Abstract
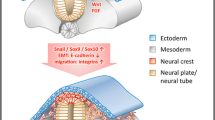
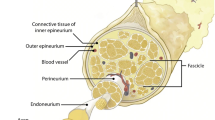
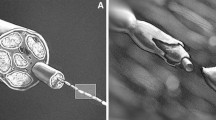
Peripheral nerves have a limited ability to regenerate and current clinical approaches involving microsurgery give suboptimal recovery. Engineered tissues using aligned cellular collagen hydrogels can be used as in vitro models through the incorporation of human Schwann cells. However, primary human Schwann cells are difficult to obtain and can be challenging to culture. The ability to generate Schwann cells from human-induced pluripotent stem cells (hiPSCs) provides a more reliable cell source for modeling peripheral nerve tissue. Here, we describe protocols for generating hiPSC-derived Schwann cells and incorporating them into 3D engineered tissue culture models for peripheral nerve research.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Huang CW, Huang WC, Qiu X et al (2017) The differentiation stage of transplanted stem cells modulates nerve regeneration. Sci Rep 7(1):17401. https://doi.org/10.1038/s41598-017-17043-4
Kim HS, Lee J, Lee DY et al (2017) Schwann cell precursors from human pluripotent stem cells as a potential therapeutic target for myelin repair. Stem Cell Rep 8(6):1714–1726. https://doi.org/10.1016/j.stemcr.2017.04.011
Phillips JB, Brown R (2011) Micro-structured materials and mechanical cues in 3D collagen gels. Methods Mol Biol 695:183–196. https://doi.org/10.1007/978-1-60761-984-0_12
O’Rourke C, Lee-Reeves C, Drake RA et al (2017) Adapting tissue-engineered in vitro CNS models for high-throughput study of neurodegeneration. J Tissue Eng 8:2041731417697920. https://doi.org/10.1177/2041731417697920
Georgiou M, Bunting SC, Davies HA et al (2013) Engineered neural tissue for peripheral nerve repair. Biomaterials 34(30):7335–7343. https://doi.org/10.1016/j.biomaterials.2013.06.025
Georgiou M, Golding JP, Loughlin AJ et al (2015) Engineered neural tissue with aligned, differentiated adipose-derived stem cells promotes peripheral nerve regeneration across a critical sized defect in rat sciatic nerve. Biomaterials 37:242–251. https://doi.org/10.1016/j.biomaterials.2014.10.009
Gonzalez-Perez F, Hernandez J, Heimann C et al (2018) Schwann cells and mesenchymal stem cells in laminin- or fibronectin-aligned matrices and regeneration across a critical size defect of 15 mm in the rat sciatic nerve. J Neurosurg Spine 28(1):109–118. https://doi.org/10.3171/2017.5.SPINE161100
Martens W, Sanen K, Georgiou M et al (2014) Human dental pulp stem cells can differentiate into Schwann cells and promote and guide neurite outgrowth in an aligned tissue-engineered collagen construct in vitro. FASEB J 28(4):1634–1643. https://doi.org/10.1096/fj.13-243980
O’Rourke C, Day AGE, Murray-Dunning C et al (2018) An allogeneic ‘off the shelf’ therapeutic strategy for peripheral nerve tissue engineering using clinical grade human neural stem cells. Sci Rep 8(1):2951. https://doi.org/10.1038/s41598-018-20927-8
Sanen K, Martens W, Georgiou M et al (2017) Engineered neural tissue with Schwann cell differentiated human dental pulp stem cells: potential for peripheral nerve repair? J Tissue Eng Regen Med 11:3362. https://doi.org/10.1002/term.2249
Phillips JB, Bunting SC, Hall SM et al (2005) Neural tissue engineering: a self-organizing collagen guidance conduit. Tissue Eng 11(9–10):1611–1617. https://doi.org/10.1089/ten.2005.11.1611
Rayner MLD, Laranjeira S, Evans RE et al (2018) Developing an in vitro model to screen drugs for nerve regeneration. Anat Rec (Hoboken) 301(10):1628–1637. https://doi.org/10.1002/ar.23918
Navarro X, Vivo M, Valero-Cabre A (2007) Neural plasticity after peripheral nerve injury and regeneration. Prog Neurobiol 82(4):163–201. https://doi.org/10.1016/j.pneurobio.2007.06.005
Deumens R, Bozkurt A, Meek MF et al (2010) Repairing injured peripheral nerves: bridging the gap. Prog Neurobiol 92(3):245–276. https://doi.org/10.1016/j.pneurobio.2010.10.002
Jessen KR, Mirsky R (2016) The repair Schwann cell and its function in regenerating nerves. J Physiol 594(13):3521–3531. https://doi.org/10.1113/JP270874
Van den Berg LH, Bar PR, Sodaar P et al (1995) Selective expansion and long-term culture of human Schwann cells from sural nerve biopsies. Ann Neurol 38(4):674–678. https://doi.org/10.1002/ana.410380419
Haastert-Talini K (2012) Culture and proliferation of highly purified adult Schwann cells from rat, dog, and man. Methods Mol Biol 846:189–200. https://doi.org/10.1007/978-1-61779-536-7_17
Rutkowski JL, Kirk CJ, Lerner MA et al (1995) Purification and expansion of human Schwann cells in vitro. Nat Med 1(1):80–83. https://doi.org/10.1038/nm0195-80
O’Rourke C, Drake RA, Cameron GW et al (2015) Optimising contraction and alignment of cellular collagen hydrogels to achieve reliable and consistent engineered anisotropic tissue. J Biomater Appl 30(5):599–607. https://doi.org/10.1177/0885328215597818
Phillips JB (2014) Monitoring neuron and astrocyte interactions with a 3D cell culture system. Methods Mol Biol 1162:113–124. https://doi.org/10.1007/978-1-4939-0777-9_9
Acknowledgments
Rebecca Powell is supported by Medical Research Council funding MR/N013867/1.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Powell, R., Phillips, J.B. (2021). Engineered Tissues Made from Human iPSC-Derived Schwann Cells for Investigating Peripheral Nerve Regeneration In Vitro. In: Stock, P., Christ, B. (eds) In Vitro Models for Stem Cell Therapy. Methods in Molecular Biology, vol 2269. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-1225-5_17
Download citation
DOI: https://doi.org/10.1007/978-1-0716-1225-5_17
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-1224-8
Online ISBN: 978-1-0716-1225-5
eBook Packages: Springer Protocols