Abstract
Predicting the impact of climate change on species distribution at different spatial and temporal scales has emerged as one of the important areas of research in invasion ecology and conservation biology. We used MaxEnt (Maximum Entropy Algorithm) to predict the distribution of a highly invasive species, namely Parthenium hysterophorus L. under four Representative Concentration Pathway scenarios (RCPs 2.6, 4.5, 6.0 and 8.5) in 2050 and 2070 at global (world), regional (India) and local (Jammu & Kashmir State) spatial scales. Model predictions indicated differences in the extent of expansion in the distribution of this species under different climate change scenarios with marginal increase in moderately suitable area at the global scale but mostly a declining trend was noticed in its suitable and highly suitable area in future. More or less similar trend was predicted for India where increase in moderately suitable area was evident but decline in suitable and highly suitable areas was observed. In respect of Jammu & Kashmir, moderately suitable as well suitable area showed increase mostly under RCP scenarios of 6.0 and 8.5 in 2050 as well as 2070. Further analysis revealed that current centroid of P. hysterophorus is in south of Jammu and Kashmir and is predicted to shift by an average of 20.48 km in the north-west direction by 2050 and by 36.83 km by 2070. The future suitable area is likely to be around Hirapora Wildlife sanctuary in Kashmir. Pairwise comparison of the niche overlap and dynamics of P. hysterophorus between the native Americas and each of the regions (Africa, Asia, Australia and Oceania) where the species is introduced using Schoener’s D revealed variations in the niche overlap which was high between native Americas and Australia (0.70) and Africa (0.69), moderate between Americas and Asia (0.59) and low between Americas and Oceania (0.24). Exclusion of 25% of rare climatic conditions did not have any effect on the niche overlap index (D). Niche similarity test was not significant for any of the pairwise comparisons of native Americas and the continents in which the species is non-native indicating that the native niche is more similar to the exotic niche than any randomly sampled niche from the exotic range. But the niche equivalency tests showed that the environmental realized niche of P. hysterophorus in its invaded range was not totally equivalent to that in the native range indicating niche differentiation. The niche dynamic indices based on analogous and the entire climatic space in the native and introduced regions revealed a very high niche stability. A very limited niche expansion was noticed only in Asia and niche unfilling was evident in Oceania. Like niche overlap index (D), niche expansion and niche stability were not affected by the exclusion of 25% of rare climatic conditions but marginal change was noticed in niche unfilling in the Oceania. The above predictions have implications for formulation of policies at local, regional and global level for the management of this invasive species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Among the plethora of impacts of climate change on flora and fauna, change in the distributional range of the species has been well reported (Gomes et al. 2018; Lamsal et al. 2018; Adhikari et al. 2019; Ahmad et al. 2019; Shrestha and Shrestha 2019). While some species may show poleward shift in their distribution, in others upward altitudinal shift in response to climate change has been reported (Pauchard et al. 2016). Of particular interest in this context are the alien (non-native/exotic) species whose fortunes may also change with the change in the climate (Bradley et al. 2010). Predicting future distribution of species over relevant spatio-temporal scales under various climate change scenarios is of particular significance in the management of invasive alien species (Porfirio et al. 2014; Fletcher et al. 2016; Chai et al. 2016). Consequently, in the last two decades several modelling tools (Elith et al. 2010; Fourcade et al. 2014; Thuiller et al. 2016; Di Cola et al. 2017) have been developed for predicting future distribution of species on the basis of current correlation between climatic variables and species occurrence records, assuming niche conservatism, no dispersal limitation or biotic interactions (Ramirez-Albores et al. 2016). Such models have become popular in predicting the distribution of invasive species (Bradley et al. 2010; Tingley et al. 2014; Kramer et al. 2017; Barbet-Massin et al. 2018), especially under changing climate conditions (Bellard et al. 2018; Ahmad et al. 2019). Changes in species distributions are likely to affect structural organization and functional integrity of communities and ecosystems with unforeseeable ecological and economic impacts and costs (Hughes 2000).
Understanding of niche dynamics in the native and introduced regions using the COUE framework of Centroid shift, Overlap, Unfilling, and Expansion (Broennimann et al. 2012; Petitpierre et al. 2012) has received lot of attention in the recent past. Results of such studies have been variable (Thuiller et al. 2005) with some species showing niche conservatism (Liu et al. 2020) and others exhibiting niche shift (Atwater et al. 2018). Based on 86 studies dealing with 434 invasive species, Liu et al. (2020) concluded that most invasive species largely conserve their climatic niche and niche conservatism not only allows for transfer of niche models to new ranges but also provides for use of ecological niche modelling in understanding the response of species to climate change.
Climate change that is driven by anthropogenic greenhouse gas emissions on account of increase in human population, economic activity, changes in life style, energy use, land use changes and other related activities is widespread and well reported (IPCC 2014). Representative concentration pathways (RCPs) of greenhouse gases including a stringent mitigation scenario (RCP 2.6), two intermediate scenarios (RCP 4.5 and RCP 6.0) and one scenario with very high GHG emissions (RCP 8.5) have been used for making future projections. Relative to 1986–2005, the increase in global mean surface temperature by the end of the twenty-first century (2081–2100) is likely to be 0.3–1.7 °C under RCP 2.6, 1.1–2.6 °C under RCP 4.5, 1.4–3.1 °C under RCP 6.0 and 2.6–4.8 °C under RCP 8.5 (IPCC 2014).
Over the last few decades, indicators of climate change in the form of increasing temperature, shrinking of glaciers and erratic precipitation pattern are easily discernible in the Himalaya (Shrestha et al. 2012; Bahuguna et al. 2014) including Kashmir Himalaya (Rashid et al. 2015; Zaz et al. 2018). Since Himalaya is a biodiversity hotspot, the impact of such changes may be more damaging on the native biodiversity (Rashid et al. 2013; Malik et al. 2015). It is very likely that new species alien to the region may gain foothold due to changing climatic conditions and P. hysterophorus is one species that is making inroads into previously uninvaded areas in Kashmir Himalaya.
Parthenium hysterophorus is an annual short-lived herbaceous species that belongs to family Asteraceae (commonly known as feverfew, carrot grass, congress grass and parthenium weed). Though native to the tropics and subtropics of Central and South America (Dhileepan and Wilmot Senaratne 2009; Adkins and Shabbir 2014), P. hysterophorus has invaded vast areas in a number of countries across five continents. Accidental or unchecked trade and transport across the borders is believed to be the primary reason for its global spread (Adkins and Shabbir 2014; Bajwa et al. 2016), in addition to its vigorous growth, high seed production and effective adaptive and dispersal mechanisms (Bajwa et al. 2016). In fact, it is one of the world’s top ten noxious weeds (Callaway and Ridenour 2004). In Asia, P. hysterophorus is believed to be unintentionally introduced with cereal and grass seed shipment from America during the 1950s (Rao 1956; Bhowmik and Sarkar 2005). However, in India it has been first reported in 1810 in Arunachal Pradesh (Gnanavel 2013) and there is mention of this weed in a book namely “Hortus Bengalensis” by Roxburgh as early as 1814. Subsequently, it became invasive in most of the Indian states but there are very few reports of occurrence of P. hysterophorus in Kashmir Himalaya (Yaqoob et al. 1988).
To predict the distribution of P. hysterophorus at global, regional (India) and local (Jammu & Kashmir) spatial scales under various RCP scenarios in future (2050 and 2070), we used Maximum Entropy Algorithm (MaxEnt), a widely used machine-learning software (Bradley 2009; Kumar and Stohlgren 2009; Elith et al. 2010; Lamsal et al. 2018), for its easy implementation and use of presence-only data as absence data is rarely available or reliable (Phillips et al. 2006). To improve the model performance, the usually spatially biased presence records (Lake et al. 2020) were corrected for sampling and latitudinal biases. We selected occurrence records only within the specified habitat range using a particular tool in SDM Toolbox. Though several studies that have modelled the distribution of P. hysterophorus at global level (Kriticos et al. 2015; Mainali et al. 2015) and in India (Ahmad et al. 2019) are available, the present study was aimed to uncover and compare the differences in the distribution of this species at different spatial scales under various climate change scenarios in two time periods of 2050 and 2070. Also in the present study, unlike previous studies, pairwise comparison in the extent of overlap in the niche of P. hysterophorus between the native and introduced regions was undertaken following the ordination technique (PCA-env) using Schoener’s D (Broennimann et al. 2012; Datta et al. 2019). Niche dynamics was further analysed using several indices (stable, unfilling and expansion) which were statistically tested using niche equivalency and niche similarity tests. Since we suspected that P. hysterophorus may move to previously uninvaded areas in Jammu & Kashmir, the rate and direction of core distributional shift in P. hysterophorus was figured out which has implications for deploying rapid response measures for the management of this species in Kashmir Himalaya.
Materials and methods
Species occurrence data
The occurrence data of P. hysterophorus with georeferenced geographic coordinates were collected from its native and alien ranges. A total of 7185 records were compiled which were obtained from different sources, such as Global Biodiversity Information Facility (GBIF) (http://data.gbif.org/), an open access database for distribution of species across the globe and for India, CABI (Centre for Agriculture and Biosciences International) (http://www.cabi.org/isc/datasheet/45573), Indian Biodiversity Portal, published records on P. hysterophorus in journals, books, reports and other published literature, herbarium records of Botanical Survey of India, JeevSampada, Kashmir University Herbarium (KASH) and field survey records of authors.
Since being important for better predictive modelling, the occurrence records were thoroughly cleaned for spatial errors using Diva GIS (Hijmans et al. 2001) which was accessed in July 2019 and only georeferenced localities were included. Data without or with obviously erroneous coordinates (e.g. 0.0) were discarded. The selected records were first converted to decimal degrees as it is the best system for the digital notation of geographic coordinates and then converted into an MS excel format with species name, latitude, longitude, altitude, location and data source. The duplicate records were then removed. The file containing remaining occurrence points was converted to a CSV format containing only Species ID, Longitude and Latitude.
Correction of sampling bias
The input data i.e. the presence records are usually spatially biased towards more easily accessed and better surveyed areas (Phillips et al. 2009; Kramer-Schadt et al. 2013) and eventually can translate into environmental bias due to greater representation of certain features of these better surveyed regions (Kramer-Schadt et al. 2013). As most of our data were derived from opportunistic observations and herbaria, the potential effect of sampling bias in our data was acknowledged and taken into account. Firstly, to reduce the bias due to spatially auto-correlated occurrence records plus spatial clusters of localities, the Graduated Filtering method (Brown 2014) was used. This method addresses the problem by spatially filtering the data to a single point within the specified (here 10 km) Euclidian Distance (Boria et al. 2014) and thus rarefied the occurrence points to 1674. After extracting values from raster at the location of presence points using QGIS, the null values were removed, reducing the data set to 1644 points. Furthermore, the bias correction features available in MaxEnt, allows the users to include bias files for background selection. These files, besides controlling the selection of background points, also facilitate to control density of background sampling. Background points when compared to presence occurrence points aid in differentiating the environmental conditions suitable for the potential occurrence of a species (Brown 2014). We used the Correcting Latitudinal Background Selection Bias Tool in the SDM Toolbox (Cao et al. 2016) to address the issue of biasing the selection of background points (or pseudoabsence points) towards the poles. The level of bias depends on the breadth of latitudes the analysis covers (Brown 2014). It provides solution in three steps: (a) Creation of a BiasFile for Coordinate Data (BFCD) in MaxEnt (downloadable from http://sdmtoolbox.org/technical-info). This file accounts for pseudoabsence sampling biases, (b) Background Selection: Sample by Buffered MCP (buffer distance was taken as 10 km). It limits selection of background points to only feasible areas of dispersal; (c) Clip BFCD by Background Selection Bias File. It merges the two bias files. The resultant bias file was used for background selection in MaxEnt.
Environmental data
To model potential global distribution of P. hysterophorus, a total of 19 grid-based bioclimatic variables (Table 1) for the current and future time periods were obtained at a spatial resolution of 30 arc-seconds which corresponds to a pixel size of approximately (~ 1 sq.km) from Worldclim version 1.4 (www.worldclim.org/; Hijmans et al. 2005). We used lowest available grain size of bioclimatic variables to increase model accuracy and transferability (Manzoor et al. 2018). Since it is widely accepted that most climatic variables are highly correlated with each other, it is essential to perform multi-collinearity analysis in habitat suitability modelling as correlated environmental variables negatively affect model performance (Pearson et al. 2007). Though MaxEnt has been found to cope well with collinearity (Elith et al. 2011), Pearson correlation analysis was used to omit highest correlative predictors (Dormann et al. 2013). A commonly used threshold of Pearson’s correlation coefficient (r > 0.7) was used (Dormann et al. 2013). All the 19 bioclimatic raster layers were tested for collinearity by calculating pairwise correlations between them and highly correlated variables (Pearson’s correlation coefficient r > 0.75) were omitted (Syfert et al. 2013; Cao et al. 2016; Wei et al. 2017). Discarding highly correlated variables reduces autocorrelation of input environmental data and minimizes the impact of multicollinearity and overfitting of the model. Finally, only nine variables (Bio 1, Bio 2, Bio 3, Bio 4, Bio 5, Bio 12, Bio 14, Bio 15 and Bio 19) were used (Table 1) to generate the niche model under current and future climatic conditions. The datasets of selected nine bioclimatic variables were converted into ASCII format in ArcGIS 10.3 as required in MaxEnt.
Owing to the fact that P. hysterophorus is widespread in all the continents except Europe and Antarctica (Mainali et al. 2015), we studied its future distribution at three spatial scales under four future greenhouse gas concentration trajectories, commonly known as RCPs (Representative Concentration Pathways: RCP 2.6, RCP 4.5, RCP 6.0 and RCP 8.5) which were used by IPCC (Intergovernmental Panel on Climate Change) in its Fifth Assessment Report (AR5). We selected a GCM i.e. Global Circulation Model, HadGEM2-AO (Hadley Global Environment Model 2–AtmosphereOcean). This model is designed to run for the major scenarios as recommended by IPCC (2014) and has been used by a number of workers for species distribution modelling (Ahmad et al. 2019; Banerjee et al. 2019). The datasets were downloaded for two time periods: 2050 (average for 2041–2060) and 2070 (average for 2061–2080) from the WorldClim website (http://www.worldclim.org). In order to maintain consistency, we selected the same independent predictors (Bio 1, Bio 2, Bio 3, Bio 4, Bio 5, Bio 12, Bio 14, Bio 15 and Bio 19) as were used for mapping current distribution of P. hysterophorus. However, all the 19 bioclimatic variables were used for niche dynamic analysis (Table 1).
Modelling approach
To model the current and future distribution of P. hysterophorus under different climate change scenarios, we used MaxEnt (Maximum Entropy Niche Modelling software, version 3.4.1), a machine-learning method that employs the principle of maximum entropy to approximate the unknown probability distribution of a target species based on presence-only data (Phillips et al. 2006). We preferred it owing to its easy use, higher predictive accuracy, functionality to use presence only data and relatively better performance than other methods (Reside et al. 2019; Chen et al. 2020), and popularity (Merow et al. 2013). Also MaxEnt has successfully outperformed classical presence-only models like DOMAIN, ENFA, BIOCLIM (Hirzel et al. 2002; Elith et al. 2006; Ward 2007) and is believed to be most reliable amongst available machine learning methods (Guisan et al. 2007; Elith and Graham 2009; Fourcade et al. 2014). We ran MaxEnt mostly with default settings but the random test percentage was kept equal to 25% and the maximum number of iterations that permit the algorithm to get close to convergence was set to 5000 instead of default 500 allowing the model to have sufficient time for convergence. MaxEnt has an ability to run a model multiple times and finally averages the results from all models created. Thus, model validation was done using sub-sampling option with 10 replications and the default “logistic” format was used which is a continuous map with an estimated probability of species’ presence ranging from 0 to 1. To further improve model performance, we used the bias file obtained using Correcting Latitudinal Background Selection Bias Tool in the SDMtoolbox (Brown 2014) instead of using default randomly selected background points. Further, to calculate significant contribution of each predictor to the model, Jackknife procedure was used. This configuration has been effectively used in a wide range of niche modelling studies (Phillips and Dudík 2008; Yang et al 2013; Yi et al. 2016; Esfanjani et al. 2018).
Model evaluation
The main outputs of MaxEnt included ROC curve, response curves, Jackknife tests of variable importance and probability maps. The capacity of the model to differentiate between presence and absence states was determined by using the Area Under the Curve (AUC) of the Receiver Operating Characteristics (ROC) plot test statistics. The strength and accuracy of the model is commonly determined by AUC values ranging between 0 and 1 whereby AUC values of 0.5–0.7 are considered low, 0.7–0.8 is ‘acceptable prediction', 0.8–0.9 is ‘excellent’ and > 0.9 is ‘outstanding prediction’ (Pittman and Brown 2011). MaxEnt output is continuous data with values ranging from 0 (representing lowest) to 1 (representing highest) probability of potential distribution. In addition, the Boyce Index commonly used for presence only models was calculated using ‘Ecospat’ package in R (version 3.5.3) for model evaluation. This index is considered to be adequate especially in case of MaxEnt as it uses background data instead of true absences (Di Cola et al. 2017). The Boyce index, a threshold independent metric, measures Spearman’s Rank correlation coefficient which ranges from -1 to 1. While negative values signify false or erroneous predictions, values close to zero imply no better than random results and positive values close to 1 indicate good or correct model predictions (Boyce et al. 2002).
For area change analysis, we imported our MaxEnt output data and reclassified it into five categories of habitat suitability, namely unsuitable (0–0.2), less suitable (0.2–0.4), moderately suitable (0.4–0.6), suitable (0.6–0.8) and highly suitable (0.8–1.0). The suitable area predicted for P. hysterophorus under present and future climate scenarios was calculated by multiplying the number of presence grid cell to their spatial resolution. Data for India and Jammu & Kashmir were extracted from world data file using ‘Extract by Mask’ in Spatial Analyst tools of ArcGIS. Further, the data were given proper projection for geometrical analysis. All GIS operations were performed in ArcGIS 10.3 (ESRI).
Core distributional shifts
To further examine the trend of suitable area change in Jammu & Kashmir, we calculated and compared the centroids of current and future suitable areas by using the python-based GIS toolkit available in the SDMtoolbox V2.4. The SDMs (current and future) were converted to binary maps [presence (1) and absences (0)] using MaxEnt produced threshold. MaxEnt software generated 10 percentile training presence threshold with a value of 0.404. The models were reclassified 0 (absence) values < 0.404 and 1 (presence) > 0.404. The core distributional shifts of the P. hysterophorus were summarised using the binary maps. In this analysis, the species distribution was reduced to a single central point known as centroid and a vector file was created depicting the direction of shift in species ranges over time. The centroid was calculated by averaging the latitude and longitude of all MaxEnt predicted suitable/presence input pixels. The distance in centroid shift was calculated using distance measure tools in GIS software and the average annual shift in the distance of P. hysterophorus was estimated.
Niche dynamics
Niche dynamics of P. hysterophorus was studied using the analytical framework proposed by Broennimann et al. (2012) and modified by Datta et al. (2019). Climatic data related to all the 19 bioclimatic variables (Table 1) of all the occurrence points of P. hysterophorus in both native (Americas) and introduced regions (Africa, Asia, Australia and Oceania) were pooled to calibrate a principal component analysis on the entire environmental space of the species (PCA-env). The first two components of the PCA-env were taken to define the “global PCA space” which was subsequently divided into 100 × 100 bins of equal size with each bin representing a unique set of climatic conditions. Gaussian Kernel density function was used for smoothing the density of occurrences and density of environmental grid cells in each bin of the global PCA space. In the native as well as each of the introduced continents, 10,000 random background points were generated separately to account for the available (background) environments using Research tools function in QGIS software. Bias due to geographic differences in the range size of P. hysterophorus was also corrected as suggested by Datta et al. (2019). This allowed us to compare and visualize niche dynamic analysis between the native and each of the introduced regions in the same ‘global PCA’ space.
Schoener’s D, a measure of the degree of niche overlap, was calculated as a bootstrapped estimate with 95% confidence interval using 100 resampling iterations of uncorrected occurrence density of P. hysterophorus as proposed by Datta et al. (2019). Schoener’s D ranges between 0 and 1 with value of 1 indicating complete overlap and no niche shift, and 0 indicating no overlap. The effect of the exclusion of 25% of rare climatic conditions on the niche overlap index (D) was also investigated. Single-tailed niche similarity and equivalency tests were carried out following the methodology described by Broennimann et al. (2012) using uncorrected occurrence densities (Datta et al. 2019). The null hypothesis for the niche similarity test assumes that the observed niche overlap is due to similarity in the climatic conditions of the two ranges for which the niche overlap is compared. Using an iterative process with 100 iterations, the niche in an exotic range was randomly shifted within the available environmental space while keeping the niche constant in the native range. Since niche overlap index was calculated for each iteration, a simulated distribution of niche overlap values was obtained. When the observed niche overlap is significantly lower than the simulated niche overlap values (P < 0.05), niche shift is indicated. For the equivalency test, the null hypothesis posits that niche overlap between the two ranges that are compared remains consistent even if the occurrence data in the two ranges are pooled and reallocated randomly between the ranges. A randomization procedure with 100 permutations was used wherein the species occurrences of the two ranges were pooled and then randomly reallocated between the two ranges and the simulated overlap index was calculated for each such iteration. The observed value of niche overlap when significantly lower (P < 0.05) than the simulated niche overlap value indicates niche shift. Niche unfilling, niche expansion and niche stability for both analogous and entire climatic space were also calculated using Ecospat package (Di Cola et al. 2017) implemented in R (version 3.5.3) to provide a more complete depiction of niche dynamics. The effect of progressive exclusion of rare climatic conditions (up to 25 percentile) on various niche dynamic indices was also studied.
Results
Current distribution
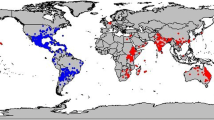
Current occurrence of Parthenium hysterophorus is presented in Fig. 1. It is apparent that in addition to its native regions of Central and South America, Parthenium is quite common in Indian sub-continent, South-eastern Asia, tropical/subtropical Australia, Southern and Eastern Africa, Madagascar and many Oceanic islands with warm climates.
Model performance and variable importance
Maxent was run with a dataset of 1644 occurrence points of P. hysterophorus using only the current climatic data. In this run, 25% of the occurrence records were withheld for model substantiation and average of ten replications was used to estimate the performance of the model on the basis of AUC (area under cover) of the ROC (receiver operating characteristic). Model calibration with a mean training AUC of 0.873 and a test AUC of 0.871 (Table 2) indicated high accuracy of our model in discriminating suitable and unsuitable habitats (Fig. 2). In order to identify contributions of each of the bioclimatic variables employed for modelling habitat suitability, we used percent contribution and Jackknife procedure. Out of the nine bioclimatic predictors’ used for the present study, Isothermality (bio 3) and Annual Mean Temperature (bio 1) were seen to be the best predicting variables contributing together about 56.8% to the predictive model, followed by Annual Precipitation (bio 12) and Temperature Seasonality (bio 4) contributing a total of 31.1% to the predictive model. However, Maximum Temperature of Warmest Month (bio 5) was the least contributing variable with percent contribution of 0.3% (Table 3).
The Maxent internal jackknife test which is yet another method of testing the importance of each predictor showed that the predictor with highest gain when used in isolation was bio 4, which, therefore, appears to contain the most useful information by itself. The explanatory variable that decreases the gain the most when it is omitted is bio 12, which, therefore, appears to have the most information that is not present in the other bioclimatic predictors (Fig. 3). The results were also validated on the basis of the analysis of omission/commission and predicted area was utilized to determine whether presence data occurred in suitable or unsuitable habitat in relation to the given threshold by MaxEnt. The omission/commission analysis (Fig. 4) showed the omission on test samples (green line) are in harmony with the predicted omission rate (black line) from the MaxEnt distribution indicating suitable habitat exists above the threshold. The omission rate and predicted area as a function of the cumulative threshold for P. hysterophorus Area Under Curve (AUC) of the Receiver Operating Characteristic (ROC) curve (Fig. 4) is a parameter that was used to evaluate the predictive ability of the generated model. It is apparent that MaxEnt showed better discrimination of suitable and unsuitable areas of the species in the analysis of AUC. Moreover, positive Boyce Index (Spearman rank correlation coefficient = 0.986) validated that our results were consistent with the significant model.
Jackknife of regularized training gain {Dark blue bars indicate how well a model performs using only that feature compared to the maximal model (red bar), and light blue bars indicate how well a model performs excluding that feature. Thus, important variables can either have (1) large dark blue bars, indicating strong (but perhaps non-unique) contribution to presences; (2) short light blue bars, indicating no other variable contains equivalent information; or (3) both, indicating the variable is independently predictive}
Predicted change in suitable habitat area
A slight increase in habitat suitability from 15.67% under current scenario to 16.02% under RCP 8.5 was predicted in 2050 and to 16.19% under RCP 8.5 in 2070 (Tables 4 and 5). In India, however, a reverse trend was seen i.e., reduction in suitable area in both the future climate scenarios as compared to present scenario. The approximate suitable area under current scenario is believed to be 65.92% and decline to 52.19% was observed under RCP 8.5 in 2070. Though shrinkage of suitable areas is predicted in India, interestingly in Jammu & Kashmir, expansion of suitable area of P. hysterophorus was observed under 2050 climate scenario followed by slight increase of the same by the year 2070 (Fig. 5, 6, 7, 8, and 9). In Jammu & Kashmir, the current estimated potential suitable area for P. hysterophorus is approximately 8.91% which is predicted to increase up to 11.15% under RCP 8.5 in 2050 and 12.16% under RCP 8.5 in 2070 (Tables 4 and 5).
Categorization of suitable area into moderately suitable, suitable and highly suitable revealed that of the total area suitable for P. hysterophorus under current climate conditions (2,11,42,336 sq km, approx. 15.67% of total global land cover excluding Antarctica) 1,07,61,810 sq. km were moderately suitable, 85,53,960 sq. km were suitable and 18,26,566 sq km. were highly suitable. Globally, the regions predicted suitable for P. hysterophorus include Indian subcontinent, Eastern parts of Africa, parts of South and North America and Australia and South-eastern parts of Asia including China. Under the current scenario, about 65.92% of India seems favourable for P. hysterophorus. The climatic simulations in the present study predicted decrease in suitable area for P. hysterophorus in India (Fig. 8) with Eastern Ghats, Western Ghats, Central India and a significant portion of Himalaya remaining suitable for P. hysterophorus while as moderately suitable areas in future would include, Karnataka, Tamil Nadu, Telangana, Maharashtra, Madhya Pradesh, Andhra Pradesh, Chhattisgarh, West Bengal, Uttar Pradesh, Uttrakhand, Himachal Pradesh, Punjab, Haryana and Delhi. Although our model does not predict highly suitable areas in Jammu & Kashmir, but all districts of Jammu and Kashmir are predicted to offer moderate as well as suitable habitats for this weed.
Core distributional shifts
The current centroid of P. hysterophorus is in south of Jammu and Kashmir within the geographic coordinates of 74.50 E longitude and 33.39 N latitude. The centroid of P. hysterophorus is predicted to shift by an average of 20.48 km in the north-west direction by 2050 and by 36.83 km in 2070. The future centroids (2050 and 2070) are mostly located around areas surrounding the Hirapora Wildlife Sanctuary (Table 6; Fig. 10) which is situated about 70 km south of Srinagar, in Shopian district of Jammu & Kashmir. This sanctuary has recently been placed in the eco-sensitive list of the Union Ministry of Environment, Forests and Climate Change. It is an abode to many rare and threatened plants and animals. Further, the estimated change in centroid was at an average of 0.68 km/year by 2050 and shift was noticed at an average of 0.73 km/year by 2070 (see Fig. 10).
Niche dynamics
The PCA-env based on all the 19 bioclimatic variables revealed that the axis 1 retained about 48.3% variation while as axis 2 retained about 32.5% variation (Fig. 11). Furthermore, niche overlap (Schoener’s D) between the native and introduced regions varied across all pairwise comparisons. The niche overlap was very low for Oceania (D = 0.24), moderate for Asia (D = 0.59) and high for Africa (D = 0.69) and Australia (D = 0.70) (Fig. 12; Table 7). Except Oceania, the observed values for equivalency test were significantly lower than the random niche overlap indicating non-equivalency of the climatic niches (Table 7). As is evident from the data (Table 7), the niche similarity test did not yield any significant result. Perusal of the data related to niche dynamic indices (Table 8) revealed very low niche expansion but considerable stability irrespective of whether the niche dynamic indices were computed using analogous or entire climates between native and non-native regions. Niche unfilling was usually low except Oceania (Table 8). Even in the case of Oceania, niche unfilling was more (0.748) when computed on the basis of entire climate as against a value of 0.370 when calculated using the analogous climate. Perusal of data in Table 9 reveals that exclusion of 25% of rare climatic conditions did not alter the niche overlap index (D). It is more or less true for other niche dynamic indices where progressive exclusion of rare climatic conditions did not affect niche dynamic indices with the exception of niche unfilling in the Oceania which decreased from 0.37 to 0.186 with the exclusion of rare climatic conditions (Table 10).
Multivariate climatic niche space of P. hysterophorus in native region of Americas (a) and pairwise comparison of niche dynamics in the introduced regions of Africa (b), Asia (c), Australia (d) and Oceania (e). Unfilled, stable and expanded niches are represented by green, blue and red shades, respectively. The grey shading shows the smoothed occurrence density in the native niche space (a) and in the introduced niche space (b–e). The bold lines mark the available environment in each range (green native, red introduced). Histograms show the observed niche overlap (d) between the native and introduced regions (bars with a diamond) and simulated niche overlap (grey bars). P values for similarity and equivalency tests are also given
Discussion
It is a well known fact that P. hysterophorus is one of the widespread invasive alien species and is known to occur in about 50 tropical and sub-tropical countries across the world (Cowie et al. 2020) which indicates that it is not only able to overcome various environmental constraints but also adapts to a wide range of environmental conditions (Kohli et al. 2006; Cowie et al. 2018). Among the climatic variables, isothermality (bio 3), annual mean temperature (bio 1), annual precipitation (bio 12) and temperature seasonality (bio 4) were the best predictors of the distribution of P. hysterophorus. Importance of temperature and precipitation in predicting the distribution of P. hysterophorus has also been reported by Ahmad et al. (2019). In fact, climatic variables, particularly temperature, rainfall and their interaction (seasonality), in conjunction with the species’ ecology, physiology and adaptability often predict the potential distribution of a species (Sakai et al. 2001). The importance of isothermality, the quotient of the difference between the daily and annual temperature ranges, in predicting the distribution is quite obvious because P. hysterophorus grows in different regions across the world where significant variation in daily and seasonal temperatures exist. Aguirre-Gutierrez et al. (2015) also reported similar relationship between isothermality and widespread distribution of Mexican white pines. The role of temperature in predicting the distribution of P. hysterophorus is because of favourable influence of spring and summer season temperature on the germination of its seeds and seedling growth in the areas of its occurrence (Friedman and Rubin 2015) but availability of water in soil, determined by the amount and frequency of precipitation, may decline with increase in temperature under the warming climate. Though this situation may apparently limit the growth and distribution of a species (McConnachie et al. 2010) but P. hysterophorus being a C3-C4 intermediate species (McConnachie et al. 2010) is known to tide over water stress through drought resistant seeds and persistence of young plants as rosettes (Annapurna and Singh 2003). Interestingly, decreasing soil moisture is reported to promote early flowering and seed set in adult plants of P. hysterophorus (Bajwa et al. 2017) which clearly establishes the role of temperature, rainfall and their interaction in the distribution of this species. Such a conclusion draws support from many other studies that have also emphasized the importance of temperature and precipitation in the geographic distribution of invasive species (Thuiller et al. 2007; Bradley et al. 2010; Priyanka and Joshi 2013; Li et al. 2019; Yan et al. 2019).
Since models built solely on the basis of occurrence data from native range are known to be poor predictors of invasive ranges (Broennimann and Guisan 2008), presence data from both native and non-native ranges was used in the present study because it improves model prediction (Mainali et al. 2015; Bocsi et al. 2016). AUC values averaging 0.871 for the current distribution also indicated a reliable performance of the predictive model with good ability to differentiate between presence and absence areas for the target species (Barik and Adhikari 2011). The highly positive value for Boyce index (Spearman’s Rank Correlation Coefficient = 0.986) further validated the predictive ability of the model. However, uncertainty associated with the accuracy of the model predictions when transferred in space and time remains a major concern and we acknowledge the fact that the predictions from SDM’s are confounded by sampling biases, traits of the target species and omission of the important predictors, such as biotic interactions, population pressure, habitat structure (Yates et al. 2018).
The present study predicted a modest expansion in the distribution range of P. hysterophorus on a global scale under future climate scenarios, especially under RCP scenario 8.5 in 2070 but decrease in the suitable area for P. hysterophorus was predicted at the regional scale (India). In contrast, increase in the suitable area was predicted at a local scale (Jammu and Kashmir). Variation in the spatial distribution under different spatial scales has also been reported by Nielsen et al. (2008). These results bring out the need for predictive modelling of species distribution at different spatial scales which is not the usual practice.
The results obtained during the present study are consistent with numerous studies that have also predicted change in the range expansion of invasive species at the global level due to climate change (Walther et al. 2009; Barbet-Massin et al. 2013; Bellard et al. 2013; Adhikari et al. 2019). Decrease in the suitable area of P. hysterophorus at the regional level has also been reported by Bezeng et al. (2017) for South Africa, Ahmad et al. (2019) for India and Lamsal et al. (2018) for the Himalayan region. Increase in the suitable area at the local level (Jammu & Kashmir) unlike India, for P. hysterophorus, was further studied to predict the trend in the suitable habitat area changes by comparing the centroids of present and future models (Brown 2014). The centroids shift at an average of 0.68 km/year around 2050 and 0.73 km/year by 2070 is quite significant and the centroids were found to be shifting within and towards surrounding areas of the Hirapora Wildlife Sanctuary which is a cause of concern keeping in view the spreading nature of the species.
Present study also revealed that P. hysterophorus shows moderate to high niche overlap (Schoener’s D) between the native Americas and the introduced continents except Oceania. Further analysis (Table 11) pointed out that higher niche overlap between Americas and Asia is due to similarity in their climatic conditions but same does not hold true for higher overlap between native Americas and introduced regions of Africa and Australia. Thus, it clearly brings out that niche overlap is not always a manifestation of climatic overlap between the native and introduced regions. Such inferences are consistent with the study of Datta et al. (2019). Furthermore, insignificant similarity test though points towards a more similar climatic niche between native and introduced regions but the equivalency test revealed that native niche is not equivalent to any of the climatic niches of this species in the introduced regions except Oceania. Such an observation is consistent with the results of Aguirre-Gutierrez et al. (2015). Thus, it can be inferred from these results that P. hysterophorus will continue to invade the areas with climate similar but not necessarily equivalent to the climate prevailing in the native Americas.
Niche dynamic analysis revealed high degree of niche stability between the native and introduced regions irrespective of whether analogous or entire climates were considered. It indicates that the species occupies more or less similar climates though not equivalent in the native and introduced regions. Low unfilling indicates reduced climatic space that could be colonized by this species in future except Oceania where less residence time or constraints to dispersal could not have allowed the species to occupy all the available climatic niches. Low expansion values also reflect that P. hysterophorus is not showing any niche shift in the introduced regions. The observation of high niche stability and little niche expansion and low unfilling is consistent with many previous studies on various invasive species (Petitpierre et al. 2012; Callen and Miller 2015).
Conclusions
Based on the detailed analysis, it can be safely concluded that the P. hysterophorus exhibits niche conservatism which may restrict its spread under climate change in the regions where it has already spread and occupied most of the climatic niche space but may spread to new areas where it finds suitable climate niche. Thus, the present study provides a strong rationale for studying niche dynamics to better predict the distribution of alien species under climate change as modelled distribution P. hysterophorus differed at different spatiotemporal scales under changing climate and more importantly in Jammu & Kashmir. Since the species is expected to spread to previously uninvaded areas in Kashmir with debilitating ecological and economic consequences, the study would serve the purpose of raising the concern with land managers about the impending threat from invasive alien species.
References
Adhikari P, Jeon J, Kim HW et al (2019) Potential impact of climate change on plant invasion in the Republic of Korea. J Ecol Environ 43:1–12
Adkins S, Shabbir A (2014) Biology, ecology and management of the invasive parthenium weed (Parthenium hysterophorus L.). Pest Manag Sci 70:1023–1029
Aguirre-Gutierrez J, Serna-Chavez M, Villalobos-Arambula AR et al (2015) Similar but not equivalent: ecological niche comparison across closely-related Mexican white pines. Divers Distrib 21:245–257
Ahmad R, Khuroo AA, Hamid M et al (2019) Predicting invasion potential and niche dynamics of Parthenium hysterophorus (Congress grass) in India under projected climate change. Biodivers Conserv 28:2319–2344
Annapurna C, Singh JS (2003) Variation of Parthenium hysterophorus in response to soil quality: implications for invasiveness. Weed Res 43:190–198
Atwater DZ, Ervine C, Barney JN (2018) Climatic niche shifts are common in introduced plants. Nat Ecol Evol 2:34–43
Bahuguna I, Rathore B, Brahmbhatt R et al (2014) Are the Himalayan glaciers retreating? Curr Sci 106:1008–1013
Bajwa AA, Chauhan BS, Farooq M (2016) What do we really know about alien plant invasion? A review of the invasion mechanism of one of the world’s worst weeds. Planta 244:39–57
Bajwa AA, Chauhan BS, Adkins S (2017) Morphological, physiological and biochemical responses of two Australian biotypes of Parthenium hysterophorus to different soil moisture regimes. Environ Sci Pollut Res 24:16186–16194
Banerjee AK, Mukherjee A, Guo W et al (2019) Spatio-temporal patterns of climatic niche dynamics of an invasive plant Mikania micrantha Kunth and its potential distribution under projected climate change. Front Ecol Evol 7:291. https://doi.org/10.3389/fevo.2019.00291
Barbet-Massin M, Rome Q, Muller F et al (2013) Climate change increases the risk of invasion by the yellow-legged hornet. Biol Conserv 157:4–10
Barbet-Massin M, Rome Q, Villemant C (2018) Can species distribution models really predict the expansion of invasive species? PLoS ONE 13:e0193085
Barik SK, Adhikari D (2011) Predicting the geographical distribution of an invasive species (Chromolaena odorata L. (King) & H E Robins) in the Indian subcontinent under climate change scenarios. In: Bhatt JR, Singh JS, Singh SP, Tripathi RS, Kohli RK (eds) Invasive alien plants: an ecological appraisal for the Indian subcontinent pp 77–88. CABI International
Bellard C, Thuiller W, Leroy B et al (2013) Will climate change promote future invasions? Glob Change Biol 19:3740–3748
Bellard C, Jeschke JM, Leroy B et al (2018) Insights from modelling studies on how climate change affects invasive alien species geography. Ecology and Evolution 8:5688–5700
Bezeng SB, Van der B, Yessoufou M et al (2017) Climate change may reduce the spread of invasive and invading species in South Africa. Ecosphere 8:e01694
Bhowmik PC, Sarkar D (2005) Parthenium hysterophorus L.: its world status and potential management. In: Proceeding of the Second International Conference on Parthenium Management, Bangalore, 5–7 December 2005, pp 1–6
Bocsi T, Allen JM, Bellemare J (2016) Plants’ native distributions do not reflect climatic tolerance. Divers Distrib 22:615–624
Boria RA, Olson LE, Goodman SM et al (2014) Spatial filtering to reduce sampling bias can improve the performance of ecological niche models. Ecol Model 275:73–77
Boyce MS, Vernier PR, Nielsen SE et al (2002) Evaluating resource selection functions. Ecol Model 157:281–300. https://doi.org/10.1016/S0304-3800(02)00200-4
Bradley BA (2009) Regional analysis of the impacts of climate change on cheat grass invasion shows potential risk and opportunity. Glob Change Biol 15:196–208
Bradley BA, Blumenthal DM, Wilcove DS et al (2010) Predicting plant invasions in an era of global change. Trends Ecol Evol 25:310–318
Broennimann O, Guisan A (2008) Predicting current and future biological invasions: both native and invaded ranges matter. Biol Let 4:585–589. https://doi.org/10.1098/rsbl.2008.0254
Broennimann O, Fitzpatrick MC, Pearman PB et al (2012) Measuring ecological niche overlap from occurrence and spatial environmental data. Glob Ecol Biogeogr 21:481–497
Brown JL (2014) SDMtoolbox: a python-based GIS toolkit for landscape genetic, biogeographic and species distribution model analyses. Methods Ecol Evol 5:694–700
Callaway RA, Ridenour WM (2004) Novel weapons: invasive success and the evolution of increased competitive ability. Front Ecol Environ 2:436e443
Callen ST, Miller AJ (2015) Signatures of niche conservatism and niche shift in the North American kudzu (Pueraria montana) invasion. Divers Distrib 21:853–863
Cao B, Bai CK, Zhang LL et al (2016) Modeling habitat distribution of Cornus officinalis with Maxent modelling and fuzzy logics in China. J Plant Ecol 9:1–12
Chai SL, Zhang J, Nixon A et al (2016) using risk assessment and habitat suitability models to prioritise invasive species for management in a changing climate. PLoS ONE 11(10):e0165292
Chen Q, Yin Y, Zhao R, Yang Y et al (2020) Incorporating local adaptation into species distribution modelling of Paeonia mairei, an endemic plant to China. Front Plant Sci 10:1717. https://doi.org/10.3389/fpls.2019.01717
Cowie BW, Witkowski ETF, Byrne MJ, Strathie LW, Goodall JM, Venter N (2018) Physiological response of Parthenium hysterophorus to defoliation by the leaf feeding beetle Zygogramma bicolorata. Biol Control 117:35–42
Cowie BW, Byrne MJ, Witkowski ETF, Strathie LW et al (2020) Parthenium avoids drought: Understanding the morphological and physiological responses of the invasive herb Parthenium hysterophorus to progressive water stress. Environ Exp Bot 171:103945. https://doi.org/10.1016/j.envexpbot.2019.103945
Datta A, Schweiger O, Kühn I (2019) Niche expansion of the invasive plant species Ageratina adeophora despite evolutionary constraints. J Biogeogr 46:1306–1315
Dhileepan K, Wilmot Senaratne KAD (2009) How widespread is Parthenium hysterophorus and its biological control agent Zygogramma bicolorata in South Asia? Weed Res 49:557–562
Di Cola V, Broennimann O, Petitpierre B et al (2017) Ecospat: an R package to support spatial analyses and modeling of species niches and distributions. Ecography 40:774–787
Dormann CF, Elith J, Bacher S et al (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36:027–046
Elith J, Graham CH (2009) Do they? How do they? Why do they differ? On finding reasons for differing performances of species distribution models. Ecography 32:66–77
Elith J, Graham CH, Anderson RP et al (2006) Novel methods improve prediction of species’ distributions from occurrence data. Ecography 29:129–151
Elith J, Kearney M, Phillips S (2010) The art of modelling range-shifting species. Methods Ecol Evol 1:330–342
Elith J, Phillips SJ, Hastie T et al (2011) A statistical explanation of MaxEnt for ecologists. Divers Distrib 17:43–57
Esfanjani J, Ghorbani A, ZareChahouki M (2018) MaxEnt modeling for predicting impacts of environmental factors on the potential distribution of Artemisia aucheri and Bromus tomentellus-Festuca ovina in Iran. Pol J Environ Stud 27(3):1041–1047
Fletcher D, Gillingham P, Britton J et al (2016) Predicting global invasion risks: a management tool to prevent future introductions. Sci Rep 6:26316
Fourcade Y, Engler JO, Rodder D et al (2014) Mapping species distributions with MAXENT using a geographically biased sample of presence data: a performance assessment of methods for correcting sampling bias. PLoS ONE 9:e97122
Friedman J, Rubin MJ (2015) All in good time: understanding annual and perennial strategies in plants. Am J Bot 102(4):497–499
Gnanavel I (2013) Parthenium hysterophorus L.: A major threat to natural and agro eco-systems in India. Sci Int 1:124–131
Gomes VHF, Stéphanie DIJFF, Raes N et al (2018) Species distribution modelling: contrasting presence-only models with plot abundance data. Sci Rep 8:1003. https://doi.org/10.1038/s41598-017-18927-1
Guisan A, Graham CH, Elith J et al (2007) Sensitivity of predictive species distribution models to change in grain size. Divers Distrib 13:332–340
Hijmans RJ, Cruz JM, Rojas E et al (2001) DIVA-GIS. A geographic information system for the management and analysis of genetic resources data. Manual (Internet). International Potato Center and International Plant Genetic Resources Institute, Lima, Peru. http://www.diva-gis.org
Hijmans RJ, Cameron SE, Parra JL et al (2005) Very high resolution interpolated climate surfaces for global land areas. Intl J Climatol 25:1965/1978
Hirzel AH, Hausser J, Chessel D et al (2002) Ecological-niche factor analysis: how to compute habitat- suitability maps without absence data? Ecology 83:2027–2036
Hughes L (2000) Biological consequences of global warming: is the signal already apparent? Trends Ecol Evol 15:56
IPCC (2014) Climate Change (2014) Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Core Writing Team, R.K. Pachauri and L.A. Meyer (eds.)]. IPCC, Geneva, Switzerland, p 151
Kohli RK, Batish DR, Singh HP, Dogra KS (2006) Status, invasiveness and environmental threats of three tropical American invasive weeds (Parthenium hysterophorus L., Ageratum conyzoides L., Lantana camara L.) in India. Biol Invasions 8:1501–1510
Kramer AM, Annis G, Wittmann ME et al (2017) Suitability of Laurentian Great Lakes for invasive species based on global species distribution models and local habitat. Ecosphere 8:e01883
Kramer-Schadt S, Niedballa J, Pilgrim JD et al (2013) The importance of correcting for sampling bias in MaxEnt species distribution models. Divers Distrib 19:1366–1379
Kriticos DJ, Brunel S, Ota N et al (2015) Downscaling pest risk analyses: identifying current and future potentially suitable habitats for Parthenium hysterophorus with particular reference to Europe and North Africa. PLoS ONE 10:e0132807
Kumar S, Stohlgren TJ (2009) Maxent modeling for predicting suitable habitat for threatened and endangered tree Canacomyrica monticola in New Caledonia. J Ecol Nat Environ 1:94–98
Lake TA, Runquist RDB, Moeller DA (2020) Predicting range expansion of invasive species: Pitfalls and best practices for obtaining biologically realistic projections. Divers Distrib 26:1767–1779
Lamsal P, Kumar L, Aryal A et al (2018) Invasive alien plant species dynamics in the Himalayan region under climate change. Ambio 34:1–14. https://doi.org/10.1007/s13280-018-1017-z
Li X, Mao H, Du G et al (2019) Spatiotemporal evolution and impacts of climate change on bamboo distribution in China. J Environ Manag 248:109265
Liu C, Wolter C, Xian W et al (2020) Most invasive species largely conserve their climatic niche. Proc Natl Acad Sci 117(38):23643–23651. https://doi.org/10.1073/pnas.2004289117
Mainali K, Dhileepan K, Warren D et al (2015) Projecting future expansion of invasive species: comparing and improving methodologies. Glob Change Biol 21:4464–4480. https://doi.org/10.1111/gcb.13038
Malik AH, Rashid I, Ganie AH et al (2015) Benefitting from Geoinformatics: estimating floristic diversity of Warwan valley in Northwestern Himalaya, India. J Mt Sci 12(4):854–863. https://doi.org/10.1007/s11629-015-3457-2
Manzoor SA, Geoffrey G, Martin L (2018) Species distribution model transferability and model grain size–finer may not always be better. Sci Rep 8(1):7168
McConnachie AJ, Strathie LW, Mersie W, Gebrehiwot L, Zewdie K, Abdurehim A, Abrha B, Araya T, Asaregew F, Assefa F, Gebre-Tsadik R, Nigatu L, Tadesse B, Tana T (2010) Current and potential geographical distribution of the invasive plant Parthenium hysterophorus (Asteraceae) in eastern and southern Africa. Weed Res 51(1):71–84
Merow C, Smith MJ, Silander Jr JA (2013) A practical guide to MaxEnt for modeling species’ distributions: what it does, and why inputs and settings matter. Ecography 36(10):1058–1069
Nielsen C, Hartvig P, Kollmann J (2008) Predicting the distribution of the invasive alien Heracleum mantegazzianum at two different spatial scales. Divers Distrib 14:307–317
Pauchard A, Escudero A, Garcia RA et al (2016) Pine invasions in treeless environments: dispersal overruns microsite heterogeneity. Ecol Evol 6:447–459
Pearson RG, Raxworthy CJ, Nakamura M et al (2007) Predicting species distributions from small numbers of occurrence records: a test case using cryptic geckos in Madagascar. J Biogeogr 34(1):102–117
Petitpierre B, Kueffer C, Broennimann O et al (2012) Climatic niche shifts are rare among terrestrial plant invaders. Science 335(6074):1344–1348. https://doi.org/10.1126/science.1215933
Phillips SJ, Dudík M (2008) Modeling of species distributions with Maxent: new extensions and a comprehensive evaluation. Ecography 31:161–175
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modelling of species geographic distributions. Ecol Model 190:231–259
Phillips SJ, Dudik M, Elith J et al (2009) Sample selection bias and presence-only models of species distributions. Ecol Appl 19:181–197
Pittman SJ, Brown KA (2011) Multi-Scale approach for predicting fish species distributions across coral reef seascapes. PLoS ONE 6(5): https://doi.org/10.1371/journal.pone.0020583
Porfirio LL, Harris RMB, Lefroy EC et al (2014) Improving the use of species distribution models in conservation planning and management under climate change. PLoS ONE 9:e113749
Priyanka N, Joshi PK (2013) Effects of climate change on invasion potential distribution of Lantana camara. Earth Sci Clim Change 4:164
Ramirez-albores JE, Bustamante RO, Badano EI (2016) Improved predictions of the geographic distribution of invasive plants using climatic niche models. PLoS ONE 11:e0156029
Rao RS (1956) Parthenium hysterophorus Linn.: a new record for India. J Bombay Nat Hist Soc 54:218–220
Rashid I, Romshoo SA, Vijayalakshmi T (2013) Geospatial modelling approach for identifying disturbance regimes and biodiversity rich areas in North Western Himalayas, India. Biodivers Conserv 22(11):2537–2566
Rashid I, Romshoo SA, Chaturvedi RK et al (2015) Projected climate change impacts on vegetation distribution over Kashmir Himalayas. Clim Change 132(4):601–613
Reside AE, Critchell K, Crayn DM, Goosem M, Goosem S, Hoskin CJ et al (2019) Beyond the model: expert knowledge improves predictions of species’ fates under climate change. Ecol Appl 29:e01824. https://doi.org/10.1002/eap.1824
Sakai AK, Allendorf FW, Holt JS, Lodge DM, Molofsky J, With KA, Baughman S, Cabin RJ, Cohen JE, Ellstrand NC et al (2001) The population biology of invasive species. Annu Rev Ecol Syst 32:305–332
Shrestha UB, Shrestha BB (2019) Climate change amplifies plant invasion hotspots in Nepal. Divers Distrib 25(10):1599–1612. https://doi.org/10.1111/ddi.12963
Shrestha UB, Gautam S, Bawa KS (2012) Widespread climate change in the Himalayas and associated changes in local ecosystems. PLoS ONE 7:e36741
Syfert MM, Smith MJ, Coomes DA (2013) The effects of sampling bias and model complexity on the predictive performance of MaxEnt species distribution models. PLoS ONE 8:e55158
Thuiller W, Richardson DM, Pysek P et al (2005) Niche-based modeling as a tool for predicting the risk of alien plant invasions at a global scale. Glob Change Biol 11:2234–2250
Thuiller W, Richardson D, Midgley G (2007) Will climate change promote alien plant invasions? In: Nentwig W (ed) Biological invasions. Springer-Verlag, Berlin
Thuiller W, Georges D, Engler R et al (2016) Package ‘biomod2’. ftp://ftp2.de.freebsd.org/ pub/ misc/cran/web/packages/biomod2/biomod2.pdf
Tingley R, Vallinoto M, Sequeira F et al (2014) Realized niche shift during a global biological invasion. Proc Natl Acad Sci USA 111:10233–10238
Walther G, Roques A, Hulme P et al (2009) Alien species in a warmer world: Risks and opportunities. Trends Ecol Evol 23:686–693
Ward DF (2007) Modelling the potential geographic distribution of invasive ant species in New Zealand. Biol Invasions 9:723–735. https://doi.org/10.1007/s10530-006-9072-y
Wei JF, Zhang H, Zhao W et al (2017) Niche shifts and the potential distribution of Phenacoccus solenopsis (Hemiptera: Pseudococcidae) under climate change. PLoS ONE 12:e0180913
Yan HY, Feng L, Zhao YF et al (2019) (2019) Predicting the potential distribution of an invasive species, Erigeron canadensis L, in China with a maximum entropy model . Glob Ecol Conserv 21:e00822
Yang XQ, Kushwaha SPS, Saran S et al (2013) Maxent modeling for predicting the potential distribution of medicinal plant, Justiciaadhatoda L: in Lesser Himalayan foothills. Ecol Eng 51:83–87
Yaqoob MB, Nisar A, Naqshi AR (1988) Extension of distribution of an obnoxious American weed, Parthenium hysterophorus L. (Asteraceae). J Econ Taxon Bot 12:375–376
Yates K, Bouchet P, Caley M (2018) Outstanding challenges in the transferability of ecological models. Trends Ecol Evol 33(10):790–802. https://doi.org/10.1016/j.tree.2018.08.001
Yi YJ, Cheng X, Yang ZF et al (2016) Maxent modeling for predicting the potential distribution of endangered medicinal plant (H. riparia Lour) in Yunnan, China. Ecol Eng 92:260–269
Zaz S, Romshoo SA, Thokuluwa R et al (2018) Climatic and extreme weather variations over Mountainous Jammu and Kashmir, India: physical explanations based on observations and modelling. Atmos Chem Phys Discuss. https://doi.org/10.5194/acp-2018-201
Acknowledgements
We thank Head, Department of Botany, University of Kashmir for providing laboratory facilities. Support under the CPEPA by the UGC, New Delhi to the University of Kashmir is also gratefully acknowledged which helped in conduct of present work as well. We also acknowledge the help extended by Dr. Alaaeldin Soultan, Swedish University of Agricultural Sciences in the analysis of data. We would also like to thank the anonymous reviewers for their constructive comments which helped us to significantly improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mushtaq, S., Reshi, Z.A., Shah, M.A. et al. Modelled distribution of an invasive alien plant species differs at different spatiotemporal scales under changing climate: a case study of Parthenium hysterophorus L.. Trop Ecol 62, 398–417 (2021). https://doi.org/10.1007/s42965-020-00135-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42965-020-00135-0