Abstract
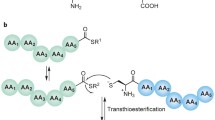
Site-specific isopeptide linkages between the ε-amino group of a lysine residue in one protein and a carboxyl group in another are central to ubiquitin-mediated protein degradation and other cellular processes. These linkages are inaccessible with common recombinant DNA techniques. Here, we describe a method to link two proteins by an authentic isopeptide bond. The method unites three techniques at the forefront of molecular biology. An azidonorleucine residue is installed at a desired site in a substrate protein by nonnatural amino acid incorporation, and a phosphinothioester is installed at the C terminus of a pendant protein by expressed protein ligation. Then, the traceless Staudinger ligation is used to link the substrate and pendant proteins via an isopeptide bond. This method facilitates the study of otherwise intractable protein structure–function relationships.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ciechanover A, Iwai K (2004) The ubiquitin system: from basic mechanisms to the patient bed. IUBMB Life 56:193–201
Li W, Ye Y (2008) Polyubiquitin chains: functions, structures, and mechanisms. Cell Mol Life Sci 65:2397–2406
Komander D (2009) The emerging complexity of protein ubiquitination. Biochem Soc Trans 37:937–953
Griffin M, Casadio R, Bergamini CM (2002) Transglutaminases: nature’s biological glues. Biochem J 368:377–396
Gentile V, Thomazy V, Piacentini M, Fesus L, Davies PJ (1992) Expression of tissue transglutaminase in Balb-C 3T3 fibroblasts: effects on cellular morphology and adhesion. J Cell Biol 119:463–474
Jones RA, Nicholas B, Mian S, Davies PJ, Griffin M (1997) Reduced expression of tissue transglutaminase in a human endothelial cell line leads to changes in cell spreading, cell adhesion and reduced polymerisation of fibronectin. J Cell Sci 110:2461–2472
Greenberg CS, Birckbichler PJ, Rice RH (1991) Transglutaminases: multifunctional cross-linking enzymes that stabilize tissues. FASEB J 5:3071–3077
Ariëns RAS, Lai T-S, Weisel JW, Greenberg CS, Grant PJ (2002) Role of factor XIII in fibrin clot formation and effects of genetic polymorphisms. Blood 100:743–754
El Oualid F, Merkx R, Ekkebus R, Hameed DS, Smit JJ, de Jong A, Hilkmann H, Sixma TK, Ovaa H (2010) Chemical synthesis of ubiquitin, ubiquitin-based probes, and diubiquitin. Angew Chem Int Ed 49:10149–10153
Eger S, Scheffner M, Marx A, Rubini M (2010) Synthesis of defined ubiquitin dimers. J Am Chem Soc 132:16337–16339
Bavikar SN, Spasser L, Haj-Yahya M, Karthikeyan SV, Moyal T, Ajish Kumar KS, Brik A (2011) Chemical synthesis of ubiquitinated peptides with varying lengths and types of ubiquitin chains to explore the activity of deubiquitinases. Angew Chem Int Ed 50:758–763
Virdee S, Kapadnis PB, Elliott T, Lang K, Madrzak J, Nguyen DP, Riechmann L, Chin JW (2011) Traceless and site-specific ubiquitination of recombinant proteins. J Am Chem Soc 133:10708–10711
Castañeda C, Liu J, Chaturvedi A, Nowicka U, Cropp TA, Fushman D (2011) Nonenzymatic assembly of natural polyubiquitin chains of any linkage composition and isotopic labeling scheme. J Am Chem Soc 133:17855–17868
Weikart ND, Sommer S, Mootz HD (2012) Click synthesis of ubiquitin dimer analogs to interrogate linkage-specific UBA domain binding. Chem Commun 48:296–298
Valkevich EM, Guenette RG, Sanchez NA, Chen Y-c, Ge Y, Strieter ER (2012) Forging isopeptide bonds using thiol–ene chemistry: site-specific coupling of ubiquitin molecules for studying the activity of isopeptidases. J Am Chem Soc 134:6916–6919
Martin LJ, Raines RT (2010) Carpe diubiquitin. Angew Chem Int Ed 49:9042–9044
Nilsson BL, Kiessling LL, Raines RT (2000) Staudinger ligation: a peptide from a thioester and azide. Org Lett 2:1939–1941
Saxon E, Armstrong JI, Bertozzi CR (2000) A “traceless” Staudinger ligation for the chemoselective synthesis of amide bonds. Org Lett 2:2141–2143
Pickart CM, Cohen RE (2004) Proteasomes and their kin: proteases in the machine age. Nat Rev Mol Cell Biol 5:177–187
Nilsson BL, Soellner MB, Raines RT (2005) Chemical synthesis of proteins. Annu Rev Biophys Biomol Struct 34:91–118
Tam A, Raines RT (2009) Protein engineering with the traceless Staudinger ligation. Methods Enzymol 462:25–44
Schilling CI, Jung N, Biskup M, Schepers U, Bräse S (2011) Bioconjugation via azide-Staudinger ligation: an overview. Angew Chem Int Ed 40:4840–4871
Soellner MB, Nilsson BL, Raines RT (2006) Reaction mechanism and kinetics of the traceless Staudinger ligation. J Am Chem Soc 128:8820–8828
Soellner MB, Nilsson BL, Raines RT (2002) Staudinger ligation of α-azido acids retains stereochemistry. J Org Chem 67:4993–4996
Nilsson BL, Hondal RJ, Soellner MB, Raines RT (2003) Protein assembly by orthogonal chemical ligation methods. J Am Chem Soc 125:5268–5269
Tam A, Soellner MB, Raines RT (2007) Water-soluble phosphinothiols for traceless Staudinger ligation and integration with expressed protein ligation. J Am Chem Soc 129:11421–11430
Tanrikulu IC, Schmitt E, Mechulam Y, Goddard WA, Tirrell DA (2009) Discovery of Escherichia coli methionyl-tRNA synthetase mutants for efficient labeling of proteins with azidonorleucine in vivo. Proc Natl Acad Sci U S A 106:15285–15290
Muir TW, Sondhi D, Cole PA (1998) Expressed protein ligation: a general method for protein engineering. Proc Natl Acad Sci U S A 95:6705–6710
Tam A, Raines RT (2009) Coulombic effects on the traceless Staudinger ligation in water. Bioorg Med Chem 17:1055–1063
Link AJ, Vink MKS, Tirrell DA (2004) Presentation and detection of azide functionality in bacterial cell surface proteins. J Am Chem Soc 126:10598–10602
McGrath NA, Raines RT (2011) Chemoselectivity in chemical biology: acyl transfer reactions with sulfur and selenium. Acc Chem Res 44:752–761
Acknowledgements
We are grateful to Drs. L. J. Martin and I. C. Tanrikulu for contributive discussions. This work was supported by Grant R01 GM044783 (NIH). K.A.A. was supported by the Molecular and Cellular Pharmacology Training Grant T32 GM008688 (NIH) and a predoctoral fellowship from the PhRMA Foundation.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this protocol
Cite this protocol
Andersen, K.A., Raines, R.T. (2015). Creating Site-Specific Isopeptide Linkages Between Proteins with the Traceless Staudinger Ligation. In: Derda, R. (eds) Peptide Libraries. Methods in Molecular Biology, vol 1248. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-2020-4_4
Download citation
DOI: https://doi.org/10.1007/978-1-4939-2020-4_4
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-2019-8
Online ISBN: 978-1-4939-2020-4
eBook Packages: Springer Protocols