Abstract
Chalcopyrite is the main mineral source of copper ore for extracting and producing copper. However, with the continuous mining of copper ore, the grade of chalcopyrite decreases year by year and its composition becomes more and more complex. The traditional pyrometallurgical extraction process has been unable to make efficient use of such resources, and it is extremely urgent to explore an efficient and environmentally friendly hydrometallurgical leaching method of chalcopyrite. In this paper, the research progress of hydrometallurgical leaching of low-grade complex chalcopyrite is reviewed, and the advantages and disadvantages of mainstream leaching processes such as oxidation leaching, coordination leaching, and biological leaching are analyzed. The analysis shows that bioleaching is suitable for the leaching copper from low-grade chalcopyrite and even original ore, but the leaching rate is not high, and the time for bacterial culture and domestication, as well as the leaching cycle is long. The time of the oxidation leaching is short, the reaction conditions are mild, but the oxidant is difficult to recycle resulting large consumption. Coordination leaching is highly selective, but the system is highly corrosive and requires high investigation in equipment. No matter what kind of process is adopted, the passivation layer composed of elemental sulfur, polysulfide, and jarosite will be produced in the process, hindering the leaching. Increasing the research on the adaptability and selectivity of microorganisms is the direction of bioleaching, and the selective leaching in coordination leaching under ammonia medium with the synergy of oxidant and coordination agent can be realized. In addition, the formation of passivated layer can be inhibited by controlling acidity and redox potential, and the passivated layer can be stripped or eroded by microwave or ultrasonic reinforcement, so as to improve the efficiency of leaching reaction.
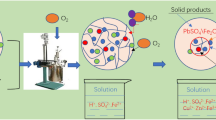
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Copper is one of the basic natural resources supporting economic development. More than 80% of industrial raw materials and 70% of agricultural materials depend on mining engineering, especially copper mining [1]. Copper sulfide is the main mineral source of copper mining and contains many mineral types, of which the common ones are as follows (Table 1).
Among them, chalcopyrite (CuFeS2) is one of the most abundant and widely distributed copper-bearing minerals, accounting for more than 70% of the earth's copper reserves [3], and belongs to primary copper sulfide minerals. Although about 80% to 85% of the world's copper output is produced by means of pyrometallurgical processes, the hydrometallurgical process routes have gradually attracted attention. Traditional pyrometallurgical copper is extracted from high-grade chalcopyrite concentrate. With the continuous mining and utilization of copper-bearing minerals, the grade of the currently mined chalcopyrite is getting lower and lower. It is becoming more and more difficult to extract copper from low-grade chalcopyrite economically and efficiently by the traditional pyrometallurgical method. The low-grade chalcopyrite ores produced after ore dressing are characterized by the low grade of copper (Cu < 18 wt%), the fine of disseminated grain size, and most of them are associated with other metals to form polymetallic (or multimetallic) sulfides ore containing pyrite, galena, sphalerite, etc., compared to chalcopyrite concentrate got by floatation with common original copper ores. The lower the content of copper in low-grade chalcopyrite is, the more the content of other metals like zinc, lead, and iron are, resulting lots refractory by-products containing copper like smelting slag, fume, and lead matte, while it was conducted with traditional pyrometallurgical process. At the same time, the pyrometallurgical process will produce a large amount of SO2 and other gases [4] harmful to the environment and the human body. In comparison, the hydrometallurgical method of extracting copper from chalcopyrite is more economical and environmentally friendly than pyrometallurgical smelting, especially when dealing with low-grade chalcopyrite combining with multimetallic sulfides such as pyrite, galena, and sphalerite [5], and hydrometallurgical smelting is used more appropriate.
The hydrometallurgical process used to treat chalcopyrite include acid leaching, ammonia leaching, high-valent iron salt leaching, chloride leaching, hydrogen peroxide leaching, biological leaching [6], acid dichromate leaching, and reduction leaching. Due to the stable structure and high lattice energy of chalcopyrite, as well as the similarity and complexity in mineralogical combination and properties are exhibited in sulfide ores [7]; the leaching rate and the recovery efficiency of valuable metals in the hydrometallurgical extraction process are not high; it is necessary to strengthen the research on the mechanism and the intensified methods of hydrometallurgical leaching performed on chalcopyrite for overcoming the bottleneck problem and obtaining the process with high-efficiency hydrometallurgical extraction. The scientific theories and research achievements about the hydrometallurgical leaching process of chalcopyrite are analyzed in this paper, such as dissolution mechanism, the formation mechanism of passivation layer during the leaching process, and the main influencing factors in the leaching process, in order to obtain the mechanism and the method for inhibiting the formation of the surface passivation layer during the chalcopyrite leaching process, which has certain reference and guiding significance for the research and industrialization of chalcopyrite leaching.
Oxidation Leaching
When the oxidation–reduction potential in the leaching system is higher than the potential required for the leaching of chalcopyrite, the leaching of chalcopyrite can be realized. From the view point of thermodynamics, the Gibbs free energy during the oxidation leaching reaction at ambient temperature is negative value, indicating the leaching method can be carried out spontaneously. The free energy of the leaching decreases with the increase of temperature, indicating that the leaching rate of chalcopyrite can be accelerated and thorough completed with the increase of temperature. At present, the oxidation leaching process of chalcopyrite includes atmospheric oxidation leaching [8,9,10,11,12,13,14] and pressure oxidation leaching [15].
Atmospheric Oxidation Leaching
In principle, for leaching copper from chalcopyrite, the sulfide of the valuable metal in the mineral must be oxidized into soluble sulfate. The mechanism of the oxidation leaching of chalcopyrite is oxidizing sulfides to oxides by oxidants with high redox potential, and then the valuable metal enters the solution in the form of ions and is separated from the impurities and gangue in the mineral. Under normal pressure conditions, O2 in the atmosphere cannot meet the potential required for leaching copper in chalcopyrite, and other oxidants need to be introduced to meet the oxidation–reduction potential of the leaching system. In recent years, Fe3+, Cr2O72−, ClO3−, MnO4−, H2O2, and other oxidants have been widely used in the oxidative leaching of chalcopyrite [16,17,18,19,20,21,22,23,24].
Fe3+ Oxidation Leaching
Fe3+ is a commonly used oxidant. Increasing the concentration of Fe3+ can increase the leaching efficiency of chalcopyrite. The action mechanism that chalcopyrite can be leached in the presence of Fe3+ can be accounted for by the reaction that occurs during leaching and is shown in Eq. (1).
It can be seen from Eq. (1) that the higher the Fe3+ concentration, that is, the higher the redox potential, the higher the leaching rate of chalcopyrite. Hirato et al. [25] testified that the leaching of chalcopyrite showed parabolic-like kinetics initially and then showed linear kinetics, and that Fe3+ influenced the leaching rate of copper and the leaching of chalcopyrite was chemically controlled. Hiroyoshi et al. [16] found that the oxidation–reduction reaction in the leaching process of chalcopyrite was not done in one step, but in sequence. Due to the formation of Fe2+ and Cu2+ during the leaching process, chalcopyrite will also be reduced to chalcocite (Cu2S). Compared with chalcopyrite, Cu2S is more likely to be oxidized by Fe3+, which is the reason why the copper leaching rate increases at a low potential of 560 ~ 600 mV (SHE) [26]. Ibanez [17] found that the solution potential was in the range of 540 ~ 630 mV, the leaching efficiency of chalcopyrite was significantly improved. In the sulfuric acid system, either FeCl3 or Fe2(SO4)3 can be used as the oxidant; however, there are different leaching effects. Dutrizac et al. [27] believed that when FeCl3 was used as an oxidant, the leaching process was controlled by diffusion. However, Olvera et al. [28] believed that when Fe2(SO4)3 was used as an oxidant, the leaching process was controlled by a chemical reaction. In addition, in the system with FeCl3 as the oxidant, the permeability of the surface products was better than that of Fe2(SO4)3, which made the copper leaching rate higher. However, the strong affinity of chloride ions makes the separation of copper from the leaching solution complicated and corrosive to equipment.
During the leaching of chalcopyrite with ferric sulfate media, the addition of iodine can increase the rate of copper dissolution and decrease the activation energy of the leaching owing to the increasing of solution potential, and the presence of iodine changed the reaction mechanism [29, 30]. Compared to conventional ferric leaching, the JX Iodine Process consisting of the irrigation of ferric iron and low-concentration iodide solutions in the column leaching test also indicated that the effectiveness of the addition of iodine in the leaching of chalcopyrite [31].
Dichromate Leaching
Murr et al. [18] carried out a study on the leaching of chalcopyrite with dichromate as an oxidant in a sulfuric acid system. The leaching rate of copper reached 97%, and the obtained leaching solution contained less iron, which was beneficial to the subsequent process of extracting copper. Of course, this leaching process also has obvious shortcomings. Aydogan et al. [32] and Hu et al. [32] found that elemental sulfur would be generated during dichromate leaching, which would then wrap minerals and hinder the leaching reaction, and that elemental sulfur would be oxidized as the reaction temperature gradually increased. They found that the surface of the chalcopyrite leached by potassium dichromate was extensively covered with elemental sulfur (Fig. 1), which would inhibit the leachate from entering the chalcopyrite for a fuller reaction.
SEM and EDX of chalcopyrite after leaching by potassium dichromate [19]
H2O2 Oxidation Leaching
Hydrogen peroxide is a strong oxidant, which has a significant effect on the leaching of insoluble copper sulfide ore. In addition, the product after oxidizing the mineral with hydrogen peroxide is water, which is highly environmentally friendly. Wu et al. [33] studied the leaching of chalcopyrite with hydrogen peroxide as the oxidant in the methanesulfonic acid system, and the leaching efficiency of copper was significantly improved. Sokic et al. [34] found that the oxidation rate of chalcopyrite was directly proportional to the concentration of hydrogen peroxide when chalcopyrite was leached with hydrogen peroxide in a sulfuric acid medium. Petrovic et al. [21] also found the same conclusion. Petrovic et al. used hydrogen peroxide as an oxidant to leach chalcopyrite in a hydrochloric acid system, and found that the maximum final copper extraction of 33% was attained with 3.0 mol/L H2O2 in 0.5 mol/L HCl at room temperature after 180 min of the reaction. The leaching process was described by the first order kinetics equation with apparent activation energy of 19.6 kJ/mol, which suggesting the dissolution process was under diffusion control. In addition the reason leading to the lower leaching efficiency of copper lays the generation of the elemental sulfur on mineral surfaces which tended to inhibit the leaching rate.
However, hydrogen peroxide is also an oxidant with obvious advantages and disadvantages. The advantage is that the reaction conditions are mild and it can be carried out at low temperature and atmospheric pressure. The disadvantage is that it consumes a lot of money and has poor economic benefits.
Other Oxidants Leaching
In addition to the above-mentioned oxidants, there are also other agents can be used as oxidant for leaching copper from chalcopyrite such as O3, S2O82−, NO3−, and Ag+. The biggest advantage of O3 is that it does not introduce other impurity ions and has a high redox potential [35]. O3 is favored as a clean oxidant to treat sulfide ore, which is also ecofriendly [36]. When O3 leaches chalcopyrite, it can quickly oxidize Fe2+ to Fe3+, increase the oxidation–reduction potential, and increase the leaching rate of copper [37]. S2O82− is also a common oxidant, which can be regenerated by electrochemistry [38, 39], and the cost is low. Nitric acid is both a strong acid and a strong oxidant, and is widely used in the leaching of copper sulfide ore. However, the nitrogen oxides produced during the leaching process of nitric acid are not easy to recover. Therefore, the economic and environmental friendliness of leaching copper with nitric acid is poor. In addition, the sulfur produced during the leaching process will be wrapped on the surface of the mineral, making the mineral passivated and difficult to leaching [40]. Fortunately, research testified that silver ion contained in the sulfuric acid solution could catalyze chalcopyrite electro-dissolution and the addition of silver to the solution increases the apparent capacitance of the chalcopyrite passive film, owing to changes in the semiconductor properties of the film [41].
Pressure Oxidation Leaching
Pressure oxidation leaching is divided into three leaching methods: low-temperature pressure leaching, medium-temperature pressure leaching, and high-temperature pressure leaching. The temperature and oxygen partial pressure ranges are shown in Table 2.
The reaction at the conditions of temperature lower than 120 °C and oxygen partial pressure between 0.5 and 0.7 Mpa is called low-temperature pressure leaching. Chaidez et al. [42] leached chalcopyrite with O2 as an oxidant in a low-pressure reactor and sulfuric acid medium, and found that the higher the oxygen partial pressure, the higher the leaching efficiency. However, the effect of leaching chalcopyrite with O2 alone as an oxidant is not ideal, and it is often necessary to cooperate with some other additives to increase the leaching rate. In addition, the particle size of chalcopyrite is also required to be as small as possible.
The main chemical reaction of pressure leaching under low temperature and pressure was shown in Eq. 2.
The existing low-temperature pressure leaching processes mainly include the Activox process, the Mt Gordon ferric sulfate process, the Pasminco process, and the BGRIMM-LPT process.
Medium-temperature pressure leaching refers to a pressure leaching process with a reaction temperature of 140–180 °C and an oxygen partial pressure of 0.6–1.2Mpa. Under this reaction condition, the initial reaction speed of chalcopyrite is relatively fast, but the elemental sulfur generated as the reaction proceeds will slowly form a coating layer on the surface of the mineral, thereby reducing the reaction rate. The main reaction of pressure leaching under medium temperature and pressure is shown in Eq. 3.
During the reaction, part of the sulfur in chalcopyrite will produce sulfuric acid due to oxidation. The reaction temperature and acidity are the main factors affecting the formation of elemental sulfur and sulfuric acid [43]. CESL process, NSC process and Dynatec process are the main medium-temperature pressurization processes. Both the CESL process and the NSC process are currently only in the experimental stage, and have not achieved industrial application. The Dynatec [43] process was only used for the pressure leaching of zinc concentrate and other minerals at the beginning, and later it was used for the pressure leaching of chalcopyrite. When the Dynatec process is used for leaching chalcopyrite, charcoal with a carbon content of 25% to 55% needs to be added to alleviate the formation of a passivation layer on the mineral surface.
When the reaction temperature is above 200 °C and the oxygen partial pressure is between 0.8 and 1.7 MPa, it is called high-temperature pressurized leaching. Under these conditions, the minerals react quickly and thoroughly, and there is no need to finely grind the minerals like low-temperature pressurization. There is no need to add additives to alleviate the passivation behavior of minerals. The main chemical reactions of pressure leaching under high temperature and pressure are shown in Eqs. 4, 5 and 6.
Phelps Dodge-Placer Dome [44], developed in 2001 and gradually used in industrial production in 2003, is a commonly used high-temperature pressure leaching process. The reaction conditions controlled by this process are reaction temperature 225 °C and oxygen partial pressure 0.7 MPa, the reaction time is 1 h. In addition, there are the Total Pressure Oxidation process and the Sherritt–Cominco process. However, due to cost constraints, these two processes are currently only in the laboratory research stage and have not achieved industrial production.
Greater than 94% copper was extracted from chalcopyrite concentrate under medium-temperature conditions at 125–150 °C. About 80–90% oxidation of sulfide to elemental sulfur occurred at 108 °C and was complete oxidation to sulfate above 180 °C. Hematite formation was favored at higher temperatures (≥ 150 °C), low acidity, and low-to-moderate salinity. Goethite formation was favored at lower temperatures (< 150 °C) and by low acidity and low salinity. Jarosite was formed under conditions of moderate-to-high acidity and its formation was enhanced in the presence of sodium ions [45, 46]. At present, pressure leaching in novel lixiviants with outstanding advantages had been reported, resulting 88.5% of copper and only 2.11% of iron extracted from chalcopyrite with 3 mol/L H2O2 and 100 g/L H2C2O4 at 170 °C for 180 min [47]. The study of Han et al. [48] showed that the pyrite addition significantly promoted on the dissolution of copper from chalcopyrite due to the production of sulfuric acid from pyrite under pressure-oxidative condition. Under the obtained optimal conditions in the study, copper extraction of 94.5% was achieved after 90 min leaching, while a dissolution of iron at 4.2% was obtained.
Process-Enhanced Leaching
Ultrasound Enhancement
Due to the stable crystal structure of chalcopyrite, it is difficult to be leached. In recent years, many scholars have done a lot of research on how to change the crystal structure of chalcopyrite and increase its leaching rate. Literature [49,50,51] reported the application of ultrasonic in the hydrometallurgical leaching of chalcopyrite, especially for low-grade chalcopyrite, which has a very ideal leaching effect. Ultrasonic enhancement is mainly through the formation of a cavity for the solution, the cavity collapses causing the instantaneous release of a large amount of energy, resulting in a reaction temperature much higher than the surroundings, and speeding up the chemical reaction. The high-speed micro-jet generated by the ultrasonic action corrodes the surface of the mineral, thereby making the reaction more complete. Ultrasonic-enhanced leaching has a very ideal effect on the leaching of low-grade chalcopyrite. Wang et al. [52] found that the copper leaching rate increased by 5.6–14.8% under different ultrasonic intensity conditions compared with the leaching experiment without ultrasonic waves. The reason accounts for this is that ultrasound can change the surface morphology of minerals (Fig. 2), further expand the fractures of minerals, and make the reaction more complete. In addition, ultrasound can reduce the surface tension and viscosity of the solution, and increase the conductivity and dissolved oxygen of the solution, thereby speeding up the reaction.
SEM images of chalcopyrite surface before and after leaching [52]: a before leaching (without ultrasound), b after leaching (without ultrasound), c before leaching (with ultrasound), d after leaching (with ultrasound)
Microwave Enhancement
Researchers have found that a passivation layer is produced during the leaching process of chalcopyrite, which inhibits its leaching efficiency. Therefore, on the basis of the best conditions for conventional leaching of chalcopyrite, microwave radiation is introduced to strengthen the leaching process of chalcopyrite. Wen [53] and others believed that microwave could modify the surface structure of chalcopyrite, remove the surface passivation layer during the leaching process, and expose the wrapped chalcopyrite, which provided power for the leaching of copper. Kang et al. [54] found through research that when the microwave heating powers introduced were 300 W and 500 W, the copper leaching rate was increased by 6.98% and 13.08%, respectively, compared with the copper leaching rate under the conventional heating system. Through SEM analysis (Fig. 3), compared with conventional heating, the leaching residue after microwave heating has more cracks, more potholes on the surface and more fluffy. It shows that chalcopyrite is eroded more severely under the action of microwave heating, and microwave can peel off the sulfur layer produced during the leaching process, which slows down the passivation of the mineral surface; all these have a positive effect on the smooth progress of chalcopyrite leaching.
SEM of chalcopyrite leaching residue [54]: a conventional heating leaching slag, b microwave 300 W heating leaching slag, c microwave 500 W heating leaching slag
Coordination Leaching
Copper forms soluble complexes with appropriate ligands to achieve the method of leaching chalcopyrite, which is called copper coordination leaching. In the treatment of low-grade insoluble copper sulfide ore, the method of synergistic leaching of oxidant and complexing agent is also used. Chloride ions and ammonium ions will form complex ions with copper ions and enter the solution. Coordination leaching in chlorine and ammonia media is a common coordination leaching process. In coordination leaching, the undesired elements such as iron, lead, and silicon are precipitated or retained in the leaching residue by controlling the reaction conditions such as temperature and acidity.
Coordination Leaching in Chlorine Medium
The Intec Process of Australia Intec and the Hydro-copper process of Finnish Outotec are currently two more mature chlorine media coordination leaching processes. The advantages of these two processes are that the leaching of chalcopyrite can be achieved under normal pressure, and that low energy consumption. The disadvantage, however, is that the leaching system is highly corrosive, and the material requirements for the leaching equipment are relatively high. The most commonly used chloride additive in the chloride leaching process is CuCl2, and its reaction equation with chalcopyrite is shown in Eq. 7.
Yevenes et al. [55] studied the leaching of chalcopyrite with CuCl2 as an additive and found that a small amount of Cu2+ would increase the leaching rate of chalcopyrite, but with the continuous increase of Cu2+ concentration, the leaching rate of copper in chalcopyrite hardly raised again. NaCl is also often used as an additive to participate in the leaching of chalcopyrite. Zhong et al. [56] studied the influence of NaCl on the leaching of chalcopyrite and found that the leaching rate of copper was proportional to the concentration of NaCl. The reason accounts for this is that chloride ions promote the formation of a porous sulfur layer on the surface of chalcopyrite, which is beneficial to the leaching of copper. In addition, chloride ions will reduce Cu2+ in chalcopyrite to Cu+, and the amount of Cu+ generated increases with the concentration of chloride ions, which increases the critical potential of Cu+/Cu2+, thereby increasing the leaching rate of chalcopyrite. In addition to the above two chlorides, HCl and FeCl3 can also be used as additives for the leaching of chalcopyrite.
Coordinated Leaching in Ammonia Medium
In the ammonia leaching system, the leaching selectivity for the target metal is relatively strong. While rejecting the leaching of iron from chalcopyrite, copper and other valuable metals are dissolved in the leaching solution in the form of ammonia complex ions [57]. Baba et al. [58] found that in the ammonia leaching system, the leaching rate of copper was greatly affected by temperature, ammonia concentration, and mineral particle size, while the oxygen partial pressure had little effect on it. However, some studies have shown that in the ammonia leaching system, the partial pressure of oxygen has a greater impact on the leaching rate of chalcopyrite. The total chemical reaction formula of coordination leaching in ammonia medium is shown as Eq. 8.
In addition, D'yachenko [59] proposed to calcinate ammonium salt and mineral first, carry out coordination reaction during the roasting process, and finally carry out solution leaching. However, as the calcination temperature increases, the complexes that have been formed will be decomposed and metal salts will be formed. Its reaction is expressed as Eq. 9.
Bell et al. [60] found that increasing the amount of oxidant could speed up the leaching reaction. In addition, the addition of organic compounds could stabilize the iron oxide particles generated during the reaction, making it difficult to wrap the mineral particles, thereby reducing the passivation effect. The schematic diagram mechanism of coordination leaching could be found in Fig. 4.
Bioleaching
The Effect of Microorganisms on Chalcopyrite
The debate on the ability of microorganisms to enhance leaching has never stopped [61]. Although the beneficial effects of microorganisms in the leaching process of chalcopyrite, cobalt copper ore, and pyrite have been confirmed, it is still not effective for the use of microorganisms in the leaching of chalcopyrite. No unanimous conclusion has been reached. A large number of studies have shown that many microorganisms have the ability to oxidize or assist in the oxidation of chalcopyrite. The mechanism of action is to realize the auxiliary leaching of chalcopyrite through iron oxide and sulfur. The reactions involved were shown in the following equations:
Among them, Eqs. 11 and 12 are affected by bacteria. It can be seen from Eq. 11 that microorganisms will oxidize Fe2+ to Fe3+ during the leaching process, which increases the redox potential in the system and accelerates the leaching efficiency. In Eq. 12, microorganisms oxidize the elemental sulfur produced during the dissolution of chalcopyrite to sulfuric acid, and H+ in the sulfuric acid cooperates with Fe3+ to dissolve chalcopyrite.
In bioleaching, the surface of minerals is usually embedded with bacterial extracellular polymer (EPS) [62]. EPS is considered to play a vital role in the oxidation of iron and sulfur, and it is mainly composed of proteins, lipids, sugars, and nucleic acids [63]. EPS is produced by microorganisms and mediates the attachment of microorganisms to the mineral surface. After the cells initially attach, a biofilm is formed within a few days, and the cells are embedded in a continuous EPS layer to cover the mineral surface [64] (Fig. 5).
Schematic diagram of bioleaching mechanism [13]
Studies have shown that the iron on the surface of minerals is enriched by the complexation of uronic acid or other EPS residues [65, 66], which makes the reaction environment in a state of net positive charge; cells are more likely to attach to negatively charged minerals. This creates conditions for the dissolution reaction and accelerates the dissolution of minerals [67], thereby increasing the leaching rate of minerals. Research by Rohwerder [68] et al. found that Fe3+ absorbed electrons from the surface of minerals in the extracellular polymer. These electrons then reduce oxygen molecules through the redox chain in the bacterial membrane, and further oxidize and decompose minerals.
Types of Microorganisms and Factors Affecting Their Activity
Generally, microorganisms are classified into mesophilic thermophiles (30–40 °C), moderate thermophiles (50 °C), and extreme thermophiles (> 65 °C) according to the differences in temperature, pH value, and ion concentration in the leaching system. Common microorganisms are Leptospirillum, Acidithiobacillus, and Sulfobacillus thermosulfidooxidans ST [69]. The bacteria used for the leaching of chalcopyrite usually include single strain leaching and mixed strain leaching. Because mixed strains can adapt to the changing leaching conditions during the leaching process, the leaching efficiency of mixed strains is often higher than that of single strains [70]. Through biological research on the gene and metabolic function of bacteria, it is found that proper enhancement of the physiological functions of the bacteria can make the leaching efficiency of chalcopyrite bacteria higher. Research by Peterson et al. [71] showed that the temperature of thermophilic heap leaching of a chalcopyrite concentrate could increase to 70 °C while successively introducing various mesophile and thermophile cultures, and that copper extractions in excess of 90% were achieved within 100 days.
The activity of microorganisms in the leaching system plays a vital role in the leaching efficiency. Environmental, physicochemical, and biological factors have a great influence on the growth activity of microorganisms. Only when the pH value, temperature, nutrition, humidity, and energy in the culture system are suitable, and there are no inhibitory factors in the whole process, can colonies that grow fast and healthily be formed. At the same time, before putting the colony into the leaching system, there needs to be a process to adapt to the pH value, pulp density, reaction temperature, and ion concentration, so that the bacteria can adapt to the reaction conditions to a greater extent and produce better leaching efficient.
The effect of microorganisms in the solution on the leaching process is not only related to the activity of the microorganisms themselves but also depends on the concentration of Fe3+. At the optimal Fe3+ concentration, microorganisms can increase the leaching rate by regenerating Fe3+, but if the produced Fe3+ concentration exceeds the consumed Fe3+ concentration, then metal-deficient polysulfides will be produced, and iron-deficient polysulfides with poor reactivity and low diffusivity will coat the mineral surface, resulting in the passivation of chalcopyrite. At this time, the microorganisms will become harmful, which is not conducive to the leaching of chalcopyrite. The resulting polysulfides are difficult to be directly oxidized by Fe3+, and can only be dissolved by using potassium dichromate or nitric acid, which are more oxidizing oxidants. Xin et al. [72] believed that in the enhanced microbial leaching, the rational use of the microbial population and the control of the Fe3+ concentration were effective methods to increase the leaching rate. Ren et al. [73] found that chalcopyrite leaching in acidic ferric sulfate media combined with iron-oxidizing bacterium (acidithiobacillus ferrooxidans) was improved by addition of ethylene thiourea (ETU), and that ETU was able to solubilize the chalcopyrite, exhibiting significant surface corrosion and enhancing the copper extraction rate from low-grade chalcopyrite ore.
Discussion and Analysis
There are many methods for leaching chalcopyrite by hydrometallurgical technology. Oxidation leaching, coordination leaching, and biological leaching are the three mainstream methods at present.
Oxidation leaching is currently the most mature and widely used process. In the process of leaching chalcopyrite, the concentration of sulfuric acid is one of the decisive factors. The acidity not only affects the leaching rate of copper but also affects the conversion of iron in the leaching solution. When the sulfuric acid concentration is low, the leaching efficiency is low. When the sulfuric acid concentration is high, the solubility of hematite in the sulfuric acid solution will increase while the leaching rate of copper is increased, which leads to the redissolution of hematite to increase the leaching rate of iron [74]. Aydogan et al. [32] studied the leaching of chalcopyrite with H2SO4 as the leaching medium and found that when the acidity in the leaching system increased, it would inhibit the hydrolysis and precipitation of Fe3+ in the solution, so the formation of alkaline sulfate passivation layer such as jarosite was inhibited, and the iron leaching rate also increased. However, the concentration of sulfuric acid in the solution cannot be increased blindly. When the pH value of the slurry is less than 0.5, the competition between Fe3+ and H+ [75] leads to iron deficiency on the surface of the ore, which promotes passivation. Therefore, increasing the concentration of sulfuric acid in the solution within a certain range has a positive effect on the leaching of chalcopyrite.
At the same time, temperature is also an important factor affecting the leaching efficiency of chalcopyrite in pressure oxidation leaching. In the medium- and low-temperature reaction system, the reaction temperature is often only increased by 20–40 °C, but the leaching rate of copper can be increased by 40–80%. However, under the high temperature in the pressure oxidation leaching system, chalcopyrite leaching efficiency can exceed 95%, or even close to 100%. Sokic [40] and Cordoba [76] also found the same phenomenon in their research. In addition, the leaching effect of chalcopyrite in the high-temperature reaction system is better than that in the medium- and low-temperature reaction system. Numerous experiments have shown that when the reaction temperature reaches 180 °C, the leaching rate of copper is basically above 95%. The reason is that temperature can greatly affect the reaction kinetics in the leaching process, and high temperature will accelerate the kinetics in the reaction process, thereby improving the leaching efficiency. It can be seen that temperature plays a vital role in improving the leaching rate of chalcopyrite. Properly controlling the reaction temperature under corresponding reaction conditions can maximize the leaching efficiency of chalcopyrite. At present, most leaching research of chalcopyrite is conducted at high temperature, while some oxidants with strong oxidation ability are generally added to cooperate with chalcopyrite leaching in medium- and low-temperature system. In addition, proper stirring rate, liquid–solid ratio, and finer mineral particle size all have a positive effect on improving the leaching rate of chalcopyrite. From the phase analysis of chalcopyrite, it is known that copper and iron in minerals will form sulfides with the same internal phase. Therefore, it is necessary to achieve copper leaching with the assistance of oxygen or other oxidants [75]. There are many types of oxidants, such as O2, Fe3+, H2O2 [77], NaNO3 [40], Cu2+ [26], and Cr6+ [32]. Among them, oxygen and Fe3+ are commonly used oxidants in the leaching process of chalcopyrite, especially when using oxygen pressure to leaching chalcopyrite, oxygen is the main oxidant, which plays a decisive role in the leaching process.
Coordination leaching of chalcopyrite is also a popular process at present, and chlorine coordination and ammonia coordination are the most common. Adding an appropriate amount of Cu2+ in a chlorine medium can increase the leaching rate of copper, but excessive addition of Cu2+ has no significant effect on increasing the leaching rate of copper. This is because when the initial Cu2+ concentration is too high, Cu+ and Cu2+ form a dynamic inert adduct in the chlorine solution, thereby inhibiting copper ions and reducing the reaction rate. In this case, the dissolution of chalcopyrite can be promoted by adding Cl−. This is because Cu2+ and Cl− form copper chloride complexes in the leaching system. In this case, Cu2+ and CuCl+ have positive promoting effect on leaching of chalcopyrite, which is consistent with the views of Torres et al. [78]. It can be seen that the leaching efficiency of chalcopyrite can be improved by controlling the initial concentration of Cu2+ in the chlorine medium.
The biggest advantage of coordinated leaching with ammonia medium is its good selectivity to copper. While leaching copper, it can also settle iron in the leaching slag. The reason is that ammonia forms soluble amine complexes with copper ions, while iron cannot form soluble amine complexes with ammonia [58]. In addition, the reaction conditions of ammonia leaching are relatively mild and not corrosive.
Biological leaching is a metallurgical technology that has only emerged in the past forty years. Its principle is to add bacteria in the leaching process to extract valuable metals from the ore. According to the different reaction conditions, the bacteria can be divided into medium temperature, moderate, and extremely thermophilic bacteria. Among sulfide ore, chalcopyrite is the most difficult to leaching. The current representative chalcopyrite bioleaching process is the BioCOP™ process developed by BHP Billiton [79]. When chalcopyrite is leached with mesophilic thermophiles or moderate thermophiles, the dissolution kinetics of chalcopyrite is very slow, which makes the reaction time longer and reduces the extraction rate of copper, resulting in higher leaching costs. Leaching chalcopyrite with extremely thermophilic bacteria can significantly accelerate the dissolution kinetics of chalcopyrite, the reason for this is the high acidity, temperature, and metal ion concentration tolerance of extremely thermophilic bacteria, especially archaea. Although leaching chalcopyrite with extremely thermophilic bacteria can reduce the leaching time, its oxygen consumption is higher, and its sensitive cell wall is easy to cause bacteria die during the leaching process, so it is not suitable for industrial production. In addition, the bioleaching process will produce polysulfide wrapped minerals to form a passivation layer, which inhibits the leaching efficiency of chalcopyrite [80].
As the grade of copper ore continues to decrease, researchers are constantly looking for methods that can effectively leach low-grade copper ore. In the 1970s, in order to improve the leaching effect of low-grade minerals and increase the leaching rate, researchers first proposed heap leaching to leach low-grade ore. The process is to crush the minerals first, then bond them with a binder, connect them for agglomeration curing, and finally stack them for leaching.
Heap leaching of chalcopyrite [81] is often accompanied by biological leaching. Adding bacteria to the heap leaching to increase the leaching rate of chalcopyrite is the current mainstream heap leaching process of chalcopyrite. However, there are many factors that affect the leaching efficiency in the process of biological heap leaching, of which temperature and climate are the main influencing factors. How to control the temperature has always been a difficult point in the heap leaching technology. The temperature at different depths of the leaching pile is different, which is related to the aeration and irrigation rate [82]. The climate is also a difficult problem to solve. Generally, the biological heap leaching is carried out in the open air, so the environment is relatively harsh. Solar radiation, wind, and extreme temperatures will all have a great impact on the leaching effect. Some researchers have found that climatic conditions have an important influence on the dissolution process of chalcopyrite [83], especially for insoluble low-grade chalcopyrite, usually the leaching time can be as long as several months or even half a year. During this period, rainfall and night-time temperature will affect the activity and growth rate of bacteria in the pile, and slow the dissolution of chalcopyrite, which intensifies the formation of a passivation layer on the mineral surface and reduces the leaching efficiency. Therefore, further research on the diversity of microorganisms in the biological heap leaching process and overcoming the passivation problem in the heap leaching process is of great significance for improving the leaching rate of chalcopyrite.
In summary, the advantages and disadvantages of oxidative leaching, coordination leaching, and biological leaching treatment of low-grade complex chalcopyrite are shown in Table 3.
In addition, the cooperative leaching along with contact and non-contact leaching is a promising method, such as leaching enhanced with ultrasonic, microwave, and mechanical activation or microwave-irradiated activation performed on chalcopyrite before leaching [84, 85]. Due to various reasons, in the process of chalcopyrite oxidative leaching, substances such as sulfur element, polysulfide, and jarosite will be produced to cover the surface of the reactive minerals to form a passivation layer, which hinders the progress of the reaction. The addition of ultrasound and/or microwaves can erode or peel off the passivation layers that encapsulate the reactive minerals, so that the encapsulated unreacted minerals can be fully contacted with the lixiviant, thereby improving the leaching efficiency. Through a large number of experiments and references, it is found that in the oxidative leaching of chalcopyrite, the leaching rate of copper enhanced with ultrasonic or microwave is 6–15% higher than that without ultrasonic or microwave. This shows that ultrasonic and microwave have a certain positive effect on the leaching of chalcopyrite.
Conclusion
Passivation is a problem that exists in all hydrometallurgical leaching processes of chalcopyrite. During the leaching process, various species will be formed on the surface of chalcopyrite. Some of them promote the leaching efficiency of chalcopyrite, while others hinder the leaching of chalcopyrite. These species that hinder leaching form a so-called passivation layer on the surface of chalcopyrite. Up to date, there is no consistent conclusion on the composition and the role of the newly formed species layer on the CuFeS2 surface in the leaching process. It can be achieved from the discussion above that elemental sulfur, polysulfide, and jarosite can all be passivation products.
In addition, alkaline iron sulfate is also a common passivation product in the leaching process of chalcopyrite. For the formation of such a passivation layer, the ratio of the redox potential Fe3+/Fe2+ in the leaching system is very important. There is a range of oxidation–reduction potential during the leaching process of chalcopyrite. When the oxidation–reduction potential is less than this range, the degree of oxidation in the leaching system is insufficient, and the oxidation and dissolution of chalcopyrite is insufficient. When the oxidation–reduction potential is greater than this range, the ions in the solution will quickly form alkaline iron sulfate, covering the chalcopyrite to passivate it, thereby inhibiting the leaching of chalcopyrite. Tian et al. [86] also obtained the same conclusion in the experiment.
The advantage of coordinated leaching under chlorine medium is leaching under normal temperature and pressure, low energy consumption, but the disadvantage is strong corrosiveness and higher requirements for leaching equipment. Coordinated leaching under ammonia medium has relatively better selectivity, and the experimental conditions are relatively mild, and the requirements for equipment are relatively low. At the same time, iron enters the leaching residue during the leaching process, but excessive oxygen partial pressure is required during the process. At the same time, there is the defect that ammonia volatilizes to the environment during the process.
Biological leaching has an ideal effect in the leaching of various low-grade minerals. Therefore, it shows great advantages for treating chalcopyrite with decreasing grade. However, due to the slow leaching rate, it is not suitable for industrial production. For biological heap leaching, it is also subject to environmental factors such as temperature and climate. Therefore, further research on the adaptability and selectivity of microorganisms, as well as the mechanism of leaching, is of great significance for tapping the potential of bioleaching and improving the leaching rate of chalcopyrite.
Oxidation leaching of ore has a history of more than one hundred years. With the continuous development and progress of science and technology, the pressure acid leaching process of chalcopyrite has become mature. Compared with biological leaching and coordination leaching, the oxygen consumption of pressurized acid leaching is only 10.45% and 83.33%, and the reaction period is short. At the same time, the pressure acid leaching reaction conditions are mild, the reaction equipment is simple to operate, the application range is wide, and the entire operation process is green and environmentally friendly, and no pollutants are discharged. For low-grade chalcopyrite, due to its complex phase structure, a passivation layer is prone to appear during the leaching process. The formation of the passivation layer can be inhibited by controlling the acidity and redox potential. On this basis, the formed passivation layer can be stripped or eroded by auxiliary means such as microwave strengthening or ultrasonic strengthening, thereby improving the reaction efficiency. Moreover, in order to optimize the leaching selectivity, further research on leaching of chalcopyrite by oxygen pressure acid leaching synergistic ammonia leaching should be strengthened. In addition, the control of the plasma concentration of Ca2+, Mg2+, Al3+, and Cl− also has a great influence on the leaching efficiency of chalcopyrite, but this requires further investigation.
As the grade of chalcopyrite is getting lower and lower, hydrometallurgical leaching of low-grade chalcopyrite has gradually become the mainstream trend. For different grades of chalcopyrite, the selected leaching method is different. Therefore, choosing a suitable method for leaching chalcopyrite, continuously optimizing the leaching process, reducing production costs, improving the operation process, and reducing the negative impact on the environment are the main directions that need to be studied in the current hydrometallurgical leaching of chalcopyrite.
References
Henckens MLCM, Biermann FHB, Driessen PPJ (2019) Mineral resources governance: a call for the establishment of an international competence center on mineral resources management. Resour Conserv Recycl 141:255–263
Zhang L, Yang HP, Zhao JW, Cao F (2015) Series study on copper resource in world-general situation and analysis of supply and demand. Conserv Util Mineral Resour 5:63–67
Wang XY, Li YB (2017) Study on the chalcopyrite leaching and its kinetics. Metal Mine 1:82–85
Wang XY (2017) Study on the leaching kinetics and mechanism of chalcopyrite. Wuhan University of Technology, Wuhan
Huang XT, Liao R, Yang BJ, Yu SC, Wu BQ, Hong MX, Wang J, Zhao HB, Gan M, Jiao F, Qin WQ, Qiu GZ (2020) Role and maintenance of redox potential on chalcopyrite biohydrometallurgy: an overview. J Central S Univ 27(5):1351–1366
Zhu P, Liu XD, Chen AJ, Liu HW, Yin HQ, Qiu GZ, Hao XD, Liang YL (2019) Comparative study on chalcopyrite bioleaching with assistance of different carbon materials by mixed moderate thermophiles. Trans Nonferrous Metals Soc China 29(6):1294–1303
Olubambi PA, Ndlovu S, Potgieter JH et al (2007) Effects of ore mineralogy on the microbial leaching of low-grade complex sulphide ores. Hydrometallurgy 86(1–2):96–104
Bai YL, Xie F, Wang W, Jiang KX (2019) Effect of manganese dioxide on leaching behavior of chalcopyrite. Nonferrous Metals (Extractive Metallurgy) 11:1–6
Zhao HB, Wang J, Gan XW, Hu MH, Tao L, Qin WQ, Qiu GZ (2016) Role of pyrite in sulfuric acid leaching of chalcopyrite: an elimination of polysulfide by controlling redox potential. Hydrometallurgy 164:159–165
Luo ZH, Li L, Hong WJ, Huang HG, Chen JH, Zhou LC (2007) Study on dissolution behavior of chalcopyrite in acidic medium. Mod Chem Ind 27(A2):153–155
Watling HR (2014) Chalcopyrite hydrometallurgy at atmospheric pressure: 2. Review of acidic chloride process options. Hydrometallurgy 146:96–110
Lane DJ, Cook NJ, Grano SR, Ehrig K (2016) Selective leaching of penalty elements from copper concentrates: a review. Miner Eng 98:110–121
Li Y, Kawashima N, Li J, Chandra AP, Gerson AR (2013) A review of the structure, and fundamental mechanisms and kinetics of the leaching of chalcopyrite. Adv Coll Interface Sci 197:1–32
Qian L (2020) Experimental study on sulphuric acid leaching of high arsenic copper sulfide concentrate. Kunming University of Science and Technology, Kunming
Zheng YF, Hua XM, Xu Q, Lu XG, Cheng HW, Zou XL (2019) Leaching and electrochemical oxidation mechanism of chalcopyrite in sulfuric acid solution. Shanghai Metals 41(3):81–88
Hiroyoshi N, Tsunekawa M, Okamoto H (2009) Improved chalcopyrite leaching through optimization of redox potential. Can Metall Q 47(3):253–258
Ibanez T, Velasquez L (2013) The dissolution of chalcopyrite in chloride media. Rev Metal 49(2):131–144
Murr LE, Hiskey JB (2007) Kinetic effects of particle-size and crystal dislocation density on the dichromate leaching of chalcopyrite. Metall Trans B 12(2):255–267
Hu JX, Zi FT, Tian GC (2021) Extraction of copper from chalcopyrite with potassium dichromate in 1-ethyl-3-methylimidazolium hydrogen sulfate ionic liquid aqueous solution. Miner Eng 172:107179
Phuong TNT, Tsuji S, Jeon S, Park I, Tabelin CB, Ito M, Net H (2020) Redox potential-dependent chalcopyrite leaching in acidic ferric chloride solutions: leaching experiments. Hydrometallurgy 194:105299
Petrovic SJ, Bogdanovic GD, Antonijevic MM (2018) Leaching of chalcopyrite with hydrogen peroxide in hydrochloric acid solution. Trans Nonferrous Met Soc China 28(7):1444–1455
Turan MD, Altundogan HS (2013) Leaching of chalcopyrite concentrate with hydrogen peroxide and sulfuric acid in an autoclave system. Metall Mater Trans B 44(4):809–819
Hidalgo T, Kuhar L, Beinlich A, Putnis A (2018) Kinetic study of chalcopyrite dissolution with iron(III) chloride in methanesulfonic acid. Miner Eng 125(1):66–74
Hu JX, Tian GC, Zi FT, Hu XZ (2017) Leaching of chalcopyrite with hydrogen peroxide in 1-hexyl-3-methyl-imidazolium hydrogen sulfate ionic liquid aqueous solution. Hydrometallurgy 169:1–8
Hirato T, Majima H, Awakura Y (1987) The leaching of chalcopyrite with ferric sulfate. Metall Trans B 18B:489–496
Nicol M, Miki H, Velasquez YL (2010) The dissolution of chalcopyrite in chloride solutions: part 3. Mech Hydrometall 103(1–4):86–95
Dutrizac JE (1981) The dissolution of chalcopyrite in ferric sulfate and ferric chloride media. Metall Trans B 12(2):371–378
Olvera OG, Rebolledo M, Asselin E (2016) Atmospheric ferric sulfate leaching of chalcopyrite: thermodynamics, kinetics and electrochemistry. Hydrometallurgy 165(1):148–158
Winarko R, Dreisinger DB, Miura A, Fukano Y, Liu W (2022) Iodine-assisted chalcopyrite leaching in ferric sulfate media: kinetic study under fully controlled redox potential and pH. Hydrometallurgy 208:105797
Winarko R, Dreisinger DB, Miura A, Tokoro C, Liu W (2020) Kinetic modelling of chalcopyrite leaching assisted by iodine in ferric sulfate media. Hydrometallurgy 197:105481
Fukano Y, Miura A (2021) Chalcopyrite leaching with iodine (JX Iodine Process) for various ore types. Hydrometallurgy 206:105752
Aydogan S, Ucar G, Canbazoglu M (2006) Dissolution kinetics of chalcopyrite in acidic potassium dichromate solution. Hydrometallurgy 81(1):45–51
Wu JJ, Ahn J, Lee J (2021) Kinetic and mechanism studies using shrinking core model for copper leaching from chalcopyrite in methanesulfonic acid with hydrogen peroxide. Miner Process Extr Metall Rev 42(1):38–45
Sokic MD, Markovic B, Stankovic S, Kamberovic Z, Strbac N, Manojlovic V, Petronijevic N (2019) Kinetics of chalcopyrite leaching by hydrogen peroxide in sulfuric acid. Metals 9(11):1173
Tian GC, Hu JX, Zi FT (2017) Research advances in applications of oxidants in oxidation leaching of chalcopyrite. Chin J Process Eng 17(4):664–676
Wang JX, Faraji F, Ghahreman A (2021) Evaluation of ozone as an efficient and sustainable reagent for chalcopyrite leaching: process optimization and oxidative mechanism. J Ind Eng Chem 104:333–344
Pedroza FRC, Aguilar MDJS, Trevino TP, Luevanos AM, Castillo MS (2012) Treatment of sulfide minerals by oxidative leaching with ozone. Miner Process Extr Metall Rev 33(4):269–279
Serrano K, Michaud PA (2002) Electrochemical preparation of peroxodisulfuric acid using boron doped diamond thin film electrodes. Electrochim Acta 48(4):431–436
Dakubo F, Baygents JC, Farrell J (2012) Peroxodisulfate assisted leaching of chalcopyrite. Hydrometallurgy 121:68–73
Sokic MD, Markovic B, Živkovic D (2009) Kinetics of chalcopyrite leaching by sodium nitrate in sulphuric acid. Hydrometallurgy 95(3):273–279
Ghahremaninezhad A, Radzinski R, Gheorghiu T, Dixon DG, Asselin E (2015) A model for silver ion catalysis of chalcopyrite (CuFeS2) dissolution. Hydrometallurgy 155:95–104
Chaidez J, Parga J, Valenzuela J, Carrillo R, Almaguer I (2019) Leaching chalcopyrite concentrate with oxygen and sulfuric acid using a low-pressure reactor. Metals 9(2):189
Li C (2015) Study on pressure leaching of chalcopyrite. Northeastern University, ShenYang
Wang SJ (2006) Copper leaching from chalcopyrite concentrates. JOM 57(7):48–51
McDonald RG, Muir DM (2007) Pressure oxidation leaching of chalcopyrite. Part I. Comparison of high and low temperature reaction kinetics and products. Hydrometallurgy 86(3–4):191–205
McDonald RG, Muir DM (2007) Pressure oxidation leaching of chalcopyrite: part II: comparison of medium temperature kinetics and products and effect of chloride ion. Hydrometallurgy 86(3–4):206–220
Turan MD, Sarı ZA, Nizamoğlu H (2021) Pressure leaching of chalcopyrite with oxalic acid and hydrogen peroxide. J Taiwan Inst Chem Eng 118:112–120
Han B, Altansukh B, Haga K, Takasaki Y, Shibayama A (2017) Leaching and kinetic study on pressure oxidation of chalcopyrite in H2SO4 solution and the effect of pyrite on chalcopyrite leaching. J Sustain Metall 3:528–542
Xia GH, Lu M, Su XL, Zhao XD (2012) Iron removal from kaolin using thiourea assisted by ultrasonic wave. Ultrason Sonochem 19(1):38–42
Xi FS, Cui HQ, Zhang Z, Yang ZM, Li SY, Ma WH, Wei KX, Chen ZJ, Lei Y, Wu JJ (2019) Novel and efficient purification of silicon through ultrasonic-Cu catalyzed chemical leaching. Ultrason Sonochem 56:474–480
Hu YT, Guo P, Wang SX, Zhang LB (2020) Leaching kinetics of antimony from refractory gold ore in alkaline sodium sulfide under ultrasound. Chem Eng Res Des 164:219–229
Wang YM, Wu AX, Ai CM (2013) Experiment and mechanism analysis on leaching process of low grade. Chin J Nonferrous Metals 7:2019–2025
Wen T, Zhao YL, Ma QL, Xiao QH, Zhang TT, Chen JX, Song SX (2020) Microwave improving copper extraction from chalcopyrite through modifying the surface structure. J Market Res 9(1):263–270
Kang SC, Zhao YL, Wen T, Ai ZQ, Ma QL, Song SX (2017) Intensified leaching of chalcopyrite by microwave and its mechanism. Metal Mine 1:86–90
Yevenes LV, Miki H, Nicol M (2010) The dissolution of chalcopyrite in chloride solutions: part 2: effect of various parameters on the rate. Hydrometallurgy 103(1–4):80–85
Zhong S, Li YB (2019) An improved understanding of chalcopyrite leaching kinetics and mechanisms in the presence of NaCl. J Market Res 8(4):3487–3494
Moyo T, Petersen J (2016) Study of the dissolution of chalcopyrite in solutions of different ammonium salts. J South Afr Inst Min Metall 116(6):509–516
Baba AA, Ghosh MK, Pradhan SR, Rao DS, Baral A, Adekola FA (2014) Characterization and kinetic study on ammonia leaching of complex copper ore. Trans Nonferrous Met Soc China 24(5):1587–1595
D’yachenko AN, Kraidenko RI (2010) Processing oxide-sulfide copper ores using ammonium chloride. Russ J Non-Ferrous Met 51(5):377–381
Bell SL, Welch GD, Bennett PG (1995) Development of ammoniacal lixiviants for the in-situ leaching of chalcopyrite. Hydrometallurgy 39(1):11–23
Panda S, Akcil A, Pradhan N, Deveci H (2015) Current scenario of chalcopyrite bioleaching: a review on the recent advances to Its heap-leach technology. Biores Technol 196:694–706
Yu ZB, Liu YJ, Wu JL, Yu MM (2016) Review on extracellular polymer substances of bacteria on bioleaching. Nonferrous Metals 2:1–6
Zeng WM, Qiu GZ, Zhou HB, Chen MA (2011) Electrochemical behaviour of massive chalcopyrite electrodes bioleached by moderately thermophilic microorganisms at 48 °C. Hydrometallurgy 105(3–4):259–263
Zeng WM, Tan SE, Chen MA, Qiu GZ (2011) Detection and analysis of attached microorganisms on the mineral surface during bioleaching of pure chalcopyrite with moderate thermophiles. Hydrometallurgy 106(1–2):46–50
Kinzler K, Gehrke T, Telegdi J, Sand W (2003) Bioleaching—a result of interfacial processes caused by extracellular polymeric substances (EPS). Hydrometallurgy 71(1–2):83–88
Yu RL, Tan JX, Gu GH, Hu YH, Qiu GZ (2010) Mechanism of bioleaching chalcopyrite by acidithiobacillus ferrooxidans in agar-simulated extracellular polymeric substances media. J Cent South Univ Technol 17(1):56–61
Fowler TA, Crundwell FK (1998) Leaching of zinc sulfide by thiobacillus ferrooxidans: experiments with a controlled redox potential indicate no direct bacterial mechanism. Appl Environ Microbiol 64(10):3570–3575
Rohwerder T, Gehrke T, Kinzler K, Sand W (2003) Bioleaching review part A: progress in bioleaching: fundamentals and mechanisms of bacterial metal sulfide oxidation. Appl Microbiol Biotechnol 63(3):239–248
Zeng WM, Cai YX, Hou CW, Liu AJ, Peng TJ, Chen M, Qiu GZ, Shen L (2020) Influence diversity of extracellular DNA on bioleaching chalcopyrite and pyrite by Sulfobacillus thermosulfidooxidans ST. J Central S Univ 27(5):1466–1476
Zhu W, Xia JL, Peng AA, Nie ZY, Qiu GZ (2013) Characterization of apparent sulfur oxidation activity of thermophilic archaea in bioleaching of chalcopyrite. Trans Nonferrous Met Soc China 23(8):2383–2388
Petersen J, Dixon DG (2002) Thermophilic heap leaching of a chalcopyrite concentrate. Miner Eng 15(11):777–785
Xin JJ, Liu JY, Wu ZL, Zhang HY, Zhang SL, Yang LH (2018) Research progress of passivation in the bioleaching of chalcopyrite. Metal Mine 9:15–21
Ren Z, Krishnamoorthy P, Sanchez PZ, Asselin E, Dixon DG, Mora N (2020) Catalytic effect of ethylene thiourea on the leaching of chalcopyrite. Hydrometallurgy 196:105–410
Zhao J, Brugger J, Pring A (2019) Mechanism and kinetics of hydrothermal replacement of magnetite by hematite. Geosci Front 10(1):29–41
Antonijevic MM, Bogdanovic GD (2004) Investigation of the leaching of chalcopyritic ore in acidic solutions. Hydrometallurgy 73(3–4):245
Cordoba EM, Munoz JA, Blazquez ML, Gonzalez F, Ballester A (2009) Leaching of chalcopyrite with ferric ion. Part I: General aspects. Hydrometallurgy 93(3–4):81–87
Zhao SR (2016) Study on mechanical activation leaching and activation mechanism of chalcopyrite. Northeastern University, Shenyang
Torres CM, Ghorbani Y, Hernandez PC, Justel FJ, Aravena ML, Herreros OO (2019) Cupric and chloride ions: leaching of chalcopyrite concentrate with low chloride concentration media. Minerals 9(10):639
Mokmeli M (2020) Pre feasibility study in hydrometallurgical treatment of low-grade chalcopyrite ores from sarcheshmeh copper mine. Hydrometallurgy 191:105215
Wu SF, Yang CR, Qin WQ, Jiao F, Wang J, Zhang YS (2015) Sulfur composition on surface of chalcopyrite during its bioleaching at 50℃. Trans Nonferrous Met Soc China 25(12):4110–4118
Zeng WM, Qiu GZ (2010) Development of heap bioleaching of copper sulphide ores. Metal Mine 8:102–107
Govender E, Bryan CG, Harrison STL (2015) A novel experimental system for the study of microbial ecology and mineral leaching within a simulated agglomerate-scale heap bioleaching system. Biochem Eng J 95(1):86–97
Sandeep P, Ata A, Nilotpala P, Haci D (2015) Current scenario of chalcopyrite bioleaching: a review on the recent advances to its heap-leach technology. Biores Technol 196:694–706
Bai YL, Wang W, Dong KW, Xie F, Lu DK, Chang YF, Jiang KX (2022) Effect of microwave pretreatment on chalcopyrite dissolution in acid solution. J Market Res 16:471–481
Yang H, Zhao S, Wang G, Zhang Q, Jin Z, Tong L, Chen G, Qiu X (2022) Mechanical activation modes of chalcopyrite concentrate and relationship between microstructure and leaching efficiency. Hydrometallurgy 207:105778
Tian ZY, Li HD, Wei Q, Jiao F, Qin WQ, Yang CR (2021) Effects of Cu2+, Fe2+, and Fe3+on bioleaching of chalcopyrite by moderate thermophilic mixed bacteria. Chin J Nonferrous Met 31(1):171–180
Acknowledgements
The authors express their sincere appreciation to the National Natural Science Foundation of China for the financial support (Project Nos. 21978122 and 21566017).
Author information
Authors and Affiliations
Corresponding author
Additional information
The contributing editor for this article was Atsushi Shibayama.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ji, G., Liao, Y., Wu, Y. et al. A Review on the Research of Hydrometallurgical Leaching of Low-Grade Complex Chalcopyrite. J. Sustain. Metall. 8, 964–977 (2022). https://doi.org/10.1007/s40831-022-00561-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40831-022-00561-5