Abstract
Background
Although a previous study has reported the relationship between intact parathyroid hormone (iPTH) and whole parathyroid hormone (wPTH) levels in patients undergoing dialysis, the w/i PTH ratio (whole/intact PTH ratio) among predialysis chronic kidney disease (CKD) patients remains unclear. The present study therefore aimed to examine the relationship between w/i PTH ratio and kidney function and determine other factors affecting the w/i PTH ratio.
Methods
An observational study including 773 predialysis CKD patients was conducted. The correlation between w/i PTH ratio and kidney function, as well as clinical factors at different CKD stages, were assessed using univariate and multivariate analyses. In addition, the relationship between w/i PTH ratio and composite renal outcome [kidney transplantation, dialysis, and 30% decline in estimated glomerular filtration rate (eGFR)] was examined.
Results
The w/i PTH ratio decreased as the CKD stage progressed. Patients in each CKD stage (1/2, 3, 4, and 5) had a w/i PTH ratio of 0.85, 0.81, 0.78, and 0.59, respectively. The inflection point in the correlation lines between eGFR and w/i PTH ratio was 24.1 mL/min/1.73 m2. In multivariate analysis, the w/i PTH ratio was significantly correlated with serum calcium levels only in the CKD5 group and with eGFR in the CKD3, CKD4 and CKD5 group. Furthermore, w/i PTH ratio, eGFR, serum phosphate levels, and urinary protein/creatinine ratio were determined to be significant independent predictors for composite renal outcome.
Conclusions
Our study demonstrated that changes in the w/i PTH ratio were associated with kidney function, abnormal mineral metabolism, and renal outcome.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The kidney is responsible for maintaining serum calcium, serum phosphate, and bone metabolism balance via several pathways. From the early stages of chronic kidney disease (CKD), serum fibroblast growth factor 23 (FGF23) levels increase, active vitamin D levels decrease, and parathyroid hormone (PTH) secretion is stimulated to keep serum calcium and phosphate levels within normal range [1]. However, as CKD progresses from early to advanced stages, physiological mechanisms gradually break down, thereby resulting in hypocalcemia, hyperphosphatemia, and hyperparathyroidism. Such mineral metabolism abnormalities have been closely associated with cardiovascular disease and mortality [2, 3]. Therefore, the Japanese Society of Dialysis Therapy (JSDT) and the 2017 Kidney Disease: Improving Global Outcomes (KDIGO) guidelines have recommended the control of serum intact PTH (iPTH) level within the appropriate ranges (JSDT, 60–240 pg/mL; KDIGO, 130–585 pg/mL) given that maintaining serum iPTH levels within such ranges has been associated with a lower risk for death in patients undergoing dialysis [4, 5].
Recent developments in the assay system for PTH measurement have led to the availability of a third–generation assay. Considering that the second–generation PTH assay (iPTH assay) measures 1–84 and 7–84 PTH fragments, such an assay would yield higher levels compared to a third–generation assay [whole PTH (wPTH) assay], which measures only 1–84 PTH fragment. However, only a few reports have suggested the superiority of a wPTH assay over an iPTH assay [6], while the current accumulation of evidence has revealed that an iPTH assay has been widely used in clinical settings.
wPTH is the biologically active form of PTH. On the other hand, 7–84 PTH is a degradation product in the parathyroid gland and liver and inhibits the wPTH activity and secretion [7, 8]. Although the detailed mechanisms and clinical implications of changes in the whole/intact (w/i) PTH ratio remain unclear, it seems to reflect not only the balance between wPTH production and degradation, but also the biological action of PTH.
In patients undergoing maintenance dialysis, the JSDT guideline recommended a target range for wPTH, which can be estimated by dividing the target iPTH range by 1.7. Considering this formula, the w/i PTH ratio could be determined as 0.58 in those patients [9].
However, only a few studies including a small number of patients have reported on the w/i PTH ratio at each CKD stage among those who did not undergo dialysis [10,11,12,13]. Therefore, the present study examines the relationship between w/i PTH ratio and kidney function, determines clinical factors related to the w/i PTH ratio, and discusses the clinical implication of the w/i PTH ratio.
Materials and methods
Study design and population
This was a retrospective observational study conducted in accordance with the principles stated in the Declaration of Helsinki. Our study protocol was approved by the appropriate institutional review committee of Kobe University Graduate School of Medicine (no. 1871). Informed consent was obtained from all participants.
This study included 954 predialysis patients at different CKD stages who visited our department between December 2014 and December 2015. All patients satisfied the KDIGO guideline criteria for CKD [14]. Patients who were diagnosed with primary hyperparathyroidism, underwent kidney transplantation, and developed acute myocardial infarction, acute heart failure, or serious infection were excluded from the present study. Those who had wPTH levels higher than their iPTH levels, had insufficient clinical data, and did not provide informed consent were also excluded. Among the 954 patients, 181 were excluded because of the aforementioned reasons (primary hyperparathyroidism, n = 4; kidney transplant, n = 45; acute myocardial infarction, n = 9; acute heart failure, n = 15; serious infection, n = 4; wPTH levels higher than iPTH levels, n = 14; insufficient clinical data, n = 90). The remaining 773 patients were classified according to CKD stage based on estimated glomerular filtration rate (eGFR); CKD stage 1 and 2 (≥ 60 mL/min/1.73 m2), stage 3 (30–59 mL/min/1.73 m2), stage 4 (15–29 mL/min/1.73 m2), and stage 5 (< 15 mL/min/1.73 m2). In the medical record, a history of diabetes mellitus (DM), coronary artery disease (CAD), chronic heart failure (CHF), hypertension (HTN), and hyperlipidemia (HL) were collected. CAD was defined as disease which causes occlusion or stenosis of the coronary arteries, such as myocardial infarction and angina pectoris. CHF was defined as chronic cardiac disorder which causes lowering exercise tolerance with dyspnea, malaise, and edema. HTN was defined as ≥ 140 mmHg systolic and/or ≥ 90 mmHg diastolic pressure and/or antihypertensive agents treatment. HL was defined as LDL cholesterol ≥ 140 mg/dL, HDL cholesterol ≤ 40 mg/dL, and/or triglyceride ≥ 150 mg/dL and/or lipid-lowering agents treatment. Among the patients with all CKD stages, 206 were able to follow the composite renal outcome (kidney transplantation, dialysis, and 30% decline in eGFR) in the two years retrospectively based on medical records.
Laboratory measurements
Laboratory tests were conducted using standardized clinical laboratory methods. The remaining EDTA–2Na plasma samples were stored at − 80 °C until analysis. iPTH and wPTH levels were determined using the Elecsys Intact PTH assay and Elecsys PTH (1–84) assay (Roche Diagnostics K.K., Tokyo, Japan).
Statistical analysis
Computer software application IBM SPSS statistics version 25.0 (SPSS Inc., IL, USA) was used for all statistical analyses. Normally distributed variables were presented as mean values ± standard deviation, while non–normally distributed variables were presented as median (quartile). Each group was compared using linear by linear analysis for nominal variables and Jonckheere–Terpstra analysis and analysis of variance for continuous variables. We used locally weighted scatter plot smoothing (LOWESS) curve on the scatter diagram of the w/i PTH ratio, the iPTH, the wPTH, corrected calcium, and phosphate levels versus eGFR and estimated the eGFR threshold value at which the slope of the w/i PTH ratio, the iPTH, the wPTH, serum calcium, and phosphate levels became steeper by minimizing the residual sum of squares. Simple linear regression analysis was performed on wPTH and iPTH in patients with each CKD stage. To determine independent predictors of the w/i PTH ratio, multiple linear regression analysis was conducted. The relationship between the composite renal outcome and w/i PTH ratio was investigated using Kaplan–Meier curves and Cox proportional hazard analysis. A P value of < 0.05 was considered statistically significant.
Results
Patient characteristics
Table 1 shows the characteristics and laboratory data for all patients according to each CKD stage. Although patients were significantly older as CKD stage progressed, no statistical difference was observed between the CKD stage 4 and 5 group. No differences in the percentage of males, smoking history, DM, and HL were observed among the study groups. The number of patients with CAD, CHF, and HTN increased significantly with declining kidney function. Increased prescription of calcium carbonate and decreased prescription of bisphosphonate were observed as CKD stage progressed. Laboratory data showed that as CKD stage progressed, iPTH, wPTH, blood urea nitrogen, creatinine, C–reactive protein, and urinary protein levels increased, while the w/i PTH ratio, hemoglobin, eGFR, and total cholesterol levels decreased. The CKD stage 5 group had significantly lower serum calcium levels and significantly higher serum phosphate levels than the other CKD groups.
Correlation between eGFR and w/i PTH ratio, serum calcium, and serum phosphate levels
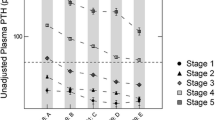
Simple linear regression analysis showed that the w/i PTH ratio was significantly correlated with eGFR in patients with CKD stage 1–5 (r = 0.675; P < 0.001). At an eGFR of 24.1 mL/min/1.73 m2, the slope of the LOWESS curve on the w/i PTH ratio versus eGFR scatter plot steepened. Similarly, the slope of the LOWESS curves on the iPTH, the wPTH, serum calcium, and serum phosphate levels versus eGFR scatter plot steepened at an eGFR of 17.5, 17.5, 20.0 and 17.8 mL/min/1.73 m2, respectively (Fig. 1). Simple linear regression analysis showed a significantly strong correlation between iPTH and wPTH in patients at each CKD stage (Stage 1/2: r = 0.985; P < 0.001, Stage 3: r = 0.986; P < 0.001, Stage 4: r = 0.990; P < 0.001, Stage 5: r = 0.994; P < 0.001) (Fig. 2).
Scatter diagram with LOWESS curve of the association between eGFR and w/i PTH ratio (a), iPTH (b), wPTH (c), serum calcium levels (d), and serum phosphate levels (e) among predialysis CKD patients. LOWESS, locally weighted scatter plot smoothing; eGFR, estimated glomerular filtration rate; w/i PTH, whole/intact parathyroid hormone; wPTH, whole PTH; iPTH, intact PTH; CKD, chronic kidney disease. The dashed lines represent statistical thresholds for eGFR (a 24.1 mL/min/1.73 m2, b 17.5 mL/min/1.73 m2, c 17.5 mL/min/1.73 m2, d 20.0 mL/min/1.73 m2, and e 17.8 mL/min/1.73 m2) at which the slopes steepened, respectively
Relationship between whole PTH and intact PTH levels among patients with each CKD stage. PTH, parathyroid hormone; CKD, chronic kidney disease. Intact PTH levels were strongly correlated with whole PTH levels at each CKD stage (Stage 1/2: r = 0.985; P < 0.001, Stage 3: r = 0.986; P < 0.001, Stage 4: r = 0.990; P < 0.001, Stage 5: r = 0.994; P < 0.001)
Correlation between clinical factors and w/i PTH ratio
Multivariate analysis showed that the w/i PTH ratio was significantly associated with male gender and use of active vitamin D agents among those with CKD stage 1 and 2; age, serum albumin levels, and eGFR among those with CKD stage 3; age, male gender, and eGFR among those with CKD stage 4; and use of active vitamin D agents, serum calcium, alkaline phosphatase levels and eGFR among those with CKD stage 5 (Table 2).
Predicting renal events using the w/i PTH ratio
A total of 206 patients with all CKD stages had available data on the composite renal outcome based on medical records and were divided into two groups based on whether or not their w/i PTH ratio was greater than or equal to the median value 0.79 among these patients. Total 66 composite renal outcomes were observed in the two years; 3 were kidney transplantation, 31 were dialysis initiation, and 32 were 30% decline in eGFR. The mean follow-up duration was 83.2 ± 33.3 weeks. The Kaplan–Meier curve showed that patients with a w/i PTH ratio of more than 0.79 had significantly better 2-year composite renal outcome compared to those with a lower w/i PTH ratio. The event free survival rate in the w/i PTH ratio > 0.79 and ≤ 0.79 group was 87% and 50%, respectively (Fig. 3). Multivariate analysis based on the Cox proportional hazards model showed that serum phosphate levels (HR 1.61, 95% CI 1.07–2.41; P < 0.05), eGFR (HR 0.92, 95% CI 0.90–0.96; P < 0.05), urinary protein/creatinine ratio (HR 1.18, 95% CI 1.11–1.26; P < 0.05), and w/i PTH ratio [hazard ratio (HR) 0.004, 95% CI 0.00002–0.96; P < 0.05], were statistically significant predictors for the composite renal outcome (Table 3).
Kaplan–Meier survival curve showing composite renal outcome according to the w/i PTH ratio. w/i PTH, whole/intact parathyroid hormone; CKD, chronic kidney disease. Predialysis CKD patients having a w/i PTH ratio ≤ 0.79 had significantly worse composite renal outcome compared to those having the w/i PTH ratio > 0.79 (Log–rank, P < 0.05)
Discussion
The present study demonstrated that (1) the w/i PTH ratio decreased as kidney function declined and rapidly decreased when eGFR reduced to less than 24.1 mL/min/1.73 m2; (2) serum calcium levels rapidly decreased and serum phosphate levels rapidly increased when eGFR reduced to less than 20.0 and 17.8 mL/min/1.73 m2, respectively; and (3) a lower w/i PTH ratio was significantly associated with poor renal outcome.
PTH is among the chronic kidney disease–mineral bone disorder (CKD–MBD) parameters that need to be controlled appropriately [5]. Serum PTH levels are elevated to keep normal active vitamin D and calcium levels within normal range. Impaired excretion of phosphate from urine due to reduced kidney function and resistance to the action of PTH under uremic conditions also increase serum PTH levels [15]. Thus, PTH has a close interaction with calcium and phosphate metabolism and greatly affects mineral and bone balance. The present study showed that the w/i PTH ratio decreased rapidly when eGFR was below 24.1 mL/min/1.73 m2. Therefore, from CKD stage 4, the balance between wPTH and iPTH seems to change. Previous studies have demonstrated that the prevalence of patients with hyperphosphatemia and hypocalcemia increased progressively in those with CKD stage 4 and 5 [16, 17]. The results of our study also showed that serum phosphate and calcium levels steeply changed around CKD stage 4 and that the w/i PTH ratio changed followed by serum calcium and phosphate levels.
A decrease in serum calcium levels, increase in serum phosphate levels, and impaired excretion from kidney due to declining kidney function led to an increase in 7–84 PTH. Although 1–84 PTH has a physiologically active property, 7–84 PTH binds to the PTH receptor and inhibits 1–84 PTH activity [7]. In addition, a previous experimental study demonstrated that 7–84 PTH induced hypocalcemia and inhibited the 1–84 PTH secretory response to hypocalcemia [8]. Furthermore, parathyroid cells predominantly secrete 7–84 PTH during hypercalcemia, whereas 1–84 PTH secretion is promoted during hypocalcemia [18, 19]. The progression of CKD impairs the excretion of 1–84 PTH and 7–84 PTH fragments from the kidney. Therefore, the increase in both 1–84 PTH and 7–84 PTH levels and the change in the balance between 1–84 PTH and 7–84 PTH results in a lower w/i PTH ratio during advanced CKD. Furthermore, our multivariate analysis showed that serum phosphate level was not a significant predictor of the w/i PTH ratio at all CKD stages, while serum calcium level was a significant predictor of the w/i PTH ratio only at CKD stage 5. Taken together, our results suggest that serum calcium level has a stronger effect on increasing PTH production compared to serum phosphate level.
A few reports have examined the clinical implication of the w/i PTH ratio in clinical settings. A previous study including predialysis Japanese patients with CKD demonstrated that the w/i PTH ratio increased with serum calcium level and GFR decline and was significantly associated with bone metabolic markers [20]. Another study performing bone biopsy reported that the w/i PTH ratio was a useful predictor of bone turnover in patients undergoing dialysis [21]. Moreover, the aforementioned study revealed that a lower w/i PTH ratio (< 1) was associated with low bone turnover. A few studies have also shown that the w/i PTH ratio was associated with hyperparathyroidism severity [22, 23]. Thus, although all previous studies had focused on the relationship between the w/i PTH ratio and bone turnover or hyperparathyroidism, no study to date has examined the relationship between the w/i PTH ratio and renal outcome. The present study showed that the w/i PTH ratio was an independent and significant predictor for composite renal outcome after adjustment for several factors, including kidney function, serum calcium levels, and serum phosphate levels in predialysis CKD patients. Therefore, we believe that the results presented herein are of considerable value.
Several possible mechanisms can explain the poor renal outcome observed with a low w/i PTH ratio. As mention above, a decrease in the w/i PTH ratio reflects an increase in the 7–84 PTH fragment. Previous experimental studies have demonstrated that the 7–84 fragment decreased phosphate excretion from urine and inhibits the conversion of 25(OH) vitamin D to 1, 25(OH)2 vitamin D [7, 24]. These physiological alterations may increase FGF23 and decrease klotho. In addition, reduced 1, 25(OH)2 vitamin D activates the renin–angiotensin–aldosterone system [25]. These changes in mineral bone metabolisms could affect the progression of kidney damage [26,27,28].
Some limitations of the present study are worth noting. Firstly, given the observational nature of this study, the associations between several factors and the w/i PTH ratio may not reflect direct effects. Furthermore, this study used the serum calcium level calculated using Payne's equation. Therefore, there may be a slight dissociation from the ionized calcium level described previously [29]. Secondly, as we used second- and third-generation parathyroid hormone assays, we could not measure oxidized and non-oxidized PTH separately. It has been reported that higher non-oxidized PTH reduced the odds ratio for death in hemodialysis patients [30]. If we also could measure these types of PTH, we may obtain further information. Thirdly, we could not measure serum 25(OH) vitamin D, 1, 25(OH)2 vitamin D, and FGF23 levels and determine their effects on the w/i PTH ratio. Future studies should therefore investigate whether such factors influence the w/i PTH ratio and renal outcome.
In conclusion, our data suggest that the w/i PTH ratio decreases as kidney function declines possibly due to disorders in PTH fragment excretion from the kidney and calcium/phosphate metabolism. Furthermore, a lower w/i PTH ratio might be associated with worse renal outcome.
References
Fukagawa M, Nakanishi S, Fujii H, Hamada Y, Abe T (2006) Regulation of parathyroid function in chronic kidney disease (CKD). Clin Exp Nephrol 10:175–179
Block GA, Hulbert-Shearon TE, Levin NW, Port FK (1998) Association of serum phosphorus and calcium × phosphate product with mortality risk in chronic hemodialysis patients: a national study. Am J Kidney Dis 31:607–617
Molony DA, Stephens BW (2011) Derangements in phosphate metabolism in chronic kidney diseases/endstage renal disease: therapeutic considerations. Adv Chronic Kidney Dis 18:120–131
Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group (2017) KDIGO clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease–mineral and bone disorder (CKD-MBD). Kidney Int Suppl 7:1–59
Japanese Society for Dialysis Therapy (2012) Guidelines for the management for CKD-MBD. J Jpn Soc Dial Ther 45:301–356 (In Japanese)
Melamed ML, Eustace JA, Plantinga LC, Jaar BG, Fink NE, Parekh RS, Coresh J, Yang Z, Cantor T, Powe NR (2008) Third-generation parathyroid hormone assays and all-cause mortality in incident dialysis patients: the CHOICE study. Nephrol Dial Transpl 23:1650–1658
Slatopolsky E, Finch J, Clay P, Martin D, Sicard G, Singer G, Gao P, Cantor T, Dusso A (2000) A novel mechanism for skeletal resistance in uremia. Kidney Int 58:753–761
Huan J, Olgaard K, Nielsen LB, Lewin E (2006) Parathyroid hormone 7–84 induces hypocalcemia and inhibits the parathyroid hormone 1–84 secretory response to hypocalcemia in rats with intact parathyroid glands. J Am Soc Nephrol 17:1923–1930
Gao P, Scheibel S, DʼAmour P, John MR, Rao SD, Schmidt-Gayk H, Cantor TL (2001) Development of a novel immunoradiometric assay exclusively for biologically active whole parathyroid hormone 1–84: implications for improvement of accurate assessment of parathyroid function. J Bone Miner Res 16:605–614
Kurajoh M, Inaba M, Yamada S, Imanishi Y, Tsuchida T, Ishimura E, Nishizawa Y (2008) Association of increased active PTH(1–84) fraction with decreased GFR and serum Ca in predialysis CRF patients: modulation by serum 25-OH-D. Osteoporos Int 19:709–716
Herberth J, Fahrleitner-Pammer A, Obermayer-Pietsch B, Krisper P, Holzer H, Malluche HH, Dobnig H (2006) Changes in total parathyroid hormone (PTH), PTH-(1–84) and large C-PTH fragments in different stages of chronic kidney disease. Clin Nephrol 65:328–334
Brossard JH, Lepage R, Cardinal H, Roy L, Rousseau L, Dorais C, DʼAmour P (2000) Influence of glomerular filtration rate on non-(1–84) parathyroid hormone (PTH) detected by intact PTH assays. Clin Chem 46:697–703
Tsuchida T, Ishimura E, Hirowatari K, Matsumoto N, Imanishi Y, Jono S, Miki T, Inaba M, Nishizawa Y (2006) Serum levels of 1–84 and 7–84 parathyroid hormone in predialysis patients with chronic renal failure measured by the intact and bio-PTH assay. Nephron Clin Pract 102:c108–c114
Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group (2013) KDIGO clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl 3:1–150
Yamamoto S, Fukagawa M (2017) Uremic toxicity and bone in CKD. J Nephrol 30:623–627
Isakova T, Wahl P, Vargas GS, Gutiérrez OM, Scialla J, Xie H, Appleby D, Nessel L, Bellovich K, Chen J, Hamm L, Gadegbeku C, Horwitz E, Townsend RR, Anderson CA, Lash JP, Hsu CY, Leonard MB, Wolf M (2011) Fibroblast growth factor 23 is elevated before parathyroid hormone and phosphate in chronic kidney disease. Kidney Int 79:1370–1378
Nakano C, Hamano T, Fujii N, Matsui I, Tomida K, Mikami S, Inoue K, Obi Y, Okada N, Tsubakihara Y, Isaka Y, Rakugi H (2012) Combined use of vitamin D status and FGF23 for risk stratification of renal outcome. Clin J Am Soc Nephrol 7:810–819
Estepa JC, Lopez I, Felsenfeld AJ, Gao P, Cantor T, Rodriguez M, Aguilera-Tejero E (2003) Dynamics of secretion and metabolism of PTH during hypo- and hypercalcaemia in the dog as determined by the ‘intact’ and ‘whole’ PTH assays. Nephrol Dial Transpl 18:1101–1107
D’Amour P (2002) Effects of acute and chronic hypercalcemia on parathyroid function and circulating parathyroid hormone molecular forms. Eur J Endocrinol 146:407–410
Kurajoh M, Inaba M, Okuno S, Nagayama H, Yamada S, Imanishi Y, Ishimura E, Shoji S, Yamakawa T, Nishizawa Y (2011) Reduction of whole PTH/intact PTH ratio as a predictor of bone metabolism in cinacalcet treatment of hemodialysis patients with secondary hyperparathyroidism. Osteoporos Int 22:923–930
Monier-Faugere MC, Geng Z, Mawad H, Friedler RM, Gao P, Cantor TL, Malluche HH (2001) Improved assessment of bone turnover by the PTH-(1–84)/large C-PTH fragments ratio in ESRD patients. Kidney Int 60:1460–1468
Tanaka M, Komaba H, Itoh K, Matsushita K, Hamada Y, Fujii H, Fukagawa M (2008) The whole-PTH/intact-PTH ratio is a useful predictor of severity of secondary hyperparathyroidism. NDT Plus 1:59–62
Komaba H, Takeda Y, Shin J, Tanaka R, Kakuta T, Tominaga Y, Fukagawa M (2008) Reversed whole PTH/intact PTH ratio as an indicator of marked parathyroid enlargement: five case studies and a literature review. NDT Plus 1:54–58
Nakajima K, Nohtomi K, Sato M, Takano K, Sato K (2009) PTH(7–84) inhibits PTH(1–34)-induced 1,25-(OH)2D3 production in murine renal tubules. Biochem Biophys Res Commun 381:283–287
Yuan W, Pan W, Kong J, Zheng W, Szeto FL, Wong KE, Cohen R, Klopot A, Zhang Z, Li YC (2007) 1,25-dihydroxyvitamin D3 suppresses renin gene transcription by blocking the activity of the cyclic AMP response element in the renin gene promoter. J Biol Chem 282:29821–29830
Schwarz S, Trivedi BK, Kalantar-Zadeh K, Kovesdy CP (2006) Association of disorders in mineral metabolism with progression of chronic kidney disease. Clin J Am Soc Nephrol 1:825–831
Sugiura H, Yoshida T, Shiohira S, Kohei J, Mitobe M, Kurosu H, Kuro-o M, Nitta K, Tsuchiya K (2012) Reduced Klotho expression level in kidney aggravates renal interstitial fibrosis. Am J Physiol Renal Physiol 302:F1252–F1264
Zhang Y, Kong J, Deb DK, Chang A, Li YC (2010) Vitamin D receptor attenuates renal fibrosis by suppressing the renin-angiotensin system. J Am Soc Nephrol 21:966–973
Kaku Y, Ookawara S, Miyazawa H, Ito K, Ueda Y, Hirai K, Hoshino T, Mori H, Yoshida I, Morishita Y, Tabei K (2015) Approximation of corrected calcium concentrations in advanced chronic kidney disease patients with or without dialysis therapy. Nephron Extra 5:39–49
Tepel M, Armbruster FP, Grön HJ, Scholze A, Reichetzeder C, Roth HJ, Hocher B (2013) Nonoxidized, biologically active parathyroid hormone determines mortality in hemodialysis patients. J Clin Endocrinol Metab 98:4744–4751
Acknowledgements
The authors thank the staff of Kobe University Graduate School of Medicine (Dr Shuhei Watanabe, Dr Kimihiko Goto, and Dr Nozomi Yamada).
Funding
None.
Author information
Authors and Affiliations
Contributions
FH drafted this study. KO analyzed the data and wrote the text. All co-authors reviewed and approved this paper.
Corresponding author
Ethics declarations
Conflict of interest
This study was partly supported by Roche Diagnostics K.K.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Okamoto, K., Fujii, H., Goto, S. et al. Changes in the whole/intact parathyroid hormone ratio and their clinical implications in patients with chronic kidney disease. J Nephrol 33, 795–802 (2020). https://doi.org/10.1007/s40620-019-00690-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-019-00690-3