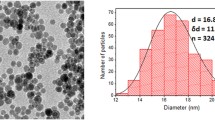
Abstract
Superparamagnetic iron oxide (SPIO) nanoparticles can function as specific, long-term multimodal contrast agents for noninvasive imaging studies. Here we describe how to achieve high-resolution, long-term, serial images of single-label transplanted cells through two complementary imaging techniques: magnetic resonance imaging (MRI) and microcomputed tomography (μCT).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Lai CP, Mardini O, Ericsson M, Prabhakar S, Maguire C, Chen JW, Tannous BA, Breakefield XO (2014) Dynamic biodistribution of extracellular vesicles in vivo using a multimodal imaging reporter. ACS Nano 8(1):483–494. https://doi.org/10.1021/nn404945r
Pacak CA, Hammer PE, MacKay AA, Dowd RP, Wang KR, Masuzawa A, Sill B, McCully JD, Cowan DB (2014) Superparamagnetic iron oxide nanoparticles function as a long-term, multi-modal imaging label for non-invasive tracking of implanted progenitor cells. PLoS One 9(9):e108695. https://doi.org/10.1371/journal.pone.0108695
Peng X, Li C, Bai Y, Wang X, Zhang Y, An Y, Teng GJ, Ju S (2018) Noninvasive evaluation of the migration effect of transplanted endothelial progenitor cells in ischemic muscle using a multimodal imaging agent. Int J Nanomedicine 13:1819–1829. https://doi.org/10.2147/IJN.S152976
Skachkov I, Luan Y, van Tiel ST, van der Steen AFW, de Jong N, Bernsen MR, Kooiman K (2018) SPIO labeling of endothelial cells using ultrasound and targeted microbubbles at diagnostic pressures. PLoS One 13(9):e0204354. https://doi.org/10.1371/journal.pone.0204354
Wang YX, Xuan S, Port M, Idee JM (2013) Recent advances in superparamagnetic iron oxide nanoparticles for cellular imaging and targeted therapy research. Curr Pharm Des 19(37):6575–6593
Li X, Zhang XN, Li XD, Chang J (2016) Multimodality imaging in nanomedicine and nanotheranostics. Cancer Biol Med 13(3):339–348. https://doi.org/10.20892/j.issn.2095-3941.2016.0055
Kim J, Piao Y, Hyeon T (2009) Multifunctional nanostructured materials for multimodal imaging, and simultaneous imaging and therapy. Chem Soc Rev 38(2):372–390. https://doi.org/10.1039/b709883a
Szpak A, Fiejdasz S, Prendota W, Straczek T, Kapusta C, Szmyd J, Nowakowska M, Zapotoczny S (2014) T1-T2 Dual-modal MRI contrast agents based on superparamagnetic iron oxide nanoparticles with surface attached gadolinium complexes. J Nanopart Res 16(11):2678. https://doi.org/10.1007/s11051-014-2678-6
Zareei L, Divband B, Mesbahi A, Khatamian M, Kiani A, Gharehaghaji N (2019) A new potential contrast agent for magnetic resonance imaging: iron oxide-4A nanocomposite. J Biomed Phys Eng 9(2):211–216
Richards JM, Shaw CA, Lang NN, Williams MC, Semple SI, MacGillivray TJ, Gray C, Crawford JH, Alam SR, Atkinson AP, Forrest EK, Bienek C, Mills NL, Burdess A, Dhaliwal K, Simpson AJ, Wallace WA, Hill AT, Roddie PH, McKillop G, Connolly TA, Feuerstein GZ, Barclay GR, Turner ML, Newby DE (2012) In vivo mononuclear cell tracking using superparamagnetic particles of iron oxide: feasibility and safety in humans. Circ Cardiovasc Imaging 5(4):509–517. https://doi.org/10.1161/CIRCIMAGING.112.972596
Riegler J, Liew A, Hynes SO, Ortega D, O'Brien T, Day RM, Richards T, Sharif F, Pankhurst QA, Lythgoe MF (2013) Superparamagnetic iron oxide nanoparticle targeting of MSCs in vascular injury. Biomaterials 34(8):1987–1994. https://doi.org/10.1016/j.biomaterials.2012.11.040
Alam SR, Shah AS, Richards J, Lang NN, Barnes G, Joshi N, MacGillivray T, McKillop G, Mirsadraee S, Payne J, Fox KA, Henriksen P, Newby DE, Semple SI (2012) Ultrasmall superparamagnetic particles of iron oxide in patients with acute myocardial infarction: early clinical experience. Circ Cardiovasc Imaging 5(5):559–565. https://doi.org/10.1161/CIRCIMAGING.112.974907
Pacak CA, Cowan DB (2009) Fabrication of myogenic engineered tissue constructs. J Vis Exp 27:1137. https://doi.org/10.3791/1137
Arbab AS, Bashaw LA, Miller BR, Jordan EK, Lewis BK, Kalish H, Frank JA (2003) Characterization of biophysical and metabolic properties of cells labeled with superparamagnetic iron oxide nanoparticles and transfection agent for cellular MR imaging. Radiology 229(3):838–846. https://doi.org/10.1148/radiol.2293021215
Cianciaruso C, Pagani A, Martelli C, Bacigaluppi M, Squadrito ML, Dico AL, De Palma M, Furlan R, Lucignani G, Falini A, Biffi A, Ottobrini L, Politi LS (2014) Cellular magnetic resonance with iron oxide nanoparticles: long-term persistence of SPIO signal in the CNS after transplanted cell death. Nanomedicine 9(10):1457–1474. https://doi.org/10.2217/nnm.14.84
Acknowledgements
We would like to acknowledge the following investigators who were all involved in the original development and assessment of this methodology: Peter E. Hammer, Allison A. MacKay, Rory P. Dowd, Kai-Roy Wang, Akihiro Masuzawa, Bjoern Sill, James D. McCully, and Douglas B. Cowan.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Sriramvenugopal, M., Pacak, C.A. (2020). Noninvasive Tracking of Implanted Cells: Superparamagnetic Iron Oxide Nanoparticles as a Long-Term, Multimodal Imaging Label. In: Basel, M., Bossmann, S. (eds) Cell Tracking. Methods in Molecular Biology, vol 2126. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0364-2_15
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0364-2_15
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0363-5
Online ISBN: 978-1-0716-0364-2
eBook Packages: Springer Protocols