Abstract
Type 2 diabetes mellitus (T2DM) and obesity represent the twin scourge of the modern era demanding urgent and long-lasting solutions. T2DM had a worldwide prevalence among adults of at least 285 million in 2010, and this will likely increase to 439 million by 2030 (Shaw et al., Diabetes Res Clin Pract 87(1):4–14, 2010). Medical management of diabetes has historically revolved around lifestyle intervention combined with pharmacotherapy to address insulin resistance and/or defective beta cell function, the core issues in the pathophysiology of this disease. Despite several advances in medical therapy, few patients are able to achieve and maintain optimal glycemic targets in the long term. Moreover, weight gain from insulin usage and the risk of hypoglycemia often preclude intensive medical management.
Several gastrointestinal procedures originally devised to produce significant and durable weight loss among the morbidly obese were shown to also ameliorate virtually all obesity-related comorbidities and reduce long-term mortality. While weight loss by any means would intuitively be expected to result in an improvement of diabetes and other metabolic disorders, evidence from both clinical and animal experimental data suggests that these effects are partly independent of weight loss. The impact on diabetes is striking; in that, 84 % of obese patients who undergo gastric bypass experience diabetes remission maintain euglycemia without diabetes medications (Rubino et al., Annu Rev Med 61:393–411, 2010). This has led to a dramatic shift in goals with regard to the treatment of diabetes from achieving good glycemic control and delaying end-organ damage to complete disease remission.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Gastric bypass
- Biliopancreatic diversion
- Laparoscopic adjustable gastric banding
- Sleeve gastrectomy
- Type 2 diabetes mellitus
- Obesity
- Endoluminal sleeve
Introduction
The concept that gastrointestinal surgery may be intentionally used to treat T2DM was formalized by a multidisciplinary international group of experts at the Diabetes Surgery Summit in Rome, 2007 [3]. In consideration of the abundant data now available to attest to the efficacy and safety of bariatric surgery to treat T2DM in obese patients, the International Diabetes Federation (IDF) more recently issued its position statement [4], recommending the use of bariatric surgery in patients with diabetes and BMI > 35 kg/m2 and as an alternative treatment option in patients with BMI 30–35 kg/m2 inadequately controlled with optimal medical regimens.
Experimental data from rodent studies [5, 6] and subsequent observational studies in humans [7, 8] have demonstrated that less obese individuals may benefit similarly to morbidly obese subjects. There are currently several small- to medium-sized randomized controlled trials (RCTs) investigating the efficacy of different bariatric procedures versus optimal medical management to treat T2DM in mild to moderately obese subjects. The outcomes of published RCTs and observational studies and the putative mechanisms of effects of bariatric surgery on diabetes are discussed in this chapter.
Physiological Mechanisms of Diabetes Remission Following Bariatric Surgery
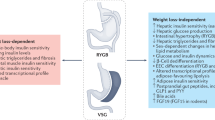
Improvement in glucose homeostasis and insulin sensitivity is an expected outcome of weight loss in obese individuals due to any intervention, whether it is medical or surgical. Concomitant with the profound weight loss following gastrointestinal surgery, there is improvement in insulin sensitivity with elevation in the levels of the insulin-sensitizing hormone adiponectin. The concentration of insulin receptors and markers of insulin signaling increase, and the lipid content within the muscle and liver also decreases after bariatric surgery [9].
It is now abundantly clear, however, that the improvement of glycemia is not determined by weight loss alone. Studies have shown that glycemic control improves within days after Roux-en-Y gastric bypass (RYGB) or biliopancreatic diversion (BPD) prior to any significant weight loss. In addition, glycemic control after equivalent weight loss is superior after RYGB as compared to that after laparoscopic adjustable gastric banding (LAGB) or caloric restriction [2, 7, 8]. Experimental evidence in favor of weight-independent mechanisms is derived from animal investigations using the duodenojejunal bypass (DJB) model developed by Rubino in rodents to study the effects of gastric bypass on glucose homeostasis [5]. In lean diabetic Goto-Kakizaki (GK) rats, Rubino and colleagues found that DJB (a bypass of the proximal intestine by the same amount as RYGB but without gastric restriction) rapidly and durably improved fasting glucose and postprandial hyperglycemia. These effects were not observed in sham-operated animals that had undergone equivalent weight loss by caloric restriction. Other investigators have subsequently made similar observations in nonobese diabetic GK rats and obese diabetic Zucker rats.
The gut is the largest endocrine organ secreting numerous hormones and factors involved not only in digestion but also regulation of body weight and glucose homeostasis. It is now recognized that surgical manipulation of the gut results in a change in the levels of various gut hormones including glucagon-like peptide-1 (GLP-1), peptide-YY (PYY), and ghrelin, which can significantly impact glycemic control and body weight.
GLP-1 is an incretin hormone that increases glucose tolerance by enhancing glucose-dependent insulin secretion, suppressing glucagon secretion, inhibiting gastric emptying, increasing β-cell mass, and possibly improving insulin insensitivity [10]. GLP-1 is produced primarily in the ileum and colon by nutrient-stimulated L cells. The postprandial GLP-1 response is consistently observed to be augmented during an oral glucose tolerance test or mixed meal test after the RYGB or BPD in obese nondiabetic and diabetic patients [9, 11]. The rise in GLP-1 level occurs as early as 2 days after surgery and has been shown to persist at 6 months and 1 year postoperatively [12]. Restrictive procedures, such as vertical banded gastroplasty (VBG) and LAGB, have not shown to be associated with any changes in the levels of GLP-1 postoperatively [13].
Ghrelin is an orexigenic hormone produced primarily bVBy the stomach, and administration of ghrelin and its analogs stimulates food intake. Ghrelin levels have an inverse relationship with body weight, and consequently obese individuals have lower ghrelin levels. Weight loss induced by caloric restriction results in an increase in the levels of ghrelin, which subsequently may contribute to the resistance to lifestyle interventions for obesity. Ghrelin may play a role in glucose homeostasis and has been shown to inhibit insulin secretion and suppress the insulin-sensitizing hormone adiponectin [14]. Ghrelin deletion in diabetic obese mice models has shown to reduce fasting glucose and insulin and thus improve glucose tolerance [15]. Several groups have reported a decrease in the levels of ghrelin after RYGB, which may partly account for the improved glycemia [12, 16]. This has not been consistently reported with other studies showing either unchanged or increased ghrelin levels [9, 17]. As expected, ghrelin levels are markedly suppressed following resection of the ghrelin-rich gastric fundus as with a sleeve gastrectomy. However, ghrelin levels show the normal physiological rise with weight loss after LAGB and VBG.
PYY is an anorexigenic hormone co-secreted with GLP-1 from intestinal L cells in response to food intake. PYY has been shown to decrease food intake in humans when injected. Experimental studies in rodents suggest that PYY may directly ameliorate insulin resistance [18]. Several studies have reported elevated PYY levels after RYGB. In a prospective, nonrandomized controlled study comparing the effects of medical and surgical treatment on PYY levels after similar weight loss, it was observed that PYY’s area under the curve increased following RYGB and sleeve gastrectomy, but remained unchanged in the medical arm [19].
Korner et al. [7] conducted a prospective study of gut hormone and metabolic changes after LAGB and RYGB and suggested that the changes in hormone secretion may contribute to the efficacy of the particular procedure in improving obesity-related comorbidities. It is established that PYY and GLP-1 are secreted shortly after food intake from L cells in the distal small bowel and colon. Both decrease appetite, increase satiety, slow gut motility, and improve insulin sensitivity. GLP-1 also functions as an incretin to potentiate glucose-stimulated insulin release. In contrast, ghrelin stimulates appetite, profoundly promotes food intake and gut motility, and decreases insulin sensitivity [20]. Korner et al. [7] found that in LAGB patients, the postprandial rise of GLP-1 measured at 30 min did not change after surgery, whereas in contrast, postprandial GLP-1 levels after RYGB were significantly greater compared with 30-min values before surgery and threefold higher compared with the same time points after LAGB. Fasting glucose, insulin, and homeostasis model assessment of insulin resistance (HOMA-IR) were also shown to decrease to a greater extent after RYGB. The degree of suppression of ghrelin levels post-meal did not differ significantly presurgery to week 52 in either the LAGB or RYGB and did not differ between groups. Laferrère et al. [8] suggested that the markedly increased incretin levels and effect observed after the RYGB may well be one of the mediators of the antidiabetic effects of the surgery, and they set out to determine whether the magnitude of change of the incretin levels is greater after the RYGB compared to a hypocaloric diet at an equivalent weight loss. Their results showed a markedly increased GLP-1 response to oral glucose after the RYGB, but GLP-1 levels tended to decrease after diet intervention, although this decrease was not significant. The incretin effect markedly increased after an RYGB, but not with diet, and suggests that this effect was not likely to be weight loss related. Therefore, this could play a key role in the remission and resolution of T2DM after an RYGB. Data from various studies has shown that beneficial changes of incretin levels occur rapidly after the RYGB, which is secondary to the bypass procedure, thus resulting in an improved insulin secretion profile and decreased postprandial glucose [21].
Another mechanism proposed in recent years is the effect of bariatric surgery on the alteration of taste. This novel weight loss mechanism involves alterations in food preferences, which may be secondary to changes in taste detection and reward. Miras et al. [22] have shown that after RYGB, patients’ eating behavior changes, and they start to adopt healthier food preferences by avoiding high-calorie and high-fat foods. Patients find sweet and fatty meals less pleasant than preoperatively, and this may be attributed to changes in the sense of taste. A putative mechanism to explain this is that RYGB may reverse the higher activation of brain taste reward and addiction centers in response to high calorie and fat tasting.
Two hypotheses have been proposed to explain improved glycemia following gastrointestinal surgery. The “hindgut hypothesis” posits that the expedited delivery of ingested nutrients to the lower bowel, due to an intestinal bypass, stimulates L cells, which results in an increased secretion of incretin hormones and an improved glucose homeostasis [5]. Consistent with the hindgut hypothesis, the bariatric operations most noted for rapid T2DM remission, RYGB and BPD, create GI shortcuts for food to access the distal bowel. However, a significant improvement in glycemia and remission of diabetes with elevated incretin levels is also noted to occur after a sleeve gastrectomy, which does not involve an intestinal bypass. It must be noted that GLP-1 secretion is stimulated not only by direct nutrient contact with distal intestinal L cells but also by proximal nutrient-related signals that are transmitted from the duodenum to the distal bowel by neural pathways and other unknown mechanisms [9]. The alternative hypothesis is the “foregut hypothesis,” which depends on the exclusion of the duodenum and proximal jejunum from the transit of nutrients, possibly preventing secretion of a putative signal that promotes insulin resistance and T2DM [6]. Rubino et al. conducted a study to investigate these mechanisms by testing both hypotheses and found that the exclusion of the proximal small intestine from contact with ingested nutrients is a critical component in the mechanism improving glucose tolerance after the DJB in GK rats, thus supporting the foregut hypothesis [6].
Other hypothesized antidiabetic mechanisms after bariatric surgery include changes in intestinal nutrient-sensing mechanisms regulating insulin sensitivity, disruption of vagal afferent and efferent innervations, bile acid perturbations, changes in gut microbiota, and alterations in undiscovered gut factors.
The Effects and Outcomes of Bariatric Surgery on Diabetes in Patients with a BMI > 35 kg/m2
The outcomes of bariatric surgery on diabetes and the metabolic profile in patients with a BMI of >35 kg/m2 have been well documented over many years.
Buchwald et al. [23] conducted a meta-analysis including 22,094 patients with T2DM reported an overall 77 % remission rate of T2DM (defined as persistent normoglycemia without diabetes medication after bariatric surgery). The mean procedure-specific resolution of T2DM was 48 % for LAGB, 68 % for VBG, 84 % for RYGB, and 98 % for BPD. The drawback of this study is that the majority of these studies included was retrospective and had a short follow-up duration of 1–3 years. The multicenter Swedish Obese Subjects (SOS) study, a large prospective observational study [24], compared bariatric surgery (LAGB n = 156, VBG n = 451, RYGB n = 34) with conservative medical management in a group of well-matched obese patients. At 2 years, 72 % of diabetic subjects in the surgical group achieved remission of T2DM compared to 21 % in the medically treated arm. At 10 years, the relative risk of incident T2DM was three times lower, and the rates of recovery from T2DM were three times greater, for patients who underwent surgery than for individuals in the control group. The proportion of subjects in whom the remission was sustained at 10 years declined to 36 % in the surgical group and 13 % in the medical group. It must be noted, however, that approximately 95 % of patients in the SOS study underwent gastric restrictive procedures rather than RYGB.
Recently, Schauer et al. [25] published results from their randomized, non-blinded, single-center trial and compared intensive medical therapy alone versus medical therapy plus RYGB or sleeve gastrectomy (SG) in 150 obese patients (BMI 27–43 kg/m2) with uncontrolled T2DM. The primary end point was the proportion of patients with a glycated hemoglobin (HbA1c) level of 6.0 % or less 12 months after treatment. The proportion of patients who reached the primary end point was 12 % for the medical group, 42 % in the RYGB group, and 37 % in the SG group. Glycemic control was demonstrated to improve in all three groups with a mean HbA1c level of 7.5 ± 1.8 % in the medical therapy group, 6.4 ± 0.9 % in the gastric bypass group, and 6.6 ± 1.0 % in the sleeve gastrectomy group. It was demonstrated that many patients in the surgical groups, particularly in the RYGB group, achieved glycemic control without the use of diabetes medications after 1-year follow-up. Secondary end points, including BMI, body weight, waist circumference, and the HOMA-IR, improved more significantly in the surgical groups than the medical therapy group. It was also shown that the reduction in the use of glycemic pharmacotherapy occurred before achievement of maximal weight loss, thus supporting the weight-independent mechanisms of surgery. This trial also supported the notion that bariatric surgery represents a useful management strategy for uncontrolled T2DM.
Mingrone et al. [26] published results from an RCT conducted including 60 patients (BMI ≥ 35 kg/m2) with a history of at least 5 years of T2DM, and an HbAlc level of 7.0 % or more. They compared conventional medical therapy to the RYGB or BPD. The primary end point was the rate of diabetes remission at 2 years (defined as a fasting glucose level of <100 mg/dL and an HbA1c level of <6.5 % in the absence of pharmacological therapy). At 2 years follow-up, diabetes remission had occurred in no patients in the medical therapy group versus 75 % in the RYGB group, and 95 % in the BPD group. All patients in the surgical groups had discontinued their pharmacotherapy (oral hypoglycemic agents and insulin) within 15 days after the operation. At 2 years, the average baseline HbA1c level (8.65 ± 1.45 %) had decreased in all groups, but patients in the two surgical groups had the greatest degree of improvement (average HbA1c levels, 7.69 ± 0.57 % in the medical therapy group, 6.35 ± 1.42 % in the RYGB group, and 4.95 ± 0.49 % in the BPD group). The findings support the conclusion that bariatric surgery may be more effective than conventional medical therapy in controlling hyperglycemia in severely obese patients with T2DM.
The Effects and Outcomes of Bariatric Surgery on Diabetes in Patients with a BMI > 35 kg/m2
The effects of bariatric surgery in the morbidly obese population (>35 kg/m2) are well documented. The field of metabolic surgery has become increasingly researched in recent years, investigating the effects and benefits of operating on those patients with a BMI of less than 35 kg/m2. The aims of metabolic surgery differ slightly from bariatric surgery; in that, there is a shift in goals of surgery from weight reduction to the control of metabolic disease, with the aim of “curing” T2DM or putting this prevalent condition into remission. Recently, prospective RCTs have been conducted in lower BMI patients (<35 kg/m2) in order to compare various gastrointestinal surgical procedures versus intensive medical management, to identify the differences between conventional treatment and metabolic surgery.
Dixon et al. [27] conducted an RCT comparing the LAGB versus conventional therapy in 60 obese patients (BMI > 30 and <40 kg/m2) with recently diagnosed (<2 years) T2DM. The primary outcome measure was the remission of T2DM (defined as fasting plasma glucose < 126 mg/dL and an HbA1c level of <6.2 % while taking no glycemic therapy). After a 2-year follow-up, remission of T2DM was achieved in 73 % in the surgical group and 13 % in the conventional therapy group, and it was found to be related to weight loss. Mean levels of fasting plasma glucose and HbA1c were significantly lower in the LAGB group at 2 years. There was also a significant reduction in the use of pharmacotherapy for glycemic control in the LAGB group at 2 years. Secondary outcome measures, such as insulin sensitivity and levels of triglycerides and HDL, were also improved in the surgical group. It was concluded that this trial presents evidence to support the early consideration of surgery in the treatment of obese patients with T2DM.
Lee et al. designed a randomized, double-blind trial [28] in order to compare the RYGB versus SG on T2DM resolution (remission defined as fasting plasma glucose < 126 mg/dL, HbA1c < 6.5 % without the use of oral hypoglycemics or insulin) in non-morbidly obese patients (BMI < 35 kg/m2) who had T2DM that was inadequately controlled. The resolution rate was 93 % in the RYGB group compared with 47 % in the SG group. These results are consistent with the RYGB being more effective than restrictive-type procedures. The RYGB group also achieved a lower waist circumference, fasting plasma glucose level, HbA1c level, and blood lipid level. Therefore, the RYGB group had a higher remission rate for the metabolic syndrome than the SG group. This study concluded that RYGB is more effective than SG for surgical treatment of inadequately controlled T2DM, suggesting that duodenal exclusion plays a role in the mechanism behind the remission of T2DM.
Cohen et al. published results from a prospective study [29] following 66 diabetic patients (BMI 30–35 kg/m2) up to 6 years who elected to have the RYGB. The percentage of patients experiencing diabetes remission (defined as an HbA1c < 6.5 % without the use of hypoglycemic medication) was identified. The mean HbA1c fell progressively throughout the duration of the study from 9.7 ± 1.5 to 5.9 ± 0.1 %, and fasting plasma glucose fell from 156 ± 11 to 97 ± 5 mg/dL. HOMA-IR also fell dramatically within the first 6 months. Remission of diabetes occurred in 88 % of patients, whose diabetes medications were discontinued 3–26 weeks after surgery. Improvement of diabetes without full remission was seen in 11 % of patients, which lead to a decrease in the usage of pharmacotherapy and withdrawal of insulin when previous used. There was found to be no correlation between the change in body weight and change in HbA1c at any postoperative time point. There was also no association between the amount of weight lost and magnitude of improvement in β-cell sensitivity to glucose.
Despite these prospective RCTs being conducted at various institutions, it is clear that longer-term follow-up of these patients is required. Larger and perhaps multicenter trials are necessary to evaluate the potential benefits of bariatric surgery for the treatment of T2DM in less obese patients. Studies are consistent in showing the efficacy of surgery with regard to the improvement in glycemic indexes as well as improving other aspects of the metabolic syndrome. The impact of surgery on microvascular and macrovascular complications of diabetes needs to be assessed with long-term follow-up.
Improvement in Cardiovascular Outcomes and Mortality in Type 2 Diabetic Patients
It is now evident that apart from its glycemic effects, bariatric surgery confers non-glycemic benefits including improvement of cardiovascular risk factors such as dyslipidemia and hypertension. More importantly, it has been established that there is a reduction in mortality. The meta-analysis by Buchwald et al. [23] showed a marked decrease in levels of total cholesterol, LDL cholesterol, and triglycerides after gastrointestinal surgery. An improvement in hyperlipidemia was shown in approximately 70 % of patients, and hypertension was shown to improve or resolve in 79 % of patients. Lee et al. have demonstrated these beneficial effects of gastrointestinal surgery in patients with a BMI < 35 kg/m2 [30]. Follow-up of participants in the SOS study after an average of 11 years found that bariatric surgery was associated with a 29 % reduction in all-cause mortality after adjusting for age, sex, and risk factors in this severely obese group [31].
In a recent study, Cohen et al. [29] prospectively followed up type 2 diabetic patients who had an RYGB, and it was demonstrated that the predicted 10-year risk of cardiovascular disease, calculated by the UKPDS risk engine, fell substantially after surgery. There was a decrease in the risk of events: 71 % decrease in coronary heart disease, 84 % decrease in fatal, 50 % decrease in stroke, and 57 % decrease in fatal stroke.
Romeo et al. recently published on the prospective, controlled SOS study, looking at the effects of bariatric surgery on cardiovascular events on participants with T2DM [32]. The mean follow-up was 13.3 years for all cardiovascular events. Bariatric surgery was associated with a reduction in the incidence of fatal and nonfatal cardiovascular events, and importantly, the benefit of surgery was present after adjusting for baseline parameters. Bariatric surgery was associated with a lower incidence of myocardial infarction. A total of 38 of the 345 individuals in the surgery group compared with 43 of the 262 individuals in the control group had myocardial infarction during follow-up. Interestingly, no significant differences in the incidence of myocardial infarction were found between the different surgical procedures (RYGB, LAGB, vertical gastroplasty). However, bariatric surgery was not found to be associated with changes in the incidence of cerebral stroke in these type 2 diabetic patients.
Adams et al. conducted a retrospective cohort study including 7,925 severely obese patients and 7,925 similarly obese matched controls [33]. After a mean follow-up of 8.4 years, it was demonstrated that surgery reduced overall mortality by 40 %, cardiovascular mortality by 56 %, cancer mortality by 60 %, and diabetes-related mortality by 92 %.
Comparative Efficacy of Different Bariatric Procedures to Treat Diabetes
Bariatric procedures differ in their ability to ameliorate T2DM, with intestinal bypass procedures (i.e., RYGB, BPD) generally associated with greater glycemic control and remission rates than purely restrictive procedures (i.e., LAGB). As indicated in the meta-analysis by Buchwald et al. [23], BPD appears to be the most efficacious closely followed by RYGB and then LAGB. A systematic review by Tice et al. [34] of 14 comparative studies, albeit of low quality (mostly retrospective and unmatched), testified to the superior efficacy of RYGB over LAGB in treating T2DM. There has been until now a paucity of data from RCTs comparing the efficacy of various bariatric procedures to treat diabetes. There are currently several ongoing small- to medium-sized RCTs comparing the efficacy of medical versus surgical treatment of type 2 diabetes including some that have more than one bariatric procedure as the surgical comparator. The results of some of these have recently been published. Lee et al. [28] have reported the results of their RCT comparing gastric bypass to sleeve gastrectomy in patients with BMI 25–35 kg/m2; the remission rate for T2DM was 93 % for patients who underwent RYGB compared to 47 % for those who underwent sleeve gastrectomy. The recently published RCT by Schauer et al. [25] also indicates superior efficacy of RYGB over sleeve gastrectomy in the treatment of diabetes in obese individuals. On the other hand, BPD produced greater remission of diabetes in morbidly obese patients compared to RYGB (95 % versus 75 %) in the RCT reported by Mingrone et al. [26].
Predictors of Remission of Type 2 Diabetes Following Bariatric Surgery
As discussed above, the choice of procedure is an important determinant of outcome with a decreasing gradient of efficacy predicted from BPD, RYGB to SG and then LAGB. Other factors that have been positively correlated with diabetes remission are percentage of excess weight loss (% EWL), younger age, lower preop HbA1c, and shorter duration of diabetes (less than 5 years) [35]. Severity of diabetes, as judged by preop treatment modality, has also been noted to be a significant factor. Schauer et al. [36] have reported in their series of 191 obese diabetic patients (the majority of whom were on oral agents or insulin) a diabetes remission rate of 97 % in diet-controlled, 87 % in oral agent-treated, and 62 % in insulin-treated subjects. This was also confirmed by a recent retrospective analysis of 505 morbidly obese diabetic patients who underwent RYGB [37]. In this study, a more recent diagnosis of T2DM and the absence of preoperative insulin therapy were significant predictors of remission, independent of the percentage of EWL. Dixon et al. [38] have recently identified diabetes duration < 4 years, BMI > 35 kg/m2, and fasting c-peptide concentration > 2.9 ng/mL as three clinically useful cutoffs and independent preoperative predictors of remission after analyzing the outcomes of 154 ethnic Chinese subjects after gastric bypass. C-peptide > 3 ng/mL has also previously been shown to be an important predictor of diabetes resolution after sleeve gastrectomy in non-morbidly obese diabetic subjects by Lee et al. [39].
The Future of Metabolic Surgery
In recent years, there has been an increasing amount of research conducted to look at minimally invasive methods with the aim of mimicking the effects of gastrointestinal surgery but providing a safer and reversible alternative to conventional surgery. Endoluminal and transgastric procedures are evolving concepts that combine the skills and techniques of flexible endoscopy with minimally invasive surgery. This is the future of bariatric and metabolic surgery in order to develop less morbid and less costly treatment options [40].
The endoluminal sleeve (ELS) was an idea, which stemmed from an experimental model in rats performed by Rubino et al. They developed an (ELS) to prevent contact between nutrients and the duodenal mucosa in rats [2], therefore producing a functional duodenal bypass without creating a rapid delivery of nutrient to the distal bowel. Rats undergoing the ELS demonstrated a dramatic improvement of glucose tolerance compared to matched controls in which the ELS had been fenestrated to allow nutrients to come into contact with the duodenal mucosa. The antidiabetic effect of ELS was also shown by Aguirre et al. [41] in a diet-induced rat model of insulin resistance. A prospective randomized trial comparing ELS to sham endoscopy plus a low-fat diet and exercise showed a dramatic decrease in HbA1c of 2.9 % in the ELS group compared to the conventional therapy group [42].
Gut hormones and the enteric nervous system are involved in the regulation of satiety signals, GI motility, and insulin sensitivity. Electrophysiologic devices interfere with vagal signals between the brain and gastrointestinal tract, through a variety of mechanisms including gastric stimulation or pacing, neuromodulation, vagal resection, and intermittent vagal nerve blockage as a means of controlling satiety [42]. The TANTALUS system is a laparoscopically implantable system, which is a gastric electrical stimulator. Bohdjalian et al. [43] conducted a multicenter open-label European feasibility trial involving 24 obese diabetic patients treated with insulin and/or oral hyperglycemic agents with a BMI between 33.3 and 49.7 kg/m2. In those subjects that reached the 1-year visit, weight was reduced by 4.5 ± 2.7 kg (p < 0.05) and HbA1c by 0.5 ± 0.3 % (p < 0.05). In a subgroup of patients on oral medications, weight was reduced by 6.3 ± 3.4 kg (p < 0.05), and HbA1c was reduced by 0.9 ± 0.4 % (p < 0.05). The group on insulin had no significant changes in either weight or the HbA1c. The study concluded that the TANTALUS system and gastric electrical stimulation can potentially lead to improvement in glucose metabolism and the lipid profile, as well as inducing weight loss and favorable changes in eating behavior, in obese diabetic individuals. Consistent results were published by Sanmiguel et al. [44] in a study involving 14 obese type 2 diabetic patients. They demonstrated that short-term therapy with the TANTALUS system improves glucose control, induces weight loss, and improves blood pressure and the lipid profile in these subjects on oral antidiabetes therapy and found that the improvement in glucose control did not correlate with weight loss.
These endoluminal procedures and other novel minimally invasive procedures offer a lot of potential for the future of bariatric and metabolic surgery. However, an extensive amount of research is required in order to take it from the experimental trial settings and actually incorporate it into standard practice. Prospective randomized trials are necessary in order to investigate these procedures further, but it certainly offers a lot of scope for the future of interventional diabetology, potentially revolutionizing this newly emerging field.
Conclusion
The incorporation of bariatric surgery into the treatment options for type 2 diabetes is currently being researched quite extensively. Type 2 diabetes is a worldwide epidemic, and current conventional methods of treatment have their limitations. Despite advancements in medical therapy, many patients still fail to achieve optimal glycemic control, and thus surgery may provide a suitable alternative for those who fail conventional methods. Continued research must be conducted in this field in order to understand the mechanisms of surgery with regard to the remission of type 2 diabetes and to further our understanding of the role of the gut. Novel procedures may also introduce minimally invasive surgery into metabolic surgery offering patients a safer and less risky procedure to treat their diabetes. Further research and prospective studies offer the potential to advance pharmacotherapy in order to develop and uncover new therapeutic targets in order to manage type 2 diabetes more effectively.
References
Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract. 2010;87(1):4–14.
Rubino F, Schauer PR, Kaplan LM, et al. Metabolic surgery to treat type 2 diabetes: clinical outcomes and mechanisms of action. Annu Rev Med. 2010;61:393–411.
Rubino F, Kaplan LM, Schauer PR, et al. The Diabetes Surgery Summit consensus conference: recommendations for the evaluation and use of gastrointestinal surgery to treat type 2 diabetes mellitus. Ann Surg. 2010;251(3):399–405.
Zimmet P, Alberti KG, Rubino F, et al. IDF’s view of bariatric surgery in type 2 diabetes. Lancet. 2011;378(9786):108–10.
Rubino F, Marescaux J. Effect of duodenal-jejunal exclusion in a nonobese animal model of type 2 diabetes: a new perspective for an old disease. Ann Surg. 2004;239:1–11.
Rubino F, Forgione A, Cummings DE, et al. The mechanism of diabetes control after gastrointestinal bypass surgery reveals a role of the proximal small intestine in the pathophysiology of type 2 diabetes. Ann Surg. 2006;244(5):741–9.
Korner J, Inabnet W, Febres G, et al. Prospective study of gut hormone and metabolic changes after adjustable gastric banding and Roux-en-Y gastric bypass. Int J Obes (Lond). 2009;33(7):786–95.
Laferrère B, Teixeira J, McGinty J, et al. Effect of weight loss by gastric bypass surgery versus hypocaloric diet on glucose and incretin levels in patients with type 2 diabetes. J Clin Endocrinol Metab. 2008;93:2479–85.
Thaler JP, Cummings DE. Minireview: hormonal and metabolic mechanisms of diabetes remission after gastrointestinal surgery. Endocrinology. 2009;150(6):2518–25.
Shukla AP, Rubino F. Secretion and function of gastrointestinal hormones after bariatric surgery: their role in type 2 diabetes. Can J Diabetes. 2011;35(2):115–22.
Laferrère B, Heshka S, Wang K, et al. Incretin levels and effect are markedly enhanced 1 month after Roux-en-Y gastric bypass surgery in obese patients with type 2 diabetes. Diabetes Care. 2007;30(7):1709–16.
Bose M, Oliván B, Teixeira J, et al. Do incretins play a role in the remission of type 2 diabetes after gastric bypass surgery: what are the evidence? Obes Surg. 2009;19(2):217–29.
Korner J, Inabnet W, Conwell IM, et al. Differential effects of gastric bypass and banding on circulating gut hormone and leptin levels. Obesity (Silver Spring). 2006;14(9):1553–61.
Cummings DE, Foster-Schubert KE, Overduin J. Ghrelin and energy balance: focus on current controversies. Curr Drug Targets. 2005;6(2):153–69.
Sun Y, Asnicar M, Saha PK, et al. Ablation of ghrelin improves the diabetic but not obese phenotype of ob/ob mice. Cell Metab. 2006;3(5):379–86.
Cummings DE, Weigle DS, Frayo RS, et al. Plasma ghrelin levels after diet-induced weight loss or gastric bypass surgery. N Engl J Med. 2002;346(21):1623–30.
Ybarra J, Bobbioni-Harsch E, Chassot G, et al. Persistent correlation of ghrelin plasma levels with body mass index both in stable weight conditions and during gastric-bypass-induced weight loss. Obes Surg. 2009;19(3):327–31.
Boey D, Lin S, Karl T, et al. Peptide YY ablation in mice leads to the development of hyperinsulinaemia and obesity. Diabetologia. 2006;49(6):1360–70.
Valderas JP, Irribarra V, Boza C, et al. Medical and surgical treatments for obesity have opposite effects on peptide YY and appetite: a prospective study controlled for weight loss. J Clin Endocrinol Metab. 2010;95(3):1069–75.
Wiedmer P, Noguerias R, Broglio F, et al. Ghrelin, obesity and diabetes. Nat Clin Pract Endocrinol Metab. 2007;3(10):705–12.
Laferrère B. Diabetes remission after bariatric surgery: is it just the incretins? Int J Obes (Lond). 2011;35 Suppl 3:S22–5.
Miras AD, Le Roux CW. Bariatric surgery and taste: novel mechanisms of weight loss. Curr Opin Gastroenterol. 2010;26(2):140–5.
Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA. 2004;292(14):1724–37.
Sjöström L, Lindroos AK, Peltonen M, et al. Lifestyle, diabetes and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 2004;351(26):2683–93.
Schauer PR, Kashyap SR, Wolski K, et al. Bariatric surgery versus intensive medical therapy in obese patients with diabetes. N Engl J Med. 2012;366(17):1567–76.
Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366(17):1577–85.
Dixon JB, O’Brien PE, Playfair J, et al. Adjustable gastric banding and conventional therapy for type 2 diabetes: a randomized controlled trial. JAMA. 2008;299(3):316–23.
Lee WJ, Chong K, Ser KH, et al. Gastric bypass vs sleeve gastrectomy for type 2 diabetes mellitus: a randomized controlled trial. Arch Surg. 2011;146(2):143–8.
Cohen RV, Pinheiro JC, Schiavon CA, et al. Effects of gastric bypass surgery in patients with type 2 diabetes and only mild obesity. Diabetes Care. 2012;35(7):1420–8.
Lee WJ, Wang W, Lee C. Effect of laparoscopic mini-gastric bypass for type 2 diabetes mellitus: comparison of BMI > 35 and < 35 kg/m2. J Gastrointest Surg. 2008;12(5):945–52.
Sjöström L, Narbro K, Sjöström CD, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357(8):741–52.
Romeo S, Maglio C, Burza MA, et al. Cardiovascular events after bariatric surgery in obese subjects with type 2 diabetes. Diabetes Care. 2012;35(12):2613–7.
Adams TD, Gress RE, Smith SC, et al. Long-term mortality after gastric bypass surgery. N Engl J Med. 2007;357(8):753–61.
Tice JA, Karliner L, Walsh J, et al. Gastric banding or bypass? A systematic review comparing the two most popular bariatric procedures. Am J Med. 2008;121(10):885–93.
Shukla AP, Ahn SM, Patel RT, et al. Surgical treatment of type 2 diabetes: the surgeon perspective. Endocrine. 2011;40(2):151–61.
Schauer PR, Burguera B, Ikramuddin S, et al. Effect of laparoscopic Roux-en Y gastric bypass on type 2 diabetes mellitus. Ann Surg. 2003;238(4):467–84.
Blackstone R, Bunt JC, Cortés MC, et al. Type 2 diabetes after gastric bypass: remission in five models using HbA1c, fasting blood glucose, and medication status. Surg Obes Relat Dis. 2012;8(5):548–55.
Dixon JB, Chuang LM, Chong K, et al. Predicting the glycemic response to gastric bypass surgery in patients with type 2 diabetes. Diabetes Care. 2012;36(1):20–6.
Lee WJ, Ser KH, Chong K, et al. Laparoscopic sleeve gastrectomy for diabetes treatment in nonmorbidly obese patients: efficacy and change of insulin secretion. Surgery. 2010;147(5):664–9.
Schauer P, Chand B, Brethauer S. New applications for endoscopy: the emerging field of endoluminal and transgastric bariatric surgery. Surg Endosc. 2007;21(3):347–56.
Aguirre V, Stylopoulos N, Grinbaum R, et al. An endoluminal sleeve induces substantial weight loss and normalizes glucose homeostasis in rats with diet-induced obesity. Obesity. 2008;16:2585–92.
Soni A, Shukla AP, Rubino F. Interventional diabetology: the evolution of diabetes care in the XXI century. Curr Atheroscler Rep. 2012;14(6):631–6.
Bohdjalian A, Prager G, Rosak C, et al. Improvement in glycemic control in morbidly obese type 2 diabetic subjects by gastric stimulation. Obes Surg. 2009;19(9):1221–7.
Sanmiguel CP, Conklin JL, Cunneen SA, et al. Gastric electrical stimulation with the TANTALUS® system in obese type 2 diabetes patients: effect on weight and glycemic control. J Diabetes Sci Technol. 2009;3(4):964–70.
Financial Support/Conflict of Interest
None.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Review Questions and Answers
Review Questions and Answers
-
1.
Factors that are positively correlated with the remission of T2DM after surgery include all except:
-
A.
Increasing age
-
B.
Lower preoperative HbA1c
-
C.
Shorter duration of diabetes
-
D.
Percentage of excess weight loss
Answer is A. Patients who have a lower preoperative HbA1c and a shorter duration of T2DM, and those who have a higher percentage of excess weight loss, have a greater chance of remission of T2DM after surgery.
-
A.
-
2.
Improvement in insulin sensitivity following bariatric surgery is accompanied by all of the following mechanisms except:
-
A.
Decrease in the lipid content of the liver and muscle
-
B.
Reduction in the levels of adiponectin
-
C.
Increase in the concentration of insulin receptors
-
D.
Increase in the markers of insulin signaling
Answer is B. There is an elevation in the levels of adiponectin following bariatric surgery, as adiponectin is an insulin-sensitizing hormone.
-
A.
-
3.
The Swedish Obese Subjects (SOS) study demonstrated the following effects of bariatric surgery on cardiovascular events on participants with T2DM except:
-
A.
Reduction in the incidence of fatal and nonfatal cardiovascular events
-
B.
Lower incidence of myocardial infarction
-
C.
Lower incidence of cerebral stroke
-
D.
Similar incidence of myocardial infarction following different surgical procedures
Answer is C. Bariatric surgery was not found to be associated with changes in the incidence of cerebral stroke in type 2 diabetic patients in the SOS study.
-
A.
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this chapter
Cite this chapter
Soni, A., Shukla, A., Rubino, F. (2015). 48 Effects of Bariatric Surgery on Diabetes. In: Brethauer, S., Schauer, P., Schirmer, B. (eds) Minimally Invasive Bariatric Surgery. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-1637-5_48
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1637-5_48
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-1636-8
Online ISBN: 978-1-4939-1637-5
eBook Packages: MedicineMedicine (R0)