Abstract
The Galapagos Islands offer a unique ecological landscape found nowhere else in the world. However, a lack of water and sanitation infrastructure within populated areas of the Galapagos Islands has the potential to affect many of these ecosystem attributes. Specifically, a rapidly growing population and expanding impacts of tourism are contaminating the coastal environments of the Galapagos Islands. Water quality monitoring is conducted globally to provide important information on the incidence of dangerous microbial contamination. Fecal indicator bacteria (such as Enterococcus spp. and Escherichia coli) are quantified as proxies for the presence of pathogens that can be found in water that is contaminated with human and animal feces. These fecal indicator bacteria, however, cannot be used to indicate a specific source of fecal contamination. Recently, advancements have been made in the quantification of other alternative markers for human fecal contamination, such as members of the Bacteroidales group. These are bacteria or gene markers in the bacteria that are predominantly found in the human gastrointestinal tract and, as such, can be more accurate indicators of the presence of human fecal pollution. In this study, we used molecular techniques for measurement of DNA to quantify both Enterococcus spp. and Bacteroides spp. specific molecular markers of human fecal contamination in the coastal waters of San Cristobal and Santa Cruz islands. Enterococcus spp. and Bacteroides spp. specific molecular marker concentrations were high enough in certain locations to warrant concern. Human fecal pollution was present in both coastal marine beach waters and a brackish water lagoon popular for bathing and recreation. Since our study, the wastewater treatment plant on San Cristobal has been upgraded, possibly resulting in reduced microbial contamination. However, these results indicate that poor wastewater management could be posing a threat to human health for both residents and tourists, and the coastal environments of the Galapagos Islands. Improvements to marine water quality are critical to protect inhabitants, tourists, and the fragile ecosystem components of the Galapagos Islands and to ensure the long-term viability and sustainability of a unique place.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Fecal Coliform
- Fecal Contamination
- Fecal Indicator Bacterium
- Galapagos Island
- Japan International Cooperation Agency
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Introduction
The Galapagos Archipelago is a UNESCO World Heritage Site and unique ecological setting of species diversity. Internationally renowned for its link to Charles Darwin’s research and seminal publication on biodiversity, “Origin of the Species,” a lesser known fact is that the archipelago is home to more than 30,000 inhabitants, a rapidly growing population, and a burgeoning tourism industry (Epler 2007; Watkins and Cruz 2007). Tourism has grown dramatically in recent years, increasing more than threefold from 1990 to 2006 (Watkins and Cruz 2007), with more than 170,000 people visiting the islands in 2010 (GNPS 2011). The increase in tourism (and economic growth) has resulted in increased immigration (Kerr et al. 2004). This interconnected growth in tourism and population has created a commensurate if not greater strain on the limited infrastructure and ecological resource of the islands (Walsh et al. 2010). Unsustainable growth threatens the islands’ resources, the tourism-driven economy, and human health through inadequate infrastructure, especially pertaining to water resources.
Water resources are critically important to the Galapagos Islands (Hennessy and McCleary 2011; Lopez and Rueda 2010; Kerr et al. 2004). Management of marine and fresh water is paramount to the success and balance of the Galapagos Island economy and ecology; however, the human impact on groundwater and marine water resources is apparent in bacterially contaminated aquifers on Santa Cruz Island, contaminated household water on San Cristobal Island, and contamination of nearshore marine waters on Isabela Island (d’Ozouville 2008; Lopez and Rueda 2010; Walsh et al. 2010). Wastewater management is one of the greatest challenges, as most population centers depend on septic (onsite wastewater treatment systems usually consisting of a collection area and subsequent leaching into the ground) or poorly constructed sewage systems (Walsh and McCleary 2009), which are not well suited for the lava-like bedrock which contains little soil and primarily fractured basalt (d’Ozouville 2008). This lack of sewage infrastructure and non-ideal subsurface create a high potential for water resource contamination.
Internationally, fecal contamination of coastal waters used for recreation and seafood production is of serious concern given the potential public health risk associated with contact and/or ingestion of fecal pathogens. In developing countries with increasing populations and insufficient infrastructure, fecal contamination can be of concern to both inhabitants and visitors (Rose 2006). Health risks often include gastrointestinal and respiratory illness, skin and eye irritation and, if symptoms go untreated, can result in more severe illness (Fleisher et al. 2010; Wade et al. 2010). Water-related illnesses have been reported in the Galapagos Islands, though are often anecdotal and poorly researched (Hennessy and McCleary 2011; Walsh et al. 2010).
The impact of fecal contamination, such as that from sewage discharge, also extends to an array of animals and plants (Fernandez 2008; Werdeman 2006). Sewage treatment plants often discharge waters that have high concentrations of heavy metals, organic compounds, detergents, endocrine disruptors, and personnel care products that can have myriad effects on wildlife (Brausch and Rand 2011; Islam and Tanaka 2004; Atkinson et al. 2003). Even the most advanced sewage treatment plants discharge highly labile organic and inorganic nutrients, often contributing directly to the formation of surface algae and phytoplankton growth in the water. These surface “blooms” can contribute to low dissolved oxygen in coastal waters (Pearl 2009), endangering the survival of fish, turtles, amphibians, and benthic organisms with consequences that are severe (Fernandez 2008).
Galapagos Islands Water Resources
Water Impairment
Fecal microbial contamination of water resources of the Galapagos Islands, though anecdotally suspected, has been difficult to demonstrate scientifically. Water quality studies have mainly focused on freshwater (groundwater and household drinking water) and have found elevated Escherichia coli (E. coli) and fecal coliform concentrations (Lopez and Rueda 2010). A recent study at eight total sites on three islands, conducted by the Galapagos National Park in combination with the Japan International Cooperation Agency (JICA), found levels of fecal coliforms (of which E. coli is a major subset) in groundwater, lagoon water, and household water at levels ranging from 102 to 103 fecal coliform bacteria per 100 ml, for all sample types (Lopez and Rueda 2010). These levels are beyond limits established by Ecuadorian national environmental legislation to protect public health (TULAS 2003). Marine and brackish water fecal contamination has been previously determined around larger towns of the islands and their associated ports (Kerr et al. 2004; Moir and Armijos 2007; d’Ozouville 2008; Lopez and Rueda 2010). For instance, a study by Armijos et al. (2002) in the coastal waters surrounding Puerto Ayora, Santa Cruz, found marine water degradation due to high E. coli concentrations at sites where contaminated groundwater leachate was flowing into Academy Bay during low tide, which is when water is being pulled from the land into the coastal environment.
Overall, long-term microbial water quality monitoring reports and robust data sets are not available for the Galapagos Islands (Walsh et al. 2010), especially for nearshore marine waters. When microbial water quality data does exist, it does not indicate sources of contamination, routes of transport, or seasonality. These are all more difficult pieces of information to derive. In addition, all previous reported monitoring has been conducted using non-molecular methods (e.g., multiple tube fermentation or most probable number methods such as direct culture plating or utilization of chromogenic substrate tests). Though these studies of microbial water quality are useful, they are generally insufficient for identifying the source(s) of contamination, which are likely to be variable and multiple. Using a sanitary survey style to visually identify potential sources of contamination, and based in previous research, the following are suspected sources of fecal contamination to marine waters: contaminated groundwater (Lopez and Rueda 2010), submarine sewage discharges, overland pipe discharges (Walsh and McCleary 2009), boat discharges (Werdeman 2006), and surface runoff during wet weather. Source-specific monitoring is needed to understand the complete picture of potential water impairment in the Galapagos Islands.
Elevated nutrient levels and decreased dissolved oxygen concentrations have also been reported within Academy, Wreck, and Turtle Bay waters proximate to some of the largest towns in the Galapagos Islands and are suspected to be related to sewage contamination (Fernandez 2008; Werdeman 2006). A study by Werdeman (2006) examined the marine waters around Puerto Ayora, Puerto Baquerizo Moreno, and Puerto Villamil and compared nutrient levels (phosphate, nitrate, and ammonium) to a non-populated reference bay (Cartago Bay). Generally, levels of nutrients were higher in all populated bays than the reference bay and showed increased eutrophication and decreased dissolved oxygen. A second study conducted by Fernandez (2008) also found increased nutrient levels in Academy Bay, the bay surrounding Puerto Ayora, Santa Cruz. This study determined a net flux of nutrients from the terrestrial to the marine environment, leading to potential increases in both eutrophication and microbial contamination. In both studies, it was concluded that these elevated nutrient concentrations were partially due to sewage inputs (Fernandez 2008; Werdeman 2006).
Water Infrastructure
Throughout the Galapagos Islands, expanding population and community sprawl are challenging an already stressed and inadequate water and sanitation infrastructure. In a domino effect scenario, increased population and sprawl leads to increased sewage waste, which increases unregulated discharge and overwhelms an already overburdened wastewater system. Without proper infrastructure, “straight piping” into fissures (Moir and Armijos 2007), septic systems in subsurfaces not ideal for this method of disposal (d’Ozouville 2008), and cesspools (latrines) are often utilized (Walsh and McCleary 2009). Fecal contamination is occuring in coastal waters near growing population centers like Puerto Ayora, where brackish lagoons have been contaminated (Kerr et al. 2004; Moir and Armijos 2007), as well as Isabela, where previous untreated waste was directly piped to lagoons and mangroves (Walsh and McCleary 2009), and even on San Cristobal (where greater than 85% of households are connected to a public sewage disposal system) where untreated sewage is pumped through town to a submarine discharge, resulting in increased eutrophication and microbial contamination of waters proximate to Punta Carola, a popular swimming and surfing area. In essence, tourism and population growth in the Galapagos Islands are out of balance with the development and demand for water and wastewater infrastructure (Hennessy and McCleary 2011).
Impacts
Water quality impairment has human health implications for users of marine waters for swimming. As described earlier, fecal contamination of water resources can have major repercussions for the public health of residents and tourists alike in the Galapagos Islands. Though a paucity of data exists to link water quality contamination directly with public illness (Walsh et al. 2010), anecdotal evidence from tourists and residents of the islands points to direct connections between water contamination and illness (Hennessy and McCleary 2011). For instance, Hennessy and McCleary (2011) reported that, based on information from a physician of Puerto Villamil, Isabela, as many as 70% of local illnesses were related to contact (either through consumption or exposure) to contaminated water. These illnesses reported are typical of fecal microbial water quality contamination, such as gastroenteritis and skin diseases, and commonly affect younger children (Wade et al. 2010; Colford et al. 2007). Generally, waterborne illnesses are underreported, indicating that this correlation between water contact and illness may be even greater than estimated.
Nearshore marine water quality degradation not only threatens the public health of tourists and residents but also impacts the wildlife of the archipelago. Previous research has shown impacts of sewage contamination to mollusk and crabs assemblages (Cannicci et al. 2009), large mammals such as sea lions (Sturm et al. 2011), general fish species through impacts to food sources such as phytoplankton (Pearl 2009), and chemicals associated with altered breeding and reproduction (Al-Bahry et al. 2009; Penha-Lopes et al. 2009). Additional impacts to wildlife have also been reported from increased heavy metals and endocrine-disrupting compounds (Brausch and Rand 2011). Within the Galapagos Islands, marine wildlife includes sally lightfoot crabs, sea lions, marine iguanas, and myriad fish species. Numerous birds, including the blue-footed boobies, the wingless cormorant, and the magnificent frigate bird, are also potentially affected through their dependence on marine-based food sources. These species are likely affected by decreased nearshore water quality, which results in fish kills and effects reproduction and recruitment of many lower trophic food species.
One example of potential sewage impacts on wildlife of the Galapagos Islands is the sea lion. An integral part of the Galapagos Islands marine landscape and a tourism icon, sea lions may be affected by sewage-related marine water impairment. Previous studies have shown that these pinnipeds are particularly susceptible to illness when inbreeding increases and pathogens are common (Sturm et al. 2011; Acevedo-Whitehouse et al. 2003), which is likely the case for sea lions in the Galapagos Islands’ marine environment. Even though sea lion populations have recovered (following a massive reduction in recruitment during the 1997–1998 El Niño), current offspring are experiencing a high degree of illness (Jiménez-Uzcátegui et al. 2007). These high rates of illness may have some links to stressors associated with human-caused marine water impairment. For instance, recent research by Sturm et al. (2011) found increased illness in sea lions on the Chilean coast due to Salmonella enterica infection and identified exposure to sewage as one of the likely causes. For a place which ecologically thrives on a tourism industry driven by the image of a pristine and healthy ecological landscape, determining whether water contamination is leading to wildlife health impacts and ameliorating such impacts is critical.
Characterizing Fecal Contamination in the Coastal Waters of Santa Cruz and San Cristobal Using Molecular Methods
Study Area and Sample Analysis
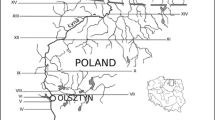
A small-scale study was conducted on two islands using molecular techniques to determine the quantities of Enterococcus spp. and Bacteroides spp. specific markers. Samples were collected within the cities of Puerto Baquerizo Moreno, San Cristobal, and Puerto Ayora, Santa Cruz (Fig. 10.1). These population centers were proximal to coastal waters that were considered impaired by visual observation. Other relatively non-impaired sites adjacent to these populated areas were also sampled as reference sites. Samples were filtered and stored frozen for later analysis at the University of North Carolina at Chapel Hill’s Institute of Marine Science, a laboratory with access to advanced tools for analysis.
Molecular analysis of samples to quantify Enterococcus spp. and source-specific Bacteroides spp. markers followed existing published protocols (Noble et al. 2010; Converse et al. 2009; Kildare et al. 2007; Seurinck et al. 2005). The trio of Bacteroidales spp. marker assays, fecal Bacteroides spp., BacHum, and HF183 (human specific), cover a gradient of specificity and sensitivity. The fecal Bacteroides spp. assay is the least specific and quantifies a cohort of anaerobic bacterial species that are found most closely associated with human feces but can also be found in some animal fecal material in lower concentrations. The BacHum and human-specific assays are more specific to human fecal contamination, with the human-specific assay having the greatest ability to discriminate between animal and human fecal contamination (94–100% discriminatory ability, Ahmed et al. 2009). In real-world samples, quantification of both the human-specific and BacHum markers, along with high concentrations of the fecal Bacteroides spp. is indicative of a strong potential for the presence of human fecal contamination in a body of water.
Study Results and Implications
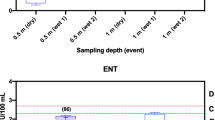
Enterococcus spp. concentrations were elevated (greater than 104 CE/100 ml) in 85.7% (6 of 7) of the samples in the coastal waters of Punta Carola Outfall and Playa de los Marinos Berm, San Cristobal. Analysis of the samples using the trio of Bacteroidales-based molecular methods demonstrated the presence of human fecal contamination in 62.5% (4 of 7) of the samples in the coastal waters of Punta Carola Outfall and Playa de los Marinos Berm, San Cristobal (two sites suspected to be impaired during an initial visual assessment). Other sites, such as Laguna de las Ninfas, showed more ephemeral signs of human fecal contamination (Fig. 10.2). This result was not surprising given the location of a nearby submarine sewage discharge pipe at Punta Carola and previous research which has determined fecal contamination in lagoons within or close to population centers in the Galapagos Islands (Lopez and Rueda 2010; Moir and Armijos 2007). The range of Enterococcus spp. concentrations is presented as circles in Fig. 10.1, while average concentrations for fecal Bacteroides spp., BacHum, and human-specific markers are presented as bar graphs for each site. Concentrations for all assays and all sites averaged 1.38 × 102, 4.74 × 105, 1.97 × 103, and 1.54 × 105 cell equivalents (CE)/100 ml for Enterococcus spp., fecal Bacteroides spp., BacHum, and human-specific markers, respectively.
The results of this small-scale study demonstrate concentrations of Enterococcus spp. that, according to current water quality standards used globally, could present a risk to public health for those using certain contaminated waters for recreation (World Health Organization 2003). Furthermore, the source-specific molecular marker prevalence and concentrations indicate a strong likelihood that human fecal contamination was present in the nearshore waters surrounding Puerto Baquerizo Moreno and Puerto Ayora during the study period. Based on previous research examining molecular analysis-based levels of Enterococcus spp. and Bacteroidales in marine bathing waters (Wade et al. 2010), levels observed at these sites could cause gastroenteritis and respiratory illness rates to exceed 10% for those swimming in these waters. Interestingly, reference clean water beaches located outside urban centers (e.g., Playa Tortuga and Las Tijeretas) showed concentrations that were generally lower for Enterococcus spp. and exhibited no apparent signs of human fecal contamination based on the trio of Bacteroides spp. markers. Unfortunately, more tourists and locals swim and recreate in areas near towns (e.g., Playa de los Marinos, Playa Carola, and Laguna de las Ninfas), making the potential incidence of disease from waterborne contact higher in these areas.
In 2011 (after this study was conducted), the Puerto Baquerizo Moreno wastewater treatment plant was upgraded for both efficiency and improvements to environmental standards for operation. Prior to this upgrade, the sewage was discharged into the coastal waters, untreated. The new treatment plant utilizes a chemical treatment, an activated sludge plant with aerobic processing, an aeration system, a disinfection process, and controlled discharge volumes. The plant has the initial capacity to serve 6,000 people, with a second phase of upgrades intended to permit service to be extended to 9,000 people. Our study has not sampled after the sewage upgrade, but future testing should be conducted to verify that there has been an improvement in water quality of the coastal waters proximal to the sewage discharge.
Recommendations
Monitoring
Consistent monitoring of fecal indicator bacteria and source-specific molecular markers is greatly needed in marine and brackish waters near residential and tourist epicenters of the Galapagos Islands. These monitoring plans should be implemented to include areas of suspected fecal contamination as well as reference (nonimpacted nearshore water comparison) sites. Sampling should include monitoring of Enterococcus spp. and more human-specific indicators such as the Bacteroidales spp. group. Additional monitoring data such as nutrient analysis for nitrogen and phosphorus, and water quality parameters, such as chlorophyll a, dissolved oxygen, turbidity, total dissolved solids, conductivity, and water temperature, should also be collected.
Initial multi-year monitoring to determine “hot spots” of contamination should be followed by targeted monitoring of contaminated sites. A combined multi-year and “hot spot”-specific approach would permit small-scale variations in contamination, such as weather patterns, and tourist and resident seasonal fluctuations to be detected. Additional changes to the monitoring plan would revolve around alterations (improvements) in wastewater infrastructure, changes in municipal boundaries (sprawl), and epidemiology (e.g., high illness rates associated with a certain marine recreational area). For instance, from January to June, heavy rain showers occasionally occur on the islands, and amounts can quadruple in El Niño years (Adelinet et al. 2008). Though the soils of the Galapagos Islands provide a low runoff potential (Adelinet et al. 2008; d’Ozouville 2008), the presence of intermittent stream beds within Puerto Baquerizo Moreno and Puerto Ayora indicate that runoff can be quite substantial. These potential storms could lead to large overland fecal contaminant flushing events and could be targeted for sampling.
Improved Infrastructure
Improved water infrastructure is essential to reducing contamination of nearshore marine and brackish waters. Septic systems and unregulated discharges are likely not permitting proper attenuation of fecal microbial pathogens before reaching nearshore waters, due to the subsurface characteristics of the Galapagos Islands. As a result, lagoons such as La Salina, Isabela (Hennessy and McCleary 2011), and Laguna de las Ninfas, Santa Cruz (Lopez and Rueda 2010), or coastal waters such as Playa de los Marinos, San Cristobal, become contaminated. Public wastewater systems, which treat waste before release, similar to the one recently completed at Puerto Baquerizo Moreno, San Cristobal, are needed for the Galapagos Islands population centers.
Education and Benefits
Though the cost of long-term monitoring and improvements to infrastructure are substantial, the overall benefits are invaluable. Many of the people of the Galapagos acknowledge the importance of water resources and the need for their monitoring, protection, and improvement (Hennessy and McCleary 2011). Continued education and awareness is the key to an engaged citizenry; one which could encourage municipal leaders to invest in improved sanitary infrastructure and water monitoring. This is especially true in developing countries, where the need for clean water and proper sanitation is ultimately up to the residents of the small municipalities (Hagedorn et al. 2011). A portion of the conservation funds that are aimed at protection of the islands and/or tourism income could be allocated for municipal sanitation improvements. Educational institutions (both Ecuadorian and international) could be utilized to assist and implement monitoring plans to reduce costs. International aid groups could also be leveraged, similar to previous studies that were conducted in collaboration between the Galapagos National Park Service and JICA on Santa Cruz Island.
Conclusions
Investment in water monitoring and infrastructure would benefit the Galapagos Islands on three fronts: through improved quality of life for residents, improved economic benefits (sustained and safe tourism), and improved ecological protection of the rare species for which the Galapagos are famous. Robust microbial contaminant monitoring is needed in nearshore and brackish waters used for recreation (beaches and lagoons) by locals and tourists to better understand the fate and transport of fecal contamination. Additional studies during the “rainy” season should also be conducted to determine inputs of runoff to these waters, and potential seasonally related bacterial reservoir populations and rainfall-associated contamination events. Wastewater infrastructure improvements (centralized wastewater treatment) could dramatically improve water quality in population centers throughout the islands. However, sewage infrastructure improvements must occur in concert with control of human population growth and sprawl to reduce unregulated waste disposal. Education and awareness of the need for clean water from the residents will ultimately influence local government, and infrastructure improvements could be funded utilizing diverse financial resources. Establishing the groundwork for better understanding of the water quality of the Galapagos Islands will inform potential long-range management strategies for improved water quality and marine water resource protection.
References
Acevedo-Whitehouse K, Gulland F, Greig D, Amos W (2003) Inbreeding: disease susceptibility in California sea lions. Nature 422:35
Adelinet M, Fortin J, d’Ozouville N (2008) The relationship between hydrodynamic properties and weathering of soils derived from volcanic rocks, Galapagos Islands (Ecuador). Environ Geol 56:45–58
Ahmed W, Goonetilleke A, Powell D, Gardner T (2009) Evaluation of multiple sewage-associated Bacteroides PCR markers for sewage pollution tracking. Water Res 43:4872–4877
Al-Bahry SN, Mahmoud IY, Al-Belushi KIA, Elshafie AE, Al-Harthy A, Bakheit CK (2009) Coastal sewage discharge and its impact on fish with reference to antibiotic resistant enteric bacteria and enteric pathogens as bio-indicators of pollution. Chemosphere 77:1534–1539
Armijos E, Palmer M, Moir FC (2002) Selection of an ocean outfall site for the town of Puerto Ayora in the Galapagos Islands. XXVII Congreso Interamericano de Ingenieria Sanitaria y Ambiental, Cancun, Mexico
Atkinson S, Atkinson MJ, Tarrant AM (2003) Estrogens from sewage in coastal marine environments. Environ Health Perspect 111:531–535
Brausch JM, Rand GM (2011) A review of personal care products in the aquatic environment: environmental concentrations and toxicity. Chemosphere 82:1518–1532
Cannicci S, Bartolini F, Dahdouh-Guebas F, Fratini S, Litulo S, Macia A, Mrabu EJ, Penha-Lopes G, Paula J (2009) Effects of urban wastewater on crab and mollusc assemblages in equatorial and subtropical mangroves of East Africa. Estuarine Coastal Shelf Sci 84:305–317
Colford JM Jr, Wade TJ, Schiff KC, Wright CC, Griffith JF, Sandhu SK, Burns S, Sobsey M, Lovelace G, Weisberg SB (2007) Water quality indicators and the risk of illness at beaches with nonpoint sources of fecal contamination. Epidemiology 18:27–35
Converse RR, Blackwood AD, Kirs MA, Griffith JF, Noble RT (2009) Rapid QPCR-based assay for fecal Bacteroides spp. as a tool for assessing fecal contamination in recreational waters. Water Res 43:4828–4837
d’Ozouville N (2008) Water resource management in Galapagos: the case of Pelican Bay Watershed. Galapagos report 2007–2008. PNG, FCD and INGALA. Puerto Ayora, Galapagos, pp 158–164
Epler B (2007) Tourism, the economy, population growth, and conservation in Galapagos. Charles Darwin Foundation, Puerto Ayora, Galapagos
Fernandez AR (2008) Coastal nutrient and water budget assessments for Puerto Ayora, Academy Bay. The Pennsylvania State University, Santa Cruz Island. SOARS Report
Fleisher JM, Fleming LE, Solo-Gabriele HM et al (2010) The BEACHES study: health effects and exposures from non-point source microbial contaminants in subtropical recreational marine waters. Int J Epidemiol 39:1291–1298
GNPS, Galapagos National Park Service (2011) Informe de Ingreso de Turistas 2010. Galapagos National Park, Puerto Ayora, Santa Cruz
Hagedorn C, Lepo JE, Hellein KN, Ajidahun AO, Xinqiang L, Li H (2011) Microbial source tracking in China and developing nations. In: Hagedorn C et al (eds) Microbial source tracking: methods, applications, and case studies, Springer, New York
Hennessy E, McCleary AL (2011) Nature’s Eden? The production and effects of ‘pristine’ nature in the Galapagos Islands. Isl Stud J 6:131–156
Islam MS, Tanaka M (2004) Impacts of pollution on coastal and marine ecosystems including coastal and marine fisheries and approach for management: a review and synthesis. Mar Pollut Bull 48:624–649
Jiménez-Uzcátegui G, Milstead B, Márquez C, Zabala J, Buitrón P, Llerena A, Salzar S, Fessl B (2007) Galapagos vertebrates: endangered status and conservation actions. Galapagos report 2006–2007. Charles Darwin Foundation, Puerto Ayora, Galapagos, Ecuador
Kerr S, Cardenas S, Hendy J (2004) Migration and the environment in the Galapagos. Motu Economic and Public Policy Research, Wellington, New Zealand
Kildare BJ, Leutenegger CM, McSwain BS, Bambic DG, Rajal VB, Wuertz S (2007) 16S rRNA-based assays for quantitative detection of universal, human-, cow-, and dog-specific fecal Bacteroidales: a Bayesian approach. Water Res 41:3701–3715
Lopez J, Rueda D (2010) Water quality monitoring system in Santa Cruz, San Cristobal, and Isabela. Galapagos report 2009–2010. Charles Darwin Foundation, Puerto Ayora, Galapagos
Moir FC, Armijos E (2007) Integrated water supply and wastewater solutions for the town of Puerto Ayora on the island of Santa Cruz–the Galapagos Islands–Ecuador. Proc Water Environ Feder 70:4816–4842
Noble RT, Blackwood AD, Griffith JF, McGee CD, Weisberg SB (2010) Comparison of rapid quantitative PCR-based and conventional culture-based methods for enumeration of Enterococcus spp. and Escherichia coli in recreational waters. Appl Environ Microbiol 76:7437–7443
Pearl HW (2009) Controlling eutrophication along the freshwater–marine continuum: dal nutrient (N and P) reductions are essential. Estuar Coast 32:593–601
Penha-Lopes G, Torres P, Narciso L, Cannicci S, Paula J (2009) Comparison of fecundity, embryo loss and fatty acid composition of mangrove crab species in sewage contaminated and pristine mangrove habitats in Mozambique. J Exp Mar Biol Ecol 381:25–32
Rose JB (2006) Identification and characterization of biological risks for water. In: Dura G et al (eds) Management of intentional and accidental water pollution. Springer
Seurinck S, Defoirdt T, Verstraete W, Siciliano SD (2005) Detection and quantification of the human-specific HF183 Bacteroides 16S rRNAgenetic marker with real-time PCR for assessment of human faecal pollution in freshwater. Environ Microbiol 7:249–259
Sturm N, Abalos P, Fernandez A, Rodriguez G, Oviedo P, Arroyo V, Retamal P (2011) Salmonella enterica in pinnipeds. Chile. Emerg Infect Dis 17:2377–2378
TULAS (2003) Texto Unificado de la Legislación Ambiental Secundaria
Wade TM, Sams E, Brenner KP, Haugland R, Chern E, Beach M, Wymer L, Rankin CC, Love D, Li Q, Noble RT, Dufour AP (2010) Rapidly measured indicators of recreational water quality and swimming-associated illness at marine beaches: a prospective cohort study. Environ Health 9:66
Walsh SJ, McCleary A (2009) Water and health in the Galapagos Islands: a spatial analysis of human-environment interactions. Presentation to UNC institute for global health and infectious diseases, University of North Carolina at Chapel Hill
Walsh SJ, McCleary AL, Heumann BW, Brewington L, Raczkowski EJ, Mena CF (2010) Community expansion and infrastructure development: implications for human health and environmental quality in the Galapagos Islands of Ecuador. J Lat Am Geogr 9:137–159
Watkins G, Cruz F (2007) Galapagos at risk: a socioeconomic analysis of the situation in the Archipelago. Charles Darwin Foundation, Puerto Ayora, Galapagos
Werdeman JL (2006) Effects of populated towns on water quality in neighboring Galapagos bays. Dissertation, University of Washington School of Oceanography, Washington
World Health Organization (2003) Guidelines for safe recreational water environments, vol 1. Feacal pollution and water quality. World Health Organization, Geneva
Acknowledgments
The research was made possible through a University of North Carolina Center for Galapagos Studies SEED grant. Special thanks to Veronica Barragan and David Hervas for assistance with sample shipping and Philip Page and Carlos Mena for assistance with logistics. Also, thanks to Monica Green for sample verification and Rodney Gaujardo and Dana Gulbransen for map development. Additional thanks to Parque Nacional Galapagos for assistance with site selection and permission to conduct this research and, particularly, Washington Tapia and Javier López from the Galapagos National Park for assistance at the beginning of the project.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media, LLC
About this chapter
Cite this chapter
Stumpf, C.H., Gonzalez, R.A., Noble, R.T. (2013). Investigating the Coastal Water Quality of the Galapagos Islands, Ecuador. In: Walsh, S., Mena, C. (eds) Science and Conservation in the Galapagos Islands. Social and Ecological Interactions in the Galapagos Islands, vol 1. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-5794-7_10
Download citation
DOI: https://doi.org/10.1007/978-1-4614-5794-7_10
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-5793-0
Online ISBN: 978-1-4614-5794-7
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)