, as discussed in Chap. 10, there is increasing evidence that conventional measures of airway function such as forced expiratory maneuvers, that focus on the conducting airways, may be insensitive to early lung disease in conditions such as cystic fibrosis (CF), which commences in the lung periphery, and which are therefore better evaluated by measuring ventilation inhomogeneity [9–12].
An ideal lung function test for infants and young children would be one that is:
-
Simple and involves no risk
-
Acceptable to both the child and the parents
-
Independent of subject cooperation
-
Applicable to any age and arousal state
-
Reproducible
-
Sensitive enough to distinguish between health and disease
-
Able to reflect the clinical situation or provide accurate and specific information about lung structure and function
-
Cheap and measurable using commercially available equipment, built to internationally approved standards, allowing standardized data collection and interpretation and for which
-
Appropriate reference ranges from healthy children, derived over a wide age span during infancy or early childhood, have been developed
Although no such test currently exists, there are a number of techniques that have been shown to be safe and feasible in sleeping infants and toddlers (generally below 2 years of age) and preschool children and which are now commercially available [13].
Special Consideration When Testing Infants
In addition to the marked developmental changes in respiratory physiology that occur during the first years of life which affect both the measurement and interpretation of results [14], the major differences in undertaking infant PFTs relate to sleep state, sedation, ethical issues, posture, and the need to miniaturize and adapt equipment for measurements in small subjects, who tend to be preferential nose breathers and who cannot be asked to undertake special breathing maneuvers [14]. Although attempts have been made to assess lung function in awake infants, measurements are normally made during sleep. A representative and stable end-expiratory level is essential for reproducible measures of tidal breathing, resting lung volume (functional residual capacity or FRC), respiratory mechanics (resistance and compliance), or partial forced expiratory flow-volume (FEFV) maneuvers. This can normally only be achieved if the child is in quiet, rather than rapid eye movement, sleep.
Unless clinically indicated, sedation is generally contraindicated for PFTs in newborn infants. Successful measurements using a full range of tests can usually be achieved during natural sleep after a feed in all infants up to at least 44 weeks postmenstrual age. Tests based on tidal breathing recordings may be applicable in the unsedated infant up to 4 months postnatal life [15, 16], whereas forced expiratory maneuvers and whole body plethysmography generally require sedation, certainly beyond 3 months of age. For all studies involving infants, strict safety precautions must be followed. In addition to adherence to local infection control procedures, resuscitation equipment, including suction, must be available, and two skilled operators, fully trained in basic life support, one of whom has prime responsibility for monitoring the well-being of the infant, must be in attendance throughout testing. Pulse oximetry is used for continuous monitoring throughout the testing session. Given the rapid rate of somatic growth during infancy and early childhood, accurate measurements of height and weight using a calibrated stadiometer and scales are essential.
Which Tests Can Be Performed in Infants and Toddlers?
Commercially available equipment is now available to assess a wide range of PFTs in sleeping infants and young children <2 years of age, including that used to assess
-
Passive respiratory mechanics (i.e., compliance and resistance; usually based on the single breath occlusion (SOT) technique) [1, 2, 18, 19]
-
Functional residual capacity (FRC) and indices of ventilation inhomogeneity (such as the Lung Clearance Index or LCI) using multiple breath inert gas washout (MBW) techniques [15, 25–27] (see Chap. 10) and
-
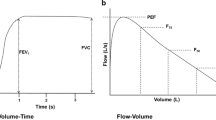
Forced expiratory flow-volume (FEFV) maneuvers, using the tidal or raised volume rapid thoraco-abdominal compression technique (Figs. 8.2 and 8.3) [8, 23, 24, 28–37]
Details of how to undertake these tests have been published elsewhere [14, 34, 38–45] and will only be summarized briefly here. Regrettably, although some groups have published results using this technique, valid measurements of plethysmographic airways resistance are not feasible using currently available commercial equipment [46, 47].
The choice of which test(s) to use in which infant, and the order in which they are performed, needs to be informed by the underlying reason for performing the test, the underlying assumptions of each test, the likely total duration of sleep (and hence testing session) and the level of expertise required for accurate measurements, rather than simple availability of equipment. Although recording of tidal breathing and respiratory mechanics using the occlusion technique appear simple, their validity can easily be compromised by leaks around the face mask, lack of quiet regular breathing or, in the case of single occlusion technique, violation of the theoretical assumptions underlying the measurements due to lack of complete relaxation during the occlusion and throughout the subsequent expiration and/or inability to represent the respiratory system by a single time constant in the presence of lung disease [14, 19, 43]. Strict quality control indicates that these conditions are frequently not met leading to a failure rate of at least 30 % [23]. Generally speaking, assessments based on quiet tidal breathing (e.g., respiratory mechanics using the occlusion technique, multiple breath washout or plethysmography) should be undertaken before those entailing forced expiratory maneuvers.
Which Tests Can Be Performed in Preschool Children?
Up until the last decade, it was commonly stated that assessment of lung function in preschool children (2–5 years) was virtually impossible, since they lacked the necessary coordination, comprehension and cooperation to participate, this period being dubbed the “dark ages” of pediatric pulmonology. Although this view is still held by many, there is now a considerable body of evidence that a wide range of lung function measurements are indeed feasible in the majority of children above 3 years of age, provided the techniques and quality control criteria are suitably adapted, and the tests are performed in a child-friendly environment by operators with suitable expertise and aptitude [9, 10, 13, 14, 32, 46, 48–68]. Success rate increases with age [13, 67, 68], with high success rates feasible from around 3.5 years, unless the child has been born very prematurely or has any signs of developmental delay.
As for all PFTs, appropriate equipment and testing conditions, skilled and experienced personnel, and rigorous adherence to published guidelines are critical for ensuring high quality, reproducible data. When assessing preschool children, it is essential to try and minimize equipment dead-space, especially if adapting that used for measurements in older subjects. For certain tests, especially those requiring long periods of tidal breathing, the use of a suitably selected video to distract and entertain the child (without inducing laughter or conversation!) can be very useful. For such tests, a face mask may be preferable to that of a mouthpiece in order to reduce risk of leaks and improve cooperation. However, the potential effect of any added dead-space when using a mask must be considered. Most young children tolerate a nose clip and mouth piece satisfactorily for short measurements such as spirometry.
PFTs that are now commonly applied in this age group include
-
Plethysmographic specific resistance (sRaw) [9, 10, 13, 14, 51, 52, 55, 72]
-
N.B. assessment of Plethysmographic FRC is also possible in specialist laboratories [48], but is generally poorly tolerated below 5–6 years of age
-
-
Interrupter Technique for assessing respiratory resistance [57, 61, 66, 73–75]
-
Multiple breath washout assessments of FRC and LCI [9–11, 13, 14, 51, 52] (see Chap. 10)
-
Forced oscillation technique or impulse oscillometry for assessing respiratory impedance [13, 14, 49, 54, 55, 57, 60, 76–79] (see Chap. 10)
Assessments using the MBW and forced oscillations are considered in Chap. 10 and will therefore not be addressed further here except in general terms.
Preschool Spirometry (Fig. 8.4): The use of carefully designed computerized incentives can be extremely helpful in gaining the young child’s cooperation in effort-dependent tests such as spirometry, but must be selected carefully and used interactively. For example, while blowing candles out is a useful means of teaching the child to blow out fast, it will rarely encourage the child to continue to blow out until residual volume is reached, for which alternative incentives such as a bowling alley may be more useful. Similarly, the operator must select the desired threshold that the child needs to reach, for which availability of appropriate reference equations is essential, as well as an appreciation of the child’s clinical condition. It is generally helpful to set targets somewhat higher than that anticipated, to increase these further to encourage maximal effort if the child readily achieves the initial target, but to always allow the child to “win” by the end of the session in order to provide encouragement and a desire to return for future assessments!
Young children have a much shorter expiratory time constant (relatively large airway caliber in relation to lung volume) than older subjects and therefore empty their lungs rapidly during forced expiration. In healthy children <5 years of age, expiration may be completed in less than a second, such that FEV1 cannot always be reported, FEV0.75 providing a useful alternative [67, 68, 80]. If undertaking spirometry in preschool children, it is essential that Quality Control (QC) criteria are adapted accordingly; stipulation that a forced expired time of at least 6 s or even 3 s must be achieved is likely to be associated with a high failure rate. Conversely, applying adult criteria with respect to repeatability criteria will likely lead to acceptance of unduly variable results [67, 68, 80].
Current data suggest that while spirometry can be successfully performed in the majority of preschool children with CF, it is far less sensitive than the lung clearance index (LCI) from multiple breath washout, such that while early lung disease can sometimes be detected, the abnormalities are on average mild and highly variable [10, 58]. Given its sensitivity in older children with Bronchopulmonary Dysplasia (BPD) [3], spirometry could potentially provide a useful longitudinal measurement for young children with BPD, in whom both lung growth and airway obstruction may be significantly abnormal in early life [2, 26, 81]. Unfortunately there is a paucity of data on preschool spirometry in children with BPD, possibly associated with the difficulty of undertaking this technique in young children if there is reduced concentration span or coordination, such as may occur following extremely preterm delivery. Spirometry is being increasingly used in children with recurrent wheezing, both to establish baseline lung function and document bronchodilator responsiveness (BDR) [14, 52, 62, 64, 82–84]. There are still limited data regarding the prevalence of BDR in normal preschool children and what constitutes a significant increase in forced expiratory volume after bronchodilator inhalation, but it has been suggested that a post-bronchodilator increase between 12 and 15 % in FEV0.75 or FEV1 is more commonly observed in preschool children with a clinical diagnosis of asthma than in healthy controls and exceeds the natural within-subject, between-test, within-occasion variability [52, 64, 67] . The use of FEF25–75 as an outcome to assess BDR is not recommended due to the high within and between-subject variability of this outcome [13]. Protocols for bronchial provocation and exercise testing in preschool children have been reported, but there is currently insufficient data to enable their use in clinical practice [13].
Plethysmographic Measurements of Specific Airway Resistance (sRaw)
sRaw is assessed while the child breathes tidally through a mouthpiece or modified facemask in a body plethysmograph (Figs. 8.5 and 8.6), without need for any special breathing maneuvers against an airway occlusion and is therefore well suited for preschool children. Since sRaw is the product of airway resistance (Raw) and FRC, both of which may increase in the presence of airway obstruction and hyperinflation, it is a potentially useful method for identifying obstructive lung disease in young children [14, 55, 72, 85]. sRaw has been found to be significantly elevated in preschool children with CF when compared with healthy controls, and appears to be more discriminative than spirometry to early lung disease, though less so than the lung clearance index [9, 10, 86]. sRaw has been used in longitudinal birth cohort studies and been found to be higher in asthmatic children than in healthy controls. Although it has been suggested that bronchial hyper-responsiveness and BDR can be successfully determined using sRaw, with fair discrimination between healthy young children and those with asthma or wheeze [55, 72], the extent to which even healthy young children demonstrate significant bronchodilator responsiveness, means that a reduction in sRaw by at least 25 % is required before the change can be considered clinically significant. A recent study concluded that the capacity of sRaw to discriminate between healthy young children and children with stable wheeze according to BDR is doubtful because of the large overlap in response between the two groups [52]. sRaw does not appear to have been used in children with prior BPD during the preschool years, possibly reflecting reduced concentration levels and delayed maturity in many of these children.
The Interrupter Technique
The interrupter resistance (Rint) technique is a quick, noninvasive measure of respiratory resistance that can be performed relatively easily in preschool children during tidal breathing. Rint is calculated from the ratio of pressure change to flow assessed at the airway opening during a brief (<100 ms) occlusion. Recent ATS/ERS guidelines recommend that occlusions for Rint occur during expiration. Although several studies have attempted to use Rint to assess lung function in preschool children with CF, the lack of discrimination between healthy children and those with CF suggests that Rint will have limited clinical utility in this population. Higher values of Rint have been observed in preschool children with severe BPD when compared without BPD, but there is considerable overlap between groups [87]. As summarized in the recent ATS document on clinical utility of PFTs in young children [13], baseline Rint does not discriminate well between healthy children and those with recurrent wheeze due to the large inter-subject variability in Rint values in health. The main application of this test may be to assess BDR in wheezy children, although a reduction in Rint of at least 30 % is required before changes are considered to be of clinical significance [56, 73].
Challenges to Using Infant PFTs in Clinical Management
Despite numerous attempts to monitor changes in lung function as a means of identifying early onset of pulmonary disease during the first year of life, the natural course of pulmonary involvement in infants with respiratory disorders remains relatively poorly understood, for the following reasons
-
Need for sedation
-
Confounding by developmental changes in respiratory physiology [14]
-
Lack of appropriate reference data
-
Lack of information regarding within- and between-subject variability, especially in those with lung disease, without which it is impossible to interpret serial measurements or response to interventions including bronchodilators
-
Difficulties in repeating measurements frequently enough to monitor change or long-term response to interventions accurately
Despite these significant challenges, among the 160 centers identified by a recent American Thoracic Society/European Respiratory Society (ATS/ERS) survey 37 (23 %) reported using infant PFTs purely for “clinical” applications and almost half of all respondents were using them to assess bronchodilator response (BDR). Given the time consuming nature of these tests (which may entail parents taking time off work), need for sedation and the considerable costs relating to equipment, consumables, and staffing costs for two specially trained and clinically qualified operators for each test [88], serious questions need to be asked regarding the clinical usefulness of results in any given child before undertaking or requesting infant PFTs “for clinical purposes.”
Challenges to Using PFTs in Clinical Management of Preschool Children
The challenges to using PFTs in the clinical management of preschool children are similar, albeit not so difficult to address, as those encountered when dealing with infants. Anyone who has tested very young children will appreciate that, in addition to needing to adapt the technique, equipment and quality control criteria as discussed above, much more time needs to be allocated to obtaining technically satisfactory results in this age group, especially during their first few visits to the laboratory. Problems relating to lack of appropriate reference data and hence confidence in what constitutes an “unusual” or “abnormal” result for a preschool child are beginning to be resolved, thanks to international efforts to collate data from healthy young children [85, 89–92]. However, with the exception of recent multi-ethnic spirometry equations [91] such reference equations remain largely limited to children of non-Hispanic White European descent. Although there remains limited information regarding within-subject, within and between-occasion variability with which to assess the clinical significance of any observed acute response to a bronchodilator or longer term response to other interventions [66, 67, 74], several recent papers have begun to address these issues [13, 52, 64, 67].
As when assessing infants, the choice of techniques must reflect the underlying research or clinical question. While techniques such as forced oscillation and interrupter technique are often considered simpler to apply in this age range, they still require considerable quality control during data collection and analysis and the wide between-subject variability in health may limit the extent to which they can identify abnormal lung function in those with lung disease. While spirometry is perfectly feasible in preschool children it has been shown to be far less sensitive than the LCI in identifying early lung disease in young children with CF [9, 10] (Box 8.1).
Box 8.1. Which PFTs Should Be Used When Assessing Infants and Preschool Children?
-
What is the research/clinical question?
-
What is the research/clinical question?
-
What is the underlying pathophysiology?
-
What is feasible in time available (duration of sleep in infants/concentration for young child)?
-
How much between-test variability may occur within any individual child?
-
Are valid reference equations available with which to interpret results?
Interpretation of Lung Function Results in Infants and Young Children and Their Role in Clinical Management
As in older subjects, the clinical usefulness of any lung function test within an individual infant or preschool child will always be enhanced if serial measures rather than a single assessment can be undertaken. However, during infancy the frequency with which PFTs can be repeated will be limited by need for sedation and the time consuming nature of the tests. When requesting such PFTs, it is essential that the choice of tests is based on the question to be answered, clinical reasoning, and a knowledge of the suspected underlying pathophysiology, rather than simply on the equipment that happens to be available in any given center [93]. Given the marked influence of factors such as preterm delivery, intrauterine growth retardation, sex, ethnic group, and maternal smoking during pregnancy, it is important to take a careful history from the parents when performing such tests in infants and young children with respiratory problems. It is also essential that such tests are only performed during periods of clinical stability, usually defined as being at least 2–3 weeks after any pulmonary exacerbation or upper respiratory tract infection. In addition to being potentially unethical due to the increased risk of sedation in a child who is acutely unwell or wheezing, attempts to assess efficacy of treatment by studying a child during the early phases of an exacerbation and then shortly after a course of antibiotics, corticosteroids, or bronchodilators, will at best reflect some natural improvement in lung function with time, but will provide minimal clinically useful information, unless part of a well-designed randomized trial with suitable placebo.
What Is Normal?
As discussed in more detail in Chap. 11, in order to identify the nature and severity of any underlying pathophysiology in an individual, it is essential to have a clear idea of the range of values to expect in a healthy child of similar age, sex, body size, and ethnicity. Reliable interpretation of pulmonary function results therefore relies on the availability of appropriate reference data to help distinguish between health and disease. The use of inappropriate reference equations and misinterpretation even when potentially appropriate equations are used, can lead to serious errors in both under and over-diagnosis, with its associated burden in terms of financial and human costs [94, 95]. It is important to remember that lung function results from healthy children and those with respiratory symptoms or disease often overlap to such an extent that a result within the normal range does not exclude disease. Similarly, while abnormal lung function results are often associated with symptoms and disease, they may simply be “atypical” and must always be interpreted in the light of all other clinically relevant information.
% Predicted or Z-Scores?
As discussed in Chap. 11, although clinicians in respiratory medicine are more familiar with the concept of expressing lung function as percent predicted, ([observed/predicted] × 100), a much better approach to interpreting lung function is to express results as Z-scores (or Standard Deviation Scores (SDS)) [94]. The Z-score is a mathematical combination of the percent predicted and the between-subject variability to give a single number that accounts for sex, age and height-related lung function variability expected within comparable healthy individuals (and, in certain cases also adjusts for ethnicity) [91]. The upper and lower limits of the normal range (ULN and LLN) are conventionally defined as Z-score of ±1.64, a range that encompasses 90 % of healthy subjects. However, due to increased uncertainty regarding reliability of reference ranges for infants and young children and the fact that multiple PFTs are often used in the assessment, these limits should be set at ±1.96 Z-scores to encompass 95 % of the healthy population. An increasing number of clinical research studies are now reporting infant and preschool lung function as Z-scores [8, 10, 21, 23, 24, 33, 52, 64, 82, 96, 97]. Particular caution is required when interpreting results which lie close to the somewhat arbitrary “cutoffs” between the normal range seen in health and suspected disease, especially when results are limited to a single test occasion. As with all tests, PFTs should be seen as only one part of the whole clinical picture.
Reference Equations for Infant PFTs
Marked biases between predicted values can occur due to alterations in equipment and protocol, differences in population characteristics, the statistical methods applied or simply as a result of sampling error due to too few healthy subjects being studied. There is currently a dearth of appropriate reference equations for infant PFTs, many users relying on outdated values, based on too few subjects and collected with different equipment and software than is now available commercially. This can result in serious misdiagnosis and adversely affect interpretation of clinical research studies. The need for sedation and the time consuming nature of the testing procedure limits the number of healthy infants who can be studied at any one center. International collaborative efforts are urgently needed to address this problem before clinical studies in individual infants can be interpreted properly. As discussed in a recent editorial [98], issues surrounding the ethics of recruiting healthy infants for PFTs need to be addressed as an urgent priority, if we are to interpret results from those with lung disease with any confidence.
It has been shown that, whether using the tidal or Raised volume technique, serious mis-interpretation of forced expiratory maneuvers that are obtained with modern, commercially available equipment will occur if based on published reference equations derived from customized equipment developed within a different laboratory [99]. While a temporary adjustment factor has been proposed to address this problem, there is an urgent need to collate data from healthy infants studied in different laboratories but using the same protocols, equipment, and quality control, so that reliable, up to date reference equations can be developed. Given the impact of ethnicity on spirometric lung function in older subjects [91], such an initiative should not be restricted to infants of white European descent. Reference equations for tidal breathing, passive respiratory mechanics and plethysmographic FRC derived from healthy white infants studied using modern equipment have been published recently [100] but still require validation using data collected in other departments.
Can We Normalize by Body Size?
Lack of appropriate reference equations has led many centers to try to adjust for the rapid growth that occurs in infancy simply by dividing results such as tidal volume, lung volumes, or compliance by body weight at time of test and expressing results as ml/kg. There is increasing evidence that this practice is misguided and misleading, especially when applied to infants with lung disease in whom growth may be disproportionate [23, 26, 100]. Generally, as in older subjects, height (or in infants, length) is a better determinant of lung function than weight, although age and sex often have to be accounted for as well. Attempts to “normalize” lung function as a ratio in relation to length (i.e., per cm) should never be undertaken as the relationship between outcomes such as tidal or lung volumes and length is not linear, nor does the regression pass close to the origin, such that no consistent ratio occurs with growth in healthy subjects, making interpretation amongst those with lung disease impossible [100] (Box 8.2).
Box 8.2. Interpretation of Infant PFTs: Key Messages
-
Limited reference data based on current commercially available equipment with which to identify abnormal lung function in individual infants.
-
Prediction equations for infant PFTs should be based on appropriate regression equations with 95 % limits of normality derived from large number of infants (>100/sex), evenly distributed over first 2 years of life.
-
Infant PFT results should not be expressed as ratio of body size.
Reference Equations for Preschool PFTs
Given the fact that it is much simpler to study healthy, conscious preschool children than sleeping, sedated infants, it is not surprising that more lung function reference data are available for young children than infants [101]. The applicability of many these equations may, however, be limited by differences in technique, equipment, quality control, number and age range of subjects studied and the type of statistical analysis applied [94] (see Chap. 11 for further details). International collaborative initiatives to address these issues has resulted in recent publication of reference equations for preschool children of White European descent with respect to spirometry [92], specific airways resistance (sRaw) [85], Interrupter resistance (Rint) [90] and multiple breath washout assessments of LCI and FRC using mass spectrometry [89]. All-age multi-ethnic spirometry equations which encompass the preschool age range and which have been endorsed by all the international respiratory societies have now been developed [91] and are currently being implemented into commercial devices (www.lungfunction.org).
Can PFTs Be Used in the Clinical Management of Individual Infants?
Although there is little doubt about the potential value of infant lung function tests as a means of providing objective outcome measures in clinical or epidemiologic research studies [1, 2, 7, 8, 14, 15, 18, 21, 23, 24, 26, 28, 29, 31, 33, 37, 50, 63, 81, 102–106], their potential usefulness with respect to influencing clinical management within an individual infant remains highly debatable. The clinical usefulness of any technique depends not only on its ability to measure parameters that are relevant to the underlying pathophysiology and to discriminate between health and disease, but also on within-subject repeatability both within and between test occasions. As discussed earlier, although highly reproducible measurements of lung function can be made in infants during the same test occasion, little is known about within-subject, between-test repeatability, which severely limits their use in the clinical management of individual infants, except in departments with a high level of expertise and contemporaneous control data collected using identical equipment and software.
Can PFTs Be Used in the Clinical Management of Individual Preschool Children?
The major clinical role of preschool PFTs would be to monitor disease severity over time, evaluate response to treatments, and serve as objective outcome measures in clinical research studies. The results should always be interpreted in the context of other clinical signs and symptoms and, as with any diagnostic test, preschool PFT results should be just one additional piece of evidence utilized by clinicians in their assessment and clinical decision making. A recent report from an official ATS workshop which reviewed five preschool lung function tests (namely spirometry, specific airway resistance, the interrupter technique, forced oscillation, and multiple breath washout), concluded that while such tests were safe and feasible in 3–6 year old children if undertaken by suitably qualified individuals, insufficient evidence currently exists to recommend incorporation of these tests into the routine clinical monitoring of infants and young children with CF, BPD or recurrent wheeze although such tests were considered to be valuable tools with which to address specific concerns [13]. Spirometry can be successfully applied to the preschool population in the clinical setting to identify disease states and track lung function over time. Assessment of sRaw is also extremely feasible in this age group, but further work is required to standardize this technique, especially with regard to the breathing pattern adopted during measurements [85].
Conclusions
Thanks to the development of commercially available devices, it is becoming increasingly feasible to perform infant PFTs, but these assessments require special expertise, are expensive to perform and are very time consuming (both for staff and families). Clinical interpretation of results in individual infants is often limited by lack of appropriate normative data and knowledge of within-subject variability, the need for sedation, and inability to repeat measurements at frequent intervals. The reason for undertaking such measurements and confidence that meaningful results are likely to be obtained should therefore be clearly established before requesting such tests in infants, particularly those who are clinically unstable. While PFTs are safe and feasible in 3–6-year-old children if undertaken by suitably qualified individuals, insufficient evidence currently exists regarding within and between occasion variability to recommend incorporation of these tests into the routine clinical monitoring of young children with lung disease. Nevertheless, such tests can be valuable tools with which to address specific concerns such as ongoing symptoms or monitoring response to treatment and as outcome measures in clinical research studies.
Abbreviations
- ATS/ERS:
-
American Thoracic Society/European Respiratory Society
- BDR:
-
Bronchodilator responsiveness
- BPD:
-
Bronchopulmonary dysplasia
- CF:
-
Cystic fibrosis
- Crs:
-
Respiratory compliance
- FEFV:
-
Forced expiratory flow volume
- FEV0.5 :
-
Forced expired volume in 0.5 s
- FRC:
-
Functional residual capacity
- FVC:
-
Forced vital capacity
- LCI:
-
Lung clearance index
- LLN:
-
Lower limit of normal
- MBW:
-
Multiple breath washout
- PFT:
-
Pulmonary function test
- QC:
-
Quality control
- Rint:
-
Interrupter resistance
- RR:
-
Respiratory rate
- Rrs:
-
Respiratory resistance
- SDS:
-
Standard deviation score (also known as Z-score)
- SOT:
-
Single occlusion technique
- sRaw:
-
Specific airways resistance
- tPTEF/tE:
-
Time to reach peak tidal expiratory flow as a ratio of expiratory time
- ULN:
-
Upper limit of normal
- VT:
-
Tidal volume
References
Prendergast M, Rafferty GF, Milner AD, Broughton S, Davenport M, Jani J, et al. Lung function at follow-up of infants with surgically correctable anomalies. Pediatr Pulmonol. 2012;47(10):973–8.
Schmalisch G, Wilitzki S, Roehr CC, Proquitte H, Buhrer C. Development of lung function in very low birth weight infants with or without bronchopulmonary dysplasia: longitudinal assessment during the first 15 months of corrected age. BMC Pediatr. 2012;12:37.
Lum S, Bush A, Stocks J. Clinical pulmonary function testing for children with bronchopulmonary dysplasia. Pediatr Allergy Immunol Pulmonol. 2011;24(2):77–88.
May C, Kennedy C, Milner AD, Rafferty GF, Peacock JL, Greenough A. Lung function abnormalities in infants developing bronchopulmonary dysplasia. Arch Dis Child. 2011;96(11):1014–9.
Prendergast M, May C, Broughton S, Pollina E, Milner AD, Rafferty GF, et al. Chorioamnionitis, lung function and bronchopulmonary dysplasia in prematurely born infants. Arch Dis Child Fetal Neonatal Ed. 2011;96:F270–4.
Roehr CC, Wilitzki S, Opgen-Rhein B, Kalache K, Proquitte H, Buhrer C, et al. Early lung function testing in infants with aortic arch anomalies identifies patients at risk for airway obstruction. PLoS One. 2011;6(9):e24903.
Balinotti JE, Chakr VC, Tiller C, Kimmel R, Coates C, Kisling J, et al. Growth of lung parenchyma in infants and toddlers with chronic lung disease of infancy. Am J Respir Crit Care Med. 2010;181(10):1093–7.
Hoo AF, Gupta A, Lum S, Costeloe KL, Huertas-Ceballos A, Marlow N, et al. Impact of ethnicity and extreme prematurity on infant pulmonary function. Pediatr Pulmonol. 2013;49(7):679–87.
Aurora P, Bush A, Gustafsson P, Oliver C, Wallis C, Price J, et al. Multiple-breath washout as a marker of lung disease in preschool children with cystic fibrosis. Am J Respir Crit Care Med. 2005;171(3):249–56.
Aurora P, Stanojevic S, Wade A, Oliver C, Kozlowska W, Lum S, et al. Lung clearance index at 4 years predicts subsequent lung function in children with cystic fibrosis. Am J Respir Crit Care Med. 2011;183:752–8.
Gustafsson PM. Inert gas washout in preschool children. Paediatr Respir Rev. 2005;6(4):239–45.
Robinson PD, Latzin P, Verbanck S, Hall GL, Horsley A, Gappa M, et al. Consensus statement for inert gas washout measurement using multiple- and single-breath tests. Eur Respir J. 2013;41(3):507–22.
Rosenfeld M, Allen J, Arets B, Aurora P, Beydon N, Calogero C, et al. An Official American Thoracic Society Workshop Report: optimal lung function tests for monitoring cystic fibrosis, bronchopulmonary dysplasia and recurrent wheezing in children <6 years of age. Ann Am Thorac Soc. 2013;10:S1–11.
Stocks J, Lum S. Pulmonary function tests in infants and preschool children. In: Wilmott RW, Boat TF, Bush A, Chernick V, Deterding R, Ratjen F, editors. Kendig’s disorders of the respiratory tract in children. 8th ed. Philadelphia, PA: Elsevier; 2012. p. 169–210.
Latzin P, Roth S, Thamrin C, Hutten GJ, Pramana I, Kuehni CE, et al. Lung volume, breathing pattern and ventilation inhomogeneity in preterm and term infants. PLoS One. 2009;4(2):e4635.
Baldwin DN, Pillow JJ, Stocks J, Frey U. Lung-function tests in neonates and infants with chronic lung disease: tidal breathing and respiratory control. Pediatr Pulmonol. 2006;41(5):391–419.
Hutten GJ, van Eykern LA, Latzin P, Kyburz M, van Aalderen WM, Frey U. Relative impact of respiratory muscle activity on tidal flow and end expiratory volume in healthy neonates. Pediatr Pulmonol. 2008;43(9):882–91.
McEvoy C, Schilling D, Peters D, Tillotson C, Spitale P, Wallen L, et al. Respiratory compliance in preterm infants after a single rescue course of antenatal steroids: a randomized controlled trial. Am J Obstet Gynecol. 2010;202(6):544–9.
Gappa M, Pillow JJ, Allen J, Mayer O, Stocks J. Lung function tests in neonates and infants with chronic lung disease: lung and chest-wall mechanics. Pediatr Pulmonol. 2006;41(4):291–317.
Malmberg LP, von WL, Kotaniemi-Syrjanen A, Malmstrom K, Pelkonen AS, Makela MJ. Methacholine-induced lung function changes measured with infant body plethysmography. Pediatr Pulmonol. 2011;46(4):362–8.
Davis SD, Rosenfeld M, Kerby GS, Brumback L, Kloster MH, Acton JD, et al. Multicenter evaluation of infant lung function tests as cystic fibrosis clinical trial endpoints. Am J Respir Crit Care Med. 2010;182:1387–97.
Hulskamp G, Pillow JJ, Dinger J, Stocks J. Lung function tests in neonates and infants with chronic lung disease of infancy: functional residual capacity. Pediatr Pulmonol. 2006;41(1):1–22.
Hoo A-F, Thia L, Nguyen TD, Bush A, Chudleigh J, Lum S, et al. Lung function is abnormal in 3 month old infants with cystic fibrosis diagnosed by newborn screening. Thorax. 2012;67:874–81.
Nguyen TT, Thia LP, Hoo AF, Bush A, Aurora P, Wade A, et al. Evolution of lung function during the first year of life in newborn screened cystic fibrosis infants. Thorax 2013. doi:10.1136/thoraxjnl-2013-204023.
Belessis Y, Dixon B, Hawkins G, Pereira J, Peat J, MacDonald R, et al. Early cystic fibrosis lung disease detected by bronchoalveolar lavage and lung clearance index. Am J Respir Crit Care Med. 2012;185(8):862–73.
Hulskamp G, Lum S, Stocks J, Wade A, Hoo AF, Costeloe K, et al. Association of prematurity, lung disease and body size with lung volume and ventilation inhomogeneity in unsedated neonates: a multicentre study. Thorax. 2009;64(3):240–5.
Pillow JJ, Frerichs I, Stocks J. Lung function tests in neonates and infants with chronic lung disease: global and regional ventilation inhomogeneity. Pediatr Pulmonol. 2006;41(2):105–21.
Bisgaard H, Loland L, Holst KK, Pipper CB. Prenatal determinants of neonatal lung function in high-risk newborns. J Allergy Clin Immunol. 2009;123(3):651. 7.
Borrego LM, Stocks J, Leiria-Pinto P, Peralta I, Romeira AM, Neuparth N, et al. Lung function and clinical risk factors for asthma in infants and young children with recurrent wheeze. Thorax. 2009;64(3):203–9.
Friedrich L, Pitrez PM, Stein RT, Goldani M, Tepper R, Jones MH. Growth rate of lung function in healthy preterm infants. Am J Respir Crit Care Med. 2007;176(12):1269–73.
Jones M. Effect of preterm birth on airway function and lung growth. Paediatr Respir Rev. 2009;10 Suppl 1:9–11.
Kozlowska WJ, Bush A, Wade A, Aurora P, Carr SB, Castle RA, et al. Lung function from infancy to the preschool years after clinical diagnosis of cystic fibrosis. Am J Respir Crit Care Med. 2008;178(1):42–9.
Linnane BM, Hall GL, Nolan G, Brennan S, Stick SM, Sly PD, et al. Lung function in infants with cystic fibrosis diagnosed by newborn screening. Am J Respir Crit Care Med. 2008;178(12):1238–44.
Lum S, Stocks J, Castile R, Davies S, Henschen M, Jones M, et al. ATS/ERS statement: raised volume forced expirations in infants: guidelines for current practice. Am J Respir Crit Care Med. 2005;172(11):1463–71.
Lum S, Hulskamp G, Merkus P, Baraldi E, Hofhuis W, Stocks J. Lung function tests in neonates and infants with chronic lung disease: forced expiratory maneuvers. Pediatr Pulmonol. 2006;41(3):199–214.
Pillarisetti N, Williamson E, Linnane B, Skoric B, Robertson CF, Robinson P, et al. Infection, inflammation, and lung function decline in infants with cystic fibrosis. Am J Respir Crit Care Med. 2011;184(1):75–81.
Rosenfeld M, Ratjen F, Brumback L, Daniel S, Rowbotham R, McNamara S, et al. Inhaled hypertonic saline in infants and children younger than 6 years with cystic fibrosis: the ISIS randomized controlled trial. JAMA. 2012;307(21):2269–77.
Stocks J, Sly PD, Tepper RS, Morgan WJ. Infant respiratory function testing. 1st ed. New York: Wiley; 1996.
Bates J, Schmalisch G, Filbrun D, Stocks J. Tidal breath analysis for infant pulmonary function testing. Eur Respir J. 2000;16:1180–92.
Frey U, Stocks J, Coates A, Sly P, Bates J. Standards for infant respiratory function testing: specifications for equipment used for infant pulmonary function testing. Eur Respir J. 2000;16:731–40.
Sly P, Tepper R, Henschen M, Gappa M, Stocks J. Standards for infant respiratory function testing: tidal forced expirations. Eur Respir J. 2000;16:741–8.
Stocks J, Sly P, Morris MG, Frey U. Standards for infant respiratory function testing: what(ever) next? Eur Respir J. 2000;16:581–4.
Gappa M, Colin AA, Goetz I, Stocks J. Passive respiratory mechanics: the occlusion techniques. Eur Respir J. 2001;17:141–8.
Morris MG, Gustafsson P, Tepper R, Gappa M, Stocks J. Standards for infant respiratory function testing: the bias flow nitrogen washout technique for measuring the functional residual capacity. Eur Respir J. 2001;17:529–36.
Stocks J, Godfrey S, Beardsmore C, Bar-Yishay E, Castile R. Standards for infant respiratory function testing: plethysmographic measurements of lung volume and airway resistance. Eur Respir J. 2001;17:302–12.
Subbarao P, Hulskamp G, Stocks J. Limitations of electronic compensation for measuring plethysmographic airway resistance in infants. Pediatr Pulmonol. 2005;40(1):45–52.
Broughton S, Rafferty GF, Milner AD, Greenough A. Effect of electronic compensation on plethysmographic airway resistance measurements. Pediatr Pulmonol. 2008;43(1):104.
Vilozni D, Efrati O, Hakim F, Adler A, Livnat G, Bentur L. FRC measurements using body plethysmography in young children. Pediatr Pulmonol. 2009;44(9):885–91.
Thamrin C, Gangell CL, Udomittipong K, Kusel MM, Patterson H, Fukushima T, et al. Assessment of bronchodilator responsiveness in preschool children using forced oscillations. Thorax. 2007;62(9):814–9.
Stocks J, Thia L, Sonnappa S. Evaluation and use of childhood lung function tests in cystic fibrosis. Curr Opin Pulm Med. 2012;18:602–8.
Sonnappa S, Bastardo C, Wade A, Seglani S, McKenzie SA, Bush A, et al. Symptom-pattern phenotype and pulmonary function in preschool wheezers. J Allergy Clin Immunol. 2010;126:519–26.
Sonnappa S, Bastardo C, Wade A, Bush A, Stocks J, Aurora P. Repeatability and bronchodilator reversibility of lung function in preschool children. Eur Respir J. 2013;42:116–24.
Ren CL, Rosenfeld M, Mayer OH, Davis SD, Kloster M, Castile RG, et al. Analysis of the associations between lung function and clinical features in preschool children with cystic fibrosis. Pediatr Pulmonol. 2012;47(6):574–81.
Oostveen E, Dom S, Desager K, Hagendorens M, De BW, Weyler J. Lung function and bronchodilator response in 4-year-old children with different wheezing phenotypes. Eur Respir J. 2010;35(4):865–72.
Nielsen KG. Lung function and bronchial responsiveness in young children. Clinical and research applications. Dan Med Bull. 2006;53(1):46–75.
Mele L, Sly PD, Calogero C, Bernardini R, Novembre E, Azzari C, et al. Assessment and validation of bronchodilation using the interrupter technique in preschool children. Pediatr Pulmonol. 2010;45(7):633–8.
Marchal F, Schweitzer C, Thuy LV. Forced oscillations, interrupter technique and body plethysmography in the preschool child. Paediatr Respir Rev. 2005;6(4):278–84.
Kerby GS, Rosenfeld M, Ren CL, Mayer OH, Brumback L, Castile R, et al. Lung function distinguishes preschool children with CF from healthy controls in a multi-center setting. Pediatr Pulmonol. 2012;47(6):597–605.
Jeng MJ, Chang HL, Tsai MC, Tsao PC, Yang CF, Lee YS, et al. Spirometric pulmonary function parameters of healthy Chinese children aged 3–6 years in Taiwan. Pediatr Pulmonol. 2009;44(7):676–82.
Gangell CL, Horak Jr F, Patterson HJ, Sly PD, Stick SM, Hall GL. Respiratory impedance in children with cystic fibrosis using forced oscillations in clinic. Eur Respir J. 2007;30(5):892–7.
Gangell CL, Hall GL, Stick SM, Sly PD. Lung function testing in preschool-aged children with cystic fibrosis in the clinical setting. Pediatr Pulmonol. 2010;45(5):419–33.
Galant SP, Nickerson B. Lung function measurement in the assessment of childhood asthma: recent important developments. Curr Opin Allergy Clin Immunol. 2010;10(2):149–54.
Brumback LC, Davis SD, Kerby GS, Kloster M, Johnson R, Castile R, et al. Lung function from infancy to preschool in a cohort of children with cystic fibrosis. Eur Respir J. 2013;41(1):60–6.
Borrego LM, Stocks J, Almeida I, Stanojevic S, Antunes J, Leiria-Pinto P, et al. Bronchodilator responsiveness using spirometry in healthy and asthmatic preschool children. Arch Dis Child. 2013;98(2):112–7.
Beydon N. Assessment of bronchial responsiveness in preschool children. Paediatr Respir Rev. 2006;7 Suppl 1:S23–5.
Beydon N, M'buila C, Bados A, Peiffer C, Bernard A, Zaccaria I, et al. Interrupter resistance short-term repeatability and bronchodilator response in preschool children. Respir Med. 2007;101(12):2482–7.
Beydon N, Davis SD, Lombardi E, Allen JL, Arets HG, Aurora P, et al. An official American Thoracic Society/European Respiratory Society statement: pulmonary function testing in preschool children. Am J Respir Crit Care Med. 2007;175(12):1304–45.
Aurora P, Stocks J, Oliver C, Saunders C, Castle R, Chaziparasidis G, et al. Quality control for spirometry in preschool children with and without lung disease. Am J Respir Crit Care Med. 2004;169(10):1152–9.
Mayer OH, Jawad AF, McDonough J, Allen J. Lung function in 3–5-year-old children with cystic fibrosis. Pediatr Pulmonol. 2008;43(12):1214–23.
Vilozni D, Livnat G, Dabbah H, Elias N, Hakim F, Bentur L. The potential use of spirometry during methacholine challenge test in young children with respiratory symptoms. Pediatr Pulmonol. 2009;44(7):720–7.
Vilozni D, Berkun Y, Levi Y, Weiss B, Jacobson JM, Efrati O. The feasibility and validity of forced spirometry in ataxia telangiectasia. Pediatr Pulmonol. 2010;45(10):1030–6.
Bisgaard H, Nielsen KG. Plethysmographic measurements of specific airway resistance in young children. Chest. 2005;128(1):355–62.
Beydon N, M’buila C, Peiffer C, Bernard A, Zaccaria I, Denjean A. Can bronchodilator response predict bronchial response to methacholine in preschool coughers? Pediatr Pulmonol. 2008;43(8):815–21.
Chan EY, Bridge PD, Dundas I, Pao CS, Healy MJ, McKenzie SA. Repeatability of airway resistance measurements made using the interrupter technique. Thorax. 2003;58(4):344–7.
Gochicoa LG, Thome-Ortiz LP, Furuya ME, Canto R, Ruiz-Garcia ME, Zuniga-Vazquez G, et al. Reference values for airway resistance in newborns, infants and preschoolers from a Latin American population. Respirology. 2012;17(4):667–73.
Nielsen KG, Bisgaard H. Discriminative capacity of bronchodilator response measured with three different lung function techniques in asthmatic and healthy children aged 2 to 5 years. Am J Respir Crit Care Med. 2001;164(4):554–9.
Song TW, Kim KW, Kim ES, Park JW, Sohn MH, Kim KE. Utility of impulse oscillometry in young children with asthma. Pediatr Allergy Immunol. 2008;19(8):763–8.
Thamrin C, Albu G, Sly PD, Hantos Z. Negative impact of the noseclip on high-frequency respiratory impedance measurements. Respir Physiol Neurobiol. 2009;165(1):115–8.
Thamrin C, Gangell CL, Kusel MM, Schultz A, Hall GL, Stick SM, et al. Expression of bronchodilator response using forced oscillation technique measurements: absolute versus relative. Eur Respir J. 2010;36(1):212.
Lum S, Stocks J. Forced expiratory manoeuvres. In: Merkus P, Frey U, editors. Paediatric lung function. Sheffield, UK: ERS Journals Ltd; 2010. p. 46–65.
Stocks J, Coates A, Bush A. Lung function in infants and young children with chronic lung disease of infancy: The next steps? Pediatr Pulmonol. 2007;42(1):3–9.
Bisgaard H, Jensen SM, Bonnelykke K. Interaction between asthma and lung function growth in early life. Am J Respir Crit Care Med. 2012;185(11):1183–9.
Sonnappa S, Bastardo CM, Saglani S, Bush A, Aurora P. Relationship between past airway pathology and current lung function in preschool wheezers. Eur Respir J. 2011;38(6):1431–6.
Vilozni D, Hakim F, Adler A, Livnat G, Bar-Yishay E, Bentur L. Reduced vital capacity after methacholine challenge in early childhood–is it due to trapped air or loss of motivation. Respir Med. 2009;103(1):109–16.
Kirkby J, Stanojevic S, Welsh L, Lum S, Badier M, Beardsmore C, et al. Reference equations for specific airway resistance in children: the asthma UK initiative. Eur Respir J. 2010;36(3):622–9.
Nielsen KG, Pressler T, Klug B, Koch C, Bisgaard H. Serial lung function and responsiveness in cystic fibrosis during early childhood. Am J Respir Crit Care Med. 2004;169(11):1209–16.
Kairamkonda VR, Richardson J, Subhedar N, Bridge PD, Shaw NJ. Lung function measurement in prematurely born preschool children with and without chronic lung disease. J Perinatol. 2008;28(3):199–204.
Lesnick BL, Davis SD. Infant pulmonary function testing: overview of technology and practical considerations—new current procedural terminology codes effective 2010. Chest. 2011;139(5):1197–202.
Lum S, Stocks J, Stanojevic S, Wade A, Robinson P, Gustafsson P, et al. Age and height dependence of lung clearance index and functional residual capacity. Eur Respir J. 2013;41:1371–7.
Merkus PJ, Stocks J, Beydon N, Lombardi E, Jones M, McKenzie SA, et al. Reference ranges for interrupter resistance technique: the Asthma UK Initiative. Eur Respir J. 2010;36(1):157–63.
Quanjer PH, Stanojevic S, Cole TJ, Baur X, Hall GL, Culver B, et al. Multi-ethnic reference values for spirometry for the 3–95 year age range: the global lung function 2012 equations. Eur Respir J. 2012;40:1324–43.
Stanojevic S, Wade A, Cole TJ, Lum S, Custovic A, Silverman M, et al. Spirometry centile charts for young Caucasian children: the Asthma UK Collaborative Initiative. Am J Respir Crit Care Med. 2009;180(6):547–52.
Stocks J, Kirkby J, Lum S. How to avoid misinterpreting lung function tests in children: a few practical tips. Pediatr Respir Rev. 2014;14(2):170–80.
Stanojevic S, Wade A, Stocks J. Reference values for lung function: past, present and future. Eur Respir J. 2010;36(1):12–9.
Stanojevic S, Stocks J, Bountziouka V, Aurora P, Kirkby J, Bourke S, et al. The impact of switching to the new global lung function initiative equations on spirometry results in the UK CF Registry. J Cyst Fibros. 2013;13(3):319–27.
Keklikian E, Sanchez-Solis M, Bonina AJ, Meneguzzi A, Pastor-Vivero MD, Mondejar-Lopez P, et al. Do risk factors for persistent asthma modify lung function in infants and young children with recurrent wheeze? Pediatr Pulmonol. 2010;45(9):914–8.
Pelkonen AS, Kotaniemi-Syrjanen A, Malmstrom K, Malmberg LP, Makela MJ. Clinical findings associated with abnormal lung function in children aged 3–26 months with recurrent respiratory symptoms. Acta Paediatr. 2010;99(8):1175–9.
Stocks J, Modi N, Tepper R. Need for healthy controls when assessing lung function in infants with respiratory disease. Am J Respir Crit Care Med. 2010;182:1340–2.
Lum S, Hoo AF, Hulskamp G, Wade A, Stocks J. Potential misinterpretation of infant lung function unless prospective healthy controls are studied. Pediatr Pulmonol. 2010;45:906–13.
Nguyen TTD, Hoo A-F, Lum S, Wade A, Thia LP, Stocks J. New reference equations to improve interpretation of infant lung function. Pediatr Pulmonol. 2013;48(4):370–80.
Stanojevic S, Wade A, Lum S, Stocks J. Reference equations for pulmonary function tests in preschool children: a review. Pediatr Pulmonol. 2007;42(10):962–72.
Pike KC, Rose-Zerilli MJ, Osvald EC, Inskip HM, Godfrey KM, Crozier SR, et al. The relationship between infant lung function and the risk of wheeze in the preschool years. Pediatr Pulmonol. 2011;46(1):75–82.
van der Zalm MM, Uiterwaal CS, Wilbrink B, Koopman M, Verheij TJ, van der Ent CK. The influence of neonatal lung function on rhinovirus-associated wheeze. Am J Respir Crit Care Med. 2011;183(2):262–7.
Llapur CJ, Martinez TM, Coates C, Tiller C, Wiebke JL, Li X, et al. Lung structure and function of infants with recurrent wheeze when asymptomatic. Eur Respir J. 2009;33(1):107–12.
Mallol J, Aguirre V, Barrueto L, Wandalsen G, Tepper R. Effect of inhaled fluticasone on lung function in infants with recurrent wheezing: a randomised controlled trial. Allergol Immunopathol (Madr). 2009;37(2):57–62.
Sly PD, Brennan S, Gangell C, de Klerk N, Murray C, Mott L, et al. Lung disease at diagnosis in infants with cystic fibrosis detected by newborn screening. Am J Respir Crit Care Med. 2009;180(2):146–52.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this chapter
Cite this chapter
Stocks, J. (2015). Infant and Preschool Pulmonary Function Tests. In: Davis, S., Eber, E., Koumbourlis, A. (eds) Diagnostic Tests in Pediatric Pulmonology. Respiratory Medicine. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-1801-0_8
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1801-0_8
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-1800-3
Online ISBN: 978-1-4939-1801-0
eBook Packages: MedicineMedicine (R0)