Abstract
Biochars can improve soil health but have been widely shown to reduce plant-available nitrogen (N) owing to their high carbon (C) content, which stimulates microbial N-immobilization. However, because biochars contain large amounts of C that are not microbially available, their total elemental C:N ratio does not correspond well with impacts on soil N. We hypothesized that impacts on soil plant-available N would relate to biochar mineralizable-C (Cmin) content, and that C:N ratios of the mineralizable biochar component could provide a means for predicting conditions of net soil N-mineralization or -immobilization. We conducted two laboratory experiments, the first measuring biochar Cmin from respiration of isotopically labeled barley biochars manufactured at 300, 500, and 750 °C, and the second characterizing Cmin by proxy measurements for ten biochars from six feedstocks at several temperatures. For both experiments, soils were incubated with 2% biochar by mass to determine impacts to soil N-mineralization. Contrary to expectation, all the biochars increased soil N-mineralization relative to unamended soils. Also unexpected, higher temperature (500 and 700 °C) barley biochars with less Cmin stimulated more soil decomposition and more soil N-mineralization than a 350 °C barley biochar. However, across diverse biochar feedstocks and production methods, none of the biochar characteristics correlated with soil N-mineralization. The finding of improved soil N-mineralization adds complexity to the range of soil N responses that can be expected in response to biochar amendment. Because of the limited ability to predict soil N responses from biochar properties, users should monitor soil N to manage soil fertility.
Highlights
-
A diverse set of biochar types all increased net soil N-mineralization in a 4-week aerobic incubation.
-
For individual feedstocks, soil N-mineralization increased with pyrolysis temperature.
-
Biochar mineralizable-C and -N did not predict soil N-mineralization across feedstocks.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Biochar incorporation into agronomic systems has become an increasingly popular management practice in the past decade. The addition of biochar has been shown to boost crop yields (Spokas et al. 2012) by altering nutrient cycling (Mia et al. 2017), increasing soil pH (Clough and Condron 2010), and in some cases altering soil water properties (Liu et al. 2017; Phillips et al. 2020a), while additionally sequestering biomass carbon in long-lived forms (Baldock and Smernik 2002; Kuzyakov et al. 2009; Woolf et al. 2010). However, increasing the carbon (C) content of the soil system through biochar application typically increases the C to nitrogen (N) ratio, which in turn impacts soil microbial function and reduces plant-available N (Liu et al. 2018; Gao et al. 2019).
Biochars are highly aromatic, high C materials produced by pyrolysis, gasification, and other low-oxygen heating processes. Many biochars are made from low-value biomass such as wood or straw feedstocks with wide C:N ratios, which confers wide C:N ratios to the resulting biochar. Exceptions are biochars produced from food waste and manures that have narrow C:N ratios. However, N retention during biochar production can also be highly variable, which further contributes to diverse C:N ratios in resulting biochars (Enders et al. 2012). Depending on the feedstock and pyrolysis conditions, biochar can have a total C:N ratio ranging from 6.5 to 640 (Bonanomi et al. 2017).
Several recent meta-analyses (Nguyen et al. 2017; Liu et al. 2018; Gao et al. 2019) showed that reductions in soil mineral N are a widespread response to biochar application. Across approximately 900 studies, Nguyen et al. (2017) found biochar application decreased NH4–N by an average of 11% and NO3–N by 10%. Evidence that microbial immobilization caused this decline in NH4–N and NO3–N (collectively dissolved inorganic nitrogen, DIN) was provided by reports of increased CO2 emissions and increases in microbial abundances associated with N cycling, indicating increased microbial N demand following biochar application (Deenik et al. 2010; Zhang et al. 2012; Zheng et al. 2012; Ducey et al. 2013).
Initial decomposition of residues with wide C:N ratios usually results in N-immobilization in soil (Vigil and Kissel 1991; Aulakh et al. 1991). In some cases a critical C:N threshold can be used to predict conditions of N-mineralization or N-immobilization in soil (Trinsoutrot et al. 2000; Andrews and Foster 2007), although such thresholds are also dependent on residue quality (Ågren et al. 2013; Manzoni 2017). In the case of biochar, a critical C:N ratio ranging between 20 and 32 for biochar and over 32 for the soil as a whole has been shown to support microbial immobilization of N (Nguyen et al. 2017). However, we expected that the C and N content of the easily mineralizable fraction of biochar would have a better ability than total biochar C:N to predict N immobilization, because a large portion of biochar C is highly resistant to microbial decay. As pyrolysis temperature is increased much of the carbon becomes heterocyclic and largely inaccessible for microbial decomposition (Budai et al. 2016; Leng et al. 2019). Although the quantity of total C in biochar generally increases with pyrolysis temperature, the quantity of mineralizable-C (Cmin) decreases, due to reduction of organic molecules into aromatic rings and loss of COOH, ROH, and C=N functional groups (Keiluweit et al. 2010; Torres-Rojas et al. 2020). The resistance of biochars to microbial decomposition has been demonstrated by numerous incubation studies. A meta-analysis of incubation studies showed that as much as 15% of biochar carbon decomposed within two years, but that on average the labile pool accounted for just 3% of total C (Wang et al. 2016). Wang et al. (2016) furthermore estimated the inert biochar C pool to have a residence time of over 500 years. Biochar effects on N availability are therefore likely to be associated with only the portion of biochar C that is easily mineralizable (Jeffery et al. 2015; Gao et al. 2019).
In this study we employed biochars from multiple feedstocks, pyrolysis temperatures, and production technologies to examine links between the mineralizable-C (Cmin) content of biochars, the C:N ratio of mineralizable component, and resulting impacts on net soil N-mineralization. We hypothesized that: (1) biochar Cmin would correlate negatively with soil N-mineralization, because most biochars would be poor sources of N, and higher quantities of Cmin would therefore add more low-N substrate to soil. We further hypothesized that (2) the C:N ratio of the mineralizable component of biochar, estimated from water-extractable organic C and N concentrations, would also be negatively correlated with soil N-mineralization.
2 Materials and methods
Two experiments were conducted to evaluate the hypotheses. The first experiment used a temperature series of 13C-labeled barley biochars produced at 350, 500, and 700 °C. Cmin was quantified by isotopic analysis of respired CO2 in a 101-day soil incubation. Soil DIN concentrations were simultaneously measured to determine net N-mineralization. To expand to more biochar types, the second experiment examined non-isotopic measures of Cmin for ten biochars produced from several feedstocks under a range of temperatures and several production technologies. Cmin was estimated by several approaches, including H:C elemental ratio which provides a measure of biochar aromaticity (Budai et al. 2013), water extractable organic C (WEOC) which estimates one of the labile pools (Whitman et al. 2014), and permanganate-oxidizable (KMnO4-oxidizable) C which estimates oxidation resistance (Calvelo Pereira et al. 2011). Net N-mineralization in biochar-amended soils was determined in a 28-day aerobic soil incubation.
2.1 Experiment 1
2.1.1 Biochar and soils
A set of barley (Hordeum vulgare L.) whole-plant biochars was produced by pyrolysis at 350, 500, and 700 °C from a mixture of 13C-labeled panicles and leaves of barley and non-labeled barley straw. The 13C-labeled barley was grown in a controlled environmental chamber and pulse-labeled 3 times with 13CO2 to achieve 13C enrichment of 0.38 atom percent excess, as described by Lifschitz et al. (2002). Due to limited quantities of this material, it was diluted with biochar made from non-labeled barley straw grown near Corvallis, Oregon. The labeled and non-labeled plant materials were ground to pass through a 6 mm sieve, pyrolyzed separately, and then mixed in a ratio of 20% labeled to 80% non-labeled material by mass, providing δ13C values for the mixtures of 32–40‰ PDB (Table 1). The biochars were pyrolyzed using a Lindburg oven with a retort at the USDA-ARS in Florence, South Carolina as outlined by Cantrell and Martin (2012). Chemical characteristics of these biochars, as well as the biochars used in Experiment 2, are shown in Table 1 and testing methods are detailed below.
The barley biochars were added at a rate of 2% by mass to two silt loam soils with contrasting organic carbon contents. A total of 8 treatments were examined: each of the three biochars was added to a low-carbon and a high-carbon soil and compared to unamended soils as controls. The higher carbon content soil was classified as fine-silty, mixed, superactive, mesic Cumulic Ultic Haploxeroll in the ‘Chehalis’ series, and the lower carbon content soil was classified as a fine-silty, mixed, superactive, mesic Pachic Ultic Argixeroll in the ‘Willamette’ series (Soil Survey Staff 2006). Additional soil characteristics are shown in Table 2. Both soils were collected from 0–15 cm depth from research farms near Corvallis, OR. Soils were sieved through a 4 mm mesh to homogenize and stored at 4 °C for 5 days prior to incubations.
2.1.2 Incubations and isotopic measures
Soil-biochar mixtures were incubated for 101 days and sampled for respired CO2 and δ13CO2 on days 1, 2, 3, 4, 7, 14, 21, 32, 42, 57, 73, and 101. Seventy-five g samples (dry-mass equivalent) of soil or soil-biochar mixture were measured into graduated polyethylene specimen cups and wetted with 0.01 M CaCl2 to bring to 60% water-filled pore space. Three replicates of the eight treatments were prepared, for a total of 24 samples. The polyethylene cups were placed inside 1.8 L glass canning jars; 10 mL of water was added to the jars outside the cups to maintain humidity; and the jars were sealed. The headspace was flushed by piercing rubber septa installed in the lids and flowing CO2-free air through at a rate of 500 mL min−1 for 15 min. Process standards for CO2 measurements were prepared by flushing empty jars with CO2-free air or a 4000 ppm CO2 standard. Standard jars were incubated alongside soils to detect and quantify air leaks. Jars were incubated at 23 °C in a darkened environmental chamber between sampling events.
At each sampling event, a subsample of headspace air was collected for δ13CO2 by removing 12 mL of air with a gastight syringe and injecting it into a borosilicate vial (Extetainer®, Labco Ltd.) that had been purged with CO2-free air and pre-evacuated. Headspace CO2 concentration was then measured by connecting one of the jar’s septa to a CO2 analyzer (Ultraportable Greenhouse Gas Analyzer, Los Gatos Research) and drawing headspace air for 2 min. Data measured between 30 and 100 s were averaged. The jar headspace was then flushed with CO2-free air to prepare for the next phase of CO2 accumulation. Respiration rate was computed as the g C–CO2 accumulated, divided by the time between each sampling event. Exetainer vials were accumulated over the study and shipped in batches after days 42 and 101 to Ft. Collins, Colorado, where δ13CO2 was determined on a gas chromatograph-isotope ratio mass spectrometer (Isoprime Inc., Manchester, UK).
Quantities of CO2 derived from soil and biochar were calculated using a 2-member mixing model, as follows:
where fbiochar is the fraction of respiration from biochar, δmixture is the δ13CO2 respired from the biochar-soil mixture, and δsoil and δbiochar are the δ13CO2 respired from soil and biochar end-members, respectively. The end-members were estimated as the δ13CO2 respired from unamended soil, and the δ13C of the solid biochars.
2.1.3 Soil N-mineralization
Soil net N-mineralization was quantified in accompanying soil incubations that were destructively extracted for N–NO3− and N–NH4+ (collectively DIN) on days 0, 1, 2, 3, 4, 7, 14, 21, 42, and 101. The incubations for N-mineralization were prepared in the same way as for Cmin measurements. To ensure the samples remained aerated during incubation, each jar was manually vented twice weekly by briefly removing the lid for a few seconds. Three replicates of the 8 treatments were prepared for each of the 9 sampling events through day 42. The jars used for respiration measurements were destructively sampled on day 101, providing a total of 240 DIN samples.
Soil DIN was measured on a moist subsample (approx. 7.5 g air-dried equivalent) extracted in a 1:4 w/v ratio with 2 M KCl. Soils were shaken for 1 h with extraction solution, and then filtered through Whatman #1 filters. The filtrate was analyzed colorimetrically using the vanadium-chloride method for NO3 (Doane and Horwath 2003) and the salicylate-nitroprusside method for NH4 (Rhine et al. 1998) with a 96-well plate spectrophotometer (BioTek Instuments). Soil DIN concentrations were corrected for moisture content, determined from oven-dry (105 °C, 24 h) sub-samples of the incubated soils. Net N-mineralization on each sampling day was calculated as the difference between average DIN for that day and for day 0 baseline samples.
2.1.4 Soil and biochar fertility
The two soils were characterized for pH (2:1 water to soil) on oven-dried (105 °C), sieved samples. Ground samples were also analyzed for total C and N by combustion-IR (CN Analyzer, LECO Corporation). Biochars were characterized for extractable nutrients, EC, pH, and liming ability as potential co-variates of N-mineralization. Extractable nutrients were determined on oven-dried (105 °C) and ground biochar following the method recommendations of Camps-Arbestain et al. (2015). Available-P was determined from a 2% formic acid extraction (Wang et al. 2012; after Rajan et al. 1992) with concentrations determined using inductively coupled plasma optical emissions spectrometry (ICP-OES, Perkin-Elmer Optima 4300 DV). Available-K, Ca, Mg, Na, and S were determined from a 1 M HCl extraction (Camps-Arbestain et al. 2015 from Rayment and Lyons 2011), followed by ICP-OES analysis. Liming equivalence (CaCO3-eq) was determined by reacting the biochar with 1 M HCl followed by titration with 0.5 M NaOH (Rayment and Lyons 2011). Biochar N–NO3− and N–NH4+ were measured by extracting 1 g biochar with 2 M KCl in a 1:10 w/v ratio and analyzing the extract colorimetrically as for soil. Biochar pH and EC were determined from a 1:20 biochar-water solution.
Inorganic carbon content was determined by measuring CO2 evolved from an acidified sample of dry (non-incubated) biochar, similar to the method of Calvelo Pereira et al. (2017). A 1 g oven-dry (105 °C) biochar sample was placed in a glass beaker, which was sealed in a 1.8 L canning jar, and the headspace was flushed with CO2-free air. Using a syringe, 5 mL water and 10 mL 2 M HCl were added to the biochar via a septum in the jar lid attached to a drainage tube. After waiting 24 h for the sample to completely react, CO2 production was measured by connecting the jar to a CO2 analyzer, as described previously for soil incubations.
2.2 Experiment 2
Seven non-isotopically labeled biochars were studied in addition to the three barley plant biochars to evaluate Cmin and soil N relationships for a greater range of biochar types (Table 1). The non-labeled biochars included: grape wood (Vitis vinifera L.) prunings pyrolyzed at 350 and 700 °C at the USDA-ARS in Florence, South Carolina (Cantrell and Martin 2012); poultry litter obtained from a commercial source (Stutzman Environmental Products, Inc.), also pyrolyzed at 350 and 700 °C in Florence; a juniper wood (Juniperus occidentalis Hook.) biochar produced by flame-cap pyrolysis at > 1250 °C (Phillips et al. 2020b), and mixed-conifer wood and wheat straw (Triticum aestivum L.) biochars, both produced by gasification at > 1250 °C as described by Phillips et al. (2020a).
Three non-isotopic assessments of biochar Cmin were performed on these biochars, including (1) H/C atomic ratio, (2) water-extractable organic carbon (WEOC) content, and (3) permanganate-oxidizable (KMnO4-oxidizable) carbon content. Biochar total C, H, N, and ash content were measured by an external laboratory (Mineral Labs Inc., Salyersville, Kentucky). WEOC and total dissolved nitrogen (TDN) were determined from a 5.0 g sample extracted with 50 mL deionized water, as suggested by Chinu et al. (2017), followed by analysis using a TOC analyzer with automated acidification to remove inorganic C (TOC-VCPN, Shimadzu Scientific Instruments). KMnO4-oxidizable C was determined in the same way as the soil method described by Weil et al. (2003), but using only 0.15–1.0 g of biochar owing to its high carbon content.
Biochar impacts on soil N-mineralization were determined by incubating each of the ten biochars with the Willamette soil, using a fresh sample of the Willamette soil collected in spring 2019. 75 gram samples of soil, either unamended or amended with 2% biochar by mass, were prepared for aerobic incubations in the same way as experiment 1. Samples were incubated at 23 °C in a darkened environmental chamber, and three replicates of each treatment were destructively sampled on days 14 and 28 following Drinkwater et al. (1996). In addition, three replicate 10 g samples were prepared for each treatment as a day 0 baseline sample. Soils were analyzed for N–NO3− and N–NH4+ as described for experiment 1. Mineralized-N on days 14 and 28 were assessed as the difference between DIN on those days and day 0 baseline samples.
2.3 Statistical analysis
For experiment 1, cumulative soil respiration, biochar-C respiration, and soil N-mineralization were analyzed by two-way analysis of variance (ANOVA) to test whether responses differed by soil type or biochar pyrolysis temperature, using the base package of R (R Core Team 2020). Tukey’s honest significant difference test was used for post-hoc tests of pairwise treatment differences. Linear regression was also used to test whether soil N-mineralization correlated with biochar respired-C, alone or normalized by biochar total-N content. Biochar respired-C, soil type, and their interaction were initially evaluated as explanatory variables, and the non-significant interaction term was subsequently excluded.
For the larger set of biochars in experiment 2, regression analysis was again used to test whether soil N-mineralization correlated with KMnO4-oxidizable C, H:C ratio, WEOC, and WEOC:TDN. Averages of three analytical replicates for each biochar type were used for regressions. All of the measures of Cmin with the exception of H:C were visually determined to have long-tailed distributions (i.e., a few high values) and were log-transformed.
A principal component analysis (PCA) was performed on the dataset from experiment 2 to evaluate what qualities distinguished the biochars from one another, and to evaluate what characteristics corresponded with stimulation of soil N-mineralization. The initial dataset included all variables shown in Table 1. To stabilize the results and reduce ordination complexity, the large number of explanatory variables were reduced to nine, one fewer than the number of biochars analyzed. Some groups of variables that represented similar functions were reduced (e.g., inorganic-C, pH, and ash), and variables that made small contributions to the first three principal components were also omitted. Long-tailed data were log-transformed, and data were centered and scaled to normalize variances. PCA was performed with the ‘FactoMineR’ (Husson et al. 2020) and ‘factoextra’ (Kassambara and Mundt 2020) packages in R.
3 Results
3.1 Experiment 1: Barley biochar temperature series impacts on soil C- and N-mineralization
The respired-C from barley plant biochars decreased as pyrolysis temperature increased, thus demonstrating lower quantities of Cmin at higher pyrolysis temperatures (Fig. 1a). The 350, 500, and 700 °C biochars respired approximately 18, 6, and 4% of their original C contents over the 101-day incubation, respectively, with similar amounts respired in both soils.
A Timeseries of respired C from barley biochar relative to total biochar C and B respired soil C relative to total soil organic C during a 101-day incubation. Inset bar charts show cumulative respiration on day 101, where treatments sharing letters of the same case had non-significant differences (p > 0.05). The Chehalis (3.7% C) and Willamette (1.7% C) soils were collected near Corvallis, Oregon, USA. Biochars were pyrolyzed at 350, 500, or 700 °C. Error bars show standard deviation (N = 3)
Biochar amendments also had positive priming effects, increasing soil C respiration relative to unamended soil (Fig. 1b). In the Willamette soil, all the biochars caused significantly more soil respiration than unamended soil (p ≤ 0.02 for each pairwise comparison), and the strength of the priming effect increased with biochar pyrolysis temperature. Cumulative 101-day soil respiration increased by 20, 35, and 54% for the 350, 500, and 700 °C biochars, respectively, compared to unamended soil. In the high-C Chehalis soil, the priming effect was only significant for the 700 °C biochar, amounting to a 34% increase in cumulative respiration relative to the control (p = 0.002).
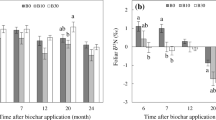
Rates of N-mineralization also increased with pyrolysis temperature (Fig. 2). Soils with no biochar or the 350 °C biochar initially immobilized N in the first days to weeks of incubation, while soils with 500 or 700 °C had net mineralization throughout the incubation (Fig. 2). Treatment differences were greatest on measurement day 42 and later converged. Focusing on day 42 to characterize treatment effects (Fig. 2 inset plots, and Fig. 3), the 350 °C biochar decreased net N-mineralization compared to unamended soil, although the effect was only significant in the Chehalis soil. The 500 and 700 °C biochars increased net N-mineralization in the Willamette soil, and were similar to the unamended control in the Chehalis soil.
Timeseries of dissolved inorganic nitrogen (DIN) concentrations over a 101-day incubation, with day 0 baseline concentrations subtracted. Inset bar charts show net N mineralized in 42 days. Treatments sharing letters of the same case had non-significant differences (p > 0.05). Error bars show 1 standard deviation (N = 3)
Net soil N-mineralization and cumulative soil-C respired on day 42 in barley biochar-amended soils, with quantities from unamended soils subtracted. Error bars are standard errors. Quadrats indicate whether biochar amendment increased or decreased N-mineralization and soil-C respiration relative to unamended soils
Figure 3 shows the relationship between cumulative soil N-mineralization and cumulative soil-C respired by day 42, relative to unamended soils. Overall, the 500 and 700 °C biochars tended to cause more positive priming of soil respiration and to stimulate more soil N-mineralization, while the 350 °C biochar had a smaller positive priming effect and immobilized N. While there was not a linear trend of more soil N-mineralized as more soil-C was respired, the biochars segregated into quadrats. The 350 °C biochar fell in the lower right quadrat—priming soil C and immobilizing soil N—while the 500 and 700 °C biochars generally fell in the upper right quadrat—priming soil C and mineralizing soil N.
Characteristics of the biochars and biochar + soil systems were evaluated as potential explanatory variables. Consistent with expectation, the C:N ratio of the biochar + soil system (Fig. 4a), and the C:N ratio of the biochars themselves (Fig. 4b) did not correlate with net N-mineralized by day 42. However, in support of hypothesis 1, a negative correlation was found between net N-mineralization and the Cmin:total N ratio (slope p-value = 0.009, R2 = 0.96 for a model with a single slope for the two soils). We note, however, the limited distribution of the data composing this regression, given the similarity between the 500 and 700 °C biochars. Because total-N quantities were similar for the three barley biochars (Table 1), the variation in soil N-mineralization was also explained well by Cmin content alone, with almost identical statistical results. Contrary to hypothesis 2, no correlation was found between the C:N ratio of biochar water extracts and soil N-mineralization (Fig. 4d).
Correlations between relative net mineralized-N after 42 days (relative = amended minus unamended soil) and characteristics of barley biochar and biochar + soil system, including A biochar + soil C to N ratio, B biochar only C to N ratio, C biochar cumulative respired-C in 42 days to N ratio, and D biochar water-extractable organic C to total dissolved N ratio. Biochars were pyrolyzed at 350, 500, and 700 °C. Error bars show standard deviations (N = 3)
3.2 Experiment 2: expanding to more biochar types
In the second experiment, the barley plant biochars and seven additional biochar types were incubated in Willamette soil (1.7% total C). The response of the barley biochars was compared in both experiments to assess repeatability. Soil N-mineralization, which was measured on day 14 in both experiments, was about 25% lower in the first experiment than in the second (data not shown), possibly related to a fresh collection of Willamette soil made in spring 2019 compared to fall 2018. Similar to experiment 1, the barley biochars in experiment 2 exhibited increased soil N-mineralization with increasing pyrolysis temperatures (Fig. 5).
N-mineralization in 28 days in Willamette soil versus C:N ratio of A the soil + biochar system, and B biochars alone. In A log-transformed N-mineralization had a significant correlation with soil + biochar C:N. N-mineralization also increased with pyrolysis temperature for individual feedstocks. In B, N-mineralization is shown as the difference between amended and unamended soil for comparison to Fig. 6. Error bars are standard deviations (too small to be visible in A, N = 3)
All but one of the biochars had a neutral or stimulatory effect on 28-day soil N-mineralization relative to unamended soil (Fig. 5b). Only the 350 °C poultry litter biochar reduced 28-day N-mineralization, but it stimulated high 14-day net N-mineralization (37.8 ± 24.3 mg N kg−1) followed by immobilization and low net N-mineralization on day 28 (6.65 ± 0.94 mg N kg−1). The rapid N-mineralization of the 350 °C poultry litter biochar was likely due to its very high N content, primarily in the form of NH4+ and organic-N (Table 1). All the other biochars had an approximate doubling of net N-mineralization from day 14 to day 28 (see supplemental materials, Fig. S2).
Soil N-mineralization in 28 days correlated positively with the C:N ratio of soil + biochar system (Fig. 5a), but not with the C:N ratio of biochar alone (Fig. 5b). When evaluated with respect to the Cmin content of the biochars, the larger set of biochars provided evidence to support the hypothesis that soil N-mineralization correlates negatively with biochar Cmin within individual feedstock types, but not across different feedstocks (Fig. 6).
Relative net N-mineralization in 28 days in Willamette soil (relative = amended minus unamended soil), versus biochar mineralizable-C characteristics. Biochar mineralizable component was characterized by A KMnO4-oxidizable C content, B atomic H:C ratio, C water-extractable organic carbon content, and D the ratio of water-extractable organic carbon to total dissolved N. Error bars are standard deviations (N = 3)
Overall, soil N-mineralization increased with higher pyrolysis temperatures while Cmin tended to decrease. Increases in soil N-mineralization with biochar pyrolysis temperature were observed for the barley plant, grape wood, and poultry litter biochar types, although variances were high for the 350 °C grape wood biochar. Simultaneously, Cmin content as quantified by all three measures—KMnO4-oxidation, H:C ratio, and WEOC—generally decreased with increasing pyrolysis temperature (Fig. 6a-c). However, the three measures of Cmin were not entirely consistent with each other (see Fig. S1). A notable exception to Cmin decreasing with pyrolysis temperature was that the poultry litter biochar had more KMnO4-oxidizable C at 700 °C than at 350 °C (Fig. 6a). The decline in Cmin with pyrolysis temperature was most consistent for the H:C measure (Fig. 6b).
Relationships between biochar Cmin and soil N-mineralization were weaker across feedstock types. A regression model of soil N-mineralization as a function of H:C and feedstock type showed a significant correlation when only pyrolysis biochars were considered (Fig. 6b, p = 0.013 for effect of H:C, R2 = 0.98) but a weaker correlation when the combustion biochars were included (p = 0.11 for effect of H:C, R2 = 0.84). The C:N ratio of biochar water extracts had no consistent relationship with soil N-mineralization, either within individual feedstocks or across the different feedstocks (Fig. 6d).
PCA was used to explore additional biochar characteristics apart from Cmin and dissolved C:N ratio that were associated with mineralization of soil N (Fig. 7). Initially, all three measures of biochar Cmin were included in the analysis, but KMnO4-oxidizable C had low loadings to the first two axes and was subsequently excluded to reduce ordination complexity. The first two axes of the PCA model explained 74% of the variance in the 9 biochar characteristics included in the model. Overall, the first principal component distinguished biochar feedstocks and the second principal component distinguished pyrolysis temperatures (Fig. 7a). The combustion biochars also segregated from pyrolysis biochars on component 2. The variables with the largest loadings in component 1 were 28-day N-mineralization and biochar C:N, which correlated positively with each other (Fig. 7b), while inorganic C content, which increased with pyrolysis temperature (Table 1), had the largest loading in component 2. Soil N-mineralization was negatively correlated with extractable-P, -K, and -Mg, and TDN. This reflected low N-mineralization rates from the three most nutrient dense biochars—the 350 °C grape wood and the two poultry litter biochars. H:C and WEOC, which were hypothesized to correlate negatively with soil N-mineralization, were orthogonal to N-mineralization in the PCA analysis. The multivariate analysis thus demonstrated possible unique responses of the most nutrient-dense biochars that were simultaneously higher in soluble N, and extractable P, Mg, and K.
4 Discussion
4.1 Generalizing soil impacts from biochar characteristics
A goal of this study was to evaluate whether biochar impacts on soil DIN content could be predicted from readily measured biochar C- and N- pools. The results suggested that generalizations of soil-N impacts were only predictable within a biochar feedstock. The hypothesis that soil N-mineralization correlates negatively with biochar Cmin was only somewhat supported for individual feedstocks produced at a range of temperatures (Figs. 4c and 6b) and was not supported across feedstocks and when pyrolysis and combustion chars were aggregated (Fig. 6b). Consistent with previous studies, we found greater variation in soil-N response across biochar feedstocks than across production temperatures (Rajkovich et al. 2012; Maaz et al. 2021).
Our results highlighted the limited utility of biochar total C:N ratios in predicting soil responses. In both experiments, soil N-mineralization tended to increase with the C:N ratio of soil + biochar system (Figs. 4a and 5a). This result directly contrasts with traditional models of decomposition and evidence from diverse ecosystems suggesting that increasing soil C:N ratios should reduce N-mineralization rates (Booth et al. 2005). Additionally, across a large range of biochar C:N ratios ranging from approximately 10 to 250, we found soil N-mineralization had no relationship with biochar C:N (Fig. 5b). This agrees with a large meta-analysis by Gao et al. (2019) that showed inconsistent impacts of C:N on soil N–NO3− and N–NH4+.
However, a limitation of our study was the lack of diverse measurements on the C:N ratio of the biologically-active component of the biochars. Biochar Cmin content was characterized with four methods (see supplemental Fig. S1 for comparisons of Cmin measures for the barley biochar temperature series). However, the N content of the mineralizable component was estimated only from water-extractable organic matter, and C data suggested that water extracts were only a small portion of the total biologically-active pool. Cmin estimates from isotopic partitioning of respired CO2 (Fig. 1a) were approximately 10 times greater than WEOC (Table 1), suggesting a considerable portion of biologically-available C and N were not contained in the water-extractable pool. Additionally, the lack of correlation between WEOC:TDN and soil N-mineralization suggested that biochar water extracts were insufficient to describe biologically-available N (Figs. 4d and 6d).
Our findings agree with others who have shown that generalizations of biochar impacts on soil N are difficult to make from simple biochar characterizations. For example, volatile matter (VM) content is an easily measured property that corresponds with a biochar’s degree of thermal alteration and Cmin content (Zimmerman 2010; Enders et al. 2012). However, the composition of the VM component has been shown to be chemically heterogenous (Spokas 2010; Spokas et al. 2011; Maaz et al. 2021), with variable impacts on soil microbial activity and N-transformations. Molecular structure measurements can help to describe the non-homogenous composition of pyrogenic organic matter (Heckman et al. 2017; Torres-Rojas et al. 2020) and will likely be useful in future work to generalize soil N-impacts from biochar characteristics. For instance, the PCA analysis (Fig. 7) showed that extractable nutrients had a negative correlation with soil N-mineralization. Molecular structure measurements may help show why the most nutrient-dense biochars had only a small impact on N-mineralization.
4.2 Linkages between soil C- and N-mineralization
A surprising result from this study was that all the biochars stimulated N-mineralization relative to unamended soil (Figs. 5b and 6). Only the low-temperature poultry litter biochar yielded less DIN than unamended soil after 28 days, but this was due to N-immobilization following a large pulse of N-mineralization earlier in the incubation (Fig. S2). Based on prior studies we expected most of the biochars to have no impact on DIN (Zheng et al. 2012) or to reduce DIN relative to unamended soil, with only high-N biochars (e.g., from manure) stimulating increases in DIN (Nguyen et al. 2017; Gao et al. 2019). Our results did not support this expectation, as the high-N (as measured by TDN) 700 °C poultry litter biochar yielded low quantities of DIN in soil incubations, while the low-N 700 °C barley biochar yielded the highest quantities of DIN (Figs. 5 and 6).
A second surprising result was that all three barley biochars in experiment 1 stimulated positive priming (Fig. 3). We had expected at least some of the biochars to cause negative priming, i.e., decreases in soil-C respiration. Although both positive and negative priming have been reported from biochar additions (Zimmerman 2010; Stewart et al. 2013; Whitman et al. 2014), a meta-analysis of 21 studies showed that incubations less than 6 months in duration generally suppressed soil respiration, and furthermore that negative priming was more common than positive priming in soils with clay contents between 10 and 40% (Wang et al. 2016), such as the Willamette and Chehalis soil types. Viewed in terms of the binary positive or negative impacts of biochar on soil N- and C-mineralization illustrated in Fig. 3, we expected at least some of the barley biochars to fall on the left side, corresponding with negative priming. Additionally, we were surprised that the 500 °C and 700 °C biochars both stimulated more positive priming than the 350 °C biochar, despite their lower Cmin content. Several studies have previously reported the reverse trend, with high-temperature biochars having less impact or reducing soil respiration compared to low temperature biochars (Cross and Sohi 2011; Ameloot et al. 2015; Wang et al. 2016).
The finding that biochars with more microbially-resistant biochemistry stimulated more soil decomposition is consistent, however, with previous isotopic studies that evaluated partitioning between soil and residue decomposition. Studying five uncharred crop residues, Stewart et al. (2015) demonstrated that a direct trade-off occurred between respiration of soil-derived C and residue-derived C in incubations. Residues that decayed the least produced the most soil-derived C, and vice versa. Because residue- and soil-respired C balanced each other, total C respired over the incubation was unrelated to residue quality. Whitman et al. (2014) also showed a trade-off between CO2 respired from biochar and soil in the first 48 h of incubations. High rates of respired soil-C corresponded with low rates of biochar-C respiration, in a highly significant linear relationship.
Our finding of more soil N-mineralization stimulated by high-temperature barley biochars was surprising given their low N-content and high C:N ratios, but was consistent with the stimulatory effect these biochars had on soil C-mineralization (Fig. 3). We also noted a difference between the two soil types, with biochar stimulating more C- and N-mineralization from the low-C Willamette soil (Figs. 1b and 2). Whitman et al. (2014) also found a difference in the responses of two soils, with stronger biochar priming effects in soils with less mineralizable-C. This stronger priming effect in the Willamette soil, as well as its higher initial C:N ratio which indicated less N-limitation (Table 1), likely explained the greater N-mineralization in the Willamette soil. These results showed that considering both biochar and soil characteristics together is important to determine amendment impacts on plant-available N.
Although uncommon, reports of biochar stimulating N-mineralization are not without precedent. For instance, additions of wood biochars to an organically-farmed soil in coastal Washington State increased potentially-mineralizable N levels (Gao et al. 2016), despite the fact that the biochars were poor sources of N.
4.3 Methodologies for biochar Cmin and soil N-mineralization
There are a large number of methods for characterizing biochar Cmin and soil N-mineralization, providing a spectrum from expediency on the one hand to mechanistic detail on the other hand. The three non-isotopic measures used to evaluate biochar Cmin—H:C ratio, KMnO4-oxidizable C, and WEOC—can be viewed as proxies for isotopic partitioning methods, which are preferred as a direct measure of biochar C mineralization (Budai et al. 2013; Leng et al. 2019). For the barley biochars, we found H:C ratio and KMnO4-oxidizable C content corresponded well with isotopic measures of Cmin, but that WEOC had a less linear relationship (Fig. S1). We also noted that both KMnO4-oxidizable C and WEOC yielded about 10% of Cmin detected by isotopic measurements (Table 1 and Fig. 1b), thus providing quasi-quantitative measures of Cmin. The H:C ratio has been recommended as a community standard for estimating biochar stability, and has been shown to correspond well with degree of aromaticity and biochar residence times from long-term incubations (Lehmann et al. 2015; Xiao et al. 2016). H:C ratio was also the Cmin measure that corresponded best with soil N-mineralization in experiment 2. Although H:C ratio does not provide a direct measure of Cmin content, we agree with its use as a non-isotopic assessment of biochar Cmin. Although we found good agreement between KMnO4-oxidizable C and respired biochar-C for the barley biochars, it was critiqued by Calvelo Pereira et al. (2011) who found it did not consistently agree with other thermal and oxidative measures of Cmin. Additionally, KMnO4-oxidizable C has been criticized for oxidizing many classes of compounds indiscriminately without reflecting microbial preference (Tirol-Padre and Ladha 2004).
Many laboratory methods are also used for characterizing soil N-mineralization, with common incubation times ranging from one to 30 weeks (Drinkwater et al. 1996; Sharifi et al. 2007). We used a 28-day aerobic incubation in experiment 2 and focused on day 42 for hypothesis testing in experiment 1, because our interest was to establish an approach for screening short-term impacts of many biochars. However, as described by Sharifi et al. (2007), different pools of mineralizable-N are recognized over different periods of incubation. Over the longer term (> 1 year), Nguyen et al. (2017) showed less impact of biochar on soil N–NH4+ but larger reductions in N–NO3− compared to short-term studies. Characterizing a wide range of biochars with inherently different mineralizable fractions in a short-term incubation likely misses important aspects, and assessing long-term outcomes is also important for completely characterizing biochar impacts on soil-N.
5 Conclusions
This study provided initial steps towards evaluating biochar characteristics that may be useful for predicting negative impacts on plant-available N. We showed that biochar C:N ratio was a poor predictor of soil N-mineralization, but that for a given feedstock, biochar Cmin content had a negative relationship with soil N-mineralization. Nevertheless, biochar Cmin was not related to soil N-mineralization across different feedstocks and biochar production methods, suggesting that Cmin is a chemically heterogenous pool with variable impacts on soil plant-available N content.
Perhaps one of the most interesting findings was that a diverse number of biochars all stimulated N-mineralization in the two soils that were studied. Based on prior studies, we expected biochar to have an overall neutral or suppressive impact on N-mineralization. However, our study indicated that despite having high C:N ratios and low N contents, biochars “unlocked” plant-available N through positive priming of soil C. These observations may be related to the fact that the two soils studied both had relatively high C contents, which readily supported the simultaneous decomposition of soil-C along with the added biochar-C. Still more surprising was that for a given feedstock, plant-available N was greater in response to high-temperature biochars that were more resistant to microbial decomposition than low-temperature biochars. These results add complexity to the range of soil-N responses that can be expected in response to biochar amendment. Because of the lack of easily measured biochar characteristics to predict soil-N responses, biochar users should monitor soil-N impacts of biochar amendment to adaptively-manage soil fertility.
Availability of data and materials
The datasets analyzed during the current study are available from the U.S.D.A. AgData Commons at the following URL: https://doi.org/10.15482/USDA.ADC/1523372.
Code availability
The code used for statistical analyses and generating figures are available from the corresponding author on reasonable request.
References
Ågren GI, Hyvönen R, Berglund SL, Hobbie SE (2013) Estimating the critical N: C from litter decomposition data and its relation to soil organic matter stoichiometry. Soil Biol Biochem 67:312–318. https://doi.org/10.1016/j.soilbio.2013.09.010
Ameloot N, Sleutel S, Das KC et al (2015) Biochar amendment to soils with contrasting organic matter level: effects on N mineralization and biological soil properties. GCB Bioenergy 7:135–144. https://doi.org/10.1111/gcbb.12119
Andrews N, Foster J (2007) Organic fertilizer calculator: a tool for comparing the cost, nutrient value, and nitrogen availability of organic materials. Oregon State University Extension Service
Aulakh MS, Walters DT, Doran JW et al (1991) Crop residue type and placement effects on denitrification and mineralization. Soil Sci Soc Am J 55:1020–1025. https://doi.org/10.2136/sssaj1991.03615995005500040022x
Baldock JA, Smernik RJ (2002) Chemical composition and bioavailability of thermally altered Pinus resinosa (Red pine) wood. Org Geochem 33:1093–1109. https://doi.org/10.1016/S0146-6380(02)00062-1
Bonanomi G, Ippolito F, Cesarano G et al (2017) Biochar as plant growth promoter: better off alone or mixed with organic amendments? Front Plant Sci 8:1570. https://doi.org/10.3389/fpls.2017.01570
Booth MS, Stark JM, Rastetter E (2005) Controls on nitrogen cycling in terrestrial ecosystems: a synthetic analysis of literature data. Ecol Monogr 75:139–157. https://doi.org/10.1890/04-0988
Budai A, Zimmerman AR, Cowie AL, et al (2013) Biochar carbon stability test method: an assessment of methods to determine biochar carbon stability. International Biochar Initiative
Budai A, Rasse DP, Lagomarsino A et al (2016) Biochar persistence, priming and microbial responses to pyrolysis temperature series. Biol Fertil Soils 52:749–761. https://doi.org/10.1007/s00374-016-1116-6
Calvelo Pereira R, Kaal J, Camps Arbestain M et al (2011) Contribution to characterisation of biochar to estimate the labile fraction of carbon. Org Geochem 42:1331–1342. https://doi.org/10.1016/j.orggeochem.2011.09.002
Calvelo Pereira R, Camps-Arbestain M, Wang T, Enders A (2017) Inorganic carbon. In: Singh B, Arbestain MC, Lehmann J (eds) Biochar: a guide to analytical methods. CRC Press, Taylor & Francis Group, Boca Raton, pp 51–63
Camps-Arbestain M, Amonette JE, Singh B et al (2015) A biochar classification system and associated test methods. In: Lehmann J, Joseph S (eds) Biochar for environmental management: science, technology and implementation, 2nd edn. Sterling, London, p 28
Cantrell KB, Martin JH (2012) Stochastic state-space temperature regulation of biochar production. Part I: theoretical development. J Sci Food Agric 92:481–489. https://doi.org/10.1002/jsfa.4618
Chinu K, Marjo CE, Joseph S, Singh B (2017) Dissolved carbon and LC-OCD of biochar. In: Singh B, Arbestain MC, Lehmann J (eds) Biochar: a guide to analytical methods. CRC Press, Taylor & Francis Group, Boca Raton London, pp 64–73
Clough TJ, Condron LM (2010) Biochar and the nitrogen cycle: introduction. J Environ Qual 39:1218–1223. https://doi.org/10.2134/jeq2010.0204
Cross A, Sohi SP (2011) The priming potential of biochar products in relation to labile carbon contents and soil organic matter status. Soil Biol Biochem 43:2127–2134. https://doi.org/10.1016/j.soilbio.2011.06.016
Deenik JL, McClellan T, Uehara G et al (2010) Charcoal volatile matter content influences plant growth and soil nitrogen transformations. Soil Sci Soc Am J 74:1259–1270. https://doi.org/10.2136/sssaj2009.0115
Doane TA, Horwath WR (2003) Spectrophotometric determination of nitrate with a single reagent. Anal Lett 36:2713–2722. https://doi.org/10.1081/AL-120024647
Drinkwater LE, Cambardella CA, Reeder JD, Rice CW (1996) Potentially mineralizable nitrogen as an indicator of biologically active soil nitrogen. In: Doran JW, Jones AJ (eds) SSSA special publications. Soil Science Society of America, Madison, pp 217–229
Ducey TF, Ippolito JA, Cantrell KB et al (2013) Addition of activated switchgrass biochar to an aridic subsoil increases microbial nitrogen cycling gene abundances. Appl Soil Ecol 65:65–72. https://doi.org/10.1016/j.apsoil.2013.01.006
Enders A, Hanley K, Whitman T et al (2012) Characterization of biochars to evaluate recalcitrance and agronomic performance. Bioresour Technol 114:644–653. https://doi.org/10.1016/j.biortech.2012.03.022
Gao S, Hoffman-Krull K, Bidwell AL, DeLuca TH (2016) Locally produced wood biochar increases nutrient retention and availability in agricultural soils of the San Juan Islands, USA. Agr Ecosyst Environ 233:43–54. https://doi.org/10.1016/j.agee.2016.08.028
Gao S, DeLuca TH, Cleveland CC (2019) Biochar additions alter phosphorus and nitrogen availability in agricultural ecosystems: a meta-analysis. Sci Total Environ 654:463–472. https://doi.org/10.1016/j.scitotenv.2018.11.124
Heckman K, Torres D, Swanston C, Lehmann J (2017) Carbon and nitrogen molecular composition of soil organic matter fractions resistant to oxidation. Soil Res 55:809. https://doi.org/10.1071/SR16182
Husson F, Josse J, Le S, Mazet J (2020) “FactoMineR”: multivariate exploratory data analysis and data mining. Version 2.4URL http://factominer.free.fr
Jeffery S, Abalos D, Spokas KA, Verheijen FGA (2015) Biochar effects on crop yield. In: Lehmann J, Joseph S (eds) Biochar for environmental management: science, technology and implementation. Sterling, London, pp 301–325
Kassambara A, Mundt F (2020) “factoextra”: Extract and Visualize the Results of Multivariate Data Analyses. Version 1.0.7URL http://www.sthda.com/english/rpkgs/factoextra
Keiluweit M, Nico PS, Johnson MG, Kleber M (2010) Dynamic molecular structure of plant biomass-derived black carbon (Biochar). Environ Sci Technol 44:1247–1253. https://doi.org/10.1021/es9031419
Kuzyakov Y, Subbotina I, Chen H et al (2009) Black carbon decomposition and incorporation into soil microbial biomass estimated by 14C labeling. Soil Biol Biochem 41:210–219. https://doi.org/10.1016/j.soilbio.2008.10.016
Lehmann J, Abiven S, Kleber M et al (2015) Persistence of biochar in soil. In: Lehmann J, Joseph S (eds) Biochar for environmental management: science, technology and implementation, 2nd edn. Routledge, Taylor & Francis Group, London
Leng L, Huang H, Li H et al (2019) Biochar stability assessment methods: a review. Sci Total Environ 647:210–222. https://doi.org/10.1016/j.scitotenv.2018.07.402
Lifschitz CH, Grusak MA, Butte NF (2002) Carbohydrate digestion in humans from a β-glucan-enriched barley is reduced. J Nutr 132:2593–2596. https://doi.org/10.1093/jn/132.9.2593
Liu Z, Dugan B, Masiello CA, Gonnermann HM (2017) Biochar particle size, shape, and porosity act together to influence soil water properties. PLoS ONE 12:1–19. https://doi.org/10.1371/journal.pone.0179079
Liu Q, Zhang Y, Liu B et al (2018) How does biochar influence soil N cycle? A meta-analysis. Plant Soil 426:211–225. https://doi.org/10.1007/s11104-018-3619-4
Maaz TM, Hockaday WC, Deenik JL (2021) Biochar volatile matter and feedstock effects on soil nitrogen mineralization and soil fungal colonization. Sustainability 13:2018. https://doi.org/10.3390/su13042018
Manzoni S (2017) Flexible carbon-use efficiency across litter types and during decomposition partly compensates nutrient imbalances—results from analytical stoichiometric models. Front Microbiol 8:661. https://doi.org/10.3389/fmicb.2017.00661
Mia S, Singh B, Dijkstra FA (2017) Aged biochar affects gross nitrogen mineralization and recovery: a 15N study in two contrasting soils. GCB Bioenergy 9:1196–1206. https://doi.org/10.1111/gcbb.12430
Nguyen TTN, Xu C-Y, Tahmasbian I et al (2017) Effects of biochar on soil available inorganic nitrogen: a review and meta-analysis. Geoderma 288:79–96. https://doi.org/10.1016/j.geoderma.2016.11.004
Phillips CL, Light SE, Gollany HT et al (2020a) Can biochar conserve water in Oregon agricultural soils? Soil till Res 198:104525. https://doi.org/10.1016/j.still.2019.104525
Phillips CL, Meyer KM, Trippe KM (2020b) Is biochar applied as surface mulch beneficial for grassland restoration? Geoderma 375:114457. https://doi.org/10.1016/j.geoderma.2020.114457
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rajan SSS, Brown MW, Boyes MK, Upsdell MP (1992) Extractable phosphorus to predict agronomic effectiveness of ground and unground phosphate rocks. Fert Res 32:291–302. https://doi.org/10.1007/BF01050366
Rajkovich S, Enders A, Hanley K et al (2012) Corn growth and nitrogen nutrition after additions of biochars with varying properties to a temperate soil. Biol Fertil Soils 48:271–284. https://doi.org/10.1007/s00374-011-0624-7
Rayment GE, Lyons DJ (2011) Soil chemical methods: Australasia. CSIRO Publishing, Collingwood
Rhine ED, Mulvaney RL, Pratt EJ, Sims GK (1998) Improving the Berthelot reaction for determining ammonium in soil extracts and water. Soil Sci Soc Am J 62:473. https://doi.org/10.2136/sssaj1998.03615995006200020026x
Sharifi M, Zebarth BJ, Burton DL et al (2007) Evaluation of some indices of potentially mineralizable nitrogen in soil. Soil Sci Soc Am J 71:1233–1239. https://doi.org/10.2136/sssaj2006.0265
Soil Survey Staff U-N (2006) Official Soil Series Descriptions [Online]. United States Department of Agriculture, Lincoln, NE
Spokas KA (2010) Review of the stability of biochar in soils: predictability of O:C molar ratios. Carbon Manag 1:289–303. https://doi.org/10.4155/cmt.10.32
Spokas KA, Novak JM, Stewart CE et al (2011) Qualitative analysis of volatile organic compounds on biochar. Chemosphere 85:869–882. https://doi.org/10.1016/j.chemosphere.2011.06.108
Spokas KA, Cantrell KB, Novak JM et al (2012) Biochar: a synthesis of its agronomic impact beyond carbon sequestration. J Environ Qual 41:973–989. https://doi.org/10.2134/jeq2011.0069
Stewart CE, Zheng J, Botte J, Cotrufo MF (2013) Co-generated fast pyrolysis biochar mitigates green-house gas emissions and increases carbon sequestration in temperate soils. GCB Bioenergy 5:153–164. https://doi.org/10.1111/gcbb.12001
Stewart CE, Moturi P, Follett RF, Halvorson AD (2015) Lignin biochemistry and soil N determine crop residue decomposition and soil priming. Biogeochemistry 124:335–351. https://doi.org/10.1007/s10533-015-0101-8
Tirol-Padre A, Ladha JK (2004) Assessing the reliability of permanganate-oxidizable carbon as an index of soil labile carbon. Soil Sci Soc Am J 68:969–978. https://doi.org/10.2136/sssaj2004.9690
Torres-Rojas D, Hestrin R, Solomon D et al (2020) Nitrogen speciation and transformations in fire-derived organic matter. Geochim Cosmochim Ac 276:170–185. https://doi.org/10.1016/j.gca.2020.02.034
Trinsoutrot I, Recous S, Bentz B et al (2000) Biochemical quality of crop residues and carbon and nitrogen mineralization kinetics under nonlimiting nitrogen conditions. Soil Sci Soc Am J 64:918–926. https://doi.org/10.2136/sssaj2000.643918x
Vigil MF, Kissel DE (1991) Equations for estimating the amount of nitrogen mineralized from crop residues. Soil Sci Soc Am J 55:757–761. https://doi.org/10.2136/sssaj1991.03615995005500030020x
Wang T, Camps-Arbestain M, Hedley M, Bishop P (2012) Predicting phosphorus bioavailability from high-ash biochars. Plant Soil 357:173–187. https://doi.org/10.1007/s11104-012-1131-9
Wang J, Xiong Z, Kuzyakov Y (2016) Biochar stability in soil: meta-analysis of decomposition and priming effects. GCB Bioenergy 8:512–523. https://doi.org/10.1111/gcbb.12266
Weil R, Islam K, Stine MA et al (2003) Estimating active carbon for soil quality assessment: a simplified method for laboratory and field use. Am J Altern Agric 18:3–17
Whitman T, Zhu Z, Lehmann J (2014) Carbon mineralizability determines interactive effects on mineralization of pyrogenic organic matter and soil organic carbon. Environ Sci Technol 48:13727–13734. https://doi.org/10.1021/es503331y
Woolf D, Amonette JE, Street-Perrott FA et al (2010) Sustainable biochar to mitigate global climate change. Nat Commun 1:56. https://doi.org/10.1038/ncomms1053
Xiao X, Chen Z, Chen B (2016) H/C atomic ratio as a smart linkage between pyrolytic temperatures, aromatic clusters and sorption properties of biochars derived from diverse precursory materials. Sci Rep 6:22644. https://doi.org/10.1038/srep22644
Zhang A, Liu Y, Pan G et al (2012) Effect of biochar amendment on maize yield and greenhouse gas emissions from a soil organic carbon poor calcareous loamy soil from Central China Plain. Plant Soil 351:263–275. https://doi.org/10.1007/s11104-011-0957-x
Zheng J, Stewart CE, Cotrufo MF (2012) Biochar and nitrogen fertilizer alters soil nitrogen dynamics and greenhouse gas fluxes from two temperate soils. J Environ Qual 41:1361–1370. https://doi.org/10.2134/jeq2012.0019
Zimmerman AR (2010) Abiotic and microbial oxidation of laboratory-produced black carbon (Biochar). Environ Sci Technol 44:1295–1301. https://doi.org/10.1021/es903140c
Acknowledgements
L. Pruessner, M. Cruse, J. Vasen, P. Mahato, and K. Sugimoto of USDA-ARS provided laboratory assistance. P. Hayes and D. Bryla provided biochar feedstocks. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Funding
This work was supported by the USDA Agricultural Research Service projects in the laboratories of K.M.T (2072-12620-001), C.L.P. (5030-12000-015), C.E.S (3012-11120-001), J.M.N. (6082-12630-001), and M.A.G. (3060-43440-016), and a Grant from the Institute for Working Forest Landscapes at Oregon State University.
Author information
Authors and Affiliations
Contributions
The study was conceptualized and designed by CLP, KMT, TW, MG-J, and CES. Material preparation was performed by MG, DW, JN, and KM. Data collection was performed by CLP, KM, CW, MG-J, TW, and CES. Analyses were performed and the first draft of the manuscript was written by CLP. All authors commented on previous versions of the manuscript and approved the final manuscript. Funding acquisition and supervision were provided by KMT.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Phillips, C.L., Meyer, K.M., Garcia-Jaramillo, M. et al. Towards predicting biochar impacts on plant-available soil nitrogen content. Biochar 4, 9 (2022). https://doi.org/10.1007/s42773-022-00137-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42773-022-00137-2