Abstract
The most common cause of liver disease worldwide is now non-alcoholic fatty liver disease (NAFLD). NAFLD refers to a spectrum of disease ranging from steatosis to non-alcoholic steatohepatitis, causing cirrhosis, and ultimately hepatocellular carcinoma. However, the impact of NAFLD is not limited to the liver. NAFLD has extra-hepatic consequences, most notably, cardiovascular and renal disease. NAFLD and chronic kidney disease share pathogenic mechanisms including insulin resistance, lipotoxicity, inflammation and oxidative stress. Not surprisingly, there has been a recent surge in efforts to manage NAFLD in an integrated way that not only protects the liver but also delays comorbidities such as chronic kidney disease. This concept of simultaneously addressing the main disease target and comorbidities is key to improve outcomes, as recently demonstrated by clinical trials of SGLT2 inhibitors and GLP1 receptor agonists in diabetes. HIF activators, already marketed in China, also have the potential to protect both liver and kidney, as suggested by preclinical data. This review concisely discusses efforts at identifying common pathogenic pathways between NAFLD and chronic kidney disease with an emphasis on potential paradigm shifts in diagnostic workup and therapeutic management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Non-alcoholic fatty liver disease (NAFLD) is the most common cause of chronic liver disease [1]. NAFLD is associated with metabolic syndrome and obesity, and the growing worldwide prevalence of obesity is driving the increased prevalence of NAFLD, currently estimated at 24% [2]. NAFLD is considered a spectrum of liver disease ranging from steatosis (only intrahepatic triglyceride accumulation) to non-alcoholic steatohepatitis (NASH) to fibrosis ultimately leading to cirrhosis [3]. At the current rate of growth in prevalence, NAFLD will likely outpace hepatitis C as the leading indication for liver transplantation [1]. Importantly, NAFLD is linked to other metabolically-related disorders, such as cardiovascular diseases (CVD), diabetes mellitus (DM), and chronic kidney disease (CKD) [2].
The pathogenic mechanisms underlying NAFLD development include increased free fatty acids accumulation, inflammatory cytokines and insulin resistance [1,2,3]. In addition, higher serum fetuin A levels and decreased serum adiponectin levels are associated with NAFLD [4]. Hepatic oxidative stress, lipotoxicity leading to cell death, mitochondrial injury, endoplasmic reticulum (ER) stress, iron overload (not in all patients), chronic immune system activation, distorted gut microbiome (increased Proteobacteria and Bacteriodetes along with a decrease in Firmicutes) are all implicated in the development of NAFLD (Fig. 1) [5].
Multi-hit model for development of non-alcoholic fatty liver disease (NAFLD). NAFLD formation has a complex pathophysiology and many components such as impaired lipid metabolism, insulin resistance, inflammatory cytokines, oxidative stress, increased fructose and uric acid levels, dysbiosis of microbiome and genetic factors may play a role on NAFLD pathophysiology
These pathogenic mechanisms may also play a role in other systemic diseases, therefore numerous recent studies evaluated the association between NAFLD and other systemic diseases. CKD prevalence is approximately 13% worldwide [1], whereas CKD prevalence in patients with NAFLD has been estimated at approximately 20–25% twofold higher than inpatients without NAFLD [6].
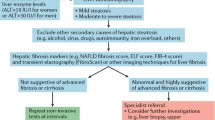
Due to the high morbidity and mortality of CKD [1, 7], early detection and treatment are key to prevent premature death. The association between NAFLD and CKD in diverse epidemiological studies (Table 1) that are associated with the prevalence and severity of CKD [8] suggests that NAFLD patients should be considered at high risk of CKD and screened for CKD by assessing eGFR and albuminuria (Table 2). Thus, histologically proven cirrhosis correlated with increased risk of CKD. In addition, NAFLD was associated with proteinuria [8]. In 1525 CKD patients followed for 10 years, NAFLD was independently associated with a larger decline in estimated glomerular filtration rate (eGFR) and with CKD progression [7]. In addition, NAFLD was associated with a higher incidence of CKD in 1760 patients with type 2 DM followed for 6.5 years and this was independent of other confounding factors such as age, gender and hypertension [9]. The prevalence of nephrolithiasis was also higher in patients with NAFLD referred to computed tomography (CT) due to clinically suspected renal colic [10].
Shared pathogenic mechanisms in NAFLD and CKD
Insulin resistance, atherogenic dyslipidemia, oxidative stress and pro-inflammatory mediators released from liver are considered key contributors in the pathogenesis of CKD [8, 11]. Thus, patients with NAFLD have increased advanced glycated end products, C-reactive protein, tumor necrosis factor-alpha and transforming growth factor-beta levels [9]. Moreover, endothelial dysfunction, decreased adiponectin and increased fetuin-A levels may interact with the renin–angiotensin–aldosterone system (RAAS) and contribute to CKD progression [1]. Gamma glutamyl transaminase (GGT) is specifically increased in NAFLD and is also associated with an increased risk for CKD [12]. Additionally, atherogenic dyslipidemia has also been linked to CKD through actions of oxidized low density lipoproteins (LDLs) on kidney cells, including mesangial cell proliferation and glomerular injury [8]. The shared pathogenic mechanisms in NAFLD and CKD and liver-kidney crosstalk are summarized in Figs. 2 and 3.
Pathogenic mechanisms potentially linking non-alcoholic fatty liver disease (NAFLD) and chronic kidney disease (CKD). Many mechanisms such as increased free fatty acid accumulation, increased inflammatory cytokines, insulin resistance, decreased hepatic oxidative stress, high uric acid and fructose levels leads to formation of fatty liver. Fatty liver increases the risk of development of CKD with activation of sympathetic nervous system, enhanced sodium retention, oxidative stress and inflammation by causing increased advanced glycated end products, CRP, tumor necrosis factor-alpha, transforming growth factor-beta levels, endothelial dysfunction, increased formation of oxidized LDL cholesterol
Potential therapeutic targets in the treatment of non-alcoholic fatty liver disease (NAFLD) and chronic kidney disease (CKD). Lifestyle modifications such as weight loss, increased physical activity, decreased salt intake and fructose intake, glucagon like peptide receptor activators and rennin angiotensin-aldosterone system blockers play a potential role for therapeutic intervention of fatty liver by anti-fibrotic, anti-oxidative and anti-inflammatory pathways
Another potential link consists of shared susceptibility gene variants between NAFLD and CKD. One example is PNPLA3: gene variants associated with decreased eGFR levels in children with NAFLD [13].
NAFLD and CKD with and without obesity
It is well known that obesity is very closely associated with NAFLD [14, 15]. On the other hand, CKD is associated with obesity [16]. In a sample of 2585 adults with a mean follow-up of 18.5 years, body mass index (BMI) was associated with a 23% (odds ratio (OR), 1.23; 95% CI 1.08–1.41) increase in developing kidney disease [17]. Similar findings were observed also in other large studies [18, 19]. Thus, it is possible that, at least in some cases, the problem is between obesity and the kidney rather than a direct link between the liver and the kidney. To say in other words, obesity may both cause NAFLD and CKD and there may not be a direct link between CKD and NAFLD. Thus it is important to underline evidence of kidney involvement in patients with fatty liver without obesity. Unfortunately, there is not satisfactory data in this regard. In one study performed in children partly explained this issue. Pacifido et al. showed that children with NAFLD had lower eGFR and higher albuminuria. More importantly, multivariate logistic regression analysis revealed that NAFLD was associated with decreased eGFR and/or microalbuminuria [OR, 2.54 (95% CI 1.16–5.57); p < 0.05] independently of anthropometric variables [20].
Besides, new data showed that there are cases of NAFLD among lean people. Seto et al. suggested that approximately 20% of the Asian population, lean NAFLD is closely linked with insulin resistance, diabetes, and other metabolic complications which are risk factors also for CKD [21]. Thus, we suggest that NAFLD is independently associated with CKD however more studies are needed in this issue.
Insulin resistance
Insulin resistance has been considered as a major pathological factor for development of both NAFLD and CKD, as well as a complication of NAFLD due to elevated liver free fatty acid uptake. Insulin resistance can accelerate CKD progression by modulating renal hemodynamics following the activation of the sympathetic nervous system, sodium retention and inactivation of the natriuretic peptide system [22]. Insulin resistance is also associated with CVD via increased oxidative stress, inflammation and endothelial dysfunction [22].
Fructose metabolism and uric acid
Several recent studies demonstrated that high fructose intake from sweetened beverages is associated with a high risk of NAFLD and progressive CKD [23,24,25,26]. Liver fructokinase phosphorylates fructose to fructose-1-phosphate, ultimately resulting in increased accumulation of uric acid. Uric acid may contribute to the development and progression of NAFLD, CKD and CVD via hepatocyte adenosine triphosphate (ATP) depletion, mitochondrial reactive oxygen species (ROS) generation, enhanced hepatic and renal lipogenesis, reduced nitric oxide (NO) bioavailability, endothelial dysfunction and proinflammatory cytokine secretion [24, 25]. Uric acid also activates aldose reductase and the polyol pathway for endogenous fructose and fat production causing fatty liver [27]. In longitudinal studies, higher uric acid levels were a risk factor for developing insulin resistance [28], dyslipidemia [29] and fatty liver [30, 31]. Hyperuricemia is also a known risk factor for developing CKD [32, 33]. This supports a role for hyperuricemia causes in both CKD and NAFLD.
Fetuin-A and adiponectin
Increased fetuin-A levels and decreased adiponectin levels are associated with NAFLD and CKD. Fetuin-A is a 64-kDa liver-secreted serum glycoprotein that promotes insulin resistance by disrupting insulin signaling through 5-AMP activated protein kinase (AMPK) inhibition after binding to insulin receptor tyrosine kinase in hepatocytes and skeletal muscle [34]. Additionally, downregulates adiponectin in adipose tissue through the Wnt-PPAR γ pathway and indeed, Fetuin-A levels are inversely correlated with adiponectin levels [35]. Adipose tissue-secreted Adiponectin improves insulin resistance, thus opposing fetuin-A actions. In this regard, adiponectin levels are inversely correlated with proteinuria [4].
Oxidative Stress
Oxidative stress is another key promoter of NAFLD and CKD. Nuclear erythroid related factor-2 (Nrf2) is a transcription factor that behaves as a master activator of the transcription of many anti-oxidant enzymes and has anti-inflammatory properties [24, 25]. In this regard, the loss of Nrf2 markedly exacerbates nonalcoholic steatohepatitis [36]. By contrast, the Nrf2 activator bardoxolone protected experimental animals from hepatic steatosis and is undergoing clinical trials for CKD, since it increases eGFR [37].The decrease of both oxidative stress and inflammation may protect from both NAFLD and CKD [25].
The FGF-Klotho axis
α Klotho and β Klotho are essential components of endocrine fibroblast growth factor (FGF) receptor complexes, and are required for binding of FGF23 and FGF19, FGF21, respectively, to their cognate FGF receptors (FGFRs) [38]. The kidney is the key source of αKlotho, a protein with anti-aging properties and kidney injury or systemic inflammation decrease αKlotho [39,40,41]. While there is little information on α Klotho levels in NAFLD, αklotho reduced liver lipid accumulation in obese mice, suggesting a liver-kidney cross-talk as discussed below [42]. By contrast, FGF19 is a satiety hormone released by the gut upon food ingestion of food that binds the β Klotho-FGFR4 complex in hepatocytes to promote metabolic responses to feeding. By contrast, fasting leads to liver secretion of the starvation hormone FGF21, which induces metabolic responses to fasting and stress responses [38]. FGF19 plays a key role in NAFLD pathogenesis and βKlotho genetic variants have been linked to NAFLD [43].
Liver-kidney crosstalk
Organ cross talk is a newly generated concept to describe and explain signals passing from organ to organ providing interactions between systems. A dysregulated cross-talk results in loss of homeostatic balance, potentially triggering organ damage. The liver and kidney have crosstalk mechanisms that are newly being understood. For example, in hepatorenal syndrome, patients with liver disease develop kidney failure in the absence of any histological abnormality cause [44]. In liver disease, activation of the RAAS and sympathetic nervous systems as compensatory mechanisms against splanchnic vasodilatation may lead to renal vasoconstriction, hypoperfusion and acute kidney injury. Moreover, increased secretion of endogenous vasopressin and the systemic inflammatory response due to translocation of intestinal bacteria are other important mechanisms promoting renal vasoconstriction and decreasing GFR [44].
Conversely, acute kidney injury can contribute to liver injury by promoting systemic inflammation and oxidative stress. Ischemia-induced damage due to decreased blood flow causes increased serum levels of alanine transaminase (ALT), aspartate transaminase (AST), lactate dehydrogenase (LDH), IL-6, IL-10 and TNF-a. Cytokine outflow results in increased vascular permeability causing neutrophil and lymphocyte migration and increased reactive oxygen species [44]. Further elucidation of the homeostatic hepatorenal crosstalk mechanisms is needed so that therapies can be targeted to this cross-talk.
Therapeutic Interventions for NAFLD Complications
Although the link between NAFLD and CKD seems evident from the above discussion, currently, CKD patients are not screened for NAFLD [7] (Table 2). But as discussed above, a diagnosis of co-existing NAFLD may influence CKD treatment and outcomes (Fig. 3).
The obvious first treatment would include lifestyle modification including weight loss, increased physical activity and smoking cessation [7, 8]. From a pharmacological point of view, some drugs have the potential to improve both NAFLD and CKD and should be first choice therapies for patients with both conditions. Thus, recent studies have tested new therapeutic modalities to delay NAFLD progression. The FANTASY open label study demonstrated that the angiotensin receptor blocker telmisartan, as opposed to losartan, may significantly decrease serum free fatty acids and improve fatty liver although there was no significant decrease in liver enzymes [45].
Specific therapies tested for NAFLD and CKD are incretin-based therapies such as glucagon like peptide receptor activators (GLP-1-RA) and dipeptidyl peptidase 4 (DPP-4) inhibitors which increase insulin secretion [46]. In a meta-analysis from the Liraglutide Effect and Action in Diabetes (LEAD) program, the GLP-1-RA liraglutide improved hepatic steatosis [47] and liver enzymes in patients with type 2 DM [48]. Besides glycemic control, GLP-1-RA may have a nephroprotective effect by inducing natriuresis with decreased angiotensin II activation and proximal tubular Na–H exchanger 3 inhibition, as shown in clinical trials [46, 49]. In this regard, GLP-1-RA have anti-fibrotic, anti-oxidative and anti-inflammatory actions in the kidneys [46]. By contrast, DPP-4 inhibitors have not provided consistent kidney protection in clinical trials.
Other key drugs are sodium-glucose cotransporter-2 (SGLT2) inhibitors. Together with GLP-1-RA, they are first choice antidiabetic drugs for the CKD patient [49]. In addition to kidney protection in dependently of glycemic control [46], most recently demonstrated in diabetic patients with overt diabetic kidney disease [50], SGLT2 inhibitors may reduce NAFLD progression via their anti-inflammatory, anti-fibrotic and anti-oxidative activities [46]. SGLT2 inhibitors reduce afferent arteriolar vasoconstriction (tubuloglomerular feedback) and may provide nephroprotection by reducing glomerular hyperfiltration, preventing glucose overload-induced oxidative stress and inflammation in proximal tubular cells and other mechanisms [49, 51].
Peroxisome proliferator activated receptor (PPAR) gamma agonists, hypoxia inducible factor (HIF) activation, mTOR complex1 inhibitors and galectin-3 inhibitors are other potential targets in kidney and liver disease [46]. However, the thiazolidinedione PPAR gamma agonists have been withdrawn from many markers due to cardiovascular safety concerns. By contrast, HIF activators are already in the market for uremic anemia in Chine, following phase 3 clinical trials and were recently reported to have a nephroprotective effect in clinical trials [52, 53]. Interestingly, in experimental animals, the HIF activator FG-4497 prevented liver steatosis [47], while JTZ-951 protected from the kidney and liver effects of a high fat diet [54].
Conclusion and future perspective
NAFLD is a major global health problem and its prevalence and impact, like that of CKD, has been growing depending on the increasing prevalence of obesity. NAFLD should be considered a multisystem disease with potential consequences for cardiac and renal health. Thus, NAFLD is associated with increased morbidity from CVD and CKD, and only the detailed characterization of common pathogenic mechanisms will allow the design of drugs that provide a holistic approach to the health problem. Many different pathogenic mechanisms may contribute to CKD progression as a complication of NAFLD. These include, but are not limited to insulin resistance, lipotoxicity, oxidative stress and inflammatory cytokines.
Besides, NAFLD is not homogenous disease and genetic factors especially in some specific patients population such as in hepatitis C patients are important. Hyperhomocysteinemia and the MTHFR C677T polymorphism promote steatosis and fibrosis in chronic hepatitis C patients therefore must be taken into consideration while taking measures regarding lifestyle and pharmacologic interventions [55,56,57]. We are also aware that above discussed mechanisms both for NAFLD and CKD exists it is not clear directly that if they are co-incidentally present or a real cause and effect relationship exists. Indeed, observational design of most studies do not enable us to establish a cause and effect relationship and it is currently not exactly known whether NAFLD carry a higher risk of incident CKD. There is also another concern that most of the studies used ultrasonography to detect NAFLD, which is the recommended first-line imaging. However liver biopsy is the gold standard method for diagnosing NAFLD as well [58]. The things become even more complicated given the fact that not all patients with NAFLD suffer from kidney damage. Thus genetic, environmental and other unknown factors probably might play a role for the development of CKD in NAFLD which further studies will show.
Future studies should address the relative contribution of these pathogenic mechanisms and the optimal, integrated way to tackle them. Lifestyle interventions such as weight loss and smoking cessation may be beneficial. From a pharmacological point of view, GLP-1-RA and SGLT2 inhibitors reduce the progression of both NAFLD and CKD and HIF activators will become available in the near future. However, only randomized clinical trials will identify the most effective and safe treatment modality. Additionally, an adequate diagnostic work-up, as suggested in Table 2, is required to identify the patients that may benefit from these novel integrated therapeutic approaches.
References
Mikolasevic I, Orlic L, Stimac D, Mavrinac V, Ferencic A, Rundic A, Babic V, Milic S (2016) Nonalcoholic fatty liver disease a multisystem disease? Lijec Vjesn 138:353–358
El Hadi H, Di Vincenzo A, Vettor R, Rossato M (2019) Cardio-metabolic disorders in non-alcoholic fatty liver disease. Int J Mol Sci 20:2215
Adams LA, Anstee QM, Tilg H, Targher G (2017) Non-alcoholic fatty liver disease and its relationship with cardiovascular disease and other extrahepatic diseases. Gut 66:1138–1153
Ix JH, Sharma K (2010) Mechanisms linking obesity, chronic kidney disease, and fatty liver disease: the roles of fetuin-A, adiponectin, and AMPK. J Am Soc Nephrol 21:406–412
Sanyal AJ (2019) Past, present and future perspectives in nonalcoholic fatty liver disease. Nat Rev Gastroenterol Hepatol 16:377–386
Han E, Lee YH (2017) Non-alcoholic fatty liver disease: the emerging burden in cardiometabolic and renal diseases. Diabetes Metab J 41:430–437
Jang HR, Kang D, Sinn DH, Gu S, Cho SJ, Lee JE, Huh W, Paik SW, Ryu S, Chang Y, Shafi T, Lazo M, Guallar E, Cho J, Gwak GY (2018) Nonalcoholic fatty liver disease accelerates kidney function decline in patients with chronic kidney disease: a cohort study. Sci Rep 8:4718
Musso G, Gambino R, Tabibian JH, Ekstedt M, Kechagias S, Hamaguchi M, Hultcrantz R, Hagstrom H, Yoon SK, Charatcharoenwitthaya P, George J, Barrera F, Hafliethadottir S, Bjornsson ES, Armstrong MJ, Hopkins LJ, Gao X, Francque S, Verrijken A, Yilmaz Y, Lindor KD, Charlton M, Haring R, Lerch MM, Rettig R, Volzke H, Ryu S, Li G, Wong LL, Machado M, Cortez-Pinto H, Yasui K, Cassader M (2014) Association of non-alcoholic fatty liver disease with chronic kidney disease: a systematic review and meta-analysis. PLoS Med 11:e1001680
Targher G, Chonchol M, Bertolini L, Rodella S, Zenari L, Lippi G, Franchini M, Zoppini G, Muggeo M (2008) Increased risk of CKD among type 2 diabetics with nonalcoholic fatty liver disease. J Am Soc Nephrol 19:1564–1570
Zeina AR, Goldenberg L, Nachtigal A, Hasadia R, Saliba W (2017) Association between nephrolithiasis and fatty liver detected on non-enhanced CT for clinically suspected renal colic. Clin Imaging 43:148–152
Ahn AL, Choi JK, Kim MN, Kim SA, Oh EJ, Kweon HJ, Cho DY (2013) Non-alcoholic fatty liver disease and chronic kidney disease in koreans aged 50 years or older. Korean J Fam Med 34:199–205
El Azeem HA, el Khalek SA, El-Akabawy H, Naeim H, Khalik HA, Alfifi AA (2013) Association between nonalcoholic fatty liver disease and the incidence of cardiovascular and renal events. J Saudi Heart Assoc 25:239–246
Di Costanzo A, Pacifico L, D'Erasmo L, Polito L, Martino MD, Perla FM, Iezzi L, Chiesa C, Arca M (2019) Nonalcoholic Fatty Liver Disease (NAFLD), but not ıts susceptibility gene variants, ınfluences the decrease of kidney function in overweight/obese children. Int J Mol Sci 20:444
Li L, Liu DW, Yan HY, Wang ZY, Zhao SH, Wang B (2016) Obesity is an independent risk factor for non-alcoholic fatty liver disease: evidence from a meta-analysis of 21 cohort studies. Obes Rev 17:510–519
Lu FB, Hu ED, Xu LM, Chen L, Wu JL, Li H, Chen DZ, Chen YP (2018) The relationship between obesity and the severity of non-alcoholic fatty liver disease: systematic review and meta-analysis. Expert Rev Gastroenterol Hepatol 12:491–502
Anastasio P, Viggiano D, Zacchia M, Altobelli C, Capasso G, Gaspare N (2017) Delay in renal hemodynamic response to a meat meal in severe obesity. Nephron 136:151–157
Fox CS, Larson MG, Leip EP, Culleton B, Wilson PW, Levy D (2004) Predictors of new-onset kidney disease in a community-based population. JAMA 291:844–850
Kramer H, Luke A, Bidani A, Cao G, Cooper R, McGee D (2005) Obesity and prevalent and incident CKD: the hypertension detection and follow-up program. Am J Kidney Dis 46:587–594
Hsu CY, McCulloch CE, Iribarren C, Darbinian J, Go AS (2006) Body mass index and risk for end-stage renal disease. Ann Intern Med 144:21–28
Pacifico L, Bonci E, Andreoli GM, Di Martino M, Gallozzi A, De Luca E, Chiesa C (2016) The ımpact of nonalcoholic fatty liver disease on renal function in children with overweight/obesity. Int J Mol Sci 17:1218
Voith K, Bruderlein FT, Humber LG (1978) Neuroleptics related to butaclamol. Synthesis and some psychopharmacological effects of a series of 3-aryl analogues. J Med Chem 21:694–698
Spoto B, Pisano A, Zoccali C (2016) Insulin resistance in chronic kidney disease: a systematic review. Am J Physiol Renal Physiol 311:F1087–f1108
Friedman SL, Neuschwander-Tetri BA, Rinella M, Sanyal AJ (2018) Mechanisms of NAFLD development and therapeutic strategies. Nat Med 24:908–922
Marcuccilli M, Chonchol M (2016) NAFLD and chronic kidney disease. Int J Mol Sci 17:562
Musso G, Cassader M, Cohney S, Pinach S, Saba F, Gambino R (2015) Emerging liver-kidney interactions in nonalcoholic fatty liver disease. Trends Mol Med 21:645–662
Targher G, Byrne CD (2017) Non-alcoholic fatty liver disease: an emerging driving force in chronic kidney disease. Nature Rev Nephrol 13:297–310
Sanchez-Lozada LG, Andres-Hernando A, Garcia-Arroyo FE, Cicerchi C, Li N, Kuwabara M, Roncal-Jimenez CA, Johnson RJ, Lanaspa MA (2019) Uric acid activates aldose reductase and the polyol pathway for endogenous fructose and fat production causing development of fatty liver in rats. J Biol Chem 294:4272–4281
Takir M, Kostek O, Ozkok A, Elcioglu OC, Bakan A, Erek A, Mutlu HH, Telci O, Semerci A, Odabas AR, Afsar B, Smits G, Sharma S, Johnson RJ, Kanbay M (2015) Lowering uric acid with allopurinol ımproves ınsulin resistance and systemic ınflammation in asymptomatic hyperuricemia. J Investig Med 63:924–929
Kuwabara M, Borghi C, Cicero AFG, Hisatome I, Niwa K, Ohno M, Johnson RJ, Lanaspa MA (2018) Elevated serum uric acid increases risks for developing high LDL cholesterol and hypertriglyceridemia: A five-year cohort study in Japan. Int J Cardiol 261:183–188
Jensen T, Niwa K, Hisatome I, Kanbay M, Andres-Hernando A, Roncal-Jimenez CA, Sato Y, Garcia G, Ohno M, Lanaspa MA, Johnson RJ, Kuwabara M (2018) Increased serum uric acid over five years is a risk factor for developing fatty liver. Sci Rep 8:11735
Kanbay M, Jensen T, Solak Y, Le M, Roncal-Jimenez C, Rivard C, Lanaspa MA, Nakagawa T, Johnson RJ (2016) Uric acid in metabolic syndrome: From an innocent bystander to a central player. Eur J Intern Med 29:3–8
Sato Y, Feig DI, Stack AG, Kang DH, Lanaspa MA, Ejaz AA, Sanchez-Lozada LG, Kuwabara M, Borghi C, Johnson RJ (2019) The case for uric acid-lowering treatment in patients with hyperuricaemia and CKD. Nature Rev Nephrol 15:767–775
Kanbay M, Afsar B, Covic A (2011) Uric acid as a cardiometabolic risk factor: to be or not to be. Contrib Nephrol 171:62–67
Pal D, Dasgupta S, Kundu R, Maitra S, Das G, Mukhopadhyay S, Ray S, Majumdar SS, Bhattacharya S (2012) Fetuin-A acts as an endogenous ligand of TLR4 to promote lipid-induced insulin resistance. Nat Med 18:1279–1285
Agarwal S, Chattopadhyay M, Mukherjee S, Dasgupta S, Mukhopadhyay S, Bhattacharya S (2017) Fetuin-A downregulates adiponectin through Wnt-PPARgamma pathway in lipid induced inflamed adipocyte. Biochim Biophys Acta Mol Basis Dis 1863:174–181
Chowdhry S, Nazmy MH, Meakin PJ, Dinkova-Kostova AT, Walsh SV, Tsujita T, Dillon JF, Ashford ML, Hayes JD (2010) Loss of Nrf2 markedly exacerbates nonalcoholic steatohepatitis. Free Radic Biol Med 48:357–371
Camer D, Yu Y, Szabo A, Dinh CH, Wang H, Cheng L, Huang XF (2015) Bardoxolone methyl prevents insulin resistance and the development of hepatic steatosis in mice fed a high-fat diet. Mol Cell Endocrinol 412:36–43
Kuro OM (2019) The Klotho proteins in health and disease. Nat Rev Nephrol 15:27–44
Fernandez-Fernandez B, Izquierdo MC, Valino-Rivas L, Nastou D, Sanz AB, Ortiz A, Sanchez-Nino MD (2018) Albumin downregulates Klotho in tubular cells. Nephrol Dial Transplant 33:1712–1722
Navarro-Gonzalez JF, Sanchez-Nino MD, Donate-Correa J, Martin-Nunez E, Ferri C, Perez-Delgado N, Gorriz JL, Martinez-Castelao A, Ortiz A, Mora-Fernandez C (2018) Effects of pentoxifylline on soluble klotho concentrations and renal tubular cell expression in diabetic kidney disease. Diabetes Care 41:1817–1820
Kanbay M, Vervloet M, Cozzolino M, Siriopol D, Covic A, Goldsmith D, Solak Y (2017) Novel Faces of Fibroblast Growth Factor 23 (FGF23): iron deficiency, inflammation, insulin resistance, left ventricular hypertrophy, proteinuria and acute kidney injury. Calcif Tissue Int 100:217–228
Rao Z, Landry T, Li P, Bunner W, Laing BT, Yuan Y, Huang H (2019) Administration of alpha klotho reduces liver and adipose lipid accumulation in obese mice. Heliyon 5:e01494
Dongiovanni P, Crudele A, Panera N, Romito I, Meroni M, De Stefanis C, Palma A, Comparcola D, Fracanzani AL, Miele L, Valenti L, Nobili V, Alisi A (2019) beta-Klotho gene variation is associated with liver damage in children with NAFLD. J Hepatol 72:411–419
Capalbo O, Giuliani S, Ferrero-Fernandez A, Casciato P, Musso CG (2019) Kidney-liver pathophysiological crosstalk: its characteristics and importance. Int Urol Nephrol 51:2203–2207
Hirata T, Tomita K, Kawai T, Yokoyama H, Shimada A, Kikuchi M, Hirose H, Ebinuma H, Irie J, Ojiro K, Oikawa Y, Saito H, Itoh H, Hibi T (2013) Effect of telmisartan or losartan for treatment of nonalcoholic fatty liver disease: fatty liver protection trial by Telmisartan or Losartan Study (FANTASY). Int J Endocrinol 2013:587140
Musso G, Cassader M, Cohney S, De Michieli F, Pinach S, Saba F, Gambino R (2016) Fatty liver and chronic kidney disease: novel mechanistic insights and therapeutic opportunities. Diabetes Care 39:1830–1845
Rahtu-Korpela L, Karsikas S, Horkko S, Blanco Sequeiros R, Lammentausta E, Makela KA, Herzig KH, Walkinshaw G, Kivirikko KI, Myllyharju J, Serpi R, Koivunen P (2014) HIF prolyl 4-hydroxylase-2 inhibition improves glucose and lipid metabolism and protects against obesity and metabolic dysfunction. Diabetes 63:3324–3333
Armstrong MJ, Houlihan DD, Rowe IA, Clausen WH, Elbrond B, Gough SC, Tomlinson JW, Newsome PN (2013) Safety and efficacy of liraglutide in patients with type 2 diabetes and elevated liver enzymes: individual patient data meta-analysis of the LEAD program. Aliment Pharmacol Ther 37:234–242
Sarafidis P, Ferro CJ, Morales E, Ortiz A, Malyszko J, Hojs R, Khazim K, Ekart R, Valdivielso J, Fouque D, London GM, Massy Z, Ruggenenti P, Porrini E, Wiecek A, Zoccali C, Mallamaci F, Hornum M (2019) SGLT-2 inhibitors and GLP-1 receptor agonists for nephroprotection and cardioprotection in patients with diabetes mellitus and chronic kidney disease. A consensus statement by the EURECA-m and the DIABESITY working groups of the ERA-EDTA. Nephrol Dial Transplant 34:208–230
Fernandez-Fernandez B, Fernandez-Prado R, Gorriz JL, Martinez-Castelao A, Navarro-Gonzalez JF, Porrini E, Soler MJ, Ortiz A (2019) Canagliflozin and Renal Events in Diabetes with Established Nephropathy Clinical Evaluation and Study of Diabetic Nephropathy with Atrasentan: what was learned about the treatment of diabetic kidney disease with canagliflozin and atrasentan? Clin Kidney J 12:313–321
Sanchez-Nino MD, Bozic M, Cordoba-Lanus E, Valcheva P, Gracia O, Ibarz M, Fernandez E, Navarro-Gonzalez JF, Ortiz A, Valdivielso JM (2012) Beyond proteinuria: VDR activation reduces renal inflammation in experimental diabetic nephropathy. Am J Physiol Renal Physiol 302:F647–657
Chen N, Hao C, Peng X, Lin H, Yin A, Hao L, Tao Y, Liang X, Liu Z, Xing C, Chen J, Luo L, Zuo L, Liao Y, Liu BC, Leong R, Wang C, Liu C, Neff T, Szczech L, Yu KP (2019) Roxadustat for anemia in patients with kidney disease not receiving dialysis. N Engl J Med 381:1001–1010
Provenzano R, Besarab A, Sun CH, Diamond SA, Durham JH, Cangiano JL, Aiello JR, Novak JE, Lee T, Leong R, Roberts BK, Saikali KG, Hemmerich S, Szczech LA, Yu KH, Neff TB (2016) Oral hypoxia-inducible factor prolyl hydroxylase inhibitor roxadustat (FG-4592) for the treatment of anemia in patients with CKD. Clin J Am Soc Nephrol 11(6):982–991. https://doi.org/10.2215/CJN.06890615
Saito H, Tanaka T, Sugahara M, Tanaka S, Fukui K, Wakashima T, Nangaku M (2019) Inhibition of prolyl hydroxylase domain (PHD) by JTZ-951 reduces obesity-related diseases in the liver, white adipose tissue, and kidney in mice with a high-fat diet. Lab Invest 99:1217–1232
Adinolfi LE, Ingrosso D, Cesaro G, Cimmino A, D'Antò M, Capasso R, Zappia V, Ruggiero G (2005) Hyperhomocysteinemia and the MTHFR C677T polymorphism promote steatosis and fibrosis in chronic hepatitis C patients. Hepatology 41:995–1003
Zampino R, Ingrosso D, Durante-Mangoni E, Capasso R, Tripodi MF, Restivo L, Zappia V, Ruggiero G, Adinolfi LE (2008) Microsomal triglyceride transfer protein (MTP) -493G/T gene polymorphism contributes to fat liver accumulation in HCV genotype 3 infected patients. J Viral Hepat 15:740–746
Zampino R, Macera M, Cirillo G, Pafundi PC, Rinaldi L, Coppola N, Pisaturo M, Adinolfi LE, Miraglia Del Giudice E, Ingrosso D, Capasso R (2018) No effect of MTP polymorphisms on PNPLA3 in HCV-correlated steatosis. Infez Med 26:244–248
Byrne CD, Targher G (2020) NAFLD as a driver of chronic kidney disease. J Hepatol 72:785–801
Sinn DH, Kang D, Jang HR, Gu S, Cho SJ, Paik SW, Ryu S, Chang Y, Lazo M, Guallar E, Cho J, Gwak GY (2017) Development of chronic kidney disease in patients with non-alcoholic fatty liver disease: a cohort study. J Hepatol 67:1274–1280
Yeung MW, Wong GL, Choi KC, Luk AO, Kwok R, Shu SS, Chan AW, Lau ESH, Ma RCW, Chan HL, Chan JC, Wong VW, Kong AP (2017) Advanced liver fibrosis but not steatosis is independently associated with albuminuria in Chinese patients with type 2 diabetes. J Hepatol. https://doi.org/10.1016/j.jhep.2017.09.020
Yasui K, Sumida Y, Mori Y, Mitsuyoshi H, Minami M, Itoh Y, Kanemasa K, Matsubara H, Okanoue T, Yoshikawa T (2011) Nonalcoholic steatohepatitis and increased risk of chronic kidney disease. Metabolism 60:735–739
Targher G, Pichiri I, Zoppini G, Trombetta M, Bonora E (2012) Increased prevalence of chronic kidney disease in patients with Type 1 diabetes and non-alcoholic fatty liver. Diabetic Med 29:220–226
Arase Y, Suzuki F, Kobayashi M, Suzuki Y, Kawamura Y, Matsumoto N, Akuta N, Kobayashi M, Sezaki H, Saito S, Hosaka T, Ikeda K, Kumada H, Ohmoto Y, Amakawa K, Tsuji H, Hsieh SD, Kato K, Tanabe M, Ogawa K, Hara S, Kobayashi T (2011) The development of chronic kidney disease in Japanese patients with non-alcoholic fatty liver disease. Internal Med (Tokyo, Japan) 50:1081–1087
Hwang ST, Cho YK, Yun JW, Park JH, Kim HJ, Park DI, Sohn CI, Jeon WK, Kim BI, Rhee EJ, Oh KW, Lee WY, Jin W (2010) Impact of non-alcoholic fatty liver disease on microalbuminuria in patients with prediabetes and diabetes. Internal Med J 40:437–442
Chang Y, Ryu S, Sung E, Woo HY, Oh E, Cha K, Jung E, Kim WS (2008) Nonalcoholic fatty liver disease predicts chronic kidney disease in nonhypertensive and nondiabetic Korean men. Metabolism 57:569–576
Mikolasevic I, Racki S, Bubic I, Jelic I, Stimac D, Orlic L (2013) Chronic kidney disease and nonalcoholic Fatty liver disease proven by transient elastography. Kidney Blood Pressure Res 37:305–310
Targher G, Mantovani A, Pichiri I, Mingolla L, Cavalieri V, Mantovani W, Pancheri S, Trombetta M, Zoppini G, Chonchol M, Byrne CD, Bonora E (2014) Nonalcoholic fatty liver disease is independently associated with an increased incidence of chronic kidney disease in patients with type 1 diabetes. Diabetes Care 37:1729–1736
Acknowledgements
MK gratefully acknowledge use of the services and facilities of the Koç University Research Center for Translational Medicine (KUTTAM), funded by the Presidency of Turkey, Presidency of Strategy and Budget. The content is solely the responsibility of the authors and does not necessarily represent the official views of the Presidency of Strategy and Budget.”AO research is supported by FIS PI19/00815, DTS18/00032, ERA-PerMed-JTC2018 (KIDNEY ATTACK AC18/00064 and PERSTIGAN AC18/00071, ISCIII-RETIC REDinREN RD016/0009 FEDER funds, Fundacion Renal Iñigo Álvarez de Toledo (FRIAT).
Funding
This study was not funded by any grant.
Author information
Authors and Affiliations
Contributions
All authors contributed to: (1) substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, and, (3) final approval of the version to be published.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kanbay, M., Bulbul, M.C., Copur, S. et al. Therapeutic implications of shared mechanisms in non-alcoholic fatty liver disease and chronic kidney disease. J Nephrol 34, 649–659 (2021). https://doi.org/10.1007/s40620-020-00751-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-020-00751-y