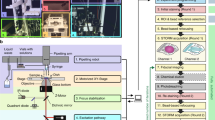
Abstract
Stimulated emission depletion (STED) microscopy is one of the optical superresolution microscopy (SRM) techniques, more recently also referred to as nanoscopy, that have risen to popularity among biologists during the past decade. These techniques keep pushing the physical boundaries of optical resolution toward the molecular scale. Thereby, they enable biologists to image cellular and tissue structures at a level of almost molecular detail that was previously only achievable using electron microscopy. All the while, they retain the advantages of light microscopy, in particular with regards to sample preparation and flexibility of imaging. Commercially available SRM setups have become more and more available and also increasingly sophisticated, both in terms of optical performance and, importantly, ease of use. Institutional microscopy core facilities now offer widespread access to this type of systems. However, the field has grown so rapidly, and keeps growing, that biologists can be easily overwhelmed by the multitude of available techniques and approaches. From this vast array of SRM modalities, STED stands out in one respect: it is essentially an extension to an advanced confocal microscope. Most experienced users of confocal microscopy will find the transition to STED microscopy relatively easy as compared with some other SRM techniques. This also applies to STED sample preparation. Nonetheless, because resolution in STED microscopy does not only depend on the wavelength of the incident light and the numerical aperture of the objective, but crucially also on the square root of the intensity of the depletion laser and, in general, on the photochemical interaction of the fluorophore with the depletion laser, some additional considerations are necessary in STED sample preparation. Here we describe the single color staining of the somatostatin receptor subtype 2A (SSTR2A) and dual color staining of the trans-Golgi-network protein TGN 38 and the t-SNARE syntaxin-6 for STED in the endocrine cell line AtT20 and STED imaging of the samples, providing the protocols in as general a form as possible. The protocols in this chapter are used in this way in an institutional microscopy core facility.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- BSA:
-
Bovine serum albumin
- DMEM:
-
Dulbecco’s Modified Eagle’s Medium
- FBS:
-
Fetal bovine serum
- NA:
-
Numerical aperture
- NGS:
-
Normal goat serum
- PFA:
-
Paraformaldehyde solution
- PBS:
-
Phosphate-buffered saline
- PSF:
-
Point spread function
- RI:
-
Refractive index
- RT:
-
Room temperature
- SNR :
-
Signal to noise ratio
- SRM:
-
Super-resolution microscopy
- SSTR2A:
-
Somatostatin receptor subtype 2A (mouse)
- STED:
-
Stimulated emission depletion
- TGN38:
-
A type 1 transmembrane protein located in the trans-Golgi network
References
Abbe E (1873) Beiträge zur Theorie des Mikroskops und der mikroskopischen Wahrnehmung. Arch Mikrosk Anat 9:413–468
Abbe E (1884) Note on the proper definition of the amplifying power of a lens or a lens-system. J R Microsc Soc 4:348–351
Inoué S (2006) Handbook of biological confocal microscopy. Springer, Boston
Hell SW, Wichmann J (1994) Breaking the diffraction resolution limit by stimulated emission: stimulated-emission-depletion fluorescence microscopy. Opt Lett 19(11):780–782
Westphal V, Hell SW (2005) Nanoscale resolution in the focal plane of an optical microscope. Phys Rev Lett 94(14):143903
Han KY et al (2009) Three-dimensional stimulated emission depletion microscopy of nitrogen-vacancy centers in diamond using continuous-wave light. Nano Lett 9(9):3323–3329
Willig KI et al (2007) STED microscopy with continuous wave beams. Nat Methods 4(11):915–918
Hama H et al (2011) Scale: a chemical approach for fluorescence imaging and reconstruction of transparent mouse brain. Nat Neurosci 14(11):1481–1488
Birk, U.J., Super-resolution microscopy.. Wiley VCH Verlag GmbH, 2017
Laflamme C et al (2019) Implementation of an antibody characterization procedure and application to the major ALS/FTD disease gene C9ORF72. elife 8
Vicidomini G et al (2011) Sharper low-power STED nanoscopy by time gating. Nat Methods 8(7):571–573
Bianchini P et al (2015) STED nanoscopy: a glimpse into the future. Cell Tissue Res 360(1):143–150
Chéreau R, Tønnesen J, Nägerl UV (2015) STED microscopy for nanoscale imaging in living brain slices. Methods 88:57–66
Tønnesen J et al (2011) Two-color STED microscopy of living synapses using a single laser-beam pair. Biophys J 101(10):2545–2552
Moneron G et al (2010) Fast STED microscopy with continuous wave fiber lasers. Opt Express 18(2):1302–1309
Deng S et al (2018) Effects of donor and acceptor's fluorescence lifetimes on the method of applying Forster resonance energy transfer in STED microscopy. J Microsc 269(1):59–65
Oracz J et al (2017) Photobleaching in STED nanoscopy and its dependence on the photon flux applied for reversible silencing of the fluorophore. Sci Rep 7(1):11354
Ding JB, Takasaki KT, Sabatini BL (2009) Supraresolution imaging in brain slices using stimulated-emission depletion two-photon laser scanning microscopy. Neuron 63(4):429–437
Wildanger D et al (2009) A compact STED microscope providing 3D nanoscale resolution. J Microsc 236(1):35–43
Göttfert F et al (2013) Coaligned dual-channel STED nanoscopy and molecular diffusion analysis at 20 nm resolution. Biophys J 105(1):L01–L03
Wurm CA et al (2012) Novel red fluorophores with superior performance in STED microscopy. Optical Nanoscopy 1(1):1–7
Fendl S et al (2017) STED imaging in drosophila brain slices. Methods Mol Biol 1563:143–150
Tam J, Merino D (2015) Stochastic optical reconstruction microscopy (STORM) in comparison with stimulated emission depletion (STED) and other imaging methods. J Neurochem 135(4):643–658
Griesbeck O et al (2001) Reducing the environmental sensitivity of yellow fluorescent protein. Mechanism and applications. J Biol Chem 276(31):29188–29194
Hein B, Willig KI, Hell SW (2008) Stimulated emission depletion (STED) nanoscopy of a fluorescent protein-labeled organelle inside a living cell. Proc Natl Acad Sci U S A 105(38):14271–14276
Wegner W et al (2017) In vivo mouse and live cell STED microscopy of neuronal actin plasticity using far-red emitting fluorescent proteins. Sci Rep 7(1):11781
Wang C et al (2019) A photostable fluorescent marker for the superresolution live imaging of the dynamic structure of the mitochondrial cristae. Proc Natl Acad Sci U S A 116(32):15817–15822
Bottanelli F et al (2016) Two-colour live-cell nanoscale imaging of intracellular targets. Nat Commun 7(1):1–5
Lukinavicius G et al (2013) A near-infrared fluorophore for live-cell super-resolution microscopy of cellular proteins. Nat Chem 5(2):132–139
Erdmann RS, Toomre D, Schepartz A (2017) STED imaging of Golgi dynamics with Cer-SiR: a two-component, photostable, high-density lipid probe for live cells. Methods Mol Biol 1663:65–78
Vicidomini G, Bianchini P, Diaspro A (2018) STED super-resolved microscopy. Nat Methods 15(3):173–182
Spahn C et al (2019) Whole-cell, 3D, and multicolor STED imaging with exchangeable fluorophores. Nano Lett 19(1):500–505
Acknowledgments
This work was supported by Discovery Grant RGPIN/06096-2016 from the Natural Sciences and Engineering Research Council of Canada (NSERC) and an operating grant to improve and develop super-resolution microscopy at the Montreal Neurological Institute by the Bachynski Family Foundation to T.S. W.A. was the holder of a Jeanne Timmins Costello Fellowship from the Montreal Neurological Institute. We are deeply thankful to Naomi Takeda for administrative support.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Alshafie, W., Stroh, T. (2022). Sample Preparation for Multicolor STED Microscopy. In: Heit, B. (eds) Fluorescent Microscopy. Methods in Molecular Biology, vol 2440. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2051-9_15
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2051-9_15
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2050-2
Online ISBN: 978-1-0716-2051-9
eBook Packages: Springer Protocols