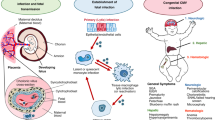
Abstract
Of the myriad of congenitally and perinatally acquired infections that can impair the neurodevelopment of the infant, cytomegalovirus (CMV) is the most important. In the developed world, congenital CMV infection occurs in approximately 1 % of all pregnancies. Long-term neurodevelopmental disabilities include developmental delay, cerebral palsy, seizure disorders, and sensorineural hearing loss. This chapter summarizes the epidemiology and impact of congenital CMV on brain development. Hypotheses regarding the pathophysiology of CNS injury are reviewed. Prospects for intervention are also summarized.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Cytomegalovirus
- Congenital cytomegalovirus infection
- Cytome-galovirus vaccine
- Cytomegalovirus neuropathogenesis
- Sensorineural hearing loss
- Inflammatory response
- Cytomegalovirus immune evasion
1 Epidemiology of Congenital CMV Infection
Human cytomegalovirus (CMV) is a ubiquitous betaherpesvirus that replicates only in human cells. CMV infections are generally asymptomatic in immunocompetent individuals but produce a mononucleosis syndrome (heterophile negative) in approximately 10 % of primary infections in older children and adults [1–3]. Similar to other herpesviruses, CMV becomes latent after primary infection, but it may reactivate from latency, particularly in the setting of immune suppression, leading to disease in HIV-infected patients [4], or in solid organ or hematopoietic stem cell transplant patients [5, 6]. Acquisition of infection typically requires intimate contact with body fluids (blood, urine, saliva, breast milk). There is no seasonality to infection. Patient populations with increased rates of primary infection include breastfeeding infants, sexually active adolescents, and childcare providers in group day care [7–13]. Seroprevalence is higher among nonwhites and among individuals of lower socioeconomic status [14].
From a public health perspective, the most important medical impact of CMV is the damage caused to the developing central nervous system (CNS) of a fetus when infection occurs in utero. A recent meta-analysis of published studies concluded that the overall birth prevalence of congenital CMV infection was approximately 0.65 %, although this study also noted that congenital infection rates varied considerably among different study populations [15]. This corresponds to over 60,000 congenital infections annually in the United States and Europe. Some of the risk factors for congenital CMV infection include nonwhite race, low socioeconomic status, premature birth, and neonatal intensive care unit admittance. The risk of fetal transmission appears to increase with gestational age, but neurological outcomes are more severe when infection occurs during the first trimester [16, 17]. However, viral transmission can occur during the entire gestation period, and adverse neurological outcomes may still be observed in the setting of infections acquired in late gestation [18], although CNS injury is much less common when fetal infections are acquired during this time point in pregnancy [19].
The prevalence of congenital CMV infection within a given population correlates directly with maternal CMV seroprevalence [20]. Indeed, preconception immunity to CMV does not confer complete protection against fetal transmission in subsequent pregnancies, although the risk of congenital CMV clearly is greater in the setting of a primary infection during pregnancy. Overall, transplacental transmission of virus occurs in about one-third of mothers with primary CMV infection [21–23], and approximately one-half of these infections acquired in utero result in a symptomatic clinical syndrome [24]. Fetal infection occurs in up to ~1.5 % of pregnancies in which there is preconception immunity [15], either due to reactivation of latent infection or, probably more commonly, due to maternal reinfection with novel strain variants of CMV [21, 25, 26]. Strain variation among clinical isolates of CMV is substantial, and the presence of preexisting maternal immunity does not appear to fully protect against different strains [27–29]. Importantly, preconception maternal immunity unfortunately does not completely protect the infected fetus from neurological injury and sequelae [30]. Indeed, in a study of 300 children with confirmed congenital CMV, investigators at the University of Alabama, Birmingham, found that the incidence of progressive hearing loss was not different in children born to mothers with preexisting immunity when compared to women who gave birth to infected newborns in the setting of a primary maternal infection during pregnancy [31]. Such observations complicate the conceptualization and design of a preventative CMV vaccine (reviewed later in this chapter). These observations suggest that infection prevention strategies should not only be targeted to seronegative but also to seropositive pregnant women.
Congenital CMV infection is the major infectious cause of birth defects and childhood neurodevelopmental disorders. Among the primary clinical manifestations associated with congenital CMV infection, the most devastating are those involving the developing CNS since, in contrast to other end-organ injury, CNS injury is generally believed to be irreversible. The presence of symptoms at birth in an infant with congenital CMV is an important harbinger of brain involvement and potential neurodevelopment sequelae. CNS injury and attendant long-term neurodevelopmental deficits are substantially more common in infants with symptoms at birth. The most commonly observed symptoms of CMV infection at birth are intrauterine growth retardation (IUGR), purpura, jaundice, hepatosplenomegaly, microcephaly, hearing impairment, and thrombocytopenia [32]. While clinical signs due to abnormalities of the reticuloendothelial system (like anemia, hepatosplenomegaly, jaundice) are transient, neurological deficits either are evident at birth and typically persist for life or tend to become evident (as sensorineural hearing loss) in early childhood. Only 10–15 % of children with congenital CMV infection exhibit clinical signs at birth, although even children who appear asymptomatic at birth are at risk for neurodevelopmental sequelae [33]. Most children (60–90 %) with symptomatic infection, and 10–15 % of those with asymptomatic infection, develop one or more long-term neurological sequelae, such as mental retardation, psychomotor retardation, cerebral palsy, developmental delay, sensorineural hearing loss, and ophthalmologic abnormalities [32, 34, 35]. Current estimates indicate that approximately 8,000 children in the United States are affected each year with one or more neurological sequelae related to in utero acquisition of CMV infection. CMV exerts a far greater impact on neurodevelopmental outcomes than that of other, better-known childhood disorders, such as Down syndrome (4,000/year), fetal alcohol syndrome (5,000/year), or spina bifida (3,500/year), although public awareness of CMV remains low [36–38]. In light of the public health significance of CMV-related long-term neurological disabilities, increased attention needs to be devoted to the study of the neuropathogenesis of this infection. Accordingly, the development of effective interventions, such as vaccines, would have a substantial and major public health impact on the prevalence of childhood disabilities [39].
2 Pathogenesis of CNS Injury Induced by Congenital CMV
Given the intrinsic limitations of performing histopathological studies on brain tissue from infants with symptomatic congenital CMV infection, the pathogenesis of CNS injury must be indirectly investigated. Brain imaging studies, cell culture models of infection, and observations from the study of cytokine and host inflammatory responses in children with CMV-induced brain injury (including cerebral palsy) have provided insights into the pathogenesis of CNS injury. In addition, animal models of perinatal and congenital CMV infection have provided important additional information about mechanisms of pathogenesis. These studies are considered in this section of the review.
2.1 Imaging Studies
A number of imaging modalities have contributed to our understanding of the natural history and pathogenesis of congenital CMV. Of particular interest are the imaging studies that have been reported of the developing brain in the CMV-infected fetus. Serial ultrasonograms or cranial CT scans have proven useful in detecting the overt pathological alterations in the fetal brain of symptomatic children and can accurately predict development of cognitive and motor deficiencies [40–42]. Importantly, the absence of detectable lesions in an asymptomatic congenitally infected newborn does not provide complete reassurance against the eventual diagnosis of CNS injury, since infants with normal CNS imaging are nevertheless at risk for developing hearing loss later in life [41].
Fetal imaging studies can demonstrate structural brain abnormalities as early as 28 weeks of gestation, using either MR images or ultrasonograms. T2- and T1-weighted MRI scans of CMV-infected fetal brains have demonstrated white matter abnormalities reflective of acute responses to infection, such as the loss of intermediate zone layer, focal necrosis, and hemorrhage. Chronic lesions due to CMV infection can also be demonstrated, including ventricular dilatation, white matter gliosis, atrophy (volume loss), parenchymal cysts, ependymal cysts, calcifications, and cortical malformations, most notably polymicrogyria [43]. Fetal sonographic studies obtained between 22 and 37 weeks of gestation have also demonstrated structural brain changes attributable to CMV. Transvaginal ultrasonograms have been reported to show abnormal periventricular hyper-/hypoechogenicity, ventricular adhesions, cystic formation around the ventricles, ependymal protrusions, abnormal sulci formation, and hypoplasia of the corpus callosum [44]. Fetal imaging studies have been useful for establishing timelines for determining the embryologic sequence of CNS infection, and these findings may in turn be useful in predicting neurodevelopmental prognosis [45]. Lanari and colleagues recently compiled an elegant summary of the pattern of neurodevelopmental injury as a function of timing of acquisition of brain infection in utero. This review noted that lesions occurring prior to 18 weeks gestational age commonly include lissencephaly with thin cerebral cortex, cerebellar hypoplasia, ventriculomegaly, periventricular calcification, and delay in myelination. At 18–24 weeks, migrational abnormalities may occur, including polymicrogyria, schizencephaly, and periventricular cysts. Third trimester infections may be associated with central nervous system (CNS) lesions that may include delayed myelination, dysmyelination, calcification, and white matter disease [46].
Neonatal imaging of children with symptomatic CMV infection is typically associated with structural brain abnormalities similar to those described in the infected fetus. The most frequent of these is the presence of intracranial calcifications, present in approximately 70 % of cases [47]. Abnormal cranial ultrasonograms (demonstrating periventricular or parenchymal calcifications, or increased ventricular size) can be performed in the neonatal period in symptomatic congenitally infected infants and are able to both identify children with overt, acute CNS injury as well as those at risk for later neurological deficits [41]. Ultrasonography, however, may miss more subtle CNS pathology in the neonate. Brain MRI of children with congenital CMV has revealed multiple intracranial pathologies, including white matter lesions, neuronal migration, and myelination abnormalities; polymicrogyria; cerebellar, cortical, and hippocampal dysplasia or hypoplasia; periventricular cysts; and ventriculomegaly [48, 49]. The finding of subtle white matter lesions with or without polymicrogyria and in combination with anterior temporal lobe cysts was described in a study of congenital CMV identified by PCR-based screening of Guthrie newborn screening cards [50]. Another recently reported study assessed the diagnostic and prognostic value of cerebral MRI in comparison to ultrasonography in predicting neurodevelopmental outcome in newborns with congenital CMV. Of note, MRI provided additional information beyond that which could be identified by ultrasound (white matter abnormalities in three cases, lissencephaly/polymicrogyria in one and a cyst of the temporal lobe in another one) in four infants who had abnormal findings in both exams. Even more significantly, three newborns had normal ultrasound exams, but had abnormal MRI exams documenting white matter abnormalities and, in one case, cerebellar hypoplasia [51]. Further studies will be required to identify the prognostic role of MRI, particularly with respect to the finding of white matter lesions currently not identifiable by ultrasonography. Figure 1 demonstrates an example of CNS pathology in an infant with symptomatic congenital CMV infection.
T1-weighted brain MRI of infant with congenital CMV infection. Axial view is demonstrated. Findings include ventriculomegaly, loss of brain volume with prominence of sulci (arrow), pachygyria (solid arrowhead) on the surface, and very thin cortex. This infant went on to manifest a seizure disorder and profound neurodevelopmental delay
2.2 Cell Culture Models of CMV Infection
Although the developing brain is the major target for end-organ damage in the setting of congenital CMV infection, the precise cellular targets of infection remain incompletely characterized. Inclusion bodies in the brain have been detected during postmortem histological analysis of fatal cases of congenital CMV infection [52], but little or no histological data identifying the different cell types infected during congenital CMV infection has been reported. Cell culture models of human brain cells are therefore vital in attempting to elucidate the pathogenesis of fetal CNS injury. Both primary human cell culture systems and studies with brain-derived cell lines have demonstrated that practically all cell types in the brain have some degree of susceptibility to CMV infection. The current state of knowledge about CNS targets of infection, including the permissiveness of various cell types for full viral replication and the putative mediators of injury, is summarized in Table 1. Brain microvascular endothelial cells [53–55], astrocytes [56], neuronal cells [57], oligodendroglial cells [58], microglia/macrophages [59], and neural progenitor/stem cells (NPCs) [60] all have a propensity for CMV infection. However, these different cell types vary in their ability to support a complete viral replication cycle, with the permissivity of any given cell type for completion of the viral infection cycle limited by host and viral transcription factors and other elements regulating viral gene expression [47].
Of the cell types of the brain that can be infected with CMV, the astrocyte, the major cell type constituting about 70 % the brain, is the cell type most supportive of productive CMV replication. Primary human fetal astrocyte cultures support cytopathic viral replication, immediate early (IE) gene expression, and β-promoter (early gene) activity, and infectious virus is readily detectable in cell supernatants [56]. Notably, these cells, in association with brain microvasculature endothelial cells (BMVEC), form the blood–brain barrier, a structure that maintains the highly regulated solute, immunologic, and cellular microenvironment in the CNS [61]. Lytic viral replication is supported by BMVEC, which in turn promotes monocyte activation, migration, and infection in the CNS [62].
In contrast to astrocytes, primary differentiated human neurons have generally been found to be refractory to CMV replication, with some exceptions as noted below. Highly purified primary neuronal cultures (>90 % neurons) contain a small percentage of dividing astrocytes that support viral replication, but viral gene products cannot be detected in neurons [56]. The block in the viral replicative cycle appears to be at the level of the major immediate early promoter (MIEP), and not due to a defect in viral entry [60]. Experiments with undifferentiated human oligodendroglioma (HOG) cells, representative of immature oligodendrocytes, demonstrate that oligodendrocytes, like neurons, may not be fully permissive for CMV infection. However, CMV IE, US11, and glycoprotein B (gB) gene expression is induced in HOG cells upon differentiation with phorbol-myristate acetate (PMA), without production of viral progeny [58]. Some studies utilizing neural progenitor cells (NPCs) have demonstrated, following in vitro differentiation and enrichment, that neurons can support productive replication of CMV [63, 64]. Taken together, it appears that the state of cell differentiation as well as its functional status may modulate permissiveness of neurons and oligodendrocytes to CMV brain infection in cell culture systems. The implications for in utero neuropathogenesis remain to be elucidated.
Microglia, the end-differentiated resident brain macrophages, also do not appear to support productive CMV infection [56]. However, CMV DNA has been demonstrated in infected microglial cells in the absence of detectable viral IE proteins [65, 66]. It has been proposed that brain microglia are replenished from bone marrow-derived precursors that migrate into the brain [47]. It has been suggested that myeloid precursor cells may be a site for CMV latency and a vehicle of viral dissemination in the host [67–70]. Although myeloid precursors and monocytes are not typically productively infected by CMV [70], they support productive CMV infection at certain stages of differentiation [71]. In addition, endothelial cell-adapted viral strains have been shown to infect both macrophages and dendritic cells [72], a process which requires the CMV gene products essential for endothelial tropism, UL128, 130, and 131 [73]. These gene products have recently emerged as key candidates for CMV subunit vaccines [74]. It has been suggested that macrophages originating in the vascular space may be an important vehicle for trafficking of virus into the CNS in the developing fetal brain [47].
NPCs have emerged as cells of particular interest in the pathogenesis of congenital CMV-induced brain injury. These cells are predominantly located in the subventricular zone and subgranular zone of the hippocampus in the mammalian brain [47]. NPCs possess the ability to migrate, proliferate, and differentiate into neurons, astrocytes, and oligodendrocytes. Figure 2 provides a schematic model of the central role of these cells in CMV-induced neuropathogenesis. In the setting of CMV brain infection (including histopathological observations discerned from fatal congenital infections), it is well recognized that virus preferentially infects cells in the ventricular or subventricular regions [52, 75]. This anatomic distribution suggests the possibility that CMV replication may be particularly well adapted to replicate in the neural stem/precursor cells residing in this region. Several studies have demonstrated that human CMV replicates efficiently in undifferentiated human neural precursor cells in cell culture [60, 63, 64, 76, 77]. It has been proposed that the extent to which these cells are infected in utero may determine the outcome of CNS sequelae associated with congenital CMV infection [47].
Schematic representation of mechanisms of brain injury following infection with CMV. Neural stem cells (in blue), found along the lateral ventricular wall of the brain, are involved in the development of new neural circuits in the developing brain. These cells differentiate into new brain cells (astrocytes, oligodendrocytes, and neurons), either directly or via an intermediate transitional progenitor cell (red cells). Formation of new neural circuitry involves migration of neuroblasts (green cells), through a directed pathway that is supported in part by astroglial cells. CMV may potentially affect any or all of these stages: (1) Infection of neural stem cells may disrupt their ability to maintain a self-renewing cycle; (2) Differentiation of neural stem cells via the transitional cells and eventually neuroblasts may also be disrupted by CMV; (3) Brain infection affects the migratory patterns of neuroblasts, particularly during cortical and cerebellar development; (4) This presumably alters the migratory patterning of other specific brain structures, causing improper layering of the neocortex; (5) Since glial cells are also susceptible to CMV, functions of glia in directing neuronal layering patterns may be affected. (6) Finally, infection can induce a myriad of inflammatory mediators, including cytokines and chemokines, and elicit inflammatory cell infiltration. Reproduced from Cheeran et al., Neuropathogenesis of congenital CMV infection: Disease mechanisms and prospects for intervention. Clin Micro Rev 2009: 22, 99–126
CMV infection of human neural precursor cells appears to inhibit their differentiation into both neurons and astrocytes, an effect that may be mediated by virus-induced apoptosis in cells undergoing differentiation [76–78]. It has been proposed that CMV replication may inhibit neural precursor cell proliferation by altering cell cycle mechanisms [60, 79] and may perturb expression of genes related to neuronal metabolism and neuronal differentiation in NPCs [78]. Indeed, disruption of these cellular processes in neural precursor cells may account for a large portion of the structural and migratory abnormalities seen during congenital human CMV brain infection [47, 78]. A recent study in a cell culture model suggests that this susceptibility does not diminish with advancing brain development. In this study, NPC cultures derived at different gestational ages were evaluated after short (3–6) or extended (11–20) in vitro passage for viral entry efficiency, viral gene expression, virus-induced cytopathic effect, and release of progeny virus. Extended passage cultures showed evidence of increased viral entry and more efficient production of infectious progeny, suggesting that CMV infection in fetal brain may continue to result in neural cell loss even with advancing brain development [80]. These observations suggest that persistent CMV infection may continue to negatively impact brain development postnatally. Extended, long-term infect of NPCs postnatally provides a mechanism by which CMV could to continue to exert an impact on neurodevelopmental processes in early childhood in the context of congenital infection of the CNS. This aspect of CMV replication in NPCs provides reinforcement for clinical trials examining the impact of extended, long-term (6 months) valganciclovir therapy in infants with congenital CMV infection [81].
2.3 Role of Inflammatory Response
In addition to the direct damage to specific cell types conferred by fetal brain infection with CMV, the inflammatory response to infection also is also increasingly becoming recognized as a major contributing factor in the pathogenesis of brain injury. Autopsies of prenatally infected fetuses with CMV have confirmed the presence of a significant inflammatory response in addition to viral inclusions in the brain [82–86]. Upon autopsy, one study recently found that nearly all fetal organs infected with CMV had evidence of inflammatory infiltrate and found that the level of organ damage was associated with the level of inflammation; intriguingly, in addition to the damaging direct effects of viral brain infection and the attendant inflammatory response, hypoxic brain injury due to severe CMV placentitis was also postulated as a contributing factor in brain injury [83]. Tissue-specific viral load has been proposed to impact the magnitude of the inflammatory response. In one study, it was noted that tissue viral load was correlated to immune response; low CMV viral load elicited only a modest immune response with mild brain damage, while tissue containing high viral load had high levels of cytotoxic CD8+ T-lymphocytes, which are associated with immune-related structural damage [82]. In total, this evidence suggests that direct fetal infection with a neurotropic pathogen like CMV not only increases the risk of neurological sequelae mediated by factors such as lytic infection, disruption of neuronal migration, and increased apoptosis, but also through a cascade of events triggering a pathological fetal immune response.
A current view of mechanisms of neuroinflammation in adults revolves around the concept of “reactive microgliosis” [87]. Microglia can be activated in several different patterns, including classical activation (M1 phenotype), alternate activation (M2a phenotype), or acquired deactivation (M2b phenotype) [88, 89]. While the M1 phenotype appears to promote a deleterious, pro-inflammatory status, it has been proposed that the M2 phenotype could favor brain repair [90, 91]. In addition to promoting a pro-inflammatory milieu, developmental brain damage mechanisms can also be driven by microglial activation [92–95]. Systemic and brain inflammation driven by activated microglia in turn impacts the development of different neural cell populations, and influences oligodendrocyte maturation [96] and survival [97, 98]. CMV-driven release of cytokines, particularly IL-6, could impact proliferation and function of neural stem cells [99]. Migrating neurons can be compromised by the release of inflammatory factors released by activated microglia, potentially leading to neuronal cell death or abnormal neuronal migration, one of the hallmarks of CMV infection of the developing fetal brain [100].
A number of studies have attempted to define the precise cytokine responses associated with fetal brain infection with CMV, predominately using cell culture models. In one model of cultured human glial cells, derived from 16- to 20-week-old fetal brain tissue, response to CMV infection is heralded by expression of a number of immune mediators, including chemokines and cytokines [101]. Microglial cells respond to CMV infection by producing TNF-α and IL-6 as well as CXCL10, CCL2, CCL3, and CCL5 [102, 103]. Many of the cytokine responses of microglial cells appear to be driven by innate responses mediated by pattern recognition receptors on microglia that recognize pathogen-associated molecular patterns [104]; for CMV, these ligands appear to be envelope glycoproteins gB and gH, which signal through Toll-like receptor (TLR) 2, and double-stranded RNA molecules generated during infection, which signal via TLR3 [105–109]. Chemokines and cytokines are also elicited by CMV infection of astrocytes. As noted, the chemokine response in astrocytes includes CCL2, CXCL8, CCL3, and CCL5 [102, 103]. In contrast, the cytokine response to CMV in astrocytes appears to be restricted to TGF-β, an anti-inflammatory cytokine [110]. Primary brain vascular pericytes have recently been shown to be a source of cytokine production in the context of CMV infection, including CXCL8/IL-8, CXCL11/ITAC, CCL5/RANTES, TNF-α, IL-1β, and IL-6 [111].
In addition to inducing the production of chemokines and cytokines following CMV infection of the fetal brain, the viral genome itself encodes homologs of several immunomodulatory proteins, including homologs of CXC (UL146, 147) and CC (UL128) chemokines [112]. CMV also encodes functional homologs of chemokine receptor-like G-protein coupled receptors [113]. The role that such gene products play in the pathogenesis of fetal brain injury is unknown. CMV also encodes a functional homolog of the IL-10 gene. Transcription of this gene product has been shown to inhibit CXCL10 production in human microglial cells, a possible mechanism by which CMV genes contribute to evasion of host immune clearance [102].
3 CMV Fetal/Neonatal Brain Infection: Prospects for Intervention
Currently, there are limited interventions for the treatment and prevention of fetal and neonatal CMV brain infections. In women with primary CMV infections complicated by intrauterine transmission, the use of an anti-CMV high-titer immunoglobulin (HIG) has been associated with improved neurodevelopmental outcomes, including regression of fetal cerebral abnormalities for fetuses treated in utero [114, 115], although these studies are uncontrolled and proof of efficacy is still uncertain. A controlled, multicenter trial of ganciclovir in infants with congenital CMV infection and neurological findings at birth indicated that 6 weeks of intravenous therapy was associated with improved short-term and long-term audiologic outcomes [116]. In a follow-up study in which infants were administered serial neurodevelopmental screening examinations, ganciclovir therapy was associated with fewer developmental delays at 6 and 12 months, compared with untreated infants [117]. Based on these encouraging observations, a trial is currently being conducted that will compare 6 weeks to 6 months of therapy, using oral valganciclovir [81], toward the goal of ascertaining whether any additional neurodevelopmental benefit might be realized by longer treatment courses.
Ultimately, the best prospects for control of congenital CMV rest with the development of an effective preconception vaccine. Several candidate vaccines are in various stages of preclinical development, and some have advanced to clinical trials [118, 119]. Most vaccines currently being examined in human studies target envelope glycoprotein B (gB) and various combinations of T-cell targets, including the pp65 (ppUL83) tegument protein and the major immediate early gene product-1 (IE1) [120]. The gB vaccine has demonstrated modest efficacy in a phase II study in young women of child-bearing age [121]. As previously noted, vaccine development has been complicated by the increasing recognition that women can become reinfected with new, novel strains of CMV during pregnancy and that these strains, in turn, can be transmitted to the fetus, resulting in attendant neurological injury and long-term sequelae [25, 122]. Ultimately, development of a CMV vaccine would therefore probably need to focus not only on prevention against primary infection in seronegative women, but also reinfection in seropositive women, in order to fully protect all infants against the neurodevelopmental sequelae of congenital infection.
References
Marshall GS, Starr SE, Witzleben CL, Gonczol E, Plotkin SA. Protracted mononucleosis-like illness associated with acquired cytomegalovirus infection in a previously healthy child: transient cellular immune defects and chronic hepatopathy. Pediatrics. 1991;87(4):556–62.
Rafailidis PI, Mourtzoukou EG, Varbobitis IC, Falagas ME. Severe cytomegalovirus infection in apparently immunocompetent patients: a systematic review. Virol J. 2008;5:47.
Cohen JI, Corey GR. Cytomegalovirus infection in the normal host. Medicine. 1985;64(2):100–14.
Kovacs A, Schluchter M, Easley K, Demmler G, Shearer W, La Russa P, Pitt J, Cooper E, Goldfarb J, Hodes D, Kattan M, McIntosh K. Cytomegalovirus infection and HIV-1 disease progression in infants born to HIV-1-infected women. Pediatric Pulmonary and Cardiovascular Complications of Vertically Transmitted HIV Infection Study Group. N Engl J Med. 1999;341(2):77–84.
Boeckh M, Fries B, Nichols WG. Recent advances in the prevention of CMV infection and disease after hematopoietic stem cell transplantation. Pediatr Transplant. 2004;8 Suppl 5:19–27.
Razonable RR. Strategies for managing cytomegalovirus in transplant recipients. Expert Opin Pharmacother. 2010;11(12):1983–97.
Collier AC, Handsfield HH, Ashley R, Roberts PL, DeRouen T, Meyers JD, Corey L. Cervical but not urinary excretion of cytomegalovirus is related to sexual activity and contraceptive practices in sexually active women. J Infect Dis. 1995;171(1):33–8.
Collier AC, Handsfield HH, Roberts PL, DeRouen T, Meyers JD, Leach L, Murphy VL, Verdon M, Corey L. Cytomegalovirus infection in women attending a sexually transmitted disease clinic. J Infect Dis. 1990;162(1):46–51.
Staras SA, Flanders WD, Dollard SC, Pass RF, McGowan Jr JE, Cannon MJ. Influence of sexual activity on cytomegalovirus seroprevalence in the United States, 1988–1994. Sex Transm Dis. 2008;35(5):472–9.
Murph JR, Baron JC, Brown CK, Ebelhack CL, Bale Jr JF. The occupational risk of cytomegalovirus infection among day-care providers. JAMA. 1991;265(5):603–8.
Pass RF, Hutto C, Ricks R, Cloud GA. Increased rate of cytomegalovirus infection among parents of children attending day-care centers. N Engl J Med. 1986;314(22):1414–8.
Schleiss MR. Role of breast milk in acquisition of cytomegalovirus infection: recent advances. Curr Opin Pediatr. 2006;18(1):48–52.
Joseph SA, Beliveau C, Muecke CJ, Rahme E, Soto JC, Flowerdew G, Johnston L, Langille D, Gyorkos TW. Cytomegalovirus as an occupational risk in daycare educators. Paediatr Child Health. 2006;11(7):401–7.
Bate SL, Dollard SC, Cannon MJ. Cytomegalovirus seroprevalence in the United States: the national health and nutrition examination surveys, 1988–2004. Clin Infect Dis. 2010;50(11):1439–47.
Kenneson A, Cannon MJ. Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev Med Virol. 2007;17(4):253–76.
Pass RF, Fowler KB, Boppana SB, Britt WJ, Stagno S. Congenital cytomegalovirus infection following first trimester maternal infection: symptoms at birth and outcome. J Clin Virol. 2006;35(2):216–20.
Picone O, Vauloup-Fellous C, Cordier AG, Guitton S, Senat MV, Fuchs F, Ayoubi JM, Grangeot Keros L, Benachi A. A series of 238 cytomegalovirus primary infections during pregnancy: description and outcome. Prenat Diagn. 2013;33:751–8.
Stagno S, Pass RF, Cloud G, Britt WJ, Henderson RE, Walton PD, Veren DA, Page F, Alford CA. Primary cytomegalovirus infection in pregnancy. Incidence, transmission to fetus, and clinical outcome. JAMA. 1986;256(14):1904–8.
Gindes L, Teperberg-Oikawa M, Sherman D, Pardo J, Rahav G. Congenital cytomegalovirus infection following primary maternal infection in the third trimester. BJOG. 2008;115(7):830–5.
de Vries JJ, van Zwet EW, Dekker FW, Kroes AC, Verkerk PH, Vossen AC. The apparent paradox of maternal seropositivity as a risk factor for congenital cytomegalovirus infection: a population-based prediction model. Rev Med Virol. 2013;23(4):241–9.
Boppana SB, Rivera LB, Fowler KB, Mach M, Britt WJ. Intrauterine transmission of cytomegalovirus to infants of women with preconceptional immunity. N Engl J Med. 2001;344(18):1366–71.
Fowler KB, Stagno S, Pass RF. Maternal immunity and prevention of congenital cytomegalovirus infection. JAMA. 2003;289(8):1008–11.
Fowler KB, Stagno S, Pass RF, Britt WJ, Boll TJ, Alford CA. The outcome of congenital cytomegalovirus infection in relation to maternal antibody status. N Engl J Med. 1992;326(10):663–7.
Adler SP, Nigro G. Interrupting intrauterine transmission of cytomegalovirus. Rev Med Virol. 2006;16(2):69–71.
Yamamoto AY, Mussi-Pinhata MM, Boppana SB, Novak Z, Wagatsuma VM, Oliveira Pde F, Duarte G, Britt WJ. Human cytomegalovirus reinfection is associated with intrauterine transmission in a highly cytomegalovirus-immune maternal population. Am J Obstet Gynecol. 2010;202(3):297. e1–8.
Ross SA, Arora N, Novak Z, Fowler KB, Britt WJ, Boppana SB. Cytomegalovirus reinfections in healthy seroimmune women. J Infect Dis. 2010;201(3):386–9.
Paradowska E, Jablonska A, Studzinska M, Suski P, Kasztelewicz B, Zawilinska B, Wisniewska-Ligier M, Dzierzanowska-Fangrat K, Wozniakowska-Gesicka T, Czech-Kowalska J, Lipka B, Kornacka M, Pawlik D, Tomasik T, Kosz-Vnenchak M, Lesnikowski ZJ. Distribution of cytomegalovirus gN variants and associated clinical sequelae in infants. J Clin Virol. 2013;58(1):271–5.
Kropff B, Burkhardt C, Schott J, Nentwich J, Fisch T, Britt W, Mach M. Glycoprotein N of human cytomegalovirus protects the virus from neutralizing antibodies. PLoS Pathog. 2012;8(10):e1002999.
Pati SK, Novak Z, Purser M, Arora N, Mach M, Britt WJ, Boppana SB. Strain-specific neutralizing antibody responses against human cytomegalovirus envelope glycoprotein N. Clin Vaccine Immunol. 2012;19(6):909–13.
Ross SA, Boppana SB. Congenital cytomegalovirus infection: outcome and diagnosis. Semin Pediatr Infect Dis. 2005;16(1):44–9.
Ross SA, Fowler KB, Ashrith G, Stagno S, Britt WJ, Pass RF, Boppana SB. Hearing loss in children with congenital cytomegalovirus infection born to mothers with preexisting immunity. J Pediatr. 2006;148(3):332–6.
Swanson EC, Schleiss MR. Congenital cytomegalovirus infection: new prospects for prevention and therapy. Pediatr Clin North Am. 2013;60(2):335–49.
Boppana SB, Fowler KB, Britt WJ, Stagno S, Pass RF. Symptomatic congenital cytomegalovirus infection in infants born to mothers with preexisting immunity to cytomegalovirus. Pediatrics. 1999;104(1 Pt 1):55–60.
Kylat RI, Kelly EN, Ford-Jones EL. Clinical findings and adverse outcome in neonates with symptomatic congenital cytomegalovirus (SCCMV) infection. Eur J Pediatr. 2006;165(11):773–8.
Gaytant MA, Steegers EA, Semmekrot BA, Merkus HM, Galama JM. Congenital cytomegalovirus infection: review of the epidemiology and outcome. Obstet Gynecol Surv. 2002;57(4):245–56.
Cannon MJ, Davis KF. Washing our hands of the congenital cytomegalovirus disease epidemic. BMC Public Health. 2005;5:70.
Cannon MJ, Westbrook K, Levis D, Schleiss MR, Thackeray R, Pass RF. Awareness of and behaviors related to child-to-mother transmission of cytomegalovirus. Prev Med. 2012;54(5):351–7.
Cordier AG, Guitton S, Vauloup-Fellous C, Grangeot-Keros L, Benachi A, Picone O. Awareness and knowledge of congenital cytomegalovirus infection among health care providers in France. J Clin Virol. 2012;55(2):158–63.
Arvin AM, Fast P, Myers M, Plotkin S, Rabinovich R. Vaccine development to prevent cytomegalovirus disease: report from the National Vaccine Advisory Committee. Clin Infect Dis. 2004;39(2):233–9.
Noyola DE, Demmler GJ, Nelson CT, Griesser C, Williamson WD, Atkins JT, Rozelle J, Turcich M, Llorente AM, Sellers-Vinson S, Reynolds A, Bale Jr JF, Gerson P, Yow MD. Early predictors of neurodevelopmental outcome in symptomatic congenital cytomegalovirus infection. J Pediatr. 2001;138(3):325–31.
Ancora G, Lanari M, Lazzarotto T, Venturi V, Tridapalli E, Sandri F, Menarini M, Ferretti E, Faldella G. Cranial ultrasound scanning and prediction of outcome in newborns with congenital cytomegalovirus infection. J Pediatr. 2007;150(2):157–61.
Boppana SB, Fowler KB, Vaid Y, Hedlund G, Stagno S, Britt WJ, Pass RF. Neuroradiographic findings in the newborn period and long-term outcome in children with symptomatic congenital cytomegalovirus infection. Pediatrics. 1997;99(3):409–14.
Barkovich AJ, Girard N. Fetal brain infections. Childs Nerv Syst. 2003;19(7–8):501–7.
Malinger G, Lev D, Zahalka N, Ben Aroia Z, Watemberg N, Kidron D, Sira LB, Lerman-Sagie T. Fetal cytomegalovirus infection of the brain: the spectrum of sonographic findings. AJNR Am J Neuroradiol. 2003;24(1):28–32.
Barkovich AJ, Lindan CE. Congenital cytomegalovirus infection of the brain: imaging analysis and embryologic considerations. AJNR Am J Neuroradiol. 1994;15(4):703–15.
Lanari M, Capretti MG, Lazzarotto T, Gabrielli L, Rizzollo S, Mostert M, Manzoni P. Neuroimaging in CMV congenital infected neonates: how and when. Early Hum Dev. 2012;88 Suppl 2:S3–5.
Cheeran MC, Lokensgard JR, Schleiss MR. Neuropathogenesis of congenital cytomegalovirus infection: disease mechanisms and prospects for intervention. Clin Microbiol Rev. 2009;22(1):99–126. Table of Contents.
Manara R, Balao L, Baracchini C, Drigo P, D’Elia R, Ruga EM. Brain magnetic resonance findings in symptomatic congenital cytomegalovirus infection. Pediatr Radiol. 2011;41(8):962–70.
Mavili E, Coskun A, Per H, Donmez H, Kumandas S, Yikilmaz A. Polymicrogyria: correlation of magnetic resonance imaging and clinical findings. Childs Nerv Syst. 2012;28(6):905–9.
van der Knaap MS, Vermeulen G, Barkhof F, Hart AA, Loeber JG, Weel JF. Pattern of white matter abnormalities at MR imaging: use of polymerase chain reaction testing of Guthrie cards to link pattern with congenital cytomegalovirus infection. Radiology. 2004;230(2):529–36.
Capretti MG, Lanari M, Tani G, Ancora G, Sciutti R, Marsico C, Lazzarotto T, Gabrielli L, Guerra B, Corvaglia L, Faldella G. Role of cerebral ultrasound and magnetic resonance imaging in newborns with congenital cytomegalovirus infection. Brain Dev. 2014;36(3):203–11.
Perlman JM, Argyle C. Lethal cytomegalovirus infection in preterm infants: clinical, radiological, and neuropathological findings. Ann Neurol. 1992;31(1):64–8.
Poland SD, Costello P, Dekaban GA, Rice GP. Cytomegalovirus in the brain: in vitro infection of human brain-derived cells. J Infect Dis. 1990;162(6):1252–62.
Lathey JL, Wiley CA, Verity MA, Nelson JA. Cultured human brain capillary endothelial cells are permissive for infection by human cytomegalovirus. Virology. 1990;176(1):266–73.
Fish KN, Soderberg-Naucler C, Mills LK, Stenglein S, Nelson JA. Human cytomegalovirus persistently infects aortic endothelial cells. J Virol. 1998;72(7):5661–8.
Lokensgard JR, Cheeran MC, Gekker G, Hu S, Chao CC, Peterson PK. Human cytomegalovirus replication and modulation of apoptosis in astrocytes. J Hum Virol. 1999;2(2):91–101.
Tsutsui Y, Kosugi I, Kawasaki H. Neuropathogenesis in cytomegalovirus infection: indication of the mechanisms using mouse models. Rev Med Virol. 2005;15(5):327–45.
Spiller OB, Borysiewicz LK, Morgan BP. Development of a model for cytomegalovirus infection of oligodendrocytes. J Gen Virol. 1997;78(Pt 12):3349–56.
Schut RL, Gekker G, Hu S, Chao CC, Pomeroy C, Jordan MC, Peterson PK. Cytomegalovirus replication in murine microglial cell cultures: suppression of permissive infection by interferon-gamma. J Infect Dis. 1994;169(5):1092–6.
Cheeran MC, Hu S, Ni HT, Sheng W, Palmquist JM, Peterson PK, Lokensgard JR. Neural precursor cell susceptibility to human cytomegalovirus diverges along glial or neuronal differentiation pathways. J Neurosci Res. 2005;82(6):839–50.
Abbott NJ, Ronnback L, Hansson E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat Rev Neurosci. 2006;7(1):41–53.
Chan G, Nogalski MT, Bentz GL, Smith MS, Parmater A, Yurochko AD. PI3K-dependent upregulation of Mcl-1 by human cytomegalovirus is mediated by epidermal growth factor receptor and inhibits apoptosis in short-lived monocytes. J Immunol. 2010;184(6):3213–22.
McCarthy M, Auger D, Whittemore SR. Human cytomegalovirus causes productive infection and neuronal injury in differentiating fetal human central nervous system neuroepithelial precursor cells. J Hum Virol. 2000;3(4):215–28.
Luo MH, Schwartz PH, Fortunato EA. Neonatal neural progenitor cells and their neuronal and glial cell derivatives are fully permissive for human cytomegalovirus infection. J Virol. 2008;82(20):9994–10007.
Pulliam L, Moore D, West DC. Human cytomegalovirus induces IL-6 and TNF alpha from macrophages and microglial cells: possible role in neurotoxicity. J Neurovirol. 1995;1(2):219–27.
Pulliam L. Cytomegalovirus preferentially infects a monocyte derived macrophage/microglial cell in human brain cultures: neuropathology differs between strains. J Neuropathol Exp Neurol. 1991;50(4):432–40.
Hahn G, Jores R, Mocarski ES. Cytomegalovirus remains latent in a common precursor of dendritic and myeloid cells. Proc Natl Acad Sci U S A. 1998;95(7):3937–42.
Jarvis MA, Nelson JA. Human cytomegalovirus persistence and latency in endothelial cells and macrophages. Curr Opin Microbiol. 2002;5(4):403–7.
Jarvis MA, Nelson JA. Mechanisms of human cytomegalovirus persistence and latency. Front Biosci. 2002;7:d1575–82.
Kondo K, Xu J, Mocarski ES. Human cytomegalovirus latent gene expression in granulocyte-macrophage progenitors in culture and in seropositive individuals. Proc Natl Acad Sci U S A. 1996;93(20):11137–42.
Soderberg-Naucler C, Streblow DN, Fish KN, Allan-Yorke J, Smith PP, Nelson JA. Reactivation of latent human cytomegalovirus in CD14(+) monocytes is differentiation dependent. J Virol. 2001;75(16):7543–54.
Sinzger C, Eberhardt K, Cavignac Y, Weinstock C, Kessler T, Jahn G, Davignon JL. Macrophage cultures are susceptible to lytic productive infection by endothelial-cell-propagated human cytomegalovirus strains and present viral IE1 protein to CD4+ T cells despite late downregulation of MHC class II molecules. J Gen Virol. 2006;87(Pt 7):1853–62.
Gerna G, Percivalle E, Lilleri D, Lozza L, Fornara C, Hahn G, Baldanti F, Revello MG. Dendritic-cell infection by human cytomegalovirus is restricted to strains carrying functional UL131-128 genes and mediates efficient viral antigen presentation to CD8+ T cells. J Gen Virol. 2005;86(Pt 2):275–84.
Fouts AE, Chan P, Stephan JP, Vandlen R, Feierbach B. Antibodies against the gH/gL/UL128/UL130/UL131 complex comprise the majority of the anti-cytomegalovirus (anti-CMV) neutralizing antibody response in CMV hyperimmune globulin. J Virol. 2012;86(13):7444–7.
Schmidbauer M, Budka H, Ulrich W, Ambros P. Cytomegalovirus (CMV) disease of the brain in AIDS and connatal infection: a comparative study by histology, immunocytochemistry and in situ DNA hybridization. Acta Neuropathol. 1989;79(3):286–93.
Odeberg J, Wolmer N, Falci S, Westgren M, Seiger A, Soderberg-Naucler C. Human cytomegalovirus inhibits neuronal differentiation and induces apoptosis in human neural precursor cells. J Virol. 2006;80(18):8929–39.
Odeberg J, Wolmer N, Falci S, Westgren M, Sundtrom E, Seiger A, Soderberg-Naucler C. Late human cytomegalovirus (HCMV) proteins inhibit differentiation of human neural precursor cells into astrocytes. J Neurosci Res. 2007;85(3):583–93.
D’Aiuto L, Di Maio R, Heath B, Raimondi G, Milosevic J, Watson AM, Bamne M, Parks WT, Yang L, Lin B, Miki T, Mich-Basso JD, Arav-Boger R, Sibille E, Sabunciyan S, Yolken R, Nimgaonkar V. Human induced pluripotent stem cell-derived models to investigate human cytomegalovirus infection in neural cells. PLoS One. 2012;7(11):e49700.
Schleiss MR. Congenital cytomegalovirus infection: molecular mechanisms mediating viral pathogenesis. Infect Disord Drug Targets. 2011;11(5):449–65.
Pan X, Li XJ, Liu XJ, Yuan H, Li JF, Duan YL, Ye HQ, Fu YR, Qiao GH, Wu CC, Yang B, Tian XH, Hu KH, Miao LF, Chen XL, Zheng J, Rayner S, Schwartz PH, Britt WJ, Xu J, Luo MH. Later passage neural progenitor cells from neonatal brain are more permissive for human cytomegalovirus infection. J Virol. 2013;87(20):10968–79.
Nassetta L, Kimberlin D, Whitley R. Treatment of congenital cytomegalovirus infection: implications for future therapeutic strategies. J Antimicrob Chemother. 2009;63(5):862–7.
Gabrielli L, Bonasoni MP, Santini D, Piccirilli G, Chiereghin A, Petrisli E, Dolcetti R, Guerra B, Piccioli M, Lanari M, Landini MP, Lazzarotto T. Congenital cytomegalovirus infection: patterns of fetal brain damage. Clin Microbiol Infect. 2012;18(10):E419–27.
Gabrielli L, Bonasoni MP, Lazzarotto T, Lega S, Santini D, Foschini MP, Guerra B, Baccolini F, Piccirilli G, Chiereghin A, Petrisli E, Gardini G, Lanari M, Landini MP. Histological findings in foetuses congenitally infected by cytomegalovirus. J Clin Virol. 2009;46 Suppl 4:S16–21.
Teissier N, Delezoide AL, Mas AE, Khung-Savatovsky S, Bessieres B, Nardelli J, Vauloup-Fellous C, Picone O, Houhou N, Oury JF, Van Den Abbeele T, Gressens P, Adle-Biassette H. Inner ear lesions in congenital cytomegalovirus infection of human fetuses. Acta Neuropathol. 2011;122(6):763–74.
Tongsong T, Sukpan K, Wanapirak C, Phadungkiatwattna P. Fetal cytomegalovirus infection associated with cerebral hemorrhage, hydrops fetalis, and echogenic bowel: case report. Fetal Diagn Ther. 2008;23(3):169–72.
Pushpalatha LDJ, Kuruvilla S. Cytomegalovirus infection with lissencephaly. Indian J Pathol Microbiol. 2008;51(3):402–4.
Block ML, Zecca L, Hong JS. Microglia-mediated neurotoxicity: uncovering the molecular mechanisms. Nat Rev Neurosci. 2007;8(1):57–69.
Colton CA. Heterogeneity of microglial activation in the innate immune response in the brain. J Neuroimmune Pharmacol. 2009;4(4):399–418.
Murray PJ, Wynn TA. Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol. 2011;11(11):723–37.
Gensel JC, Kigerl KA, Mandrekar-Colucci SS, Gaudet AD, Popovich PG. Achieving CNS axon regeneration by manipulating convergent neuro-immune signaling. Cell Tissue Res. 2012;349(1):201–13.
Kigerl KA, Gensel JC, Ankeny DP, Alexander JK, Donnelly DJ, Popovich PG. Identification of two distinct macrophage subsets with divergent effects causing either neurotoxicity or regeneration in the injured mouse spinal cord. J Neurosci. 2009;29(43):13435–44.
Tahraoui SL, Marret S, Bodenant C, Leroux P, Dommergues MA, Evrard P, Gressens P. Central role of microglia in neonatal excitotoxic lesions of the murine periventricular white matter. Brain Pathol. 2001;11(1):56–71.
Chew LJ, Takanohashi A, Bell M. Microglia and inflammation: impact on developmental brain injuries. Ment Retard Dev Disabil Res Rev. 2006;12(2):105–12.
Dean JM, Wang X, Kaindl AM, Gressens P, Fleiss B, Hagberg H, Mallard C. Microglial MyD88 signaling regulates acute neuronal toxicity of LPS-stimulated microglia in vitro. Brain Behav Immun. 2010;24(5):776–83.
Verney C, Pogledic I, Biran V, Adle-Biassette H, Fallet-Bianco C, Gressens P. Microglial reaction in axonal crossroads is a hallmark of noncystic periventricular white matter injury in very preterm infants. J Neuropathol Exp Neurol. 2012;71(3):251–64.
Favrais G, van de Looij Y, Fleiss B, Ramanantsoa N, Bonnin P, Stoltenburg-Didinger G, Lacaud A, Saliba E, Dammann O, Gallego J, Sizonenko S, Hagberg H, Lelievre V, Gressens P. Systemic inflammation disrupts the developmental program of white matter. Ann Neurol. 2011;70(4):550–65.
Li J, Baud O, Vartanian T, Volpe JJ, Rosenberg PA. Peroxynitrite generated by inducible nitric oxide synthase and NADPH oxidase mediates microglial toxicity to oligodendrocytes. Proc Natl Acad Sci U S A. 2005;102(28):9936–41.
Li J, Ramenaden ER, Peng J, Koito H, Volpe JJ, Rosenberg PA. Tumor necrosis factor alpha mediates lipopolysaccharide-induced microglial toxicity to developing oligodendrocytes when astrocytes are present. J Neurosci. 2008;28(20):5321–30.
Covey MV, Loporchio D, Buono KD, Levison SW. Opposite effect of inflammation on subventricular zone versus hippocampal precursors in brain injury. Ann Neurol. 2011;70(4):616–26.
Leviton A, Gressens P. Neuronal damage accompanies perinatal white-matter damage. Trends Neurosci. 2007;30(9):473–8.
Rock RB, Gekker G, Hu S, Sheng WS, Cheeran M, Lokensgard JR, Peterson PK. Role of microglia in central nervous system infections. Clin Microbiol Rev. 2004;17(4):942–64. Table of contents.
Cheeran MC, Hu S, Sheng WS, Peterson PK, Lokensgard JR. CXCL10 production from cytomegalovirus-stimulated microglia is regulated by both human and viral interleukin-10. J Virol. 2003;77(8):4502–15.
Cheeran MC, Hu S, Yager SL, Gekker G, Peterson PK, Lokensgard JR. Cytomegalovirus induces cytokine and chemokine production differentially in microglia and astrocytes: antiviral implications. J Neurovirol. 2001;7(2):135–47.
Kaushik DK, Gupta M, Basu A. Microglial response to viral challenges: every silver lining comes with a cloud. Front Biosci. 2011;16:2187–205.
Kang SH, Abdel-Massih RC, Brown RA, Dierkhising RA, Kremers WK, Razonable RR. Homozygosity for the toll-like receptor 2 R753Q single-nucleotide polymorphism is a risk factor for cytomegalovirus disease after liver transplantation. J Infect Dis. 2012;205(4):639–46.
Chaudhuri S, Lowen B, Chan G, Davey A, Riddell M, Guilbert LJ. Human cytomegalovirus interacts with toll-like receptor 2 and CD14 on syncytiotrophoblasts to stimulate expression of TNFalpha mRNA and apoptosis. Placenta. 2009;30(11):994–1001.
Juckem LK, Boehme KW, Feire AL, Compton T. Differential initiation of innate immune responses induced by human cytomegalovirus entry into fibroblast cells. J Immunol. 2008;180(7):4965–77.
Isaacson MK, Juckem LK, Compton T. Virus entry and innate immune activation. Curr Top Microbiol Immunol. 2008;325:85–100.
Mezger M, Bonin M, Kessler T, Gebhardt F, Einsele H, Loeffler J. Toll-like receptor 3 has no critical role during early immune response of human monocyte-derived dendritic cells after infection with the human cytomegalovirus strain TB40E. Viral Immunol. 2009;22(6):343–51.
Kossmann T, Morganti-Kossmann MC, Orenstein JM, Britt WJ, Wahl SM, Smith PD. Cytomegalovirus production by infected astrocytes correlates with transforming growth factor-beta release. J Infect Dis. 2003;187(4):534–41.
Alcendor DJ, Charest AM, Zhu WQ, Vigil HE, Knobel SM. Infection and upregulation of proinflammatory cytokines in human brain vascular pericytes by human cytomegalovirus. J Neuroinflammation. 2012;9:95.
Miller-Kittrell M, Sparer TE. Feeling manipulated: cytomegalovirus immune manipulation. Virol J. 2009;6:4.
Vink C, Smit MJ, Leurs R, Bruggeman CA. The role of cytomegalovirus-encoded homologs of G protein-coupled receptors and chemokines in manipulation of and evasion from the immune system. J Clin Virol. 2001;23(1–2):43–55.
Nigro G, Torre RL, Pentimalli H, Taverna P, Lituania M, de Tejada BM, Adler SP. Regression of fetal cerebral abnormalities by primary cytomegalovirus infection following hyperimmunoglobulin therapy. Prenat Diagn. 2008;28(6):512–7.
Nigro G, Adler SP, La Torre R, Best AM. Passive immunization during pregnancy for congenital cytomegalovirus infection. N Engl J Med. 2005;353(13):1350–62.
Kimberlin DW, Lin CY, Sanchez PJ, Demmler GJ, Dankner W, Shelton M, Jacobs RF, Vaudry W, Pass RF, Kiell JM, Soong SJ, Whitley RJ. Effect of ganciclovir therapy on hearing in symptomatic congenital cytomegalovirus disease involving the central nervous system: a randomized, controlled trial. J Pediatr. 2003;143(1):16–25.
Oliver SE, Cloud GA, Sanchez PJ, Demmler GJ, Dankner W, Shelton M, Jacobs RF, Vaudry W, Pass RF, Soong SJ, Whitley RJ, Kimberlin DW. Neurodevelopmental outcomes following ganciclovir therapy in symptomatic congenital cytomegalovirus infections involving the central nervous system. J Clin Virol. 2009;46 Suppl 4:S22–6.
Griffiths P, Plotkin S, Mocarski E, Pass R, Schleiss M, Krause P, Bialek S. Desirability and feasibility of a vaccine against cytomegalovirus. Vaccine. 2013;31 Suppl 2:B197–203.
Griffiths PD. Burden of disease associated with human cytomegalovirus and prospects for elimination by universal immunisation. Lancet Infect Dis. 2012;12(10):790–8.
Sung H, Schleiss MR. Update on the current status of cytomegalovirus vaccines. Expert Rev Vaccines. 2010;9(11):1303–14.
Pass RF, Zhang C, Evans A, Simpson T, Andrews W, Huang ML, Corey L, Hill J, Davis E, Flanigan C, Cloud G. Vaccine prevention of maternal cytomegalovirus infection. N Engl J Med. 2009;360(12):1191–9.
Mussi-Pinhata MM, Yamamoto AY, Moura Brito RM, de Lima Isaac M, de Carvalho e Oliveira PF, Boppana S, Britt WJ. Birth prevalence and natural history of congenital cytomegalovirus infection in a highly seroimmune population. Clin Infect Dis. 2009;49(4):522–8.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this chapter
Cite this chapter
Schleiss, M.R. (2014). Congenital Cytomegalovirus Disease. In: Peterson, P., Toborek, M. (eds) Neuroinflammation and Neurodegeneration. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-1071-7_14
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1071-7_14
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-1070-0
Online ISBN: 978-1-4939-1071-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)