Abstract
In the present study, we studied the morphological variations in four populations of Oxynoemacheilus angorae distributed in three basins of Turkish inland waters using a geometric morphometric technique. For this purpose, a total of 55 specimens were collected from the Kızılırmak, Sakarya and Marmara basins. To extract body shape data, the left side of the specimens was photographed, and 15 landmark points on the 2D pictures were defined and digitized. After generalized procrustes analysis, the body shape data were analyzed using multivariate analyses, such as principal component analysis, canonical variation analysis and cluster analysis. The deformation grids were used to show how the body shape changed. Based on the results, two groups, namely Nevşehir and Ankara, by having a deeper body and head and a somewhat longer caudal peduncle, and Yalova and Eskişehir due to lower body and shorter caudal peduncle were clustered. Based on the results, O. angorae adapts itself to different habitats by adjusting head and body depth and caudal peduncle length based on habitat parameters.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The morphological features in fishes are used for identifying population units and morphological variations (Zelditch et al. 2004; Mouludi-Saleh and Eagderi 2021a; Yedier and Bostanci 2021; Rodrigues-Oliveira et al. 2022). Traditional morphometric is a set of distance measurements of the biological structures, whereas, in the geometric morphometric technique, the landmark points or outlines are used to extract shape data (Mouludi-Saleh et al. 2020a). The geometric method has been described as an effective method in morphological studies compared to the traditional one (Loy et al. 1996; Radkhah et al. 2016) due to its higher efficiency in extracting shape data (Mouludi-Saleh et al. 2019; Nasri et al. 2019).
In Turkey, the nemacheilid fishes possess 59 species belonging to five genera, including Oxynoemacheilus, Paracobitis, Schistura, Seminemacheilus, and Turcinoemacheilus of which the genus Oxynoemacheilus is the most diverse one, with 48 species (Çiçek et al. 2015, 2018, 2019, 2020, 2021; Yedier et al. 2021a, b; Freyhof et al. 2021; Yoğurtçuoğlu et al. 2021). Among the members of the Oxynoemacheilus, Angora loach, O. angorae (Steindachner, 1897), described from the Çubuk Stream (Sakarya Basin), has a wide distribution covering other inland waters of Turkey, including the Marmara, Susurluk, Sakarya and Kızılırmak basins (Çiçek et al. 2019). This species has been reported from Iranian inland waters; however, Freyhof et al. (2011) rejected its occurrence in the Caspian Sea Basin of Iran and considered it as an endemic species of the western and Central Anatolian and Black Sea basins. Nemacheilid species particularly some member of the genus Oxynoemacheilus show high morphological variations as reported in O. seyhanensis (Seçer et al. 2020), and O. bergianus (Mohammadi et al. 2020; Mouludi-Saleh and Eagderi 2021b), revealing their high phenotypic plasticity in their different populations.
Given the wide distribution range of this species, the aim of our study was to assess whether O. angorae populations display morphological variations in different river basins and these probable morphological variations are habitat-dependent. Therefore, we specifically compared the morphological characteristics in four populations of O. angorae from three Turkish inland basins, and explored whether some habitat characteristics such as river width or altitude could potentially explain inter-population morphological differences.
2 Martial and Methods
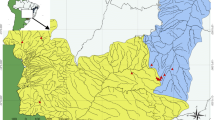
During the spring of 2019, a total of 55 specimens of O. angorae were collected from three basin of Turkish inland waters, including 14 from output of the Doyduk Dam Lake in Nevşehir (Kızılırmak Basin), 15 from Çubuk Stream (Ankara), 11 from Düden Stream (Eskişehir) (Sakarya Basin) and 15 from Altınova Stream (Yalova) (Marmara Basin) using an electrofishing device (Figs. 1, 2 and Table 1). After anesthesia, the specimens were fixed in 4% buffered formalin and transferred to the Hydrobiology Laboratory at Nevsehir Haci Bektas Veli University.
During sampling, some environmental characteristics of the habitats, including altitude, river depth, width, the average size of the substrate stones, slope, and water velocity, were measured. The coordinates and altitude of the sampling point were recorded using a hand-held GPS (Garmin eTrex 30x). The river widths were measured using a tape measure at the anterior, middle, and posterior points of the sampling station, and their average was considered the river width of the station. The river depth was measured across the river at twenty points with about 1 m distance using a graduated wooden pole that was averaged and considered as the depth of a given sampling point. The slope of the river bed was measured using a Suunto device (PM-PC Clinometer). The water velocity was measured three times and averaged at each sampling point using the floating object method (Hasanli 1999).
For extracting shape data in geometric morphometric, the left sides of the fresh specimens were photographed using a copy-stand equipped with a digital camera (Canon EOS 700D with 18 MP resolution). Then, 15 homologous landmark-points were digitized using tpsDig2 software (Rohlf 2004 version 1.04) on 2D images (Fig. 3). The extracted data were analyzed using Generalized Procrustes Analysis (GPA) to remove non-shape data, including size, position and direction (Zeldtich et al. 2004). Then, data were analyzed using Principal Components Analysis (PCA), Canonical Variate Analysis (CVA) and Cluster Analysis (CA) as a complement to CVA by adopting the Euclidean square distance as a measure of dissimilarity. Permutation test was used to prepare a pairwise distance of the studied populations based on MANOVA/CVA analysis with 10,000 repetitions in MorphoJ software version 1.02j.
The 15 defined landmark-points to extract the body shape data of Oxynoemacheilus angorae. 1. Anterior-most point of the snout tip on the upper jaw. 2. Center of eye. 3. Dorsal edge of the head perpendicular to the center of eye. 4. The line extends perpendicularly to the ventral edge of the opercle above the head. 5. Origin and 6. Insertion point of the dorsal-fin base. 7. Postero-dorsal end of the caudal peduncle at its connection to caudal fin. 8. Postero-ventral end of the caudal peduncle at its connection to caudal fin. 9. insertion and 10. Origin point of the anal-fin base. 11. Most anterior point of the ventral fin. 12. Most anterior point of the pectoral fin. 13. Ventral end of the gill slit and 14. Ventral edge of the head perpendicular to the center of eye. 15. Mandibular barbel
The Mahalanobis and Procrustes distances (as the degree of differentiation of the body shape in canonical variate analysis) were calculated to explore the degree of the morphological distance between the studied populations. For shape differences, each population was compared with the consensus configuration visualized as deformation grids presented with CA. All analyses were performed in PAST V2.17b (Hammer et al. 2001) and MorphoJ (version 1.02j) (Klingenberg 2011) software. A procrustes regression was used to find the environmental variables that significantly influenced the morphology of the populations (Sherratt 2014). For morphological data, we used procrustes distance as a descriptor for the variation in shape (Bookstein 1991). The regression was performed in R 3.6.1 using the package geomorph (Adams et al. 2021).
3 Results
In PCA, the first three principal components accounted for a total of 66.54% (PC1 + PC2 + PC3) of the variance (PC1 = 42.59, PC2 = 14.66 and PC3 = 9.28; Fig. 4). The distribution of the studied populations is presented in Fig. 5, and the body shape changes along the PC1 and PC2 axes in Fig. 6. By moving along the PC1 axis, the position of the nape (landmark-points: LM 4), the position of the dorsal (LM 5 and 6) and anal (LM 9 and 10) fins’ bases are altered, and in the PC2 axis, the position of the pectoral (LM 12), dorsal (LM and 6) and ventral fins’ bases were altered.
Canonical Variate Analysis (CVA) based on the P-value obtained from a permutation test with 10,000 replications showed a significant difference (Wilks-lambda = 0.001, F = 8.878, P < 0.01) in the body shape of the four studied populations (Fig. 7). The results of the through permutation test showed that the studied populations have significant differences in their morphological patterns (P < 0.001, Table 2).
The Mahalanobis and Procrustes distances as the degree of body shape differentiation between the studied populations are given in Tables 3 and 4. According to the results, the greatest Mahalanobis and Procrastes were found between the Yalova and Ankara populations. The cluster analysis of the studied populations based on the body shape pattern showed two main branches, including the Nevşehir + Ankara due to having a higher body and head depth and a longer caudal peduncle, and Yalova + Eskişehir by having lower body and a shorter caudal peduncle (Fig. 8).
The procrustes regression indicated that the width and depth of the rivers had a significant effect on the morphology of the specimens (Table 5). The rest of the environmental variables were redundant and therefore eliminated from the analysis.
4 Discussion
Morphological variations between different populations of a single species can be due to environmental characteristics, i.e., water flow, substrate and vegetation types and genetic differences (Nicieza 1995; Guill et al. 2003; Nasri et al. 2015), leading to morphological flexibility (Cadrin and Silva 2005; Mouludi-Saleh et al. 2020b). In the present study, four populations of O. angorae were studied to investigate the body shape variations for understanding the pattern of their morphological adaptations in different habitats. Based on the results, the differences between the body shapes of the studied populations were those of the body and head depth and caudal peduncle length. The higher body depth is more efficient in low flow areas and marginal vegetation to increase rapid maneuverability (Blake 1983) as seen in the Nevşehir and Ankara populations, whereas the lower body depth is reported to be proper in water bodies with high flow to reduce friction (Barlow 1961) as seen in the Yalova and Eskişehir populations.
As seen in Fig. 7, Angora loach has an eel-like body shape and, based on the results, this species is adapted to different habitats by adjusting its caudal peduncle length and body depth, i.e., as its morphological adaptation pattern. The tail is a serious locomotive organ in fish species, and its size and shape effect its swimming performance (Plaut 2000; Yang et al. 2013). A longer caudal area is an adaptation to have higher swimming performance but higher drag, whereas in a shorter one, both swimming performance and drag force are decreased (Fu et al. 2013). In Yalova and Eskişehir populations, the shorter tail is accompanied by deeper body, i.e., having a fusiform body shape that is proper for the swift current of Yalova and Eskişehir water bodies (Donley and Dikson 2000). In Nevşehir and Ankara samples, longer caudal peduncles can provide powerful swimming performance in the slower currents of their habitats (Watson and Balon 1984). In addition, a longer caudal peduncle enhances fast-start swimming performance (Langerhans 2009), and this is elicited by increasing prediction and prey capture (Langerhans 2009; Langerhans and Reznick 2010). As seen in the Eskişehir and Yalova populations, the smaller head, longer caudal peduncle and relatively lower body are suitable for living in fast-flowing water at a lower depth (Darcy 1985; Zamani-Faradonbe et al. 2015). The results revealed that two environmental factors, i.e., depth and width of rivers, have an effect on the morphology of the studied populations of O. angorae. These results of environmental parameters are in agreement with morphological changes in the Yalova and Eskişehir populations. However, in the Eskişehir, having deeper habitat with a wider river and increasing the river width can lead to decreasing its depth or providing some microhabitat with lower depths, especially in the littoral area, as was the case with this habitat (Fig. 9). In previous studies on the member of Oxynoemacheilus species, important morphological traits in separating their populations were those of inter orbital and maxillary barbell lengths (Seçer et al. 2020), pectoral fin height, distance between pelvic and anal fins (Mohammadi et al. 2020) and caudal peduncle length and depth, body depth, head length and position of the dorsal, anterior and pectoral fins (Mouludi-Saleh and Eagderi 2021b). These variations in the morphological variation of other members of this genus show different strategies to adapt their morphology to various habitats.
Various environmental parameters (temperature, turbidity, salinity, depth, and velocity of water bodies) can cause a change in the morphological traits, resulting in the morphological separation of populations (Eagderi et al. 2013, 2020). Based on the results, habitat-dependent morphological patterns in the studied species were head and body depth and caudal peduncle length, i.e., this species adapts itself to different habitats by adjusting these morphological features based on habitat parameters. However, the morphological change patterns indicate the high plasticity ability of this species to develop its distribution area in different inland waters of Turkey.
Data Availability
The data that support the findings of this study are available on request from the corresponding author.
References
Adams D, Collyer M, Kaliontzopoulou A, Sherratt E (2021) Geomorph: software for geometric morphometric analyses. R Package Version 3(2):1
Barlow GW (1961) Social behavior of the desert pupfish, Cyprinodon macularius, in the field and in the aquarium. Am Midl Nat 65(2):339–359. https://doi.org/10.2307/2422959
Blake RW (1983) Fish locomotion. CUP Archive, p 77
Bookstein FL (1991) Morphometric tools for landmark data. Geometry and biology. Cambridge University Press, Cambridge, p 435
Cadrin SX, Silva VM (2005) Morphometric variation of yellowtail flounder. ICES J Mar Sci 62(4):683–694. https://doi.org/10.1016/j.icesjms.2005.02.006
Çiçek E, Sungur-Birecikligil S, Fricke R (2015) Freshwater fishes of Turkey: a revised and updated annotated checklist. Bih Biol 9(2):141–157
Çiçek E, Fricke R, Sungur S, Eagderi E (2018) Endemic freshwater fishes of Turkey. Fishtaxa 3(4):1–39
Çiçek E, Eagderi S, Sungur S (2019) Oxynoemacheilus phoxinoides (Erk’akan, Nalbant & Özeren, 2007): a junior synonym of Oxynoemacheilus angorae (Steindachner, 1897). FishTaxa 4(1):13–17
Çiçek E, Sungur S, Fricke R (2020) Freshwater lampreys and fishes of Turkey; a revised and updated annotated checklist 2020. Zootaxa 4809(2):241–270. https://doi.org/10.11646/zootaxa.4809.2.2
Çiçek E, Eigdari S, Sungur S, Secer B (2021) Species of Oxynoemacheilus Bănărescu & Nalbant, 1966 (Actinopterygii: Nemacheilidae) in the Turkish Part of the Kura-Aras River System, with the First Detailed Evidence for the Occurrence of O. bergianus (Derjavin, 1934) and O. cf. elsae Eagderi et al., 2018. Acta Zool Bul 73(2):171–178
Darcy GH (1985) Synopsis of biological data on the sand perch, Diplectrum formosum (Pisces, Serranidae). NOAA Tech Rep Circ NMFS 26:1–21
Donley JM, Dickson KA (2000) Swimming kinematics of juvenile kawakawa tuna (Euthynnus affinis) and chub mackerel (Scomber japonicus). J Exp Biol 203(20):3103–3116
Eagderi S, Esmaeilzadegan E, Madah A (2013) Body shape variation in riffle minnows (Alburnoides eichwaldii De Filippii, 1863) populations of Caspian Sea basin. J Taxon Biosys 5(4):1–8
Eagderi S, Mouludi Saleh A, Poorbagher H (2020) Sexual dimorphism comparison of body shape of Esmaeilius sophiae using geometric morphometric method. J Anim Env 12(4):311–316
Freyhof J, Erk’akan F, Özeren C, Perdices A (2011) An overview of the western Palaearctic loach genus Oxynoemacheilus (Teleostei: Nemacheilidae). Ichthyol Expl Fresh 22(4):301–312
Freyhof J, Yoğurtçuoğlu B, Kaya C (2021) Oxynoemacheilus sarus/, a new nemacheilid loach from the lower Ceyhan and Seyhan in southern Anatolia (Teleostei: Nemacheilidae). Zootaxa 4964(1):123–139. https://doi.org/10.11646/zootaxa.4964.1
Fu C, Cao ZD, Fu SJ (2013) The effects of caudal fin loss and regeneration on the swimming performance of three cyprinid fish species with different swimming capacities. J Exp Bio 216(16):3164–3174. https://doi.org/10.1242/jeb.084244
Guill JM, Hood CS, Heins DC (2003) Body shape variation within and among three species of darters (Perciformes: Percidae). J Ecol Fresh 12(2):134–140. https://doi.org/10.1034/j.1600-0633.2003.00008.x
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palae Elect 4(1):1–9
Hasanli AM (1999) Diverse methods to water measurement (Hydrometry). Shiraz University publication, Iran, p 265
Klingenberg CP (2011) MorphoJ: an integrated software package for geometric morphometrics. Mol Ecol Resour 11:353–357. https://doi.org/10.1111/j.1755-0998.2010.02924.x
Langerhans RB (2009) Morphology, performance, fitness: functional insight into a post-Pleistocene radiation of mosquitofish. Biol Lett 5(4):488–491. https://doi.org/10.1098/rsbl.2009.0179
Langerhans RB, Reznick DN (2010) Ecology and evolution of swimming performance in fishes: predicting evolution with biomechanics. Fish Locomot Eco-Ethol Perspect 200:248
Loy A, Cataudella S, Corti M (1996) Shape changes during the growth of the sea bass, Dicentrarchus labrax (Teleostea: Perciformes), in relation to different rearing conditions. Advances in morphometrics. Springer, Boston, MA, pp 399–405. https://doi.org/10.1007/978-1-4757-9083-2_33
Mohammadi S, Eagdari S, Pourbagher H, Mouludi-Saleh A (2020) Morphological variation of Oxynoemacheilus bergianus (Derzhavin, 1934) from the Namak Lake and Caspian Sea basins using traditional morphometric method. J Fish Scie Tech 9(4):285–292
Mouludi-Saleh A, Eagderi S (2021a) Habitat-associated morphological divergence of Gasterosteus aculeatus in the Southern Caspian Sea Basin. Iran J Sci Technol Trans Sci 45(1):121–125. https://doi.org/10.1007/s40995-020-01005-z
Mouludi-Saleh A, Eagderi S (2021b) Morphological variations of Oxynemacheilus bergianus (Derzhavin, 1934) in two inland water basins of Iran using geometric morphometric method. J Appl Ichthyol Res 8(4):71–76
Mouludi-Saleh A, Eagderi S, Poorbagher H, Kazemzadeh S (2019) The effect of body shape type on differentiability of traditional and geometric morphometric methods: a case study of Channa gachua (Hamilton. Eur J Biol 78(2):165–168. https://doi.org/10.26650/EurJBiol.2019.0011
Mouludi-Saleh A, Eagderi S, Cicek E, Sungur S (2020a) Morphological variation of Transcaucasian chub, Squalius turcicus in southern Caspian Sea basin using geometric morphometric technique. Biol 75(10):1585–1590. https://doi.org/10.2478/s11756-019-00409-6
Mouludi-Saleh A, Eagderi S, Latif-Nejad Sh, Nasri M (2020b) The morphological study of trancaspian marinka (Schizothorax pelzami) in Harirud and Dasht-e Kavri basins using geometric morphometric technique. Nova Biol Rep 7(2):185–191
Nasri M, Eagderi S, Farahmand H (2015) Interspecies morphological variation of Cyprinion watsoni (Day, 1872) from southern and southeastern inland water basins of Iran based on Geometric morphometrics method. J Appl Ichthyol Res 2(2):1–14
Nasri M, Eagderi S, Farahmand H, Nezhadheydari H (2019) Interspecific morphological variation among members of the genus Cyprinion Heckel, 1843 (Teleostei: Cyprinidae) in Iran, using landmark-based geometric morphometric technique. Iran J Ichthyol 6(1):54–64. https://doi.org/10.22034/iji.v6i1.404
Nicieza AG (1995) Morphological variation between geographically disjunct populations of Atlantic salmon: the effects of ontogeny and habitat shift. Fun Eco 9:448–456. https://doi.org/10.2307/2390008
Plaut I (2000) Effects of fin size on swimming performance, swimming behaviour and routine activity of zebrafish Danio rerio. J Exp Biol 203(4):813–820
Radkhah A, Poorbagher H, Eagderi S (2016) Investigation of morphological differences of Capoeta capoeta populations in the upstream and downstream of Zarinerood River in Urmia Lake Basin. J Anim Env 8(3):167–174
Rodrigues-Oliveira IH, Kavalco KF, Pasa R (2022) Body shape variation in the Characid Psalidodon rivularis from São Francisco river, Southeast Brazil (Characiformes: Stethaprioninae). Acta Zool 00:1–10. https://doi.org/10.1111/azo.12415
Rohlf FJ (2004) tpsDIG. Version 1.40. Department of Ecology and Evolution. State University of New York at Stony Brook, New-York
Seçer B, Mouludi-Saleh A, Eagderi S, Çiçek E, Sungur S (2020) Morphological flexibility of Oxynoemacheilus seyhanensis in different habitats of Turkish inland waters: a case of error in describing a populations as distinct species. Iran J Ichthyol 7(3):258–264
Sherratt E (2014) Quick guide to geomorph v.2.1.4
Watson DJ, Balon EK (1984) Ecomorphological analysis of fish taxocenes in rainforest streams of northern Borneo. J Fish Biol 25(3):371–384. https://doi.org/10.1111/j.1095-8649.1984.tb04885.x
Yang H, Cao ZD, Fu SJ (2013) Effect of caudal amputation on swimming capacity, energy expenditure and behavior in juvenile Chinese bream, Parabramis pekinensis. Acta Hydrobiol Sin 37:157–163
Yedier S, Bostanci D (2021) Intra- and interspecific discrimination of Scorpaena species from the Aegean, Black, Mediterranean and Marmara seas. Sci Mar 85(3):197–209. https://doi.org/10.3989/scimar.05185.018
Yedier S, Bostanci D, Polat N (2021) First record of Oxynoemacheilus angorae (Steindachner, 1897) from perşembe plateau meandering streams in the Ordu-Turkey. Karadeniz Fen Bilimleri Dergisi 11(1):161–167. https://doi.org/10.31466/kfbd.905681
Yedier S, Bostanci D, Polat N (2021b) Some population characteristics of Oxynoemacheilus angorae (Steindachner, 1897) from the Perşembe Plateau meandering streams in Ordu. Turkey Fish Aqua Life 29(2):100–107. https://doi.org/10.2478/aopf-2021-0012
Yoğurtçuoğlu B, Kaya C, Özuluğ M, Freyhof J (2021) Oxynoemacheilus isauricus, a new nemacheilid loach from Central Anatolia (Teleostei: Nemacheilidae). Zootaxa 4975(2):369378. https://doi.org/10.11646/zootaxa.4975.2.7
Zamani-Faradonbe M, Eagderi S, Moradi M (2015) Patterns of body shape variation in Capoeta gracilis (Pisces: Cyprinidae) in relation to environmental variables in Sefidrud river Basin. Iran J Appl Biol Sci 9(1):36–42
Zelditch M, Swiderski D, Sheets DH, Fink WL (2004) Geometric morphometrics for biologists: a primer. Academic Press, New York, p 478
Acknowledgements
This study was financially supported by the Nevsehir Hacı Bektas Veli University and University of Tehran.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
Design of study contributed by SE, AMS, and BS. Data acquisition contributed by BS and EC. Data analysis/interpretation contributed by SE, HP, and AMS. Drafting manuscript contributed by AMS and SE. Critical revision of manuscript contributed by SE and HP. Final approval and accountability contributed by SE. Technical or material support contributed by EC and SS. Supervision contributed by SE.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
We confirm that all procedures performed in this study involving animals were in accordance with the ethical standards.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Secer, B., Mouludi-Saleh, A., Eagderi, S. et al. Phenotypic Plasticity of Angora Loach, Oxynoemacheilus angorae (Steindachner, 1897) in Inland Waters of Turkey. Iran J Sci Technol Trans Sci 46, 1317–1326 (2022). https://doi.org/10.1007/s40995-022-01348-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40995-022-01348-9