Abstract
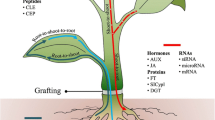
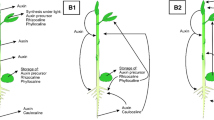
The plant Catharanthus roseus is well known for its spatial separation of iridoid and monoterpenoid indole alkaloid (MIA) biosynthesis at both intracellular and intercellular levels, collectively suggested by RNA in situ hybridization, enzymatic and transcriptomic studies using leaf epidermis, and fluorescent protein tagging studies. Although documented in other plant species, the long-distance transport of iridoid glycosides, such as secologanin, has not been known in C. roseus until a recent study suggested that secologanin is transported from root to shoot, by grafting low iridoid/MIA mutant scions onto wild-type stock plants. This chapter describes the in vitro cultivation of C. roseus plants and grafting techniques to enable studies concerning iridoid/MIA transport between organs. The iridoid and MIA analysis methods are also provided.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Morita M, Shitan N, Sawada K, Van Montagu MCE, Inzé D, Rischer H, Goossens A, Oksman-Caldentey KM, Moriyama Y, Yazaki K (2009) Vacuolar transport of nicotine is mediated by a multidrug and toxic compound extrusion (MATE) transporter in Nicotiana tabacum. Proc Natl Acad Sci U S A 106:2447–2452. https://doi.org/10.1073/pnas.0812512106
Courdavault V, Papon N, Clastre M, Giglioli-Guivarc’h N, St-Pierre B, Burlat V (2014) A look inside an alkaloid multisite plant: the Catharanthus logistics. Curr Opin Plant Biol 19:43–50. https://doi.org/10.1016/j.pbi.2014.03.010
St-Pierre B, Vazquez-Flota F, De Luca V (1999) Multicellular compartmentation of Catharanthus roseus alkaloid biosynthesis predicts intercellular translocation of a pathway intermediate. Plant Cell 11(5):887–900. https://doi.org/10.1105/tpc.11.5.887
Levac D, Murata J, Kim WS, De Luca V (2008) Application of carborundum abrasion for investigating the leaf epidermis: molecular cloning of Catharanthus roseus 16-hydroxytabersonine-16-O-methyltransferase. Plant J 53(2):225–236. https://doi.org/10.1111/j.1365-313X.2007.03337.x
Murata J, De Luca V (2005) Localization of tabersonine 16-hydroxylase and 16-OH tabersonine-16-O-methyltransferase to leaf epidermal cells defines them as a major site of precursor biosynthesis in the vindoline pathway in Catharanthus roseus. Plant J 44(4):581–594. https://doi.org/10.1111/j.1365-313X.2005.02557.x
Murata J, Roepke J, Gordon H, De Luca V (2008) The leaf epidermome of Catharanthus roseus reveals its biochemical specialization. Plant Cell 20(3):524–542. https://doi.org/10.1105/tpc.107.056630
Payne RME, Xu D, Foureau E, Carqueijeiro MIST, Oudin A, de Bernonville TD, Novak V, Burow M, Olsen CE, Jones DM, Tatsis EC (2017) An NPF transporter exports a central monoterpene indole alkaloid intermediate from the vacuole. Nat Plants 3(2):1–9. https://doi.org/10.1038/nplants.2016.208
Stavrinides A, Tatsis EC, Caputi L, Foureau E, Stevenson CEM, Lawson DM, Courdavault V, O’Connor SE (2016) Structural investigation of heteroyohimbine alkaloid synthesis reveals active site elements that control stereoselectivity. Nat Commun 7(1):1–14. https://doi.org/10.1038/ncomms12116
Guirimand G, Courdavault V, Lanoue A, Mahroug S, Guihur A, Blanc N, Giglioli-Guivarc’h N, St-Pierre B, Burlat V (2010) Strictosidine activation in Apocynaceae: towards a “nuclear time bomb”? BMC Plant Biol 10(1):1–20. https://doi.org/10.1186/1471-2229-10-182
Qu Y, Easson MLAE, Froese J, Simionescu R, Hudlicky T, De Luca V (2015) Completion of the seven-step pathway from tabersonine to the anticancer drug precursor vindoline and its assembly in yeast. Proc Natl Acad Sci U S A 112(19):6224–6229. https://doi.org/10.1073/pnas.1501821112
Yu F, De Luca V (2013) ATP-binding cassette transporter controls leaf surface secretion of anticancer drug components in Catharanthus roseus. Proc Natl Acad Sci U S A 110(39):15830–15835. https://doi.org/10.1073/pnas.1307504110
Abouzeid S, Hijazin T, Lewerenz L, Hänsch R, Selmar D (2019) The genuine localization of indole alkaloids in Vinca minor and Catharanthus roseus. Phytochemistry 168:112110. https://doi.org/10.1016/j.phytochem.2019.112110
Yamamoto K, Takahashi K, Caputi L, Mizuno H, Rodriguez-Lopez CE, Iwasaki T, Ishizaki K, Fukaki H, Ohnishi M, Yamazaki M, Masujima T (2019) The complexity of intercellular localisation of alkaloids revealed by single-cell metabolomics. New Phytol 224(2):848–859. https://doi.org/10.1111/nph.16138
Larsen B, Fuller VL, Pollier J, Van Moerkercke A, Schweizer F, Payne R, Colinas M, O’Connor SE, Goossens A, Halkier BA (2017) Identification of iridoid glucoside transporters in Catharanthus roseus. Plant Cell Physiol 58(9):1507–1518. https://doi.org/10.1093/pcp/pcx097
Kidd T, Easson MLAE, Qu Y, De Luca V (2019) Inter-organ transport of secologanin allows assembly of monoterpenoid indole alkaloids in a Catharanthus roseus mutant. Phytochemistry 159:119–126. https://doi.org/10.1016/j.phytochem.2018.12.017
Gowan E, Lewis BA, Turgeon R (1995) Phloem transport of antirrhinoside, an iridoid glycoside, in Asarina scandens (Scrophulariaceae). J Chem Ecol 21(11):1781–1788. https://doi.org/10.1007/BF02033676
Beninger CW, Cloutier RR, Monteiro MA, Grodzinski B (2007) The distribution of two major Iridoids in different organs of Antirrhinum majus L. at selected stages of development. J Chem Ecol 33(4):731–747. https://doi.org/10.1007/s10886-007-9253-x
Van Moerkercke A, Steensma P, Schweizer F, Pollier J, Gariboldi I, Payne R, Bossche RV, Miettinen K, Espoz J, Purnama PC, Kellner F (2015) The bHLH transcription factor BIS1 controls the iridoid branch of the monoterpenoid indole alkaloid pathway in Catharanthus roseus. Proc Natl Acad Sci U S A 112(26):8130–8135. https://doi.org/10.1073/pnas.1504951112
Van Moerkercke A, Steensma P, Gariboldi I, Espoz J, Purnama PC, Schweizer F, Miettinen K, Vanden Bossche R, De Clercq R, Memelink J, Goossens A (2016) The basic helix-loop-helix transcription factor BIS2 is essential for monoterpenoid indole alkaloid production in the medicinal plant Catharanthus roseus. Plant J 88(1):3–12. https://doi.org/10.1111/tpj.13230
Acknowledgment
This work was supported by an NSERC Discovery Grant to YQ and an NSERC Discovery Grant to VDL. We thank Dr. Alan Blowers and colleagues from Ball Horticultural Company for preparing the M2-mutant seeds. YQ is supported by a Cannabis Health Research Chair (New Brunswick Health Research Foundation/Tetra Bio Pharma Inc.). VDL is supported by an NSERC Tier 1 Canada Research Chair in Plant Biotechnology.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Farzana, M., Shahsavarani, M., De Luca, V., Qu, Y. (2022). Studying Iridoid Transport in Catharanthus roseus by Grafting. In: Courdavault, V., Besseau, S. (eds) Catharanthus roseus. Methods in Molecular Biology, vol 2505. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2349-7_5
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2349-7_5
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2348-0
Online ISBN: 978-1-0716-2349-7
eBook Packages: Springer Protocols