Abstract
Objective
It is uncertain whether increasing 25-hydroxyvitamin D (25-D) levels in chronic kidney disease (CKD) patients above those recommended by current guidelines result in progressive amelioration of secondary hyperparathyroidism. Our objective was to identify a potential therapeutic 25-D target which optimally lowers plasma parathyroid hormone (PTH) without producing excessive hypercalcemia or hyperphosphatemia in CKD.
Methods
We performed a cross-sectional analysis of 14,289 unselected stage 1–5 CKD patients from US primary care and nephrology practices utilizing a laboratory-based CKD clinical decision support service between September 2008 and May 2012. Estimated glomerular filtration rate (eGFR), plasma PTH, and serum 25-D, calcium, and phosphorus results were analyzed.
Results
In CKD stages 3–5, progressively higher 25-D pentiles contained progressively lower mean PTH levels. Regression analysis of log PTH on 25-D was significant in all CKD stages with no evidence of a decreasing effect of 25-D to lower PTH until 25-D levels of 42–48 ng/ml. Progressively higher 25-D concentrations were not associated with increased rates of hypercalcemia or hyperphosphatemia.
Conclusions
We found evidence for an optimal level of 25-D above which suppression of PTH progressively diminishes. This level is more than twice that currently recommended for the general population. We found no association between these higher 25-D levels and hyperphosphatemia or hypercalcemia. Additional prospective trials seem appropriate to test the idea that 25-D levels around 40–50 ng/ml could be a safe and effective treatment target for secondary hyperparathyroidism in CKD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It is well established that 25-hydroxyvitamin D (25-D) deficiency is prevalent in chronic kidney disease (CKD) patients [1–5], and likely exacerbates the deficiency of 1, 25-dihydroxyvitamin D (1, 25-D) [6–8]. Data suggest that repletion of 25-D in CKD stages 3 and 4 improves levels of 1, 25-D [9] and therefore may improve gut calcium (Ca) absorption [10] and bone histology [11], and suppress secondary hyperparathyroidism [12]. However, the optimal 25-D treatment target in CKD is unclear.
Kidney Disease: Improving Global Outcomes (KDIGO) guidelines recommend that “vitamin D deficiency be corrected using treatment strategies for the general population [13].” For the general population, the Institute of Medicine (IOM) has recently concluded that increasing 25-D levels above 20 ng/ml does not confer further benefits as judged by reduction of parathyroid hormone (PTH) [14]. It is unclear whether the IOM recommendation is applicable to patients with CKD, in whom secondary hyperparathyroidism is pronounced and multifactorial. The argument against more aggressive replacement would be concern for precipitating hypercalcemia or hyperphosphatemia.
Recent studies of treatment with nutritional vitamin D in CKD patients suggest a reduction in PTH with increasing levels of 25-D [9, 12, 15–25]. The majority of these studies focused on patients in CKD stages 3 and 4 [9, 15, 16, 18–20, 23]. However, no study has documented an optimal level of 25-D above which further reductions in PTH become minimal. We have analyzed a large, national database of patients in all 5 stages of CKD, from a variety of clinical practices, in order to identify what that optimal level of 25-D might be, and if such a level is associated with elevations in serum Ca or phosphorus (P). In addition, we sought to determine if the optimal level might be CKD stage-specific.
Materials and methods
Patients
For this cross-sectional analysis, we studied 14,289 patients drawn from unselected US physician practices (76.9 % nephrology, 22.8 % primary care, 0.3 % other specialty) making use of a laboratory-based CKD clinical decision support service (Litholink® Corporation, Chicago, IL, a subsidiary of LabCorp®) from September 2008 through May 2012. For each patient we selected the first complete set of laboratory data that contained serum creatinine for calculation of estimated glomerular filtration rate (eGFR), serum Ca, P, and 25-D, and plasma PTH all drawn on the same day. Only one set of labs per patient was utilized for this analysis. PTH was analyzed using Roche Diagnostics Operations, Inc. (Roche) reagent on a Roche Elecsys® or COBAS® E platform. 25-D was analyzed with DiaSorin Liaison® 25 OH Vitamin D Total reagent on the Diasorin Liaison® instrument. Serum Ca and P were analyzed with Roche reagent [1]. Serum creatinine was analyzed using the kinetic Jaffe method on a Roche platform.
All laboratory data were ordered for clinical management and treatment decisions were at the discretion of the ordering clinician. eGFR calculations were made using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) formula and were corrected for race when available. CKD stage was assigned based upon level of eGFR. Because clinicians chose to enroll patients in the Litholink® CKD program, it was assumed that the diagnosis of CKD was made a priori by the clinician based upon available clinical data. No patient in stage 5 received renal replacement therapy. Review exemption was granted from Western Institutional Review Board, Olympia, WA.
Analysis
Serum 25-D values were divided into pentiles using conventional methods. For convenience, we have labeled the pentiles in alphabetical order from A to E and refer to them in that manner in Results (A <17.8 ng/ml, B 17.8–24.9 ng/ml, C 25–31 ng/ml, D 31.1–39 ng/ml, E >39 ng/ml). ANOVA, general linear models, univariate regression, and comparison of group means were performed with conventional statistical software (SYSTAT® Software, Inc., San Jose, CA). Tukey’s Honestly Significant Difference Test was used to adjust for multiple pairwise comparisons within ANOVA models. Because PTH distributions depart markedly from normality [1], we log transformed PTH and performed both the ANOVAs and regression analyses on log transformed data. Untransformed PTH values were plotted on a log scale for visual clarity. Significance was taken as P < 0.05.
In order to determine the optimal 25-D level, we fitted log transformed PTH vs 25-D using nonlinear regression. Graphically, we inspected our data using both density-weighted least square and LOWESS non-parametric smoothers. Both revealed a flattening tendency of the regression compared to simple linear modeling. To determine the point at which linear regression deviated from the data, we performed cubic nonlinear regression and estimated the significance of the second and third order terms. We found significance and therefore obtained the approximate points of departure from linear using graphical interpolation from function plotting of the linear and cubic regressions. To sharpen the resolution, we calculated the difference function between linear and cubic, plotted the difference function, and determined the points at which it passed through zero. The full set of polynomial coefficients are submitted as supplemental information (Table S1); the graphs used for interpolation of separation points are included as supplemental figures S1 and S2.
Results
Patients
We studied 14,289 unselected patients (52 % female; Table 1). Mean age and all laboratory variables except 25-D varied progressively with CKD stage by linear trend analysis (P < 0.001). As expected, Ca values were lower, and P and plasma PTH values were higher with progressive fall in eGFR.
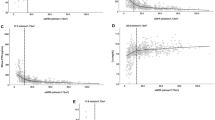
Plasma PTH by 25-D pentile
PTH changes between CKD stages within 25-D pentile
Within each 25-D pentile, PTH values for each CKD stage are distributed vertically in ascending order (stages 1–5). PTH was progressively higher with advancing CKD stage (Fig. 1). Within each pentile, all comparisons between CKD stages were significant expect for stages 1 vs. 2, and, in pentile E, between stages 1 and 3.
Plasma PTH by 25-hydroxyvitamin D pentile and CKD stage. PTH values (y-axis) are mean ± SEM. Dashed horizontal line is at 65 pg/ml, the upper limit of the PTH reference interval. Dashed lines connect symbols within CKD stage for ease of visualization. Horizontal gray bars are a visual guide to PTH values by CKD stage within 25-D pentile. Statistical analyses are in “Results”
PTH changes across 25-D pentiles within CKD stage
Within stages 3, 4, and 5, all differences between 25-D pentiles were significant except for the following: pentiles B vs C and D vs E in stage 3, D vs E in stage 4, and in stage 5, B vs C and D, C vs D, and D vs E (Fig. 1). Within stage 2, pentile A differed from C, D, and E. In stage 1, there were no significant differences between 25-D pentiles. With advancing CKD stage, progressively higher 25-D pentiles contained progressively lower mean PTH values. Adjustment of PTH levels for Ca did not alter the results (data not shown).
Regression of PTH on 25-D by CKD Stage
Linear regression analysis
The variations of mean PTH values within 25-D pentiles imply that PTH should show significant regression on 25-D levels generally and within CKD stage. This conjecture is supported by regression analysis (Fig. 2). In all 5 CKD stages, the regression coefficient for log PTH on 25-D differed significantly from zero (Fig. 2, compare regression equations across panels). The magnitude of the regression coefficient was, in general, lower with descending CKD stage. Stages 4 and 5 did not differ from each other, but both differed from stages 1 through 3 as noted by the lack of overlap of 95 % confidence limits (CI). Stages 1 through 3 did not differ from each other. Stages 1 and 2 were plotted in one panel (Fig. 2, lower right panel). The regression y-intercepts all differed significantly; 95 % CI did not overlap between any of the adjacent stages. This is the analytical homolog of the observation that PTH differences between CKD stages are the largest in the lowest 25-D pentile (pentile A, Fig. 1).
Regression of log plasma PTH vs 25-D by CKD Stage. Values for the intercept (α) and slope (β) are shown on the individual panels (95 % CI in parentheses), along with the least-squares linear regression line. Nonparametric locally weighted scatterplot (LOWESS) smoothers are overlayed. Upward departure of the LOWESS smoother suggests the log linear regression is not an ideal fit to the underlying data. Lines for stages 1 and 2 lie one below the other, respectively, in the bottom right panel
Polynomial regression analysis
In addition to the linear smoother on each panel of Fig. 2, we have plotted a locally weighted scatterplot smoother that responds to the point density in the vertical plane as one sweeps from left to right along the horizontal plane. Simple inspection reveals an obvious deviation from linearity of PTH as 25-D levels increase above 40 ng/ml. In order to analyze this departure quantitatively, we performed cubic polynomial regression and tested for the significance of the second and third order coefficients. In general, these were significant (detailed in Supplemental Table S1). Therefore, we calculated the separation point between linear and cubic regression using graphical interpolation (“Methods”, supplemental figures S1 and S2). Values (ng/ml) were: 42, 42, 46, 48, and 48 for stages 1–5, respectively.
Calcium and phosphorus
Serum Ca within 25-D pentile across CKD stage
In the lowest 25-D pentile (A), serum Ca levels in stages 4 and 5 differed from all other stages, and from each other (Fig. 3, left panel). The next lowest 25-D pentile (B) was the same except that Ca values no longer differed between stages 1 and 4. In pentiles C and D, differences were present only between stages 2–5 and 3–5, and 2–4 and 3–4, respectively. Within the highest pentile (E), Ca values did not differ between any CKD stage. All mean Ca values in this figure remained within the reference range for the laboratory (8.6–10.2 mg/dl).
Serum Ca within CKD stage across 25-D pentiles
Within stages 3–5, serum Ca in the lowest pentile (A) differed from pentiles C–E, and pentiles B and C differed within stages 4 and 5. Within stages 1 and 2, serum Ca levels did not differ between pentiles. As is apparent visually, serum Ca levels were lower with decreasing 25-D level and increasing CKD stage.
Occurrence of hypercalcemia
Hypercalcemia (serum Ca >10.2 mg/dl) occurred within 5–8 % of samples within each of the 25-D pentiles and CKD stages (not shown). Overall percentages of hypercalcemia for stages 1–5 were 4.7, 6.8, 6.6, 6.4, and 6.8, respectively. Values were not heterogeneous between 25-D pentiles except in stage 4, Χ2 = 10.5, P = 0.033. In stage 4, the heterogeneity arose from high values in the pentiles C and E (8.2 and 7.9 %, respectively). There was no evidence of heterogeneity between CKD stages within individual 25-D pentiles. Put another way, we found no evidence to suggest that increasing values of 25-D were associated with significant increase in the frequency of hypercalcemia.
Serum P
Values differed by CKD stage within all five pentiles (Fig. 3, right panel): stages 4 and 5 differed from all other stages; stage 3 differed from stage 2 in pentile A but otherwise stages 1–3 did not differ from one another. Serum P did not differ by 25-D pentile except within CKD stage 5 (pentile A differed from pentile E).
Occurrence of hyperphosphatemia
The frequency of hyperphosphatemia (P > 4.6 mg/dl, stages 1–4; P > 5.5 mg/dl, stage 5) increased with advancing CKD stage (1.3, 1.6, 5.1, 5.3, and 8.4 %, respectively). We found no heterogeneity in the prevalence of hyperphosphatemia across 25-D pentiles within CKD stage (Fig. 3). Within 25-D pentile by CKD stage, we found the expected heterogeneity created by the high P values in stages 4 and 5. As in the case of Ca, we found no evidence for worsening hyperphosphatemia with increasing 25-D levels.
Discussion
In normal individuals, 25-D deficiency is associated with elevated PTH [26]. 25-D deficiency is the rule in patients with CKD in the United States, and this is doubtless one factor in their secondary hyperparathyroidism [27]. However, the relationship between 25-D and PTH in CKD is complicated by the impaired conversion of 25-D to 1,25-D that results from reduced glomerular filtration rate, elevations of serum P and fibroblast growth factor 23 (FGF-23), and perhaps other mechanisms [28]. Nonetheless, correction of 25-D deficiency is recommended as an important initial step in treatment of elevated PTH in CKD although the optimal target level for 25-D in CKD is unclear [13], and may not be the same as in healthy individuals.
In the current study, PTH concentrations in stages 3–5 were progressively lower in ascending 25-D pentiles. We found a significant inverse log linear regression of PTH on 25-D in all 5 CKD stages which confirms and extends the pentile analysis. We found evidence of a plateau in this regression beyond a 25-D level of about 42–48 ng/ml in all 5 CKD stages. We did not find evidence for increased rates of hypercalcemia or hyperphosphatemia with higher 25-D levels in any stage. Of interest, mean P values were lowest in the highest 25-D pentile in stage 5. This may be an artifact of differential treatment with phosphate binders, but raises the question of possible effects of 25-D supplementation on phosphate balance, a topic best addressed with a dedicated trial.
In general, PTH is taken as a reporter for adequacy of 25-D: 25-D levels beyond which PTH does not further fall are considered optimal [14]. It would appear from this large data set that this point can be identified reasonably for both patients with normal kidney function and those with CKD.
Several recent studies have tested the efficacy of 25-D replacement on suppression of PTH in CKD [9, 12, 15–25]. In each study, patients with 25-D deficiency (values less than 30 ng/ml) were treated with either ergocalciferol or cholecalciferol for varying lengths of time, and the changes in 25-D and PTH determined. Although the results varied, repletion tended to raise 25-D concentrations, and in many cases also decreased PTH. This effect was demonstrated mainly in patients with stages 3 and 4 CKD; stages 2 and 5 were seldom included [15, 17, 21, 22, 24, 25]. Hypercalcemia and hyperphosphatemia occurred infrequently with 25-D repletion [9, 17, 20, 23, 25]. Comparable studies in dialysis patients were confounded by the simultaneous use of vitamin D receptor agonists [29, 30].
The few studies that measured 1,25-D levels found an increase with 25-D repletion, however the relationship between change in 25-D and either 1,25-D or PTH was inconsistent [9, 17, 19, 25]. Furthermore, the 25-D levels achieved in most of these studies were less than 40 ng/ml, thus it is unclear if achieving higher levels would have had greater impact on PTH suppression. None of these studies was designed to determine if there is an optimal level of 25-D above which no further PTH suppression will occur, or hypercalcemia or hyperphosphatemia become evident. Likewise, it is unclear from the studies if the effect of 25-D is present in CKD stages 1, 2 and 5.
One way to place our study in the larger perspective of the other 12 studies is to over plot published mean results on our log linear regression figures for PTH vs. 25-D (Fig. 4). For comparison, we calculated the regression of log plasma PTH vs 25-D using the mean values from each of the individual prior studies. Intercepts and regression coefficients are shown on the panels and can be compared with those of Fig. 2. PTH values in prior studies were all generally higher than in ours for any given level of 25-D. Linear regression analysis shows a significant increase of the intercept terms for stages 3 and 4 in prior studies vs. ours. Data are too few for conclusions about the other stages. Possibly the higher PTH values reflect assay differences. Of course, our cross-sectional analysis of diverse patients differs in design from these other studies, which were evaluating a response to therapy.
Relationship between log plasma PTH and 25-D in prior published studies compared to the linear regression values obtained in the present study. Correspondence between numbers on the figure and manuscript references: 1-Ref [15], 2-Ref [16], 3-Ref [9], 4-Ref [17], 5-Ref [18], 6-Ref [19], 7-Ref [20], 8-Ref [21], 10-Ref [23], 11-Ref [24], 12-Ref [25]. For comparison, we calculated the regression of log plasma PTH on 25-D using the mean 25-D values of the individual prior studies (dashed lines) and compared them to the regressions from the present study (solid lines)
The slope of log PTH on 25-D also differed, being more negative in past studies than in ours for patients in stage 3 (Fig. 4, compare slope coefficients to those of Fig. 2). None of the other stages had significant slopes; values did not differ significantly from 0. We have done our analysis only on mean values not having access to the full data sets from these studies. With complete data sets differences in slopes could be estimated with much higher precision. A recent meta-analysis of observational studies and trials also concluded that 25-D repletion led to significant decreases in PTH in CKD, but could not differentiate effects by stage [12].
Limitations of the present study are cross-sectional design and lack of information concerning diseases other than CKD which can influence mineral metabolism, ethnicity, medication use, and diet. For this reason we cannot determine why, for example, some patients in stages 1–3 had hypercalcemia or hyperphosphatemia. Likewise, we did not perform an external validation study. Even so, we can detect very significant regression of log PTH on 25-D and find evidence for an optimal 25-D level around 42–28 ng/ml above which further reduction of PTH diminishes. We find no evidence for significant hypercalcemia or hyperphosphatemia in higher 25-D pentiles, in accord with past studies. Missing to-date is a large scale prospective vitamin D trial to more accurately determine the optimal 25-D target level. Even at present it would seem the current 20 ng/ml recommendation from the IOM appears too low for CKD, even if it is indeed suitable for the general population.
References
Ennis J, Worcester E, Coe F (2012) Contribution of calcium, phosphorus and 25-hydroxyvitamin D to the excessive severity of secondary hyperparathyroidism in African-Americans with CKD. Nephrol Dial Transplant 27:2847–2853
Levin A, Bakris GL, Molitch M et al (2007) Prevalence of abnormal serum vitamin D, PTH, calcium, and phosphorus in patients with chronic kidney disease: results of the study to evaluate early kidney disease. Kidney Int 71:31–38
De Boer IH, Gorodetskaya I, Young B, Hsu CY, Chertow GM (2002) The severity of secondary hyperparathyroidism in chronic renal insufficiency is GFR-dependent, race-dependent, and associated with cardiovascular disease. J Am Soc Nephrol 13:2762–2769
Vassalotti JA, Uribarri J, Chen SC et al (2008) Trends in mineral metabolism: Kidney Early Evaluation Program (KEEP) and the National Health and Nutrition Examination Survey (NHANES) 1999–2004. Am J Kidney Dis 51:S56–S68
Gutierrez OM, Isakova T, Andress DL, Levin A, Wolf M (2008) Prevalence and severity of disordered mineral metabolism in Blacks with chronic kidney disease. Kidney Int 73:956–962
Reichel H, Deibert B, Schmidt-Gayk H, Ritz E (1991) Calcium metabolism in early chronic renal failure: implications for the pathogenesis of hyperparathyroidism. Nephrol Dial Transplant 6:162–169
Ishimura E, Nishizawa Y, Inaba M et al (1999) Serum levels of 1,25-dihydroxyvitamin D, 24,25-dihydroxyvitamin D, and 25-hydroxyvitamin D in nondialyzed patients with chronic renal failure. Kidney Int 55:1019–1027
Pitts TO, Piraino BH, Mitro R et al (1988) Hyperparathyroidism and 1,25-dihydroxyvitamin D deficiency in mild, moderate, and severe renal failure. J Clin Endocrinol Metab 67:876–881
Zisman AL, Hristova M, Ho LT, Sprague SM (2007) Impact of ergocalciferol treatment of vitamin D deficiency on serum parathyroid hormone concentrations in chronic kidney disease. Am J Nephrol 27:36–43
Colodro IH, Brickman AS, Coburn JW, Osborn TW, Norman AW (1978) Effect of 25-hydroxy-vitamin D3 on intestinal absorption of calcium in normal man and patients with renal failure. Metabolism 27:745–753
Eastwood JB, Stamp TC, Harris E, de Wardener HE (1976) Vitamin-D deficiency in the osteomalacia of chronic renal failure. Lancet 2:1209–1211
Kandula P, Dobre M, Schold JD, Schreiber MJ Jr, Mehrotra R, Navaneethan SD (2011) Vitamin D supplementation in chronic kidney disease: a systematic review and meta-analysis of observational studies and randomized controlled trials. Clin J Am Soc Nephrol 6:50–62
Kidney Disease: Improving Global Outcomes (KDIGO) CKD–MBD Work Group (2009) KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease–mineral and bone disorder (CKD-MBD). Kidney Int 76(Suppl 113):S1–S130
Institute of Medicine (IOM) (2011) Dietary reference intakes for calcium and vitamin D. The National Academies Press, Washington, DC
DeVille J, Thorp ML, Tobin L, Gray E, Johnson ES, Smith DH (2006) Effect of ergocalciferol supplementation on serum parathyroid hormone and serum 25-hydroxyvitamin D in chronic kidney disease. Nephrology (Carlton) 11:555–559
Al-Aly Z, Qazi RA, Gonzalez EA, Zeringue A, Martin KJ (2007) Changes in serum 25-hydroxyvitamin D and plasma intact PTH levels following treatment with ergocalciferol in patients with CKD. Am J Kidney Dis 50:59–68
Oksa A, Spustova V, Krivosikova Z et al (2008) Effects of long-term cholecalciferol supplementation on mineral metabolism and calciotropic hormones in chronic kidney disease. Kidney Blood Press Res 31:322–329
Dogan E, Erkoc R, Sayarlioglu H, Soyoral Y, Dulger H (2008) Effect of depot oral cholecalciferol treatment on secondary hyperparathyroidism in stage 3 and stage 4 chronic kidney diseases patients. Ren Fail 30:407–410
Chandra P, Binongo JN, Ziegler TR et al (2008) Cholecalciferol (vitamin D3) therapy and vitamin D insufficiency in patients with chronic kidney disease: a randomized controlled pilot study. Endocr Pract 14:10–17
Moe SM, Saifullah A, LaClair RE, Usman SA, Yu Z (2010) A randomized trial of cholecalciferol versus doxercalciferol for lowering parathyroid hormone in chronic kidney disease. Clin J Am Soc Nephrol 5:299–306
Trakarnvanich T, Chalapipat O, Disthabanchong S et al (2010) Effect of high dose ergocalciferol in chronic kidney disease patients with 25-hydroxyvitamin D deficiency. J Med Assoc Thai 93:885–891
Qunibi WY, Abdellatif A, Sankar S et al (2010) Treatment of vitamin D deficiency in CKD patients with ergocalciferol: are current K/DOQI treatment guidelines adequate? Clin Nephrol 73:276–285
Kovesdy CP, Lu JL, Malakauskas SM, Andress DL, Kalantar-Zadeh K, Ahmadzadeh S (2012) Paricalcitol versus ergocalciferol for secondary hyperparathyroidism in CKD stages 3 and 4: a randomized controlled trial. Am J Kidney Dis 59:58–66
Alvarez JA, Law J, Coakley KE et al (2012) High-dose cholecalciferol reduces parathyroid hormone in patients with early chronic kidney disease: a pilot, randomized, double-blind, placebo-controlled trial. Am J Clin Nutr 96:672–679
Marckmann P, Agerskov H, Thineshkumar S et al (2012) Randomized controlled trial of cholecalciferol supplementation in chronic kidney disease patients with hypovitaminosis D. Nephrol Dial Transplant 27:3523–3531
Holick MF (2007) Vitamin D deficiency. N Engl J Med 357:266–281
LaClair RE, Hellman RN, Karp SL et al (2005) Prevalence of calcidiol deficiency in CKD: a cross-sectional study across latitudes in the United States. Am J Kidney Dis 45:1026–1033
Nigwekar SU, Bhan I, Thadhani R (2012) Ergocalciferol and cholecalciferol in CKD. Am J Kidney Dis 60:139–156
Saab G, Young DO, Gincherman Y, Giles K, Norwood K, Coyne DW (2007) Prevalence of vitamin D deficiency and the safety and effectiveness of monthly ergocalciferol in hemodialysis patients. Nephron Clin Pract 105:c132–c138
Tokmak F, Quack I, Schieren G et al (2008) High-dose cholecalciferol to correct vitamin D deficiency in haemodialysis patients. Nephrol Dial Transplant 23:4016–4020
Conflict of interest
I (J. Ennis) am an employee of Litholink Corporation, a subsidiary of Laboratory Corporation of America (LabCorp), and hold stock in LabCorp. The research conducted was sponsored by this company. E. Worcester, F. Coe, and S. Sprague are consultants for Litholink Corporation and members of the Litholink CKD Scientific Advisory Board. S. Sprague is a consultant for and received honoraria from Amgen, Cytochroma, Kai, and Roche. S. Sprague received grant support from Amgen, Abbott, Cytochroma, NIH, Reata, Shire, and Vifor. All work for this project was undertaken at Litholink Corporation. All authors have made important contributions to the design and execution of this study, the analysis and interpretation of data, and the writing of this manuscript.
Ethical standard
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
For this type of study formal consent is not required.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
40620_2015_186_MOESM1_ESM.jpg
Supplemental Figure S1: Plot of the difference function between linear and cubic regressions of Log Plasma PTH vs 25-D. Coefficients are in Supplemental Table S1. The form of the function is in the footnote to Table S1. Y axis: C = cubic, L = linear predicted values. Zero crossing points are indicated along the horizontal dashed line. The cubic equation departs upward from the linear regression at x values between 42 -48 ng/ml. Stages are represented by panels as in Figure 2 (JPEG 241 kb)
40620_2015_186_MOESM2_ESM.jpg
Supplemental Figure S2: Individual log linear and cubic regressions of Plasma PTH vs 25-D. The crossing points of the linear and cubic equations occur at the points where the difference equation (Figure S1) cross zero. This graph is meant simply to illustrate the 2 regressions visually (JPEG 237 kb)
Rights and permissions
About this article
Cite this article
Ennis, J.L., Worcester, E.M., Coe, F.L. et al. Current recommended 25-hydroxyvitamin D targets for chronic kidney disease management may be too low. J Nephrol 29, 63–70 (2016). https://doi.org/10.1007/s40620-015-0186-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-015-0186-0