Abstract
Splenic infection and inflammation are uncommon and both the clinical and imaging findings are most often nonspecific. It is during the acute phase in which radiological studies can be helpful in the detection of splenic lesions, as well as to help guide clinical treatment. More commonly, imaging features related to benign conditions are seen in their chronic state and are often clinically not significant. Because the imaging features are nonspecific with much overlap between differing pathologies, it is imperative to combine radiographic findings and clinical histories. This should include, but not be limited to fever, sepsis, immune status, prior trauma, prior transplant surgery, and chemotherapy. Geographic background and serologic testing may also aid in the diagnosis.
Access provided by Autonomous University of Puebla. Download reference work entry PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
Introduction
Splenic infection and inflammation are uncommon and both the clinical and imaging findings are most often nonspecific. It is during the acute phase in which radiological studies can be helpful in the detection of splenic lesions, as well as to help guide clinical treatment. More commonly, imaging features related to benign conditions are seen in their chronic state and are often clinically not significant. Because the imaging features are nonspecific with much overlap between differing pathologies, it is imperative to combine radiographic findings and clinical histories. This should include, but not be limited to fever, sepsis, immune status, prior trauma, prior transplant surgery, and chemotherapy. Geographic background and serologic testing may also aid in the diagnosis.
The differential diagnosis of a splenic focal lesion with malignant diseases such as metastatic disease and lymphoma is not problematic clinically and radiologically, as they are relatively rare and often manifest in multiple systems, usually in the later phase of the disease.
There are four distinct patterns of inflammation and infection of the spleen: splenomegaly without focal lesion, single lesion, diffuse multiple lesions, and multiple calcifications.
Splenomegaly Without Focal Lesion
Infectious or inflammatory causes of splenomegaly are varied and diagnosis often requires clinical information. The most common cause is mononucleosis, which is generally self-limited. Splenic rupture can be seen with even a minor contact injury (Fig. 97.1). The causes of massive splenomegaly are much fewer and include chronic malaria and Kala Azar. Others are listed in the Table 97.1.
Single Focal Lesion
Pyogenic Abscess
Hematogenous spread is the most common route of transmission, usually from endocarditis or other source of infection (Urrutia et al. 1996; Landaw and Schrier 2011). Other less common causes stem from penetrating trauma, direct infectious spread from adjacent organs, or prior splenic infarction (Urrutia et al. 1996). Clinical diagnosis can be difficult as the symptoms are often nonspecific, including fever and left upper quadrant abdominal pain, and can be absent in some patients (Thanos et al. 2002; Landaw and Schrier 2011). Thus, imaging can be very helpful in establishing early diagnosis in these patients.
Imaging features depend on the age of the abscess and follow a natural course from an inflammatory phlegmon to the gradual development of necrosis and liquefaction (Figs. 97.2, 97.3). Peripheral wall enhancement can be present (Rabushka et al. 1994). The presence of air bubbles is highly suggestive of an abscess, although uncommon (Rabushka et al. 1994) (Fig. 97.3).
Patient with history of prosthetic valve endocarditis by MRSA. Contrast-enhanced CT shows a large low attenuation lesion within the spleen, as well as a large irregular hepatic abscess with multiple smaller hepatic abscesses. The splenic abscess was eventually drained by ultrasound guided catheter placement
Patient with history of myleofibrosis and persistent thrombocytopenia who underwent selective embolization of the spleen. (a) Contrast-enhanced CT shows a large area of low attenuation in an enlarged spleen with multiple foci of air approximately 3 weeks after initial embolization. (b) Subsequent CECT shows a large necrotic cavity with a large air fluid level and layering necrotic tissue and liquefaction. Because the patient was septic, an abscess was suspected and confirmed by splenectomy
Surgical splenectomy and antibiotics constitute the mainstay of treatment. Percutaneous drainage is emerging as a safe and effective alternative mode of treatment (Thanos et al. 2002).
Hydatid Disease
Parasitic infection of the spleen is most commonly due to the pathogen Echinococcus granulosus and usually stems from disseminated spread or from a ruptured liver cyst (Ilica et al. 2007). It is not endemic in the United States, but immigration and international travel compel physicians everywhere to be more aware of its clinical features, diagnosis, and management.
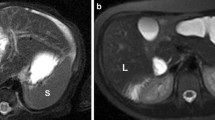
Typical imaging features are similar to echinococcal infestation of the liver, where there are well-defined cystic lesions with contents of fluid density and multiple daughter cysts (Fig. 97.4). A specific finding is partial separation of the endocyst from the pericyst, as manifested as a focal splitting of the wall, the so-called “floating membrane” sign. If there is complete dehiscence of the membrane, this is referred to as the “water lily” sign (Ilica et al. 2007). Total or peripheral rim of calcification may be present after the death of the parasite (Ilica et al. 2007; Franquet et al. 1990) (Fig. 97.5).
Pseudocyst
Pseudocysts lack a true epithelial wall and are commonly the result of prior trauma (Taylor et al. 1991). They can also result from previous infection or infarction (Freeman et al. 1993). Differentiation from a true congenital cyst on imaging can be difficult as they share the same imaging features (Rabushka et al. 1994). Typically, they are of fluid density with a well- circumscribed, thin, or imperceptible wall without enhancement of the internal contents (Rabushka et al. 1994) (Fig. 97.6). Internal septations are extremely rare and wall calcifications may be seen (Urrutia et al. 1996).
Patient with a history of lupus and immunosuppressants, who then developed spontaneous hemorrhage and subsequent pseudocyst. (a) CECT shows mild splenomegaly. (b) Subsequent CECT shows high-density hemorrhage adjacent to the spleen. (c) Approximately 3-month follow-up CECT shows a nonenhancing cystic mass
Diffuse Multiple Lesions
Fungal Infection
Candida is the most frequently encountered pathogen, followed by Aspergillus and Cryptococcus and is mainly seen in immunocompromised patients (Rabushka et al. 1994). AIDS patients more frequently exhibit extrathoracic manifestations of disseminated Pneumocystis carnii (now P. jiroveci) infection, rather than common fungal microorganisms (Freeman et al. 1993).
In general, fungal abscesses are often multiple with small, so-called microabscesses, usually 5–10 mm, and are associated with splenomegaly (Urrutia et al. 1996) (Figs. 97.7, 97.8). On CT, these are primarily of low attenuation (Freeman et al. 1993). On MR imaging, they are of fluid signal intensity with minimal enhancement of a developed capsule, if present (Elsayes et al. 2005).
Specifically, candidal infection can have a varied appearance on ultrasound. A common appearance is uniformly hypoechoic lesions. Another presentation is lesions with a “wheel within a wheel” appearance, which corresponds to alternating layers of differing echogenicities (Fig. 97.7). The outer layer is typically hypoechoic surrounding an inner layer of hyperechogenicity, which in turn surrounds a central area of hypoechogenicity. This appearance correlates with central necrosis surrounded by outer layers of inflammation and fibrosis, respectively. An alternative appearance includes a hyperechoic lesion surrounded by a peripheral rim that is hypoechoic and is thought to have evolved from a “wheel within a wheel” lesion (Pataki et al. 1988). In later stages, the abscesses become more uniformly hyperechoic and often calcify. On CT, these lesions are often well defined, of low attenuation, and without enhancement (Chew et al. 1991). MR typically shows these lesions to be hypointense with a peripheral rim of enhancement on gadolidium-enhanced sequences and is a better modality for detection (Elsayes et al. 2005).
Extrathoracic infection with P. jiroveci (recently classified as a fungus) usually involves the liver and spleen (Freeman et al. 1993). This manifests as multiple low attenuation lesions of varying sizes, which can then decrease in size and calcify in the later stages (Pantongrag-Brown et al. 1995) (Fig. 97.9). Pneumocystis lesions in the spleen are typically larger (often >2 cm) than typical fungal microabscesses and most other infectious splenic lesions in AIDS patients. (Radin 1995).
Tuberculosis
Tuberculosis is increasingly prevalent in developed countries due to HIV infection, increasing drug resistance, as well as immigration from endemic areas. Splenic involvement by tuberculosis is typically seen in immunocompromised patients who have the disseminated form of tuberculosis, more commonly, the miliary form (De Backer et al. 2006).
The disseminated miliary form is usually below the resolution of CT or ultrasound and can appear as splenomegaly without discrete lesions. However, if the lesions are discrete, they may appear on CT as innumerable, tiny low-density lesions scattered throughout the organ (Fig. 97.10.). MR imaging shows these lesions to have a variable appearance as well as varying enhancement characteristics depending on the stage of disease (De Backer et al. 2006). Associated findings such as necrotic lymphadenopathy as well as involvement of the liver can be helpful for differential diagnosis (Fig. 97.11).
Other bacterial species are known to cause multiple visceral lesions including the spleen. Glanders and pseudoglanders (melioidosis) both start with an acute respiratory syndrome but can subsequently present in the viscera after a variable latency (Srinivasan et al. 2001; Weller and Dickerson 1992). Brucellosis (undulant fever), predominantly a disease of barnyard animals, causes a non-caseating granulomatous infection of the human reticuloendothelial system sometimes complicated by multiple splenic lesions (Weller and Dickerson 1992). Calcified granulomas due to Brucellosis are described under the section of multiple splenic calcifications. The infectious agents of glanders, melioidosis, and undulant fever appear on the Centers for Disease Control list of potential weapons of bioterrorism.
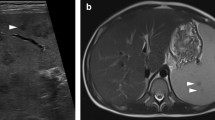
Cat scratch disease, or Bartonellosis, is a febrile illness that can be associated with splenomegaly. Usually mild in immunocompetent patients, it can sometimes cause visceral abscesses or microabscesses (Dunn et al. 1997) (Fig. 97.12). Finding multiple small splenic lesions in a teenaged cat-owner should prompt serologic evaluation. In immunocompromised patients, especially those with AIDS, the term bacillary angiomatosis identifies a serious cutaneous and systemic Bartonella infection that can include visceral lesions (Bass et al. 1997) (Fig. 97.13).
Sarcoidosis
Sarcoidosis is an inflammatory granulomatous disease of unknown etiology with a wide variety of systemic manifestations involving many organ systems. It typically occurs in middle-aged women and is more prevalent in African Americans. Diagnosis requires clinical and imaging features, as well as pathological evidence (Koyoma et al. 2004). Even if the liver is also involved, splenic involvement predominates. (Warshauer and Lee 2004).
On imaging, there may be organ enlargement and sometimes multiple small uniform low attenuating lesions in a diffuse distribution (Koyoma et al. 2004; Warshauer and Lee 2004). Although the spleen is histiologically involved in up to 50% of cases, focal lesions can be detected in only about 15%. The size of the lesions in the spleen can range from between 0.1 and 3.0 cm and typically do not enhance (Warshauer and Lee 2004) (Figs. 97.14, 97.15). Associated lymphadenopathy was seen in 76% of patients with hepatosplenic involvement in a single institution’s study (Warshauer and Lee 2004). The spleen in sarcoidosis can exhibit diffuse or multifocal hypermetabolic activity on PET, making the disease difficult to distinguish from lymphoma (Fig. 97.16).
Patient with history of sarcoidosis again showing multiple low attenuation lesions, which are larger than those shown in Fig. 97.14 (Image courtesy of Dr Peter F. Hahn, MGH, Boston, MA, USA)
Gamma Gandy Bodies
These are not inflammatory lesions, but siderotic nodules that are organized foci of hemorrhage, most often caused by portal hypertension. These manifest on MR as multiple small lesions that are low in signal intensity on T2 or gradient echo sequences due to the paramagnetic effects of hemosiderin (Katsuyoshi et al. 1997) (Fig. 97.17).
Patient with history of cirrhosis and portal hypertension. (a, b) MR GRE in-phase and out-of-phase sequences show innumerable low signal intensity lesions scattered throughout the spleen. Lesions are better seen on the longer TE image (a) because of the greater sensitivity to magnetic susceptibility
Multiple Splenic Calcifications
Splenic calcifications are the most commonly encountered splenic lesion and often are nonspecific. This is usually the result of prior granulomatous disease (histoplasmosis and tuberculosis) and other infections including P. jiroveci (Fig. 97.18). Disseminated P. jiroveci can result in few or diffuse microcalcifications to total organ calcification (Freeman et al. 1993). Prior traumatic or vascular insult may also be an alternative etiology (Taylor et al. 1991). In endemic areas such as the Mediterranean basin, multiple large calcified splenic granulomas readily visible on plain radiographs typically identify patients with prior Brucellosis infection (Fig. 97.19).
Other entities such as sickle cell disease can cause calcification, in which there is global splenic infarction over time and replacement of normal splenic tissue by fibrosis, calcium, and hemosiderin deposition (Longeran et al. 2001) (Fig. 97.20).
An unusual, recently reported cause of diffuse splenic microcalcification is systemic lupus erythematosis (Tieng et al. 2011) (Fig. 97.21). The exact pathophysiology is unknown.
Clinical Pearls
-
1.
Manifestation of splenic disease is often varied and requires clinical information to narrow the differential diagnosis.
-
2.
There are four distinct patterns of disease of the spleen which include splenomegaly without focal lesion, single lesion, diffuse multiple lesions, and multiple calcifications.
-
3.
A specific sign of hydatid disease is the “floating membrane” sign, where there is a focal split in the wall.
-
4.
Fungal microabscesses are often characterized by multiple small lesions (5–10 mm) and associated splenomegaly. The most common pathogen is Candida, followed by Aspergillus and Cryptococcus.
-
5.
Tuberculosis can appear with multiple tiny lesions and can be differentiated by its association with necrotic lymphadenopathy and involvement of the liver.
-
6.
In sarcoidosis, splenic lesions occur in a minority of patients. These are often uniform in size with primary involvement of the spleen.
-
7.
Gamma Gandy bodies are siderotic nodules primarily caused by portal hypertension that are low in signal intensity on T2 or gradient echo sequences because of their paramagnetic effects.
References
Bass JW, Vincent JM, Person DA. The expanding spectrum of Bartonella infections: II. Cat-Scratch disease. Pediatr Infect Dis J. 1997;16:163–79.
Chew FS, Smith PL, Barboriak D. Candidal splenic abscesses. AJR. 1991;156:474.
De Backer AI, Vanhoenacker FM, Mortele KJ, et al. MRI features of focal splenic lesion in patients with disseminated tuberculosis. AJR. 2006;186:1097–102.
Dunn MW, Berkowitz FE, Miller JJ, et al. Heptatosplenic Cat-Scratch disease and abdominal pain. Pediatr Infect Dis J. 1997;16:269–72.
Elsayes KM, Narre VR, Mukundan G, et al. MR imaging of the spleen: spectrum of abnormalities. Radiographics. 2005;25:967–82.
Franquet T, Montes M, Lecumberri FJ, et al. Hydatid disease of the spleen: imaging findings in nine patients. AJR. 1990;154:525–8.
Freeman JL, Jafri SZH, Roberts JL, et al. CT of congenital and acquired abnormalities of the spleen. Radiographics. 1993;13:597–610.
Ilica AT, Kocaoglu M, Zeybek N, et al. Extrahepatic abdominal hydatid disease caused by Echinococcus granulosus: imaging findings. AJR. 2007;189:337–43.
Katsuyoshi I, Mitchell DG, Honjo K, et al. MR imaging of acquired abnormalities of the spleen. AJR. 1997;168:697–702.
Koyoma T, Ueda H, Togas K, et al. Radiologic manifestations of sarcoidosis in various organs. Radiographics. 2004;24:87–104.
Landaw SA, Schrier SL, Approach to the adult patient with splenomegaly and other splenic disorders. In: UpToDate, Mentzer, WC (Ed), UpToDate, Waltham, MA, 2011.
Longeran GJ, Cline DB, Abbondanzo SL. From the archives of AFIP, sickle cell anemia. Radiographics. 2001;21:971–94.
Pantongrag-Brown L, Nelson AM, Brown AE, et al. From the archives of the AFIP, gastrointestinal manifestations of acquired immunodeficiency syndrome: radiologic-pathological correlation. Radiographics. 1995;15:1155–78.
Pataki B, Shaker TH, Thaler M, et al. Hepatosplenic candidacies: wheels within wheels. Radiology. 1988;166:417–21.
Rabushka LS, Kawashima A, Fishman EK. Imaging of the spleen: CT with supplemental MR examination. Radiographics. 1994;14:307–32.
Radin R. HIV infection: analysis in 259 consecutive patients with abnormal abdominal CT findings. Radiology. 1995;197:712–22.
Srinivasan A, Kraus CN, Dashier D, et al. Ganders in a military research microbiologist. N Engl J Med. 2001;345:256–8.
Taylor AJ, Dodds WJ, Erickson SJ, et al. CT of acquired abnormalities of the spleen. AJR. 1991;157:1213–9.
Thanos L, Dailiana T, Papaioannou G, Nikita A, et al. Percutaneous CT-guided drainage of splenic abscess. AJR. 2002;179:629–32.
Tieng AT, Sadow CA, Hochsztein JG, et al. Diffuse calcifications of the spleen: a novel association with systemic lupus erythematosus. Semin Arthritis Rheum. 2011;41(2):187–93. Epub 2010 Dec 23.
Urrutia M, Mergo PJ, Ros LH, et al. Cystic masses of the spleen: radiologic-pathologic correlation. Radiographics. 1996;16:107–29.
Warshauer DM, Lee JK. Imaging manifestations of abdominal sarcoidosis. AJR. 2004;182:15–28.
Weller PF, Dickerson GR. Case 40-1992 – a 43-year-old Cambodian man with several years of recurrent bouts of fever and abdominal pain. N Engl J Med. 1992;327:1081–7.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer-Verlag Berlin Heidelberg
About this entry
Cite this entry
Chun, J.Y., Kim, Y.H. (2013). Spleen Infectious and Inflammatory Disorders. In: Hamm, B., Ros, P.R. (eds) Abdominal Imaging. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-13327-5_132
Download citation
DOI: https://doi.org/10.1007/978-3-642-13327-5_132
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-13326-8
Online ISBN: 978-3-642-13327-5
eBook Packages: MedicineReference Module Medicine