Abstract
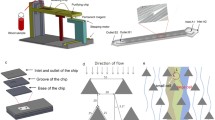
Circulating tumor cells (CTCs) are an important biomarker and their analysis can be considered a form of “liquid biopsy.” The purpose of this book chapter is to describe the use of the 4-channel CMx (cells captured in maximum) microfluidic chip, containing special micropatterns coated with an antibody-conjugated supported lipid bilayer (SLB) on its surface, to capture and isolate CTCs from the blood of cancer patients. Captured CTCs are subsequently released by an air foam to an immunofluorescence (IF) staining panel that enables further analysis, including the identification of the primary cancer source of the CTCs.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65(2):87–108
Siegel RL, Miller KD, Jemal A (2015) Cancer statistics, 2015. CA Cancer J Clin 65(1):5–29
Yap TA, Lorente D, Omlin A, Olmos D, de Bono JS (2014) Circulating tumor cells: a multifunctional biomarker. Clin Cancer Res 20(10):2553–2568
Green BJ, Saberi Safaei T, Mepham A, Labib M, Mohamadi RM, Kelley SO (2015) Beyond the capture of circulating tumor cells: next-generation devices and materials. Angew Chem Int Ed 55(4):1252–1265
Chen J-Y, Tsai W-S, Shao H-J, Wu J-C, Lai J-M, Lu S-H et al (2016) Sensitive and specific biomimetic lipid coated microfluidics to isolate viable circulating tumor cells and microemboli for cancer detection. PLoS One 11(3):e0149633
Tsai W-S, Chen J-S, Shao H-J, Wu J-C, Lai J-M, Lu S-H et al (2016) Circulating tumor cell count correlates with colorectal neoplasm progression and is a prognostic marker for distant metastasis in non-metastatic patients. Sci Rep 6:24517
Chang M-C, Chang Y-T, Chen J-Y, Jeng Y-M, Yang C-Y, Tien Y-W et al (2016) Clinical significance of circulating tumor microemboli as a prognostic marker in patients with pancreatic ductal adenocarcinoma. Clin Chem 62(3):505–513
Hou J-M, Krebs MG, Lancashire L, Sloane R, Backen A, Swain RK et al (2012) Clinical significance and molecular characteristics of circulating tumor cells and circulating tumor microemboli in patients with small-cell lung cancer. J Clin Oncol 30(5):525–532
Tien YW, Kuo HC, Ho BI, Chang MC, Chang YT, Cheng MF et al (2016) A high circulating tumor cell count in portal vein predicts liver metastasis from periampullary or pancreatic cancer: a high portal venous CTC count predicts liver metastases. Medicine (Baltimore) 95(16):e3407
Lu S-H, Tsai W-S, Chang Y-H, Chou T-Y, Pang S-T, Lin P-H et al (2016) Identifying cancer origin using circulating tumor cells. Cancer Biol Ther 17(4):430–438
Wu J-C, Tseng P-Y, Tsai W-S, Liao M-Y, Lu S-H, Frank CW et al (2013) Antibody conjugated supported lipid bilayer for capturing and purification of viable tumor cells in blood for subsequent cell culture. Biomaterials 34(21):5191–5199
Tseng PY, Chang YC (2012) Tethered fibronectin liposomes on supported lipid bilayers as a prepackaged controlled-release platform for cell-based assays. Biomacromolecules 13(8):2254–2262
Huang C-J, Tseng P-Y, Chang Y-C (2010) Effects of extracellular matrix protein functionalized fluid membrane on cell adhesion and matrix remodeling. Biomaterials 31(27):7183–7195
Liao M-Y, Lai J-K, Kuo MY-P, Lu R-M, Lin C-W, Cheng P-C et al (2015) An anti-EpCAM antibody EpAb2-6 for the treatment of colon cancer. Oncotarget 6:24947–24968
Chang YC, Wu HC, Tseng PY (2014) Capture, purification, and release of biological substances using a surface coating. US Patent and Trademark Office
Lai J-M, Shao H-J, Wu J-C, Lu S-H, Chang Y-C (2014) Efficient elusion of viable adhesive cells from a microfluidic system by air foam. Biomicrofluidics 8(5):052001
Lai C-H, Lim SC, Wu L-C, Wang C-F, Tsai W-S, Wu H-C, Chang Y-C (2017) Site-specific antibody modification and immobilization on a microfluidic chip to promote the capture of circulating tumor cells and microemboli. Chem Commun 53(29):4152–4155
Huh D, Matthews BD, Mammoto A, Montoya-Zavala M, Hsin HY, Ingber DE (2010) Reconstituting organ-level lung functions on a chip. Science 328(5986):1662–1668
Abe M, Havre PA, Urasaki Y, Ohnuma K, Morimoto C, Dang LH et al (2011) Mechanisms of confluence-dependent expression of CD26 in colon cancer cell lines. BMC Cancer 11(1):1–10
Chan CWM, Wong NA, Liu Y, Bicknell D, Turley H, Hollins L et al (2009) Gastrointestinal differentiation marker cytokeratin 20 is regulated by homeobox gene CDX1. Proc Natl Acad Sci U S A 106(6):1936–1941
Acknowledgment
This work was supported by Genomics Research Center, Academia Sinica, Taiwan and Ministry of Science and Technology of Taiwan under contract 105-0210-01-13-01, MOST-104-2113-M-001-015-MY3, MOST 105-2113-M-037-MY2, AS-105-TP-A04. We thank Katherine Yih Ruey Chen for proof reading.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Lai, CH., Chang, YC. (2017). Microfluidic Capture and Multiplex Immunofluorescence of Circulating Tumor Cells to Identify Cancer of Origin. In: M. Magbanua, M., W. Park, J. (eds) Circulating Tumor Cells. Methods in Molecular Biology, vol 1634. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7144-2_1
Download citation
DOI: https://doi.org/10.1007/978-1-4939-7144-2_1
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-7143-5
Online ISBN: 978-1-4939-7144-2
eBook Packages: Springer Protocols



