Abstract
Gait speed is now recognized as an important clinical tool in the older adult population. However, fast gait speed appears to better reflect lower-extremity muscle performance and declines more rapidly in advanced age than comfortable gait speed. We examined the ability of leg lean mass and absolute and rapid strength characteristics to predict fast gait speed in younger versus older adults. Twenty-four younger (12 men, 12 women; age = 22 ± 3 years) and 22 older (11 men, 11 women; age = 72 ± 6 years) adults participated in the study. Fast gait speed was assessed at 10- and 400-m distances. Leg lean mass was quantified with dual X-ray absorptiometry. Isometric squat peak torque and the rate of torque development at 200 ms (RTD200) were tested with a 120° knee joint angle. Statistical analyses included independent samples t tests, partial correlations, and stepwise regression. Large differences between age groups were observed for peak torque, RTD200, and gait speed (P ≤ 0.006, d ≥ 0.79), whereas the difference in leg lean mass was small (P = 0.246, d = 0.35). In older adults only, the partial correlation for 400-m speed versus RTD200 was significant (r = 0.463, P = 0.040). Stepwise regression revealed that peak torque was a significant predictor of 10-m (R2 = 0.257, P = 0.016) and 400-m (R2 = 0.239, P = 0.021) gait speed in older adults. As adults age, lower-extremity, multi-joint muscle strength becomes increasingly important in regulating fast gait speed, whereas lean mass is not predictive.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Muscle weakness in older adults independently predicts illness [1], quality of life [35], and all-cause mortality [31]. Historically, scientists have attributed much of the loss of physical function observed during aging to a decline in muscle mass, or sarcopenia. However, longitudinal studies have demonstrated that aging has a considerably greater influence on muscular strength than what can be accounted for by a decline in muscle mass alone [12, 19]. Two common explanations for the loss of muscle strength in the absence of declining muscle mass include intramuscular adipocyte infiltration [18] and impairments in the central nervous system’s ability to recruit and discharge motor units [6, 16, 22]. For example, Clark et al. [7] reported that weaker older adults exhibited a ~ 20% reduction in voluntary activation, as well as lower and higher corticospinal excitability and inhibition, respectively. Other studies carried out in older adults have shown exceptions to the typical inverse relationship between motor unit recruitment thresholds and firing rates [16], as well as inefficient motor unit derecruitment during voluntary contractions [22]. For these reasons, experts have recently called attention to the importance of maintaining neuromuscular function with advanced age [3, 26].
Gait speed is becoming increasingly recognized as an important clinical mobility assessment tool in older adults [11]. While comfortable (or usual) gait speed is commonly studied, fast gait speed declines more so during old age and may therefore be a more sensitive outcome measure [21, 23]. While only a few studies have examined the physiological underpinnings of fast gait speed, evidence suggests that the neuromuscular system’s capacity to generate force/torque rapidly is important. For example, Clark et al. [8] reported noteworthy differences in the rate of force development for the plantarflexors, as well as the rate of muscle activation for the gastrocnemius, in older adults with slow versus fast 10-m gait speed. These authors also reported no differences in muscle cross-sectional area of the triceps surae, quadriceps, and hamstrings muscle groups between slow and fast walkers, suggesting that muscle mass not play a critical role in gait performance [8]. Similarly, using data from the Baltimore Longitudinal Study of Aging, Osawa et al. (2018) reported that the rate of torque development for the knee extensors was a significant, independent predictor of gait speed in adults. Collectively, there is growing evidence to suggest that fast gait speed is an important clinical tool that is mediated by neuromuscular function.
As interest in the ability to evaluate muscle function continues to grow, so too do methodological advancements that allow for scientific inquiry. In particular, dynamometers that provide information about the neuromuscular system’s ability to generate force during large muscle mass, multi-joint movements are becoming increasingly utilized. Palmer et al. [33] recently reported acceptable test–retest reliability statistics for maximal absolute and rapid strength characteristics using a novel isometric squat device. Theoretically, multi-joint strength assessments that allow for analysis of rapid force or torque signals may better reflect function during activities of daily living and therefore provide greater clinical utility.
The objective of this study was to compare the ability of absolute and rapid strength characteristics, as well as leg lean mass, to predict fast gait speed. To determine whether predictability of the independent variables was dependent on age, we examined groups of both younger and older adults. We hypothesized that fast gait speed performance would be best predicted by the rate of torque development, followed by maximal absolute strength and leg lean mass, and that these findings would be unique to older adults.
Methods
Participants
Twenty-four younger adults (12 men, mean ± SD age = 23 ± 3 years, mass = 70.2 ± 17.1 kg, height = 1.72 ± 0.07 m; 12 women, age = 21 ± 2 years, mass = 63.4 ± 11.7 kg, height = 1.64 ± 0.06 m) and 22 older adults (11 men, mean ± SD age = 74 ± 4 years, mass = 77.4 ± 12.5 kg, height = 1.72 ± 0.28 m; 11 women, age = 71 ± 5 years, mass = 69.3 ± 7.6 kg, height = 1.54 ± 0.17 m) volunteered to participate in this study. Preliminary screening was conducted over the telephone to qualify participants based on the following eight inclusion criteria: (1) age between 18 and 30 or 65 and 85 years; (2) did not require the use of an assistive walking device; (3) no surgery on the hip or knee joints within the previous 12 months; (4) had no known neuromuscular or metabolic diseases; (5) had not experienced a myocardial infarction within the previous 12 months; (6) did not regularly engage in resistance training (< three times monthly in the previous 3 months) or other structured aerobic exercise more than 20 min per day, twice per week [24]; (7) did not have uncontrolled hypertension (> 150/90 mm Hg); (8) could not have had their primary physician advise them not to engage in exercise. Once screened for the above qualifications, participants reported to the laboratory where they were further screened for the inclusion criteria of a body mass index between 19 and 33 kg/m2, after which they completed health, medical history, and physical activity questionnaires. All participants gave written informed consent prior to participation in this investigation. This study was approved by the University Institutional Review Board following an expedited review process.
Study Design
Participants reported to the laboratory on two occasions separated by 48–96 h. The initial visit involved completion of study paperwork, body composition analysis, and gait speed testing. The participants were familiarized with isometric squat strength testing during the initial visit to the laboratory. The second visit only involved isometric strength data collection. Participants were asked to refrain from exercise 48 h prior to each laboratory visit.
Dual X-Ray Absorptiometry (DXA)
DXA has been proposed to serve as the reference technique for estimating muscle mass and body composition in older adults [2]. The participants completed one total body scan using the DXA (Lunar Prodigy Primo, GE Healthcare, Madison, WI, USA). All scans were performed by a trained technician that had completed both university radiation training and a training session held by the device’s manufacturer. Automated methods from the manufacturer-provided software (Lunar Radiation Body Composition, version 13.60, GE Healthcare, Wauwatosa, WI, USA) were used to estimate leg lean mass (kg).
Isometric Squat Strength
Isometric squat testing was performed using a commercially-available multi-joint isokinetic strength training device (Exerbotics Squat [eSQ], Exerbotics, LLC, Tulsa, OK, USA). Test–retest reliability statistics for these methods have been reported previously [33, 36]. During testing, the participants stood with their feet shoulder width apart and shoulders positioned under the padded frame. The participants were able to gently hold onto handles directly in front of them for balance and postural support. The researcher then adjusted the frame height such that the knee joint was aligned at a 120° angle. Each participant performed two maximal voluntary contractions separated by 1 min of rest. The participants were specifically instructed to perform the maximal voluntary contractions “as hard and as fast as possible until I instruct you to stop”, with each lasting a total of 4 s.
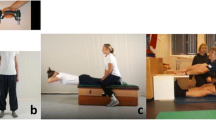
The torque signal was sampled at 2000 Hz and processed off-line using custom LabVIEW software (version 8.5, National Instruments, Austin, TX, USA). The raw signals were scaled to units (Nm) and filtered using a zero-phase shift, second-order Butterworth filter with a 20 Hz low-pass cutoff frequency. The signal onsets were determined manually via visual inspection as the point when the signal first deflected from the baseline [17], with the aid of a horizontal cursor, which helped the inspector objectively assess the collective baseline level. Peak torque (Nm) was calculated as the highest 500 ms epoch throughout the duration of the contraction. The RTD200 was quantified from the linear slope of the ascending portion of the torque-time curve at 200 ms from onset (Fig. 1). We elected to study RTD200 rather than earlier torque-time intervals because it tends to be more reliable [43], particularly using the equipment of the present investigation [33].
Examples of the isometric squat data collection and analysis procedures. The image on the left shows a younger adult male performing an isometric squat as described herein. The load cell used to acquire the torque data has been circled. On the right, an example torque-time curve has been displayed. The dotted area to the left shows the 200 ms region where the rate of torque development was calculated, whereas the shaded area corresponds to where peak torque was noted
Fast Gait Speed
We assessed fast gait speed at distances of both 10-m and 400-m. These distances were selected because they are common in the gait speed literature, as they show high sensitivity for identifying declines in lower-extremity function [8, 23] and are associated with mortality and morbidity rates [32]. The 10-m walk test was conducted according to the procedures of Clark et al. [8], in which the 10-m distance was centered between 5-m acceleration and deceleration zones to help achieve a steady state. Three trials separated by 2-min rest periods were performed. The mean of the three speeds was utilized for analysis (m/s). The 400-m test was performed in accordance with the procedures described by Newman et al. [32], in which the participants walked a total of ten laps between two cones set at 20-m apart (40-m per lap). Participants were instructed to “walk as quickly as you can, without running, at a pace you can maintain.” [32] Only one 400-m test was performed. A stopwatch was used to measure times to complete the tests. All gait speed tests were performed in an indoor, air-conditioned facility.
Statistical Analyses
Differences between younger and older adults were examined with independent samples t tests, Cohen’s d effect size statistics, and 95% confidence intervals (CIs) for mean differences. As both age [9] and height [15] have been shown to be important regulators of gait speed, partial correlations (controlling for age and height) were used to examine the associations among leg lean mass, peak torque, and RTD200 versus 10- and 400-m fast gait speed for each age group. Four separate stepwise regression analyses were performed to examine the ability for leg lean mass, peak torque, RTD200, age, and height to explain variance in 10- and 400-m fast gait speed in younger and older adults. An α level of P ≤ 0.05 was used to determine statistical significance for all data analyses. To monitor multicollinearity, the variance inflation factor (VIF) was calculated using the equation VIF = 1/(1 − R2). A VIF greater than 10 has been suggested to indicate problematic multicollinearity [46]. All statistical analyses were performed using SPSS software (IBM SPSS Statistics, Version 25.0. Armonk, NY: IBM Corp). Univariate scatterplots displaying individual participant data were created using templates provided by Weissgerber et al. [48].
Results
As shown in Fig. 2, the mean difference in leg lean mass between younger and older adults was small [P = 0.246, d = 0.35, 95% CI (−0.74, 2.82 kg)]. In contrast, as shown in Figs. 3 and 4, there were large differences for peak torque [P < 0.001, d = 1.24, 95% CI (250.7, 554.2 Nm)], RTD200 [P < 0.001, d = 1.07, 95% CI (823.9, 2308.1 Nm/s)], 10-m fast gait speed [P = 0.006, d = 0.79, 95% CI (0.08, 0.43 m/s)], and 400-m fast gait speed [P < 0.001, d = 1.43, 95% CI (0.27, 0.50 m/s)]. As shown in Table 1, none of the partial correlations for the younger adults were significant. For the older adults, however, a significant partial correlation between 400-m fast gait speed and RTD200 was demonstrated (r = 0.463, P = 0.040). Similar nonsignificant findings for 10-m fast gait speed and peak torque (r = 0.422, P = 0.064), as well as 400-m fast gait speed and peak torque (r = 0.421, P = 0.065), were demonstrated for the older adults. The results from the stepwise regression analyses indicated that, for the younger adults, none of the independent variables were significant predictors of gait speed. For the older adults, peak torque was a significant predictor of both 10-m (R2 = 0.257; adjusted R2 = 0.220; P = 0.016) and 400-m (R2 = 0.239; adjusted R2 = 0.200; P = 0.021) fast gait speed. Stepwise regression demonstrated that leg lean mass and RTD200 were not useful predictors of fast gait speed.
Individual participant data for (a) squat peak torque and (b) the rate of torque development at 200 ms, with the younger and older adults shown on the left and right, respectively, of each graph. The thick black symbol corresponds to the mean value for each age group. The most relevant statistical results are shown at the top of the graph
Individual participant data for (a) 10-m and (b) 400-m fast gait speed, with the younger and older adults shown on the left and right, respectively, of each graph. The thick black symbol corresponds to the mean value for each age group. The most relevant statistical results are shown at the top of the graph
Discussion
The primary finding of this study was that measures of absolute and rapid squat strength were associated with short and long-distance fast gait speed in older adults, but leg lean mass was not observed to be an important predictor. More specifically, similar partial correlations were demonstrated for peak torque and RTD200, whereas the stepwise regression analysis showed that peak torque was the best overall predictor of fast gait speed. Furthermore, there were large differences in absolute and rapid squat strength and gait speed between age groups, but there was no difference in leg lean mass between younger and older adults. Combined with the fact that none of the examined variables explained variance in fast gait speed among the younger adults, these findings highlight the importance of maintaining lower-extremity squat strength throughout the lifespan. We believe that these findings may highlight a potential dissociation between leg lean mass and functional performance in older adults.
Our findings demonstrated that leg lean mass did not significantly differ between younger and older adults. Similarity of DXA-derived leg lean mass among younger and older adults has been reported in some [10, 40], but not all [25, 45], studies. These results are also similar in nature to a three-year study in older adults [34], as well as those reported by Clark et al. [8], who reported no differences between slow and fast walkers for MRI-derived muscle cross sectional area of the triceps surae, quadriceps, and hamstrings muscle groups. Given that such dramatic age-related differences were observed for our muscle strength and gait speed measures (see Figs. 3 and 4), but not leg lean mass, these results highlight the dissociation between muscle mass and functional performance in older adults. When one considers that older adults may lose significant muscle strength in the absence of muscle atrophy [12, 19], we suspect that our findings provide further support for the notion that fast gait speed is characterized by the neuromuscular system’s control of muscle force, which may be mediated by parameters such as voluntary activation, corticospinal excitability/inhibition, and/or motor unit firing rates [4]. Clinicians seeking to prevent musculoskeletal declines in aging adults should be concerned with muscle tissue quality, as the most recent sarcopenia guidelines highlight the fact that a single body composition assessment provides little information about a patient’s functional status [11].
Previous studies that sought to characterize gait performance have typically focused on individual muscles or muscle groups. In particular, an emphasis has been placed on understanding the relationship between the gait cycle and ankle plantarflexion performance. These studies have demonstrated the importance of maintaining neuromuscular control of the triceps surae [8, 29], with weakness being directly associated with reduced gait speed in older adults with functional limitations [39]. A unique aspect of our approach to this study, however, was the assessment of absolute and rapid strength characteristics during maximal isometric squats. Isometric multi-joint tests involving the lower extremities have been used to study muscle strength in young adults and athletes [14], but this approach has not been utilized in older adults. We sought to study the isometric squat rather than isolated joint movements because we felt that analysis of a lower-extremity, multi-joint movement involving the activation of dozens of muscles would better characterize fast gait performance. Squatting movements are also commonly utilized in resistance training interventions [37] and physical therapy and rehabilitation clinics [27]. By focusing on muscle performance and lean mass of the entire lower extremity, rather than isolated muscles, we would contend that our approach better reflects measures that may typically be used in a clinical setting. We should concede, however, that while the absolute and rapid strength measurements utilized herein explained roughly 25% of the variance in fast gait speed, other previous studies have demonstrated stronger prediction [9, 27, 47]. For example, Clark et al. [9] reported that the combination of age, muscle and fat mass variables, and the rate of electromyographic rise accounted for 72.4% and 43.7% of the variance in fast gait speed for men and women, respectively. More recently, Mantel et al. [27] was able to explain more than 80% of the variance in fast gait speed utilizing a comprehensive assessment battery focused on both strength and balance. Given the totality of evidence, it seems likely that fast gait speed is best predicted by multiple outcome measures.
Although our study design was not specifically focused on differences in absolute and rapid strength characteristics, we should highlight these findings. As shown in Table 1, the associations between peak torque and RTD200 versus both short- and long-distance fast gait speed were quite similar for the older adults. Stepwise regression analysis revealed that RTD200 was the best single predictor of fast gait speed, with peak torque likely being removed due to high collinearity. While rapid neuromuscular activation should be emphasized in maintaining functional outcomes across the lifespan [8, 9, 42, 44], our findings suggest that absolute strength should not be completely discounted.
We should note that our investigation had limitations. Specifically, it should be noted that the optimal measurement tools for assessing body composition in older adults is still a matter of debate. Like many other previous studies [19, 28, 38, 47], our approach relied on DXA to quantify lean mass, primarily because of its availability and ease of use. Very recently, over 30 experts in the fields of aging and body composition from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis working group on frailty and sarcopenia proposed that DXA be universally adopted as the reference standard for measuring muscle mass [2]. These authors noted the benefits of using DXA as a reference measure, which include low radiation, low precision errors, and its rapid assessment of three body compartments [2]. However, this proposal has been met with some skepticism. Clark et al. [5] noted that while DXA has several advantages as a tool to quantify appendicular lean soft tissue mass, it offers lower accuracy than MRI and CT scans relative to cadaveric measurements and is prone to measurement error. Furthermore, studies have reported only a modest relationship between increases in DXA- and MRI- or CT-derived measures of percent change in muscle size [13, 20, 41]. Thus, we concede that there are a variety of methodological techniques for assessing body composition in both younger and older adults, and measurement via ultrasound, MRI, or CT may yield differing conclusions from those of the present study. Other limitations of the present study may include too small of a sample to comprehensively study sex differences, lacking sophisticated neuromuscular measurements (e.g., electromyography, motor unit analysis, transcranial magnetic stimulation, etc.), which may have helped explain variance in fast gait speed, and only studying fast gait speed. Finally, as our investigation utilized a cross-sectional design, these data should not be extrapolated to longitudinal changes that occur throughout the process of aging or interventions that may affect mobility.
In conclusion, our findings indicate that absolute and rapid multi-joint squat strength, but not leg lean mass, are important predictors of both 10- and 400-m fast gait speed in older adults. The fact that leg lean mass was not at all associated with gait speed further highlights the potential dissociation between skeletal muscle mass and function. Additional studies including larger samples, other measures of muscle morphology, and more variables well-grounded in neuromuscular physiology may help replicate these findings, as well as aid in elucidating the unexplained variance in fast gait speed. Understanding the neuromuscular determinants of mobility has important implications for creating assessments and interventions that can be used in practice to prevent functional impairments among older adults.
References
Ali NA, O'Brien JM Jr, Hoffmann SP, Phillips G, Garland A, Finley JC, Almoosa K, Hejal R, Wolf KM, Lemeshow S, Connors AF Jr, Marsh CB, Midwest Critical Care C. Acquired weakness, handgrip strength, and mortality in critically ill patients. Am J Respir Crit Care Med. 2008;178(3):261–8. https://doi.org/10.1164/rccm.200712-1829OC.
Buckinx F, Landi F, Cesari M, Fielding RA, Visser M, Engelke K, Maggi S, Dennison E, Al-Daghri NM, Allepaerts S, Bauer J, Bautmans I, Brandi ML, Bruyere O, Cederholm T, Cerreta F, Cherubini A, Cooper C, Cruz-Jentoft A, McCloskey E, Dawson-Hughes B, Kaufman JM, Laslop A, Petermans J, Reginster JY, Rizzoli R, Robinson S, Rolland Y, Rueda R, Vellas B, Kanis JA. Pitfalls in the measurement of muscle mass: a need for a reference standard. J Cachexia Sarcopenia Muscle. 2018;9(2):269–78. https://doi.org/10.1002/jcsm.12268.
Clark BC. Neuromuscular changes with aging and sarcopenia. J Frailty Aging. 2019;8(1):7–9. https://doi.org/10.14283/jfa.2018.35.
Clark BC, Manini TM. Sarcopenia =/= dynapenia. J Gerontol A Biol Sci Med Sci. 2008;63(8):829–34.
Clark BC, Tavoian D, Goodpaster BH, Cawthon PM, Hansen RD, Manini TM. Comment on: “Pitfalls in the measurement of muscle mass: a need for a reference standard” by buckinx. J Cachexia Sarcopenia Muscle. 2018;9(7):1269–71. https://doi.org/10.1002/jcsm.12372.
Clark BC, Taylor JL. Age-related changes in motor cortical properties and voluntary activation of skeletal muscle. Curr Aging Sci. 2011;4(3):192–9.
Clark BC, Taylor JL, Hong SL, Law TD, Russ DW. Weaker seniors exhibit motor cortex hypoexcitability and impairments in voluntary activation. J Gerontol A Biol Sci Med Sci. 2015;70(9):1112–9. https://doi.org/10.1093/gerona/glv030.
Clark DJ, Manini TM, Fielding RA, Patten C. Neuromuscular determinants of maximum walking speed in well-functioning older adults. Exp Gerontol. 2013;48(3):358–63. https://doi.org/10.1016/j.exger.2013.01.010.
Clark DJ, Reid KF, Patten C, Phillips EM, Ring SA, Wu SS, Fielding RA. Does quadriceps neuromuscular activation capability explain walking speed in older men and women? Exp Gerontol. 2014;55:49–53. https://doi.org/10.1016/j.exger.2014.03.019.
Crane JD, Macneil LG, Tarnopolsky MA. Long-term aerobic exercise is associated with greater muscle strength throughout the life span. J Gerontol A Biol Sci Med Sci. 2013;68(6):631–8. https://doi.org/10.1093/gerona/gls237.
Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyere O, Cederholm T, Cooper C, Landi F, Rolland Y, Sayer AA, Schneider SM, Sieber CC, Topinkova E, Vandewoude M, Visser M, Zamboni M, Writing Group for the European Working Group on Sarcopenia in Older People 2 (EWGSOP2), and the Extended Group for EWGSOP2. Sarcopenia: revised european consensus on definition and diagnosis. Age Ageing. 2019. https://doi.org/10.1093/ageing/afz046.
Delmonico MJ, Harris TB, Visser M, Park SW, Conroy MB, Velasquez-Mieyer P, Boudreau R, Manini TM, Nevitt M, Newman AB, Goodpaster BH, Health A, Body. Longitudinal study of muscle strength, quality, and adipose tissue infiltration. Am J Clin Nutr. 2009;90(6):1579–85. https://doi.org/10.3945/ajcn.2009.28047.
Delmonico MJ, Kostek MC, Johns J, Hurley BF, Conway JM. Can dual energy x-ray absorptiometry provide a valid assessment of changes in thigh muscle mass with strength training in older adults? Eur J Clin Nutr. 2008;62(12):1372–8. https://doi.org/10.1038/sj.ejcn.1602880.
Drake D, Kennedy R, Wallace E. The validity and responsiveness of isometric lower body multi-joint tests of muscular strength: a systematic review. Sports Med Open. 2017;3(1):23. https://doi.org/10.1186/s40798-017-0091-2.
Elbaz A, Artaud F, Dugravot A, Tzourio C, Singh-Manoux A. The gait speed advantage of taller stature is lost with age. Sci Rep. 2018;8(1):1485. https://doi.org/10.1038/s41598-018-19882-1.
Erim Z, Beg MF, Burke DT, de Luca CJ. Effects of aging on motor-unit control properties. J Neurophysiol. 1999;82(5):2081–91. https://doi.org/10.1152/jn.1999.82.5.2081.
Gerstner GR, Thompson BJ, Rosenberg JG, Sobolewski EJ, Scharville MJ, Ryan ED. Neural and muscular contributions to the age-related reductions in rapid strength. Med Sci Sports Exerc. 2017;49(7):1331–9. https://doi.org/10.1249/MSS.0000000000001231.
Goodpaster BH, Chomentowski P, Ward BK, Rossi A, Glynn NW, Delmonico MJ, Kritchevsky SB, Pahor M, Newman AB. Effects of physical activity on strength and skeletal muscle fat infiltration in older adults: a randomized controlled trial. J Appl Physiol. 2008;105(5):1498–503. https://doi.org/10.1152/japplphysiol.90425.2008.
Goodpaster BH, Park SW, Harris TB, Kritchevsky SB, Nevitt M, Schwartz AV, Simonsick EM, Tylavsky FA, Visser M, Newman AB. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol A Biol Sci Med Sci. 2006;61(10):1059–64.
Hansen RD, Williamson DA, Finnegan TP, Lloyd BD, Grady JN, Diamond TH, Smith EU, Stavrinos TM, Thompson MW, Gwinn TH, Allen BJ, Smerdely PI, Diwan AD, Singh NA, Singh MA. Estimation of thigh muscle cross-sectional area by dual-energy x-ray absorptiometry in frail elderly patients. Am J Clin Nutr. 2007;86(4):952–8. https://doi.org/10.1093/ajcn/86.4.952.
Jahn K, Zwergal A, Schniepp R. Gait disturbances in old age: classification, diagnosis, and treatment from a neurological perspective. Dtsch Arztebl Int. 2010;107(17):306–15. https://doi.org/10.3238/arztebl.2010.0306.
Kamen G, De Luca CJ. Unusual motor unit firing behavior in older adults. Brain Res. 1989;482(1):136–40.
Ko SU, Hausdorff JM, Ferrucci L. Age-associated differences in the gait pattern changes of older adults during fast-speed and fatigue conditions: results from the baltimore longitudinal study of ageing. Age Ageing. 2010;39(6):688–94. https://doi.org/10.1093/ageing/afq113.
Lanza IR, Larsen RG, Kent-Braun JA. Effects of old age on human skeletal muscle energetics during fatiguing contractions with and without blood flow. J Physiol. 2007;583(Pt 3):1093–105. https://doi.org/10.1113/jphysiol.2007.138362.
Larson RD, Misic MM, Evans EM. Association of adiposity and muscle quality with physical function differs in young and old women. Menopause. 2015;22(3):337–41. https://doi.org/10.1097/GME.0000000000000333.
Lavin KM, Roberts BM, Fry CS, Moro T, Rasmussen BB, Bamman MM. The importance of resistance exercise training to combat neuromuscular aging. Physiology (Bethesda). 2019;34(2):112–22. https://doi.org/10.1152/physiol.00044.2018.
Mantel A, Trapuzzano A, Chizmar S, Haffke L, Dawson N. An investigation of the predictors of comfortable and fast gait speed in community-dwelling older adults. J Geriatr Phys Ther. 2018. https://doi.org/10.1519/JPT.0000000000000216.
Moon JR, Stout JR, Smith-Ryan AE, Kendall KL, Fukuda DH, Cramer JT, Moon SE. Tracking fat-free mass changes in elderly men and women using single-frequency bioimpedance and dual-energy x-ray absorptiometry: a four-compartment model comparison. Eur J Clin Nutr. 2013;67(Suppl 1):S40–6. https://doi.org/10.1038/ejcn.2012.163.
Morse CI, Thom JM, Davis MG, Fox KR, Birch KM, Narici MV. Reduced plantarflexor specific torque in the elderly is associated with a lower activation capacity. Eur J Appl Physiol. 2004;92(1–2):219–26. https://doi.org/10.1007/s00421-004-1056-y.
Morse CI, Thom JM, Reeves ND, Birch KM, Narici MV. In vivo physiological cross-sectional area and specific force are reduced in the gastrocnemius of elderly men. J Appl Physiol. 2005;99(3):1050–5. https://doi.org/10.1152/japplphysiol.01186.2004.
Newman AB, Kupelian V, Visser M, Simonsick EM, Goodpaster BH, Kritchevsky SB, Tylavsky FA, Rubin SM, Harris TB. Strength, but not muscle mass, is associated with mortality in the health, aging and body composition study cohort. J Gerontol A Biol Sci Med Sci. 2006;61(1):72–7.
Newman AB, Simonsick EM, Naydeck BL, Boudreau RM, Kritchevsky SB, Nevitt MC, Pahor M, Satterfield S, Brach JS, Studenski SA, Harris TB. Association of long-distance corridor walk performance with mortality, cardiovascular disease, mobility limitation, and disability. JAMA. 2006;295(17):2018–26. https://doi.org/10.1001/jama.295.17.2018.
Palmer TB, Pineda JG, Durham RM. Effects of knee position on the reliability and production of maximal and rapid strength characteristics during an isometric squat test. J Appl Biomech. 2018;34(2):111–7. https://doi.org/10.1123/jab.2017-0213.
Sergi G, Sarti S, Mosele M, Ruggiero E, Imoscopi A, Miotto F, Bolzetta F, Inelmen EM, Manzato E, Coin A. Changes in healthy elderly women’s physical performance: a 3-year follow-up. Exp Gerontol. 2011;46(11):929–33. https://doi.org/10.1016/j.exger.2011.08.008.
Silva Neto LS, Karnikowiski MG, Tavares AB, Lima RM. Association between sarcopenia, sarcopenic obesity, muscle strength and quality of life variables in elderly women. Rev Bras Fisioter. 2012;16(5):360–7.
Stock MS, Luera MJ. Consistency of peak and mean concentric and eccentric force using a novel squat testing device. J Appl Biomech. 2014;30(2):322–5. https://doi.org/10.1123/jab.2013-0191.
Stock MS, Olinghouse KD, Drusch AS, Mota JA, Hernandez JM, Akalonu CC, Thompson BJ. Evidence of muscular adaptations within 4 weeks of barbell training in women. Hum Mov Sci. 2016;45:7–22. https://doi.org/10.1016/j.humov.2015.11.004.
Stout JR, Fukuda DH, Kendall KL, Smith-Ryan AE, Moon JR, Hoffman JR. Beta-hydroxy-beta-methylbutyrate (hmb) supplementation and resistance exercise significantly reduce abdominal adiposity in healthy elderly men. Exp Gerontol. 2015;64:33–4. https://doi.org/10.1016/j.exger.2015.02.012.
Suzuki T, Bean JF, Fielding RA. Muscle power of the ankle flexors predicts functional performance in community-dwelling older women. J Am Geriatr Soc. 2001;49(9):1161–7. https://doi.org/10.1046/j.1532-5415.2001.49232.x.
Tanner RE, Brunker LB, Agergaard J, Barrows KM, Briggs RA, Kwon OS, Young LM, Hopkins PN, Volpi E, Marcus RL, LaStayo PC, Drummond MJ. Age-related differences in lean mass, protein synthesis and skeletal muscle markers of proteolysis after bed rest and exercise rehabilitation. J Physiol. 2015;593(18):4259–73. https://doi.org/10.1113/JP270699.
Tavoian D, Ampomah K, Amano S, Law TD, Clark BC. Changes in dxa-derived lean mass and mri-derived cross-sectional area of the thigh are modestly associated. Sci Rep. 2019;9(1):10028. https://doi.org/10.1038/s41598-019-46428-w.
Thompson BJ, Ryan ED, Herda TJ, Costa PB, Herda AA, Cramer JT. Age-related changes in the rate of muscle activation and rapid force characteristics. Age (Dordr). 2014;36(2):839–49. https://doi.org/10.1007/s11357-013-9605-0.
Thompson BJ, Ryan ED, Herda TJ, Costa PB, Walter AA, Sobolewski EJ, Cramer JT. Consistency of rapid muscle force characteristics: influence of muscle contraction onset detection methodology. J Electromyogr Kinesiol. 2012;22(6):893–900. https://doi.org/10.1016/j.jelekin.2012.05.008.
Thompson BJ, Ryan ED, Sobolewski EJ, Conchola EC, Cramer JT. Age related differences in maximal and rapid torque characteristics of the leg extensors and flexors in young, middle-aged and old men. Exp Gerontol. 2013;48(2):277–82. https://doi.org/10.1016/j.exger.2012.10.009.
Vigelso A, Gram M, Wiuff C, Andersen JL, Helge JW, Dela F. Six weeks’ aerobic retraining after two weeks’ immobilization restores leg lean mass and aerobic capacity but does not fully rehabilitate leg strength in young and older men. J Rehabil Med. 2015;47(6):552–60. https://doi.org/10.2340/16501977-1961.
Vincent WJ, Weir JP. Statistics in kinesiology. 4th ed. Champaign: Human Kinetics; 2012.
Visser M, Kritchevsky SB, Goodpaster BH, Newman AB, Nevitt M, Stamm E, Harris TB. Leg muscle mass and composition in relation to lower extremity performance in men and women aged 70–79: the health, aging and body composition study. J Am Geriatr Soc. 2002;50(5):897–904. https://doi.org/10.1046/j.1532-5415.2002.50217.x.
Weissgerber TL, Milic NM, Winham SJ, Garovic VD. Beyond bar and line graphs: time for a new data presentation paradigm. PLoS Biol. 2015;13(4):e1002128. https://doi.org/10.1371/journal.pbio.1002128.
Acknowledgements
The authors would like to thank the research participants for taking the time to be part of this study. Funding for this study was provided by the National Strength and Conditioning Association Foundation. The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stock, M.S., Thompson, B.J. & Sobolewski, E.J. Age-Related Differences in the Predictability of Fast Gait Speed with Absolute and Rapid Squat Strength. J. of SCI. IN SPORT AND EXERCISE 1, 273–280 (2019). https://doi.org/10.1007/s42978-019-00032-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42978-019-00032-6