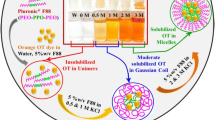
Abstract
The tremendous attention given to micelles in recent technological advancements and industries is due to its amazingly stable and flexible physicochemical properties exhibited upon exposure to different stimuli. A concise review of micelle structures and the effects of various stimuli on the structural properties of micelles with a particular focus on the effect of ultrasound are provided. While the use of conventional stimuli such as temperature, shear, etc., for controlling micelle structures is widely reported, the use of ultrasound as a stimulus has not been studied extensively. For this reason, a detailed discussion on the possibility of designing a variety of micelle nanostructures using ultrasound is provided. Using ultrasound as a stimulus is an advantage as it eliminates the need for adding external chemicals to the micellar system and the experimental parameters could be easily controlled. A case study of using cetyltrimethylammonium salicylate (CTASal) prepared from ion exchange process of equimolar mixture of cetyltrimethylammonium bromide (CTABr) and sodium salicylate (NaSal) is used in order to evaluate the efficiency of ultrasonics on controlling the micelles’ aggregational structures. Further experiments and discussion imply that the transformation is mainly driven by the physical effect generated from sonication. Evidence from cryo-TEM indicates that the structural transformation took place according to the reptation and reaction Model proposed before.
Similar content being viewed by others
References
Schryver SB et al (1913) Discussion. Trans Faraday Soc 9:93–107. doi:10.1039/TF9130900093
Croce V, Cosgrove T, Maitland G, Hughes T, Karlsson G (2003) Rheology, cryogenic transmission electron spectroscopy, and small-angle neutron scattering of highly viscoelastic wormlike micellar solutions. Langmuir 19:8536–8541. doi:10.1021/la0345800
Khatory A et al (1993) Entangled versus multiconnected network of wormlike micelles. Langmuir 9:933–939. doi:10.1021/la00028a010
Dreiss CA (2007) Wormlike micelles: where do we stand? Recent developments, linear rheology and scattering techniques. Soft Matter 3:956–970. doi:10.1039/B705775J
Guo L, Colby RH, Lin MY, Dado GP (2001) Micellar structure changes in aqueous mixtures of nonionic surfactants. J Rheol 45:1223–1243. doi:10.1122/1.1389315
Pérez-Juste J, Pastoriza-Santos I, Liz-Marzán LM, Mulvaney P (2005) Gold nanorods: synthesis, characterization and applications. Coord Chem Rev 249:1870–1901. doi:10.1016/j.ccr.2005.01.030
Vasudevan M, Shen A, Khomami B, Sureshkumar R (2008) Self-similar shear thickening behavior in CTAB/NaSal surfactant solutions. J Rheol 52:527–550. doi:10.1122/1.2833594
Yusof NSM, Khan MN (2010) Determination of an ion exchange constant by the use of a kinetic probe: a new semiempirical kinetic approach involving the effects of 3-F- and 4-F-substituted benzoates on the rate of piperidinolysis of anionic phenyl salicylate in aqueous cationic micelles. Langmuir 26:10627–10635. doi:10.1021/la100863q
Khan MN, Ismail E (2009) Kinetic evidence for the occurrence of independent ion-exchange processes in the cationic micellar-mediated reaction of piperidine with anionic phenyl salicylate. J Phys Chem A 113:6484–6488. doi:10.1021/jp902886z
Kim W-J, Yang S-M (2000) Effects of sodium salicylate on the microstructure of an aqueous micellar solution and its rheological responses. J Colloid Interface Sci 232:225–234. doi:10.1006/jcis.2000.7207
Magid LJ, Gee JC, Talmon Y (1990) A cryogenic transmission electron microscopy study of counterion effects on hexadecyltrimethylammonium dichlorobenzoate micelles. Langmuir 6:1609–1613. doi:10.1021/la00100a015
Arleth L, Bergström M, Pedersen JS (2002) Small-angle neutron scattering study of the growth behavior, flexibility, and intermicellar interactions of wormlike SDS micelles in NaBr aqueous solutions. Langmuir 18:5343–5353. doi:10.1021/la015693r
Magid LJ, Li Z, Butler PD (2000) Flexibility of elongated sodium dodecyl sulfate micelles in aqueous sodium chloride: a small-angle neutron scattering study. Langmuir 16:10028–10036. doi:10.1021/la0006216
Nakahara Y, Kida T, Nakatsuji Y, Akashi M (2005) New fluorescence method for the determination of the critical micelle concentration by photosensitive monoazacryptand derivatives. Langmuir 21:6688–6695. doi:10.1021/la050206j
Cates ME, Candau SJ (1990) Statics and dynamics of worm-like surfactant micelles. J Phys Condens Matter 2:6869
Cates ME (1990) Nonlinear viscoelasticity of wormlike micelles (and other reversibly breakable polymers). J Phys Chem 94:371–375. doi:10.1021/j100364a063
Magid LJ (1998) The surfactant−polyelectrolyte analogy. J Phys Chem B 102:4064–4074. doi:10.1021/jp9730961
Davies TS, Ketner AM, Raghavan SR (2006) Self-assembly of surfactant vesicles that transform into viscoelastic wormlike micelles upon heating. J Am Chem Soc 128:6669–6675. doi:10.1021/ja060021e
Koehler RD, Raghavan SR, Kaler EW (2000) Microstructure and dynamics of wormlike micellar solutions formed by mixing cationic and anionic surfactants. J Phys Chem B 104:11035–11044. doi:10.1021/jp0018899
Schubert BA, Kaler EW, Wagner NJ (2003) The microstructure and rheology of mixed cationic/anionic wormlike micelles. Langmuir 19:4079–4089. doi:10.1021/la020821c
Candau SJ, Hirsch E, Zana R (1985) Light scattering investigations of the behavior of semidilute aqueous micellar solutions of cetyltrimethylammonium bromide: analogy with semidilute polymer solutions. J Colloid Interface Sci 105:521–528. doi:10.1016/0021-9797(85)90327-3
Magid LJ, Han Z, Li Z, Butler PD (2000) Tuning microstructure of cationic micelles on multiple length scales: the role of electrostatics and specific ion binding. Langmuir 16:149–156. doi:10.1021/la990686c
Raghavan SR, Kaler EW (2000) Highly viscoelastic wormlike micellar solutions formed by cationic surfactants with long unsaturated tails. Langmuir 17:300–306. doi:10.1021/la0007933
Khatory A, Lequeux F, Kern F, Candau SJ (1993) Linear and nonlinear viscoelasticity of semidilute solutions of wormlike micelles at high salt content. Langmuir 9:1456–1464. doi:10.1021/la00030a005
Clausen TM et al (1992) Viscoelastic micellar solutions: microscopy and rheology. J Phys Chem 96:474–484. doi:10.1021/j100180a086
Gravsholt S (1976) Viscoelasticity in highly dilute aqueous solutions of pure cationic detergents. J Colloid Interface Sci 57:575–577. doi:10.1016/0021-9797(76)90236-8
Hassan PA et al (1996) Vesicle to micelle transition: rheological investigations. Langmuir 12:4350–4357
Mendes E et al (1997) Shear-induced vesicle to wormlike micelle transition. J Phys Chem B 101:2256–2258. doi:10.1021/jp962790y
Mahmood ME, Al-Koofee DAF (2013) Effect of temperature changes on critical micelle concentration for tween series surfactant. Glob J Sci Front Res Chem 13:1–7
Mendes E et al (1997) Shear-induced vesicle to wormlike micelle transition. J Phys Chem B 101:2256–2258
Prötzl B, Springer J (1997) Light scattering experiments on shear induced structures of micellar solutions. J Colloid Interface Sci 190:327–333. doi:10.1006/jcis.1997.4866
Chen S, Rothstein JP (2004) Flow of a wormlike micelle solution past a falling sphere. J Non-Newtonian Fluid Mech 116:205–234. doi:10.1016/j.jnnfm.2003.08.005
Cressely R, Hartmann V (1998) Rheological behaviour and shear thickening exhibited by aqueous CTAB micellar solutions. Eur Phys J B 6:57–62. doi:10.1007/s100510050526
Zheng Y et al (2000) Cryo-TEM imaging the flow-induced transition from vesicles to threadlike micelles. J Phys Chem B 104:5263–5271. doi:10.1021/jp0002998
Wang J, Pelletier M, Zhang H, Xia H, Zhao Y (2009) High-frequency ultrasound-responsive block copolymer micelle. Langmuir 25:13201–13205. doi:10.1021/la9018794
Pitt WG, Husseini GA, Staples BJ (2004) Ultrasonic drug delivery – a general review. Expert Opin Drug Deliv 1:37–56. doi:10.1517/17425247.1.1.37
Kost J, Leong K, Langer R (1989) Ultrasound-enhanced polymer degradation and release of incorporated substances. Proc Natl Acad Sci 86:7663–7666
Ghosh S et al (2013) Spontaneous transition of micelle–vesicle–micelle in a mixture of cationic surfactant and anionic surfactant-like ionic liquid: a pure nonlipid small unilamellar vesicular template used for solvent and rotational relaxation study. Langmuir 29:10066–10076. doi:10.1021/la402053a
Miskolczy Z, Sebők-Nagy K, Biczók L, Göktürk S (2004) Aggregation and micelle formation of ionic liquids in aqueous solution. Chem Phys Lett 400:296–300. doi:10.1016/j.cplett.2004.10.127
Silva BFB, Marques EF (2005) Thermotropic behavior of asymmetric chain length catanionic surfactants: the influence of the polar head group. J Colloid Interface Sci 290:267–274. doi:10.1016/j.jcis.2005.04.012
Kaler EW, Murthy AK, Rodriguez BE, Zasadzinski JA (1989) Spontaneous vesicle formation in aqueous mixtures of single-tailed surfactants. Science 245:1371–1374
Yatcilla MT et al (1996) Phase behavior of aqueous mixtures of cetyltrimethylammonium bromide (CTAB) and sodium octyl sulfate (SOS). J Phys Chem 100:5874–5879. doi:10.1021/jp952425r
Marques EF (2000) Size and stability of catanionic vesicles: effects of formation path, sonication, and aging. Langmuir 16:4798–4807. doi:10.1021/la9908135
Yusof NSM, Ashokkumar M (2013) Ultrasound-induced formation of high and low viscoelastic nanostructures of micelles. Soft Matter 9:1997–2002. doi:10.1039/C2SM27423J
Cates ME (1987) Reptation of living polymers: dynamics of entangled polymers in the presence of reversible chain-scission reactions. Macromolecules 20:2289–2296. doi:10.1021/ma00175a038
Messager R, Ott A, Chatenay D, Urbach W, Langevin D (1988) Are giant micelles living polymers? Phys Rev Lett 60:1410–1413
de Gennes PG (1971) Reptation of a polymer chain in the presence of fixed obstacles. J Chem Phys 55:572–579. doi:10.1063/1.1675789
Leger L, Hervet H, Rondelez F (1981) Reptation in entangled polymer solutions by forced Rayleigh light scattering. Macromolecules 14:1732–1738
Tanford C (1974) Theory of micelle formation in aqueous solutions. J Phys Chem 78:2469–2479. doi:10.1021/j100617a012
Turner MS, Marques C, Cates ME (1993) Dynamics of wormlike micelles: the “bond-interchange” reaction scheme. Langmuir 9:695–701. doi:10.1021/la00027a015
Candau SJ, Hirsch E, Zana R, Adam M (1988) Network properties of semidilute aqueous KBr solutions of cetyltrimethylammonium bromide. J Colloid Interface Sci 122:430–440. doi:10.1016/0021-9797(88)90377-3
Wunderlich I, Hoffmann H, Rehage H (1987) Flow birefringence and rheological measurements on shear induced micellar structures. Rheol Acta 26:532–542. doi:10.1007/BF01333737
Rehage H, Hoffmann H (1988) Rheological properties of viscoelastic surfactant systems. J Phys Chem 92:4712–4719. doi:10.1021/j100327a031
Leger L, Hervet H, Rondelez F (1981) Reptation in entangled polymer solutions by forced Rayleigh light scattering. Macromolecules 14:1732–1738. doi:10.1021/ma50007a023
Ashokkumar M, Mason TJ (2000) In: Kirk-Othmer encyclopedia of chemical technology. Wiley
Leighton T (1994) Academic Press, London
Tho P, Manasseh R, Ooi A (2007) Cavitation microstreaming patterns in single and multiple bubble systems. J Fluid Mech 576:191–233. doi:10.1017/S0022112006004393
Shoh A (1975) Industrial applications of ultrasound – a review I. High-power ultrasound. IEEE Trans Sonics Ultrason 22:60–70. doi:10.1109/T-SU.1975.30780
Patist A, Jha BK, Oh S g, Shah DO (1999) Importance of micellar relaxation time on detergent properties. J Surfactant Deterg 2:317–324. doi:10.1007/s11743-999-0083-6
Ziserman L, Abezgauz L, Ramon O, Raghavan SR, Danino D (2009) Origins of the viscosity peak in wormlike micellar solutions. 1. mixed catanionic surfactants. A cryo-transmission electron microscopy study. Langmuir 25:10483–10489. doi:10.1021/la901189k
Mohanty A, Patra T, Dey J (2007) Salt-induced vesicle to micelle transition in aqueous solution of sodium N-(4-n-octyloxybenzoyl)-l-valinate. J Phys Chem B 111:7155–7159. doi:10.1021/jp071312s
Brotchie A, Grieser F, Ashokkumar M (2009) Effect of power and frequency on bubble-size distributions in acoustic cavitation. Phys Rev Lett 102:084302
Ashokkumar M, Maspm TJ (2007) In: Kirk Othmer encyclopedia chemical tenchnology
Yusof NSM, Ashokkumar M (2015) Ultrasonic transformation of micelle structures: effect of frequency and power. Ultrason Sonochem. 24:8–12. doi:10.1016/j.ultsonch.2014.11.003
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media Singapore
About this entry
Cite this entry
Yusof, N.S.M., Ashokkumar, M. (2016). Ultrasonic Modification of Micelle Nanostructures. In: Ashokkumar, M. (eds) Handbook of Ultrasonics and Sonochemistry. Springer, Singapore. https://doi.org/10.1007/978-981-287-470-2_18-2
Download citation
DOI: https://doi.org/10.1007/978-981-287-470-2_18-2
Received:
Accepted:
Published:
Publisher Name: Springer, Singapore
Online ISBN: 978-981-287-470-2
eBook Packages: Springer Reference Chemistry and Mat. ScienceReference Module Physical and Materials ScienceReference Module Chemistry, Materials and Physics
Publish with us
Chapter history
-
Latest
Ultrasonic Modification of Micelle Nanostructures- Published:
- 12 April 2016
DOI: https://doi.org/10.1007/978-981-287-470-2_18-2
-
Original
Ultrasonic Modification of Micelle Structures- Published:
- 08 January 2016
DOI: https://doi.org/10.1007/978-981-287-470-2_18-1