Abstract
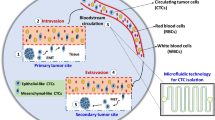
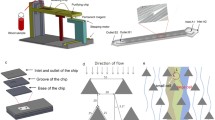
Selectively capturing and releasing viable circulating tumor cells (CTCs) from the peripheral blood of cancer patients is advantageous for investigating the molecular hallmarks of metastasis and developing personalized therapeutics. CTC-based liquid biopsies are flourishing in the clinical setting, offering opportunities to track the real-time responses of patients during clinical trials and lending accessibility to cancers that are traditionally difficult to diagnose. However, CTCs are rare compared to the breadth of cells that reside in the circulatory network, which has encouraged the engineering of novel microfluidic devices. Current microfluidic technologies either extensively enrich CTCs but compromise cellular viability or sort viable CTCs at low efficiencies. Herein we present a procedure to fabricate and operate a microfluidic device capable of capturing CTCs at high efficiencies while ensuring high viability. The microvortex-inducing microfluidic device functionalized with nanointerfaces positively enriches CTCs via cancer-specific immunoaffinity, while a thermally responsive surface chemistry releases the captured cells by raising the temperature to 37 °C.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Steeg PS (2006) Tumor metastasis: mechanistic insights and clinical challenges. Nat Med 12:895–904
Plaks V, Koopman CD, Werb Z (2013) Circulating tumor cells. Science 341:1186–1188
Moose DL, Krog BL, Kim TH, Zhao L, Williams-Perez S, Burke G et al (2020) Cancer cells resist mechanical destruction in circulation via RhoA/Actomyosin-dependent Mechano-adaptation. Cell Rep 30:3864–74.e6
Sun YF, Wu L, Liu SP, Jiang MM, Hu B, Zhou KQ et al (2021) Dissecting spatial heterogeneity and the immune-evasion mechanism of CTCs by single-cell RNA-seq in hepatocellular carcinoma. Nat Commun 12:4091
Aguirre-Ghiso JA (2007) Models, mechanisms and clinical evidence for cancer dormancy. Nat Rev Cancer 7:834–846
Aceto N, Bardia A, Miyamoto DT, Donaldson MC, Wittner BS, Spencer JA et al (2014) Circulating tumor cell clusters are Oligoclonal precursors of breast cancer metastasis. Cell 158:1110–1122
Li W, Reátegui E, Park MH, Castleberry S, Deng JZ, Hsu B et al (2015) Biodegradable nano-films for capture and non-invasive release of circulating tumor cells. Biomaterials 65:93–102
Aguirre-Ghiso JA (2021) Translating the science of cancer dormancy to the clinic. Cancer Res 81:4673–4675
Ignatiadis M, Sledge GW, Jeffrey SS (2021) Liquid biopsy enters the clinic — implementation issues and future challenges. Nat Rev Clin Oncol 18:297–312
Yu M, Bardia A, Aceto N, Bersani F, Madden MW, Donaldson MC et al (2014) Ex vivo culture of circulating breast tumor cells for individualized testing of drug susceptibility. Science 345:216–220
Kim TH, Wang Y, Oliver CR, Thamm DH, Cooling L, Paoletti C et al (2019) A temporary indwelling intravascular aphaeretic system for in vivo enrichment of circulating tumor cells. Nat Commun 10:1478
El-Ali J, Sorger PK, Jensen KF (2006) Cells on chips. Nature 442:403–411
Elodie Sollier E, Go D, James Che R, Gossett D, Sean O’Byrne M, Weaver W et al (2013) Size-selective collection of circulating tumor cells using vortex technology. Lab Chip 14:63–77
Hur SC, Henderson-MacLennan NK, McCabe ERB, Di CD (2011) Deformability-based cell classification and enrichment using inertial microfluidics. Lab Chip 11:912–920
Varsano G, Wang Y, Wu M (2017) Probing mammalian cell size homeostasis by channel-assisted cell reshaping. Cell Rep 20:397–410
Yu M, Bardia A, Wittner BS, Stott SL, Smas ME, Ting DT et al (2013) Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science 339:580–584
Keller L, Pantel K (2019) Unravelling tumour heterogeneity by single-cell profiling of circulating tumour cells. Nat Rev Cancer 19:553–567
Alvarado-Estrada K, Marenco-Hillembrand L, Maharjan S, Mainardi VL, Zhang YS, Zarco N et al (2021) Circulatory shear stress induces molecular changes and side population enrichment in primary tumor-derived lung cancer cells with higher metastatic potential. Sci Rep 11:2800
Nagrath S, Sequist LV, Maheswaran S, Bell DW, Irimia D, Ulkus L et al (2007) Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature 450:1235–1239
Stott SL, Hsu CH, Tsukrov DI, Yu M, Miyamoto DT, Waltman BA et al (2010) Isolation of circulating tumor cells using a microvortex-generating herringbone-chip. Proc Natl Acad Sci U S A 107:18392–18397
Park MH, Reátegui E, Li W, Tessier SN, Wong KHK, Jensen AE et al (2017) Enhanced isolation and release of circulating tumor cells using nanoparticle binding and ligand exchange in a microfluidic Chip. J Am Chem Soc 139:2741–2749
Reátegui E, Aceto N, Lim EJ, Sullivan JP, Jensen AE, Zeinali M et al (2015) Tunable nanostructured coating for the capture and selective release of viable circulating tumor cells. Adv Mater 27:1593–1599
Rima XY, Walters N, Nguyen LTH, Reátegui E (2020) Surface engineering within a microchannel for hydrodynamic and self-assembled cell patterning. Biomicrofluidics 14:014104
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Rima, X.Y., Zhang, J., Reátegui, E. (2023). Capture and Selective Release of Viable Circulating Tumor Cells. In: Garcia-Cordero, J.L., Revzin, A. (eds) Microfluidic Systems for Cancer Diagnosis . Methods in Molecular Biology, vol 2679. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-3271-0_5
Download citation
DOI: https://doi.org/10.1007/978-1-0716-3271-0_5
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-3270-3
Online ISBN: 978-1-0716-3271-0
eBook Packages: Springer Protocols