Abstract
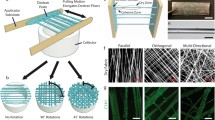
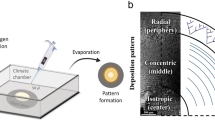
Collagen gels provide a versatile and widely used substrate for three-dimensional (3D) cell culture. Here we describe how cell-seeded Type-I collagen gels can be adapted to provide powerful 3D models to support a wide range of research applications where cell/substrate alignment, density, stiffness/compliance, and strain are critical factors. In their fully hydrated form, rectangular collagen gels can be tethered such that endogenous forces generated as resident cells attach to and remodel the fibrillar collagen network can align the substrate in a controllable, predictable, and quantifiable manner. By removing water from collagen gels (plastic compression), their density increases towards that of body tissues, facilitating the engineering of a range of biomimetic constructs with controllable mechanical properties. This dense collagen can be used in combination with other components to achieve a range of functional properties from controlled perfusion, or tensile/compressive strength to new micro-structures. Detailed methodology is provided for the assembly of a range of 3D collagen materials including tethered aligned hydrogels and plastic compressed constructs. A range of techniques for analysing cell behaviour within these models, including microscopy and molecular analyses are described. These systems therefore provide a highly controllable mechanical and chemical micro-environment for investigating a wide range of cellular responses.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Eastwood, M., Mudera, V. C., McGrouther, D. A., and Brown, R. A. (1998) Effect of precise mechanical loading on fibroblast populated collagen lattices: morphological changes. Cell Motil. Cytoskeleton 40, 13–21.
Eastwood, M., McGrouther, D. A., and Brown, R. A. (1994) A culture force monitor for measurement of contraction forces generated in human dermal fibroblast cultures: evidence for cell-matrix mechanical signalling. Biochim. Biophys. Acta 1201, 186–192.
Brown, R. A., Prajapati, R., McGrouther, D. A., Yannas, I. V., and Eastwood, M. (1998) Tensional homeostasis in dermal fibroblasts: mechanical responses to mechanical loading in three-dimensional substrates. J. Cell. Physiol. 175, 323–332.
Brown, R. A., Wiseman, M., Chuo, C.-B., Cheema, U., Nazhat, S. N. (2005) Ultrarapid engineering of biomimetic materials and tissues: fabrication of nano- and microstructures by plastic compression. Adv. Funct. Mater. 15, 1762–1770.
Brown, R. A., Sethi, K. K., Gwanmesia, I., Raemdonck, D., Eastwood, M., and Mudera, V. (2002) Enhanced fibroblast contraction of 3D collagen lattices and integrin expression by TGF-beta1 and -beta3: mechanoregulatory growth factors? Exp. Cell Res. 274, 310–322.
Cheema, U., Yang, S. Y., Mudera, V., Goldspink, G. G., and Brown, R. A. (2003) 3-D in vitro model of early skeletal muscle development. Cell Motil. Cytoskeleton 54, 226–236.
Marenzana, M., Pickard, D., MacRobert, A. J., and Brown, R. A. (2002) Optical measurement of three-dimensional collagen gel constructs by elastic scattering spectroscopy. Tissue Eng. 8, 409–418.
Phillips, J. B., Bunting, S. C., Hall, S. M., and Brown, R. A. (2005) Neural tissue engineering: a self-organizing collagen guidance conduit. Tissue Eng. 11, 1611–1617.
Porter, R. A., Brown, R. A., Eastwood, M., Occleston, N. L., and Khaw, P. T. (1998) Ultrastructural changes during contraction of collagen lattices by ocular fibroblasts. Wound Repair Regen. 6, 157–166.
Phillips, J. B., Bunting, S. C. J., Hall, S. M., and Brown, R. A. (2005) Neural tissue engineering: a self-organizing collagen guidance conduit. Tissue Eng. 11, 1612–1618.
East, E., Golding, J., and Phillips, J. B. (2009) A versatile 3D culture model facilitates monitoring of astrocytes undergoing reactive gliosis. J. Tissue Eng. Regen. Med. 3(8), 634–646.
East, E., Golding, J., and Phillips, J. B. (2007) Increased GFAP immunoreactivity by astrocytes in response to contact with dorsal root ganglia cells in a 3D culture model. Neuron Glia Biol. 3, S119.
East, E. and Phillips, J. B. (2008) Tissue Engineered Cell Culture Models for Nervous System Research. In: Tissue Engineering Research Trends (Greco, G. N., Ed.), Nova Science Publishers, New York.
Hadjipanayi, E., Mudera, V., and Brown, RA. (2009) Guiding cell migration in 3D: A collagen matrix with graded directional stiffness. Cell Motil. Cytoskeleton 66(3), 121–128.
Mudera, V. C., Pleass, R., Eastwood, M., Tarnuzzer, R., Schultz, G., Khaw, P., McGrouther, D. A., and Brown, R. A. (2000) Molecular responses of human dermal fibroblasts to dual cues: contact guidance and mechanical load. Cell Motil. Cytoskeleton 45, 1–9.
Cheema, U., Brown, R. A., Alp, B., and MacRobert, A. J. (2008) Spatially defined oxygen gradients and vascular endothelial growth factor expression in an engineered 3D cell model. Cell. Mol. Life Sci. 65, 177–186.
Abou Neel, E. A., Cheema, U., Knowles, J. C., Brown, R. A., and Nazhat, S. N. (2006) Use of multiple unconfined compression for fine control of collagen gel scaffold density and mechanical properties. Soft Matter 2, 986–992.
Nazhat, S. N., Neel, E. A., Kidane, A., Ahmed, I., Hope, C., Kershaw, M., Lee, P. D., Stride, E., Saffari, N., Knowles, J. C., and Brown, R. A. (2007) Controlled microchannelling in dense collagen scaffolds by soluble phosphate glass fibers. Biomacromolecules 8, 543–551.
Ananta, M., Aulin, C. E., Hilborn, J., Aibibu, D., Houis, S., Brown, R. A., and Mudera, V. (2009) A poly(lactic acid-co-caprolactone)-collagen hybrid for tissue engineering applications. Tissue Eng. Part A 15, 1667–1675.
East, E., Blum de Oliveira, D., Golding, J. P. and Philips, J. B. (2010) Alignment of Astrocytes Increases Neuronal Growth in Three-Dimensional Collagen Gels and Is Maintained Following Plastic Compression to Form a Spinal Cord Repair Conduit Tissue Engineering Part A. doi:10.1089/ten.tea.2010.0017
Buxton, P. G., Bitar, M., Gellynck, K., Parkar, M., Brown, R. A., Young, A. M., Knowles, J. C., and Nazhat, S. N. (2008) Dense collagen matrix accelerates osteogenic differentiation and rescues the apoptotic response to MMP inhibition. Bone 43, 377–385.
Cheema, U., Chuo, C.-B., Sarathchandra, P., Nazhat, S. N., and Brown, R. A. (2007) Engineering functional collagen scaffolds: cyclical loading increases material strength and fibril aggregation. Adv. Funct. Mat. 17, 2426–2431.
Acknowledgements
Work supported in part (RAB) by grants from BBSRC and TSB-EPSRC and (JBP) the Wellcome Trust. The authors would like to thank Emma East, Daniela Blum de Oliveira, Nelomi Anandagouda, and Tijna Alekseeva for contributing data used in figures.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Phillips, J.B., Brown, R. (2011). Micro-structured Materials and Mechanical Cues in 3D Collagen Gels. In: Haycock, J. (eds) 3D Cell Culture. Methods in Molecular Biology, vol 695. Humana Press. https://doi.org/10.1007/978-1-60761-984-0_12
Download citation
DOI: https://doi.org/10.1007/978-1-60761-984-0_12
Published:
Publisher Name: Humana Press
Print ISBN: 978-1-60761-983-3
Online ISBN: 978-1-60761-984-0
eBook Packages: Springer Protocols