Abstract
The present study aimed to assess the role of homegarden (HG) in safeguarding the tree diversity and carbon (C) density or storage in the Thodupuza urban region of Idukki district in Kerala, India. A stratified random approach was used for selecting the four homegardens (HGs) with a size of 1hectare (ha). The study mainly focused on species richness, diversity, above ground biomass, carbon, correlation and distribution of carbon with various variables, species and group wise carbon storage of the system. A total of 992 trees from 66 species belonging to 31 families were enumerated with representation of 4 endemic, 1 vulnerable, 1 endangered and 23 exotics. The diversity indices obtained were closer to those of the forest ecosystem. Above ground biomass and carbon density were estimated to be 67.06 t/ha (tonne/hectare) and 31.85 6 t/ha respectively. Species Tectona grandis showed dominancy in carbon and Important Value Index. Correlation analysis among species revealed that carbon exhibited a strong positive trend with basal area and tree density, but in the case of plot (HG)-wise examination only basal area had a strong positive relationship. The diametric class analysis showed skewed type of distribution for carbon and tree density while diversity had reverse j-shaped curve. Among the two plant groups, cultivated species had an edge over native in storing carbon. Overall, this assessment shows the potential of using homegardens as a socio-ecological systems for sustainable development particularly in terms of land availability and climate mitigation options in the face of rapid urbanization.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urbanizing regions pose enormous challenges to the ecosystem’s capacity to deliver important ecological functions (Alberti 2010). At current rates of urban growth, global urban land cover will increase by 1.2 million km2 by 2030, nearly tripling the global urban land area of 2000, with considerable loss of habitats in key biodiversity hotspots (Seto et al. 2012). Over the recent years there has been growing interest in studying the role of urban green spaces in both mitigating urbanization’s impact and providing a variety of ecosystem services. Consequently, there is much attention towards urban agro-ecosystems as a strategy that improves the quality of life of our increasingly urbanized community. The quality of the urban communities depends on the ecological, social, economic, recreational, cultural, services provided by these ecosystems (Egerer et al. 2018). One type of urban agro-ecosystem that has experienced a renaissance in recent years and has piqued the interest of local governments and urban planners are homegardens.
Homegardens, defined as the oldest form of land-use system having a multi-story combination of trees along with shrubs and herbs and sometimes in association with livestock around the homesteads (Fernandes and Nair 1986). Being a multi-species and multilayered system of land use, they offer food items (vegetables, fruits), fodder, medicines, fuel and insurance against crop failure besides generating revenue and occupation (Kumar and Nair 2004; Gbedomon 2017). Besides, they conserve water and soil, increase fertility of the soil, nutrient cycling and watershed protection (Pandey 2002). In view of these multiple functions, many researchers have concluded that homegardens are one of the sustainable production systems in the terrestrial environment (Nair 2001; Blanckaert et al. 2004; Kumar and Nair 2004; Sanyé-Mengual et al. 2018).
However, in the current scenario, in addition to climate change, these traditional homegardens are subject to rapid urbanization and increased economic activities that caused many changes to these ecosystems in well-developed city regions worldwide. To meet future challenges of land and water scarcity, biodiversity loss, emission of greenhouse gases and to ensure food security as a result to adverse fallouts of climate change, future mitigation efforts and adaptation strategies are essential for local people with the active support from decision makers that necessitate further attention (Murthy et al. 2013). Moreover, urban researchers and practitioners have been thinking carefully about how to create and manage green spaces that support native species diversity, ecological processes (e.g., stormwater infiltration and decomposition), and ecosystem services under an ecology for the city framework (Grove et al. 2016). In order to identify such strategies, it is vital to analyze quantifiable estimates of tropical homegardens ability to various ecosystem services that are important for sustainability.
The state of Kerala that lies in the Western Ghats portion (global biodiversity hotspot) of southern India (Myers et al. 2000), is one of the tropical regions where homegardening has been a part of the culture for centuries and is still critical for the local economy and greater food security (Kumar and Nair 2004). Kerala recording almost 4.32 million homegardens covering an area of 1.4 Mha (Kumar 2006) have been recognized for their ecological, social and economic sustainability values (Kumar et al. 1994; Peyre et al. 2006; Unnithan et al. 2017). Thus, homegardens play a substantial role in climate change mitigation and environmental amelioration in Kerala.
However, it may be noted that the urban populace of Kerala has registered a huge growth over the last decade as the number of towns in the State increased three times (Government of Kerala 2016). Kerala Economic Report 2018 indicates that the share of urban population in Kerala was 47.7 per cent of the total population, representing a decade increase of 21.74 per cent between 2001 and 2011. The report in its 13th, 5-year plan emphasis on the issues arising due to fast moving urbanisation and recognizes the importance of developing appropriate strategies and programmes for coping up with the challenges and opportunities thrown up by the process of urban growth in Kerala (Government of Kerala 2018).
In this context, the role of home gardens in shaping the urban environment is addressed through the investigation of important ecological functions which are essential to the quality of community life and act as a key component of sustainable development. Hence the present study has been taken up with a vision to depict the regional role of tropical homegardens as a mechanism for conserving biodiversity and its role in carbon storage, where a paucity of information still exist not only in Kerala but also from the Southern Western Ghats portion of India.
In this context, the main objectives of the present study are -
-
1.
to quantify the phytosociological aspects of homegarden by determining species diversity, composition, structure and uses of trees.
-
2.
to estimate the aboveground biomass and carbon storage capacity of trees in homegarden.
-
3.
to examine the relationship and distribution of tree carbon with various stand variables in homegardens and plant groups identified.
Materials and methods
Study area
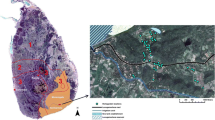
The present study was carried out in the urban region of Thodupuza, Idukki district of Kerala state which is situated in the Southern Western Ghats portion of peninsular India (Fig. 1). Thodupuza is a major urbanized town characterized by hilly terrain endowed with green vegetation. The average net population density of the district is 586 people/square kilometer (km2) and that of Thodupuzha is 1305 people/km2. (DUR 2011). The average annual temperature of Thodupuza region is about 27.6 degrees celsius (°C) having a mean precipitation of 3713 mm (Climate-data.org 2018).
Sampling approach and analysis
Four large homegardens each having the size of 1 ha (100 × 100 m2) were selected from the region based on the stratified random procedure and tree inventory was carried out in this particular plot size. The flora was identified as per Sasidharan (2004) and Nayar et al. (2014). The investigation was conducted during 2011–2012 period. The important quantitative analysis indicating the structural role of tree species (Important Value Index-IVI) was determined using a percentage of relative abundance, relative dominance and relative frequency (Whittaker 1970). Shannon–Wiener index of diversity (Shannon and Wiener 1949) and Pielou’s evenness index (Pielou 1975) were calculated using methods specified by Magurran (2004). Based on the interaction with farmers and with input from various studies (Kumar et al. 1994; Muraleedharan et al. 2005) major uses of trees were identified from the garden.
The standing tree biomass (aboveground) was computed based on the volume, specific gravity relationship (volume of tree X specific gravity) (Ravindranath and Ostwald 2008). This equation is generally employed to find biomass of trees having ≥ 10 cm dbh (diameter at breast height) or ≥ 30 cm gbh (girth at breast height). In order to find the volume of a particular tree species (≥ 10 cm dbh), species-specific volume equation formulated by Forest Survey of India (FSI 1996) was used and for those species for which equations were not available area specific generalized volume equation developed by FSI was used in the analysis (Table 1). Wood specific gravity data obtained from Forest Research Institute (ICFRE 1996–2002) was employed for calculation of biomass. But for species having no specific gravity specified, average specific gravity was considered for biomass estimation (Table 1). A regression equation developed based on the relationship between basal area (cross-sectional area of the tree) and tree biomass in the plots (HGs) (Dadhwal et al. 2009) was taken for biomass estimation of trees having < 10 cm (3–9 cm) dbh. The belowground biomass (BGB) was estimated by multiplying AGB with a factor of 0.26 which is considered as the root to shoot ratio (Cairns et al. 1997). An average value of 47% of carbon in the tree biomass is considered for regional level estimation of the carbon pool (Raghubansi et al. 1990). Hence biomass values were converted into carbon by multiplying biomass with a factor of 0.47. In the case of individual plots, the biomass and carbon tabulated from each species were summed up to obtain respective plot level (homegarden) above ground biomass and carbon and is stated in t-dry weight/ha. Finally the average value of four homegardens was thus considered as total biomass and carbon density of the homegarden ecosystem and is expressed in t-dry weight/ ha wth standard error (SE).
Results and discussion
Phytosociological analysis of homegarden
In the study, altogether 992 trees from 66 species belonging to 31 families were enumerated from the selected 4 homegardens (Tables 2 and 3). Average dbh, height and basal area (cross-sectional area of a tree) of the trees were found to be 20.42 ± 2.25 cm (range 16.20–25.66 cm), 9 ± 0.47 m (range 8–10 m), 10.01 ± 0.67 m2 (range 8.44–11.71 m2) respectively (Table 2). However, there are reports on the wide variation in ecological characteristics of homegardens both in different geographic/eco-climatic regions and within a given region (Pandey et al. 2007). Although many factors were involved it, size of homegarden is one such important factor that makes a difference in the species assemblages in different systems (Kumar and Nair 2004). Moreover, these variations especially due to garden size make it difficult in comparing results of the present study with other inventories.
Species richness
The number of species varied between different homegardens and ranged from 23 to 35 with a mean of 30.75 ± 3.2 (Table 2). Combining all the plots, a total of 66 species were enumerated from the homegardens of Thodupuza. An investigation by Rugalema et al. (1994) registered 53 species from the homegardens of Tanzania whereas 60 tree species were noted from the Mexican homegardens (Guillaumet et al. 1990). In the Indian sub-continent, Shastri et al. (2002) observed 68 tree species from the homegardens of Karnataka. In Meghalaya comparatively higher value of 187 species in homegardens of War Khasi community were reported by Tynsong and Tiwari (2010). In the present study, the number of species from the homegardens of Thodupuza were found to be comparable with the range of species (30–208 species) reported from the Kerala homegardens by different researchers (Kumar et al. 1994; Kumar 2011). The result clearly indicates that homegardens are not scarce in species richness but may change in tune with the interest of farmers.
Tree density
In the case of tree density, the results (Table 2) were found slightly higher and consistent with the range of values (76–319 trees/ha) reported by most of the earlier studies in other regions of Kerala, India (Kumar et al. 1994; Kumar 2011; Kunhamu et al. 2015). However, these estimates differ vastly with the findings of several works in the north east portion of India. Devi and Das (2012) from Assam, Islam et al. (2014) and Jaman et al. (2016) from Bangladesh and have reported the occurrence of larger number of trees per hectare from these regions. Fairly high tree density in north east India compared to southern portion reflects the vast regional differences in plant density among homegardens across the tropics (Kunhamu et al. 2015). Variations in tree density can also be attributed to the non-static nature of homegardens in response to socio-economic dynamics (Peyre et al. 2006).
Endemism, Important Value Index (IVI) and exotics
Among the species identified, 4 species were found to be endemic which include Artocarpus hirsutus, Cinnamomum malabatrum, Tabernaemontana alternifolia and Litsea quinqueflora (Table 3). The occurrence of endemic species reflects biogeography of the area, center of speciation and adaptive evolution (Nayer 1996). Among the 4,679 species of flowering plants estimated in Kerala, 1637 are endemic to Southern Western Ghats of which 263 are reported to occur only in Kerala (Sasidharan 2004). On the conservation point of view, species like Dalbergia latifolia and Cycas circinalis found in the homegardens comes under vulnerable and endangered categories as per the International Union for Conservation of Nature (Asian Regional Workshop 1998; Varghese et al. 2010) (Table 3). This calls for imminent and stringent conservation strategies to avoid further extinction of above-said species and to conserve their natural habitat status. Four species (Tectona grandis, Cocos nucifera, Artocarpus hirsutus, Artocarpus heterophyllus) constituting 50% of IVI were considered as the most important tree species in the area (Table. 3). The species with highest IVI were considered as the leading dominants of the community and are best adapted to the environmental conditions in the area (Jahantigh and Efe 2010). The specific needs (timber, food value) and the preferences of the farmers in addition to easily growing nature of species might have contributed to the greater dominance (in terms of IVI) of above four species in the site.
Another important characteristic feature of homegarden was the presence of exotic species (Table 3). In the study site, 23 exotics were enumerated and these species introduced either accidently or deliberately were found to offer economic and intrinsic benefits to the livelihood needs of local communities. Meanwhile Kumar and Nair (2006) stated that spread of aggressive exotics lead to structural changes in rural bioproduction systems that could deplete floristic diversity in the homegarden. Hence landowner’s access to knowledge on valuable native tree species need to be improved to increase indigenous species diversity on homegardens and possibly to replace some of the exotic species in the future. In general the exotics along with natives make homegardens interesting sites for studies on the domestication of plant species (Blanckaert et al. 2004). The present study also recommends a need to carry out total value assessments of homegardens by considering both positive and negative benefits of exotic species for formulating and implementing sustainable management alternatives that will allow biodiversity restoration, carbon storage and economic sustainability.
Diversity indices
The diversity indices obtained in the study are depicted in Table 2. In the case of Shannon–Wiener diversity index, the results found to be comparable with the range of values (1.12–3.87) obtained in other studies conducted in different homegardens of Kerala. (Balooni et al. 2011; Kunhamu et al. 2015). Observation from Bangladesh revealed Shannon index in between 2.99 to 3.50 (Bardhan et al. 2012). Tree diversity described by the Shannon–Wiener index showed a variation between 0.76 and 3.01 with a mean value of 2.05 ± 0.07 from homegardens of Sri Lanka (Mattson et al. 2015). Shannon–Wiener index of diversity of the current study (2.73) could be compared with tropical forest index values (2.6–4.8) for the Indian sub-continent as observed by many investigators (Visalakshi 1995; Tripathi et al. 2004). This confirms that diversity of homegarden is close to, although not as high, those of forest ecosystems even though it differs with function (Mohan 2004). Evenness index value obtained in the study is in support with the observation made by Kumar and Nair (2004) from the homegardens of Kerala. Another study from Peri-urban homegardens from southern Kerala showed evenness values in between 0.83 to 0.84 (Kunhamu et al. 2015). The evenness index reported from the homegarden of Thodupuza found to be within the range of tropical forests, between 0.64–1.34 (Sahu et al. 2012). All these diversity indices support the findings by Swift and Anderson (1993) that high plant diversity in homegarden makes these systems top among all human-made agro-ecosystems after natural forest. In addition, these indices indicate the effective conservation and sustainable management of the homegarden that would provide goods and services necessary for communities inhabiting in the rural and urban areas.
Uses
In analogous to other studies, current work also recorded numerous tree species with multiple uses from the homegarden both for consumption and income from marketable items such as coffee, passion fruit and ornamentals by the homegardeners (Table 3). This is indicative of the status of homegardens as a sustainable life support systems evolved as a traditional practice. Our findings also concur with other studies that deal with socio-economic aspects of homegardens from India and other parts of the world (Kumar 2011; Mendez et al. 2001). Balooni et al. (2011) have also noted the efficiency of homegardens in complementing livelihood sustainability.
Biomass and carbon storage of homegarden
Biomass and carbon of different homegardens were estimated and listed in Table 4. Total AGB of the home garden varied from 46.93 t/ha to 65.75 t/ha whereas carbon ranged from 22.29 to 31.23 t/ha in the area. BGB estimated found to be in the range of 12.48–17.48 t/ha. Averaging the four homegarden biomass (combining the AGB and BGB) and carbon values and extrapolating into hectare area, the total tree biomass and carbon of the homegarden are found to be 67.06 ± 5.49 t/ha and 31.85 ± 2.61 t/ha. Kumar (2011) made an attempt to compute the aboveground carbon (AGC) density of homegarden trees (> 20 cm gbh) in Central Kerala, India and found that the average standing density of carbon ranged from 16 to 36 t/ha. The AGC density of the present study is well within this limit. Woodlots in Palakkad district, Kerala, India, also showed carbon density in the range of 7.8–163.2 t/ha, implying that profound species-related variations are possible in this respect (Nair et al. 2009). The results obtained in the study are comparable with studies in Sri Lanka (Mattson et al. 2015) and Philippines (Labata et al. 2012). The study conducted in homegarden from Gunugo watershed at Wolayitta zone in Ethiopia estimated the amount of carbon stored in tree biomass as 6.63 t/ha (Bajigo et al. 2015).
However, the values obtained in the current investigation are lower than the values reported from the tropical wet forest of India (Baishya et al. 2009; Mohanraj et al. 2011; Devagiri et al. 2013). This is presumably because the forest is more or less untouched natural system with larger trees and minimal disturbances whereas homegardens are managed human landscape with severe disturbances such as tillage, manual weeding and removal of trees, all of which affect the process of carbon accumulation (Kumar and Nair 2006).
Species-wise contribution of carbon storage and its relationship with other tree variables
Here, home gardens with trees having ≥ 10 cm dbh were considered for determining the dominant carbon storing species. For this, all the homegardens were combined (Total area–4 ha) and species-specific volume and specific gravity relationship was used for this category to compute the biomass carbon. Whereas in the case of trees with < 10 cm diameter, regression equations were employed to estimate carbon, hence it was not possible to find out species-specific carbon in this range of trees. Among the 47 species identified from the class of ≥ 10 cm diameter, species Tectona grandis had carbon storage of 22% followed by Artocarpus hirsutus (18%), Cocos nucifera (18%) and Artocarpus heterophyllus (16%). From this estimate, it was understood that these four species store substantial portion of carbon (74%) in their biomass making a prominent carbon storing species in the homegarden system of Thodupuza (Fig. 2). The greater carbon storage in these four species may be attributed to the role played by tree density and sufficient size of these species in the region. This is further clarified through correlation analysis revealing the relationship between carbon and other tree variables (tree density and basal area). By considering all the species (by taking all 47 species together from 4 plots) in the homegarden and examining the relationship, the analysis found that tree carbon had a strong positive correlation with tree density (R2 = 0.85, p < 0.001) and basal area (R2 = 0.96, p < 0.001) (Fig. 3 a, b). This further establishes our conjecture that tree size (basal area) and stand density is having a significant role in maintaining species-wise carbon within the system. Aboveground carbon storage considered to be the important indicator of forest structure, can be indicated by the relationship between tree density and tree size (Nagendra 2012). Furthermore, our present understanding of these relationships can help to decide what species and species combinations are the most suitable for maximizing carbon sequestration.
Plot (HG)-wise correlation between carbon and other tree variables
Plot (HG)-wise relationship between carbon and other tree variables by combining all the trees in each plot (≥ 3 cm dbh) showed that tree carbon had a strong positive relationship with basal area in the site (Fig. 4a). Since basal area integrates the effect of both the number and size of trees, it is considered as a surrogate for biomass and carbon (Burrows et al. 2000; Ali et al. 2014). Therefore a change in the total stem area occupied by the tree during growth strongly affects the C density of the system. Apart from this, researchers from India and abroad had also indicated the positive relationship between carbon and basal area especially from forest areas (Slik et al. 2010; Chaturvedi et al. 2011).
Unlike in early observation (species-wise) (Fig. 3a) where a strong positive correlation was observed with tree density, plot (HG)-wise analysis indicated a weak negative relationship between carbon and tree density (Fig. 4b). This might be due to the inclusion of more young trees (3 cm to 9 cm dbh) in the homegarden that may have swayed the overall relationship. In the present study, the dominance of young trees is having a pivotal role in shaping the overall tree density of homegardens (Fig. 5). Therefore the contribution of tree density towards carbon storage may not be encouraging which may leads to weak or poor relationship in homagarden.
The result also implies that the increase in tree density does not guarantee a corresponding increase in carbon moreover the ecosystem indicator, such as carbon density, is not determined solely by the number of trees, but are more likely to be determined by the size characteristics of the tree species existing in the area. It is reported that the carbon storage of a tree is directly proportional to its stem size (Rutishauser et al. 2010; Feldpausch et al. 2011; Shirima et al. 2015), and therefore the total carbon of homegarden or a system is mainly controlled by density of large trees instead of young trees (low gbh) or overall tree density. According to Pragasan (2020), the sum of trees in larger stem size category is generally influencing the total carbon storage of a forest compared to total tree density. Another study by Slik et al. (2013) that used a pan-tropical tree inventory data set from 120 lowland tropical moist forest locations observed 70% of the site variation in AGB contributed mainly by the density of large trees (dbh > 70 cm) while paucity of large trees in Neotropical forest resulted in less carbon storage (30% less biomass carbon) than Palaeotropical forests. Hence the density of large trees having maximum tree size can be considered as major drivers of variation in AGB in an ecosystem. The current finding is consistent with the idea that the carbon storage in tree biomass increases with gbh or dbh, basal area, height but not with tree density (Hui et al. 2012) among homegardens.
Species richness showed a moderate negative relationship with carbon (Fig. 4c). However, a great deal of uncertainty still exists in the relationship with tree diversity as most of the studies were unable to establish any tight link between diversity and carbon storage. As per Soto-Pinto et al. (2010) coffee agroforestry system with two shade trees had large carbon storage than diverse polyculture. Mandal et al. (2013) study from Nepal also found a weak positive relationship between carbon and diversity and suggests that increase in forest carbon density or stock doesn’t/cannot assure the biodiversity conservation and promotion. Henry et al. (2009) failed to establish any kind of relationship between carbon and diversity in different land uses of Kenya. However, some authors found that species diversity tends to have a negative relationship with carbon sequestration in forest ecosystem (Sharma et al. 2010; Zhang et al. 2011). The results of the current inventory are also in conformity with the above findings showing the decrease in species diversity with increasing carbon density. This can be due to the prominent role played by the morphological characteristics of species in determining stand carbon than diversity in these homegardens (Zhang et al. 2011). Few other studies mentioned that a gradual increase in biomass lead to competition which may decrease species diversity leaving few dominant ones in the area (Keddy 2005; Healy et al. 2008). According to Horn (1974) succession arising out of disturbances lead to more number of stems, and a mix of early and late successional species are considered to be one of the reasons underlying the negative relationship between these two variables. This observation becomes much stronger in the case of homegardens where frequent interference always exists at various scales. Since homegarden is a fully managed ecosystem there can be a species difference in terms of structure and composition as some tree species grows first and another is incorporated later based on the need and preference by the farmers. All these species may not have sufficient size in determining the carbon storage of the system. This is actually upsetting the natural balance of the ecosystem which will make it difficult to establish a clear pattern in the relationship of these variables in the system. Taking account of many factors like regeneration, succession, disturbance, sample size, time and region etc. a comprehensive study considering the above-mentioned factors is essential in order to improve our understanding of these observed relationships.
Distribution of carbon, number of trees, number of species and basal area with respect to diametric classes
The work also examined the variation in carbon in different diameter classes along with other tree variables in the homegarden. According to Baishya et al. (2009) tree carbon and its distribution in different girth (diameter) class is considered to be an important factor when examining vegetation carbon of area. At the site, tree carbon increased with an increase in the dbh up to 20–29 cm before gradually decreasing at greater dbh upto 70–79 cm (Fig. 5), beyond which no further species or trees were recorded in the homegarden.
An almost similar pattern (positively skewed) as that of C was observed in the case of basal area (Fig. 5). As basal area incorporates both number and girth of the tree, the higher basal area found in this particular class (20–29 cm dbh) is leading to maximum carbon storage in this diameter category. Large number of trees (tree density) act as the leading factor for the higher basal area in this particular diametric class. The research also found that major portion of the basal area in this particular class (20–29 cm dbh) is contributed by Cocos nucifera (Fig. 6) compared to rest of the species suggesting the role played by trees of monocrop in maintaining the carbon storage of the system. From the study it can concluded that the smaller or medium trees are having foremost role in contributing major share of carbon towards the total carbon storage of the homegarden unlike in forest where large trees (> 70 cm dbh) were found to be holding maximum carbon storage (Baishya et al. 2009) of the system. This discrepancy in two systems could be due to the effect of selective logging of dominant species in the higher diameter classes for various purposes in the homegarden.
The diameter distribution of tree density (number of trees) has often been used to represent the stand structure of a forest as well as a complex agroforestry system (Khan et al. 1987). As observed in many other studies especially from forest areas, the pattern of diameter class distribution of number of trees descending exponentially with increasing dbh is considered to be the major indicator of species with continuous regeneration (Tripathi et al. 2004). Although very little information exists in the regeneration dynamics of homegardens, Kumar et al. (1994) study from Kerala revealed a slightly skewed (positive) distribution pattern having the maximum number of trees in the 20–30 cm dbh class indicating the adequate regeneration of homegarden species. In accord with preceding observation the findings from the present exploration also showed that the homegarden found to have some kind of regeneration potential where the density of the trees drops with an increase in diameter (Fig. 5).
Apart from regeneration, the present study has a different opinion regarding the skewed type of distribution in the case of tree density and carbon storage. The present research presumes that skewed type of distribution can also arise due to disturbance occurring at both stages (i.e. young stage and mature stage) of tree development in the homegarden. In the current investigation, there occurs a low density of trees in the < 10 cm dbh class with subsequent classes showing a downhill trend from 10–19 diameter class. This suggests that in the managed system, natural recruitment of young ones is very much restrained as farmers target species that serve multiple provisioning services such as timber, food, firewood etc. (De Clerck and Negreros-Castillo 2000). For the harvest of commercial products such as coconut, arecanut, rubber and other products and also to have a constant attention of the whole area farmers try to keep their homegarden less dense by removing especially younger stands of unwanted individuals or overcrowding individual of species like Macaranga peltata, Swietenia macrophylla, Ailanthus excelsa. This is in line with the observation made by Saikia and Khan (2013) that maintenance of homegardens stands to be a one of the reasons for the poor survival of seedlings. Grazing of domestic animals will also have a strong impact on the structure and composition of the vegetation in the area. At the same time, larger trees are also extremely valuable, as they are harvested and converted into economic products. So in the case homegarden, disturbance is very evident both at younger as well as at a mature stage of tree development. Although species generation is strongly influenced by both natural and anthropogenic factors (Barik et al. 1996; Tripathi and Khan 2007), it’s the man-made interference that is acting as the dominant player which is adversely affecting the survival of individuals of species in homegardens.
Even though the sufficient natural regeneration status of trees is lacking in the homegardens unlike in forest ecosystems, farmers try to accommodate maximum species in relative small numbers based on the utilitarian value which they considered as important for their subsistence and livelihood in the area (Kumar and Nair 2011). This actually reflects the species wise distribution in different diameter classes of trees with maximum species noticed in the younger classes with respect to other large classes. The trend of decreasing species with increasing diameter obtained in the present study (Fig. 5) is in conformity with works of Prasad et al. (2007) and Rajkumar and Parthasarathy (2008). When it comes to the selection of tree, the farmer depends on many factors that include economic aspects, religious and cultural beliefs, customs, food, environmental benefits and taboos of the villagers which may influence the diversity/composition of homegardens (Millat-e-Mustafa et al. 1996). Market demand and profitability of species prompt farmers to go for a new species in the area (Kumar and Nair 2011). All these practices make the homegarden much more complex in terms of species diversity making it as a man-made feature, unlike in natural systems (Kumar and Nair 2006).
Different plant groups and carbon
As homegarden is a heterogeneous land use type with a mix of cultivated and native/wild species, the present study classified the entire species (≥ 10 cm dbh trees) into two main classes such as native and cultivated and evaluated its carbon holding capacity with respect to other tree parameters (number of tree species, tree density, dbh, height and basal area). The cultivated trees include plantation (monocrops), ornamentals and exotic species whereas natives consist rest of the species including wild categories. The result suggests that cultivated species had verge over native species in almost all parameters except dbh and height (Table 5). This may be due to the wide variance in stand density that makes two groups highly distinguishable and has an impact on their carbon density (Table 5). In the case of cultivated species tree density had a prominent role in carbon storage along with diameter and height whereas in the case of native, it is the huge size of trees that is playing a vital role along with tree density. Huge size that incorporates both diameter and height can be the reason for the carbon storage of native trees that found closer (eventhough it is lesser) to the cultivated one irrespective of their (native) low tree density. This once again reiterates the role played by tree density, diameter and height in carbon storage of trees within the system. However, still there is a lot of uncertainty exists in the carbon accumulation rates of different species or plant groups (native vs. cultivated) in the agroforestry system (Nair et al. 2010). Moreover, if tree density difference between these two groups gets reduced or become much closer to one another then the dominancy in carbon accumulation and also the role of underscoring factors like tree density, dbh, height and basal area in determining C storage become more uncertain in the homegarden with respect to these two groups. Therefore it is quite ambiguous whether native or cultivated would be more superior in terms of carbon sequestration in the homegarden. Fast growing species mostly cultivated may accumulate more C within a short-term before they were harvested but slower-growing accumulates more C in the long-term (Redondo-Brenes 2007). The researchers are of opinion that higher specific gravity found in slow-growing species increase carbon sequestration potential in long-term that constitutes longer-term sink for stable carbon in the form of timber, furniture, wooden crafts material than low wood density (specific gravity) trees used for short-lived purposes such as packaging cases and poles (Bunker et al. 2005). According to Schroth et al. (2011) high and long-term accumulation of biomass with the early generation of income from annual and perennial intercrops is considered to be the characteristic feature of agroforestry. A detailed analysis covering various aspects of the homegarden can throw light in this regard which will become beneficial for the designing of management strategies for these socio-ecological systems.
Conclusion
Homegarden would be a simple and innovative approach to deal with the problems arising due to anthropogenic activities and unforeseen weather events that are critical to the functioning of the urban ecosystems. The present study sheds light on the importance of homegarden in biodiversity conservation and also in mitigating climate change for which solutions are being widely discussed around the world. The study from the homegarden system of the Thodupuza urban region, Kerala, India, holds significance in the current scenario as the quantification tree diversity and carbon accruals in these systems will provide a strong basis for management programmes prioritizing biodiversity conservation and maximum carbon sequestration which will strengthen conservation efforts in the hotspot (Southern Western Ghats) and thus enhancing regional contribution towards the protection of biodiversity as well as offsetting of CO2, a major greenhouse gas.
Our result confirms that homegarden is a more or less integrated farming system which has the potential of retaining more species despite the addition of many cultivated species. This clearly indicates that this agroforestry system can maintain a large number of species outside their native forest habitat. Besides the large number native species in comparison to exotics, presence of endemic and vulnerable species in the system calls for adequate conservation strategies to avoid further extinction of these important species in the context of climate change and other land disturbances. The diversity indices obtained in the study suggest that among all managed agroecosystems, homegarden is found to be the most important after natural forest. From the analysis, it can be concluded that the biomass and carbon storage of the homegarden is quite substantial, which is agreeing with earlier works. Results also showed that species with high carbon accumulation rates can well be considered for enhancing carbon sequestration in the area. As the study failed to establish any tight link between carbon storage with species richness or density, the basal area found to be a better predictor of carbon in the homegarden, but all relationship may vary with conditions of the garden. Skewed typed distribution of carbon and tree density in the diameter class suggests the kind of disturbance happening in the system. In contrary to this observation the numbers of species recorded high in lower diameter classes with a drop in the subsequent higher classes indicate the interest shown by the farmers in planting new species. The huge difference in stand density of two groups (native vs cultivated) has an influence on their carbon holding capacity that results in slight dominance of cultivated group over native in carbon storage in the homegarden. However, the data presented here support only a tentative conclusion, because other factors which include vegetation other than trees (shrub, herb, sapling and seedling), deadwood, litter, soil, climatic and socio-economic factors might influence phytosociology and carbon storage capacity of the ecosystem.
By employing various kinds of plant diversity measures the present study tries to enhance the understanding of how biodiversity measures and carbon storage are related in a homegarden agroforestry system that are critical for the designing of more sustainable environmental policies and developmental planning in urban ecosystems. There exists a dearth of information regarding the relationship between biodiversity and carbon storage in homegarden especially from an urban perspective. Despite the significant environmental changes that humans have caused in an urban environment, the study shows that homegardens still contain many plant species and forms that possess the potential to provide and influence various ecosystem services including carbon sequestration. However, that does not assure the simplistic assumption that higher biodiversity leads to more biomass especially in a managed agroforestry systems like homegarden. Future studies considering various aspects may provide further details into the role of homegarden systems in biodiversity conservation and carbon sequestration. In short, results provide insights for decision-makers and the public to better understand the role of homegardens in conserving biodiversity as well as reducing atmospheric CO2 in the urban regions and make better management plans and policies that can significantly improve environmental quality for these ecological sustainable systems.
References
Alberti M (2010) Maintaining ecological integrity and sustaining ecosystem function in urban areas. Curr Opin Env Sust 2(3):178–184
Ali A, Ma WJ, Yang XD, Sun BW, Shi QR, Xu MS (2014) Biomass and carbon stocks in Schima superba dominated subtropical forests of eastern China. J For Sci 55060:198–207
Asian Regional Workshop (1998) Asian Regional Workshop (Conservation & Sustainable Management of Trees, Viet Nam, August 1996) Dalbergia latifolia. IUCN Red List Threat Species. https://doi.org/10.2305/IUCN.UK.1998.RLTS.T32098A9675296.en
Baishya R, Barik SK, Upadhaya K (2009) Distribution pattern of aboveground biomass in natural and plantation forests of humid tropics in northeast India. Trop Ecol 50:295–304
Bajigo A, Tadesse M, Moges Y, Anjulo A (2015) Estimation of carbon stored in agroforestry practices in Gununo watershed, Wolayitta Zone, Ethiopia. J Ecosyst Ecograph 5:57
Balooni K, Gangopadhyay K, Kumar BM (2011) Urban sustainability and changing private green spaces: some insights from an Indian City, Asia Research Institute, Working Paper Series No. 169
Bardhan S, Jose S, Biswas S, Kabir K, Rogers W (2012) Homegarden agroforestry systems: an intermediary for biodiversity conservation in Bangladesh. Agrofor Syst 85:29–34
Barik SK, Tripathi RS, Pandey HN, Rao P (1996) Tree regeneration in a subtropical humid forest: Effect of cultural disturbance on seed production, dispersal and germination. J Appl Ecol 33:1551–1560
Blanckaert I, Swennen RL, Paredes-Flores M et al (2004) Floristic composition, plant uses and management practices in Homegardens of San Rafael Coxcatlán, Valley of Tehuacán-Cuicatlán, Mexico. J Arid Environ 57:39–62
Bunker DE, DeClerk F, Bradford JC, Colwell RK, Perfecto Y, Phillips OL, Sankaran M, Naeem S (2005) Species loss and above-ground carbon storage in a tropical forest. Science 310:1029–1031
Burrows WH, Hoffmann MB, Compton JF, Back PV, Tait LJ (2000) Allometric relationships and community biomass estimates for some dominant eucalypts in central Queensland woodlands. Aust J Bot 48:707–714
Cairns MA, Brown S, Helmer EH, Baumgartner GA (1997) Root biomass allocation in the world’s upland forests. Oecol 111:1–11
Chaturvedi RK, Raghubanshi AS, Singh JS (2011) Leaf attributes and tree growth in a tropical dry forest. J Veg Sci 22(5):917–931
Climate-data. Org (2018) Climate: Thodupuza. https://en.climatedata.org/asia/india/kerala/thodupuzha-53338/
Dadhwal VK, Singh S, Patil P (2009) Assessment of phytomass carbon pools in forest ecosystems in India. NNRMS Bulletin. Indian Institute of Remote Sensing, Dehradun, pp 41–47
De Clerck FAJ, Negreros-Castillo P (2000) Plant species of traditional Mayan Homegardens of Mexico as analogs for multi strata agro forests. Agrofort Syst 48:303–317
Devagiri GM, Money S, Singh S, Dadhawal VK, Patil P, Khaple AAS, Deva Kumar AS, Hubballi S (2013) Assessment of above ground biomass and carbon pool in different vegetation types of south western part of Karnataka, India using spectral modeling. Trop Ecol 54(2):149–165
Devi NL, Das AK (2012) Diversity and utilization of tree species in Meitei Homegardens of Barak valley, Assam. J Env Biol 34:211–217
DUR (2011) District urbanization report (DUR) of Idukki district, Kerala. Department of Town and Country Planning, Government of Kerala, India
Egerer M, Ossola A, Lin BB (2018) Creating socioecological novelty in urban agroecosystems from the ground up. Bioscience 68(1):25–34
Feldpausch TR, Banin L, Phillips OL et al (2011) Height-diameter allometry of tropical forest trees. Biogeosciences 8:1081–1106
Fernandes ECM, Nair PKR (1986) An evaluation of the structure and function of tropical homegardens. Agrofor Syst 21:279–310
FSI (1996) Volume Equations for Forests of India, Nepal and Bhutan. Forest Survey of India, Dehradun
Gbedomon RC, Salako VK, Fandohan AB, Idohou AFR, Kakaї RG, Assogbadjo AE (2017) Functional diversity of home gardens and their agrobiodiversity conservation benefits in Benin, West Africa. J Ethnobiol Ethnomed 13(1):66
Grove JM, Childers DL, Galvin M, Hines S, Muñoz-Erickson T, Svendsen ES (2016) Linking science and decision making to promote an ecology for the city: practices and opportunities. Ecosyst Health Sustain 2(9):
Government of Kerala (2016) Economic Review 2016. State Planning Board, Government of Kerala, Thiruvananthapuram
Government of Kerala (2018) Economic review 2018. State Planning Board, Government of Kerala, Thiruvananthapuram
Guillaumet JL, Grenand P, Bahri S, Grenand F, Lourd M, dos Santos AA, Gely A (1990) The homegarden orchards of the central Amazon: an example of space utilization. Turrialba 40(1):63–81
Healy C, Gotelli NJ, Potvin C (2008) Partitioning the effects of biodiversity and environmental heterogeneity for productivity and mortality in a tropical tree plantation. J Ecol 96:903–913
Henry M, Tittonell P, Manlay RJ, Bernoux M, Albrecht A, Vanlauwe B (2009) Biodiversity, carbon stocks and sequestration potential in aboveground biomass in smallholder farming systems of western Kenya. Agric Ecosyst Environ 129:238–252
Horn HS (1974) The ecology of secondary succession. Annu Rev Ecol Syst 5:25–37
Hui D, Wang J, Le X, Shen W, Ren H (2012) Influences of biotic and abiotic factors on the relationship between tree productivity and biomass in China. For Ecol Manag 264:72–80
ICFRE (1996–2002). Indian woods: their identification, properties and uses. Volumes I–VI, Revised Edition, Indian Council of Forestry Research and Education, Dehradun, India
Islam M, Dey A, Rahman M (2014) Effect of Tree Diversity on Soil Organic Carbon Content in the homegarden agroforestry System of North-Eastern Bangladesh. Small Scale For 14(1):91–101
Jahantigh M, Efe R (2010) Effect of waste water irrigation on phytosociological characteristics of the vegetation: a case study in Sistan Region. Amer-Euras J Agric Environ Sci 7(4):406–414
Jaman MS, Hossain MF, Islam MS, Helal MGJ, Jamil M (2016) Quantification of carbon stock and tree diversity of homegardens in Rangpur District, Bangladesh. Int J Agric For 6:169–180
Keddy P (2005) Putting the plants back into plant ecology: six pragmatic models for understanding and conserving plant diversity. Ann Bot 96:177–189
Khan ML, Rai JPN, Tripathi RS (1987) Population structure of some tree species in disturbed and protected subtropical forests of North-East India. Acta Oecol 8:247–255
Kumar BM (2006) Carbon sequestration potential of tropical Homegardens. In: Nair PKR, Kumar BM (eds) Tropical Homegardens: a time-tested example of sustainable agroforestry. Springer, Berlin, pp 185–204
Kumar BM, Nair PKR (eds) (2006) Tropical homegardens: A Time-tested example of sustainable agroforestry. Advances in Agroforestry. Springer Science, Dordrecht
Kumar BM (2011) Species richness and aboveground carbon stocks in the homegardens of central Kerala, India. Agric Ecosyst Environ 140:430–440
Kumar BM, Nair PKR (2004) The enigma of tropical homegardens. Agrofor Syst 61:135–152
Kumar BM, Nair PKR (2011) Carbon sequestration potential of agroforestry systems: opportunities and challenges. Springer, Berlin
Kumar BM, George SJ, Chinnamani S (1994) Diversity, structure and standing stock of wood in the homegardens of Kerala in Peninsular India. Agrofor Syst 25:243–262
Kunhamu TK, Ajeesh R, Kumar V (2015) Floristic analysis of peri-urban homegardens of Southern Kerala. Indian J Ecol 42:2
Labata MM, Aranico EC, Tabaranza ACE, Patricio JHP, AmparadoJr RF (2012) Carbon stock assessment of three selected agroforestry systems in Bukidnon, Philippines. Adv Environ Sci 4:5–11
Magurran AE (2004) Measuring biological diversity. Blackwell Science, Oxford
Mandal RA, Dutta IC, Jha PK, Karmacharya SB (2013) Relationship between carbon stock and plant biodiversity in collaborative forests in Tarai, Nepal. ISRN Botany, Article ID 625767
Mattson E, Ostwald M, Nissanka SP, Pushpakumara DKNG (2015) Quantification of carbon stock and tree diversity of home gardens in a dry zone area of Moneragala district, Sri Lanka. Agrofor Syst 89(3):435–445
Mendez VE, Lok R, Sommarriba E (2001) Interdisciplinary analysis of Homegardens in Nicaragua: micro-zonation, plant use and socioeconomic importance. Agrofor Syst 51:85–96
Millat-e-Mustafa MD, Hall JB, Teklehaimanot Z (1996) Structure and floristics of Bangladesh Homegardens. Agrofor Syst 33:263–280
Mohan S (2004) An Assessment of the ecological and socioeconomic benefits provided by the homegardens: a case study from Kerala, India. PhD Dissertation, University of Florida, Gainesville, FL, USA
Mohanraj R, Saravanan J, Dhanakumar S (2011) Carbon stock in Kolli forests, Eastern Ghats (India) with emphasis on aboveground biomass, litter, woody debris and soils. iForest 4:61–65
Muraleedharan PK, Sasidharan N, Kumar BM, Sreenivasan MA, Seethalakshmi KK (2005) Non-wood forest products in the Western Ghats of Kerala, India: floristic attributes extraction, regeneration and prospects for sustainable use. J Trop For Sci 17(2):243–257
Murthy IK, Gupta M, Tomar S, Munsi M, Tiwari R, Hegde GT, Ravindranath NH (2013) Carbon sequestration potential of agroforestry systems in India. J Earth Sci Clim Change 4(1):1–7
Myers N, Mittermeier RA, Mittermeier CG, de Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Nagendra H (2012) Assessing relatedness and redundancy of forest monitoring and change indicators. J Environ Manag 95(2012):108–113
Nair PKR (2001) Do tropical Homegardens elude science or is it the other way around? Agrofor Syst 53:239–245
Nair PKR, Kumar BM, Nair VD (2009) Agroforestry as a strategy for carbon sequestration. J Plant Nutr Soil Sci 172:10–23
Nair PKR, Nair VD, Kumar BM, Showalter JM (2010) Carbon sequestration in agroforestry systems. Adv Agron 108:237–307
Nayar MP (1996) “Hot Spots” of Endemic Plants of India, Nepal and Bhutan. Tropical Botanic Garden and Research Institute, Palode
Nayar TS, Rasiya BA, Sibi M (2014) Flowering plants of the Western Ghats, India (2 Volumes). Jawaharlal Nehru Tropical Botanic Garden and Research Institute, Palode
Pandey DN (2002) Carbon sequestration in agroforestry systems. Clim Policy 2:367–377
Pandey CB, Rai RB, Singh L, Singh AK (2007) Homegardens of Andaman and Nicobar, India. Agric Syst 92(1–3):1–22
Peyre A, Guidal A, Wiersum KF, Bongers F (2006) Dynamics of homegarden structure and function in Kerala, India. Agrofor Syst 66:101–115
Pielou EC (1975) Ecological Diversity. John Wiley & Sons, New York
Pragasan LA (2020) Tree carbon stock and its relationship to key factors from a tropical hill forest of Tamil Nadu, India. Geol Ecol Landsc. https://doi.org/10.1080/24749508.2020.1742510
Prasad PRC, Reddy CS, Dutt CBS (2007) Phytodiversity assessment of tropical rainforest of North Andaman Islands. India J of For 1(1):27–39
Raghubansi AS, Jha CS, Pandey CB, Singh L, Singh JS (1990) Effects of forest conversion on vegetation and soil carbon and functional trait of resulting vegetation. Impact of Global Climate Change on Photosynthesis and Plant Productivity, pp 723–749
Rajkumar M, Parthasarathy N (2008) Tree diversity and structure of Andaman Giant Evergreen Forests, India. Taiwania 53:356–368
Ravindranath NH, Ostwald M (2008) Carbon Inventory methods handbook for greenhouse gas inventory, carbon mitigation and round wood production projects. Springer, Heidelberg
Redondo-Brenes A (2007) Growth, carbon sequestration and management of native tree plantations in humid regions of Costa Rica. New For 34(3):253–268
Rugalema GH, Okting AA, Johnson FH (1994) The homegarden agro-forestry System of Bukoba District. North West Tanzan Farm Syst 26:53–64
Rutishauser E, Wagner F, Herault B, Nicolini EA, Blanc L (2010) Contrasting above-ground biomass balance in a Neotropical rain forest. J Veg Sci 21(4):672–682
Sahu SC, Dhal NK, Lal B, Mohanty RC (2012) Differences in tree species diversity, distribution and population structure in a tropical dry deciduous forest of Malayagiri hill range, Eastern Ghats, India. J Mount Sci 9:492–500
Saikia P, Khan ML (2013) Population structure and regeneration status of Aquilaria malaccensis L. am in Homegardens of Upper Assam, northeast India. Trop Eco 54(1):1–13
Sanyé-Mengual E, Gasperi D, Michelon N, Orsini F, Ponchia G, Gianquinto G (2018) Eco-efficiency assessment and food security potential of home gardening: a case study in Padua, Italy. Sustainability 10:2124
Sasidharan N (2004) Biodiversity documentation for Kerala. Part 6: flowering plants. Kerala Forest Research Institute, Peechi
Schroth G, da Mota MSS, Hills T, Soto-Pinto L, Wijayanto I, Arief CW, Zepeda Y (2011) Linking carbon, biodiversity and livelihoods near forest margins: the role of agroforestry. In: Kumar BM, Nair PKR (eds) Carbon sequestration potential of agroforestry systems: opportunities and challenges. Springer, Dordrecht, pp 179–200
Seto KC, Güneralp B, Hutyr LR (2012) Global forecasts of urban expansion to 2030 and direct impacts on biodiversity and carbon pools. Proc Natl Acad Sci 109(40):16083–16088
Shannon CE, Wiener W (1949) The mathematical theory of communication. University of Illinois Press, Urbana, IL
Sharma CM, Baduni NP, Gairola S, Ghildiyal SK, Suyal S (2010) Tree diversity and carbon stocks of some major forest types of Garhwal Himalaya, India. Forest Ecol Manag 260(12):2170–2179
Shastri CM, Bhat DM, Nagaraja BC, Murali KS, Ravindranath NH (2002) Tree species diversity in a village ecosystem in Uttara Kannada district in Western Ghats, Karnataka. Curr Sci 82:1080–1084
Shirima DD, Totland Ø, Munishi PK, Moe SR (2015) Relationships between tree species richness, evenness and aboveground carbon storage in montane forests and miombo woodlands of Tanzania. Basic Appl Ecol 16(3):239–249
Slik JWF, Aiba SI, Brearley Q, Cannon CH, Forshed O, Kitayama K, Nagamasu H, Nilus R, Payne J, Paoli G, Poulsen AD, Raes N, Sheil D, Sidiyasa KE, Suzuki E, Van Valkenburg JLCH (2010) Environmental correlates of tree biomass, basal area, wood specific gravity and stem density gradients in Borneo’s tropical forests. Global Ecol Biogeogr 19:50–60
Slik JF, Paoli G, McGuire K, Amaral I et al (2013) Large trees drive forest aboveground biomass variation in moist lowland forests across the tropics. Glob Ecol Biogeogr 12:1261–1271
Soto-Pinto L, Anzueto M, Mendoza J, Ferrer G, de Jong B (2010) Carbon sequestration through agroforestry in indigenous communities of Chiapas, Mexico. Agrofor Syst 78:39–51
Swift MJ, Anderson JM (1993) Biodiversity and ecosystem function in agroecosystems. In: Schultze E, Mooney HA (eds) Biodiversity and ecosystem function. Spinger, New York, pp 57–83
Tripathi RS, Khan KL (2007) Regeneration dynamics of natural forests: a review. Proc Indian Natl Sci 73:167–195
Tripathi KP, Tripathi S, Selvan T, Kumar K, Singh KK, Mehrotra S, Pushegadan P (2004) Community structure and species diversity of Saddle Peak forest in Andaman Island. Trop Ecol 45:241–250
Tynsong H, Tiwari BK (2010) Plant diversity in the homegardens and their significance in the livelihoods of War Khasi community of Meghalaya, North-East. India J Biodivers 1(1):1–11
Unnithan SR, Kunhamu TK, Sunanda C, Anoop EV, Jamaludheen V, Santhoshkumar AV (2017) Floristic diversity and standing stock of timber in homegardens of Thrissur district, Kerala. Indian J Agrofor 19(2):8–12
Varghese A, Krishnamurthy V, Garnesan R, Manu K (2010) Cycas circinalis. IUCN Red List Threat Species. https://doi.org/10.2305/IUCN.UK.2010-3.RLTS.T42089A10627275.en
Visalakshi N (1995) Vegetation analysis of two tropical dry evergreen forests in southern India. Trop Ecol 36(1):117–127
Whittaker RH (1970) Communities and ecosystems. McMillan, New York
Zhang Y, Duan B, Xian J, Korpelainen H, Li C (2011) Links between plant diversity, carbon stocks and factors along a successional gradient in a subalpine coniferous forest in Southwest China. Forest Ecol Manag 262:361–369
Acknowledgements
We gratefully acknowledge the funding from the Indian Institute of Remote Sensing (IIRS), Dehradun, Indian Space Research Organisation (ISRO), Department of Space and Government of India. Authors acknowledge Directorate of Environment and Climate Change, Government of Kerala for the support extended for this work. We are grateful to members of the School of Environmental Sciences for assisting us during data collection. Authors also thank Forest Department, Government of Kerala for their service in connection with the work. We are also thankful to Dr Sarnam Singh for his invaluable contributions to the work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Padmakumar, B., Sreekanth, N.P., Shanthiprabha, V. et al. Unveiling tree diversity and carbon density of homegarden in the Thodupuzha urban region of Kerala, India: a contribution towards urban sustainability. Trop Ecol 62, 508–524 (2021). https://doi.org/10.1007/s42965-021-00149-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42965-021-00149-2