Abstract
Purpose
To describe the clinical and biochemical profile of patients with primary hyperparathyroidism (PHPT) of the Swiss Hyperparathyroidism Cohort, with a focus on neurobehavioral and cognitive symptoms and on their changes in response to parathyroidectomy.
Methods
From June 2007 to September 2012, 332 patients were enrolled in the Swiss PHPT Cohort Study, a nationwide prospective and non-interventional project collecting clinical, biochemical, and outcome data in newly diagnosed patients. Neuro-behavioral and cognitive status were evaluated annually using the Mini-Mental State Examination, the Hospital Anxiety and Depression Scale, and the Clock Drawing tests. Follow-up data were recorded every 6 months. Patients with parathyroidectomy had one follow-up visit 3–6 months’ postoperatively.
Results
Symptomatic PHPT was present in 43 % of patients. Among asymptomatic patients, 69 % (131/189) had at least one of the US National Institutes for Health criteria for surgery, leaving thus a small number of patients with cognitive dysfunction or neuropsychological symptoms, but without any other indication for surgery. At baseline, a large proportion showed elevated depression and anxiety scores and cognitive dysfunction, but with no association between biochemical manifestations of the disease and test scores. In the 153 (46 %) patients who underwent parathyroidectomy, we observed an improvement in the Mini-Mental State Examination (P = 0.01), anxiety (P = 0.05) and depression (P = 0.05) scores.
Conclusion
PHPT patients often present elevated depression and anxiety scores and cognitive dysfunction, but rarely as isolated manifestations. These alterations may be relieved upon treatment by parathyroidectomy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Primary hyperparathyroidism (PHPT) is a common disorder characterized by an excessive and abnormal secretion of parathyroid hormone (PTH). It is usually diagnosed through routine blood chemistry screening, investigation of osteoporosis, or kidney stones. The disease is characterized biochemically by chronically elevated serum calcium in the presence of inadequately elevated or inappropriately normal serum PTH levels. The classic presentation of PHPT involves the presence of renal stones, a decrease of bone mineral density, resulting in low-trauma fractures, and neuromuscular weakness. Some reports have described an increased mortality due to cardiovascular disease and malignancy [1, 2]. An increased risk of death has been observed both among patients with mild PHPT who did not undergo surgery and seriously affected individuals who underwent parathyroidectomy (PTx) [3]. However, this has not been consistently found and one study even demonstrated that neither overall mortality nor mortality due to cancer or cardiovascular disease were increased among patients with PHPT [4].
Subjective neuro-behavioral and cognitive symptoms have been well documented in patients with advanced disease [5–7], but a wide variety of mild, non-specific symptoms have been described in patients at an early stage of the disease, thus making it difficult to identify a causal relationship in these individuals. Although unrecognized in most cases, neuro-behavioral manifestations may impact on quality of life [8–11]. Some studies have addressed psychological or neurocognitive manifestations of PHPT, but only a few have used validated psychological instruments in routine clinical practice [12–15]. Finally, geographic and disease stage-dependent phenotypic expression may exist [16].
The aim of this multicentre cohort study was to describe the clinical and biochemical profile of patients with PHPT in Switzerland, particularly their neuro-behavioral and cognitive symptoms, changes in calcium and PTH over time and treatment modalities, and to determine whether cognitive and mood symptoms improve after PTx.
Subjects and methods
Study design
The Swiss PHPT Cohort Study is a multicentre, prospective, longitudinal project aimed at collecting clinical, densitometric and biochemical parameters, including outcome data, from patients with this disorder. Cohort recruitment was initiated in June 2007 and terminated in September 2012. The study was entirely non-interventional, without any influence on patient management. The observational period was planned for at least 3 years. At inclusion, baseline demographics, medical history, and the clinical and biochemical status of all study participants were recorded. During the follow-up period, changes in therapeutic interventions, biochemical parameters, bone health, and any symptoms/complications of PHPT were regularly documented according to the standard clinical procedures of the participating centers.
Ethics Committees’ approval was obtained prior to the study commencement in each center and all subjects gave informed written consent before study inclusion. The study was conducted according to the principles of the Declaration of Helsinki and the requirements of Good Clinical Practice. All registered patients were anonymously identified by a number. Each participating center was able to access their data via internet and a secure password on a web-based data capture (WBDC) system. Data transfer was encrypted to guarantee secrecy obligations and to prohibit data abuse. Members of the operational committee had full access to data (read-only) and permission to extract data from the study database for analysis. However, the center-specific, detailed patient identification was retrievable. Password and helpline support were provided by InfoSnake GmbH (Zug, Switzerland).
Participants
A total of 332 patients with sporadic PHPT diagnosed within the last 12 months were recruited in 15 centers in Switzerland located in Basel, Bern, Geneva, Lausanne, Lucerne, St. Gall, Locarno, and Zürich. Only patients (male and female) with confirmed hypercalcemia (serum calcium levels above the respective laboratory reference range) and diagnosed with PHPT after exclusion of other causes of hypercalcemia were eligible for inclusion. Patients with normocalcemic PHPT or active non-parathyroid malignancy were excluded.
Procedures
Physicians treated patients at their discretion and recorded patient follow-up according to the data collection sheet using a WBDC system. At the time of inclusion, baseline demographics (gender, date of birth with month and year), medical history, biochemical parameters, bone mineral density (BMD) measured routinely at the lumbar spine and left hip, biomarkers for bone and calcium metabolism (serum calcium, serum parathyroid hormone level, 25-hydroxyvitamin D), and current treatment regimens were recorded. Retrospective data were entered into the patient’s file if contemporary data were not available. During follow-up, data were recorded at least every 6 months according to usual clinical procedures and guidelines for the management of asymptomatic PHPT [17–19]. If a PTx was decided upon during follow-up, a final visit took place within 3⎼6 months after surgery. Deaths and information on subjects lost to follow-up were documented.
Changes in neuropsychological functioning were evaluated prospectively at baseline, at yearly intervals, and within 3⎼6 months after parathyroid surgery. Patients in all participating centres underwent a minimal set of neuro-psychological tests that are routinely used in the primary care setting, i.e., the Mini-Mental Status Examination (MMSE) [20] and the Clock Drawing Test (CDT) [21] for cognitive testing, and the Hospital Anxiety and Depression scale (HADS) for depression and anxiety evaluation [22, 23]. The HADS contains seven items scored in a 4-point range from 0 to 3, with higher scores indicating more symptoms [22]. A cut-off score of 8 or greater defined mild to severe symptoms of anxiety or depression. We used a score of 11 or greater to qualify symptoms as severe. All tests were sent anonymously to the Geneva study centre for a central reading and scoring by a trained geriatrician (GG).
Statistical analyses
Analyses of epidemiological data and results of local clinical procedures related to the treatment of PHPT patients were performed in a descriptive manner. Descriptive statistics [mean and standard deviation, median and 95 % confidence interval (CI), or interquartile range (IQR)] were evaluated for continuous variables. Count and percentage were evaluated for dichotomous and categorical variables. Parameters were analyzed at baseline and until at least 3 months later or until lost to follow-up. Wilcoxon matched-pairs signed-rank test was applied to compare two time point assessments within the same group. Chi-squared tests were used to compare proportions. Normality was verified using a Shapiro–Francia test. Correlations were determined by Spearman rank correlation for non-normally distributed variables and by simple linear regression analyses for Gaussian variables. Logistic regression was used to predict the probability of improvement in cognitive and neuropsychological tests, based on various clinical and biological manifestations. The effect of predictor variables was expressed by odds ratio (OR) and 95 % CI. There was no power calculation since this was a post hoc analysis in a cohort study. The significance level for two-sided p values was 0.05 in all tests. All statistical analyses were performed using STATA software (version 11.0; Stata Corporation, College Station, TX, USA).
Results
Patient characteristics
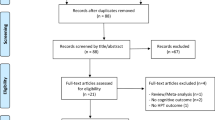
Among the 332 patients (mean age 67 ± 15 years; 78 % female) included in the cohort analysis, 42 (13 %) were younger than 50 years and 70 (21 %) were over 80 years old. Eight (2 %) had potentially other additional causes of hypercalcemia as follows: four patients received thiazides; two were on lithium therapy; and two had hyperthyroidism (Table 1; Fig. 1). Most patients had biochemically mild PHPT with median calcium and PTH levels of 2.68 mmol/l (95 % CI 2.66–2.70) and 12.4 pmol/l (95 % CI 11.7–13.40), respectively (Table 1).
Clinical presentation
One hundred forty-three patients (43 %) presented with a classic symptomatic form of PHPT (history of renal lithiasis, low-trauma fractures, or muscle weakness) (Table 2). One hundred eighty-nine patients were classified as “asymptomatic”. Of these, 69 % (131/189) presented with at least one of the criteria for PTx according to the guidelines for the management of asymptomatic PHPT [17–19]. Fifty-eight patients had no criteria for surgery. Among the latter, only 11 patients were operated because of the presence of non-specific symptoms (psychological and general symptoms as opposed to organ-specific symptoms) (Fig. 1).
Patient follow-up and PTx
We performed a prospective follow-up of 112 patients. During the study follow-up period, 153 (46 %) patients were operated; four had undergone a PTx before study inclusion and experienced persistent or recurrent disease. Twelve percent (18/153) had simultaneous thyroid surgery including total thyroidectomy (n = 6), thyroid lobectomy (n = 9), or subtotal thyroidectomy (n = 3). The related diagnoses were multinodular goiter (n = 13) with an associated Graves’ disease (n = 1), thyroid cancer (n = 2), and a single benign thyroid nodule (n = 3). PTx achieved a 97 % cure rate as indicated by postoperative serum calcium and PTH normalization. Three patients (2 %) who underwent PTx during study follow-up had persistent disease and two had recurrent disease (1 %). No patient sustained the following postoperative complications: recurrent laryngeal nerve injuries, hematomas, or permanent hypoparathyroidism. Patients undergoing PTx were younger (63 ± 13 vs 71 ± 15 years; P < 0.0001). Serum calcium and PTH levels were significantly higher in the PTx group (serum calcium, 2.8 ± 0.3 vs 2.6 ± 0.20 mmol/l, P < 0.0001; serum PTH, 20.3 ± 20.20 vs 12.8 ± 7.2 pmol/l, P < 0.0001). Forty-four patients reported a short-term improvement of one or more symptoms following PTx: fatigue (n = 28); musculoskeletal pain (n = 7); articular pain (n = 4); constipation or abdominal pain (n = 3); muscle weakness (n = 2); sleep disorders (n = 2); polydipsia (n = 2); nausea (n = 1); concentration (n = 1); mobility disability (n = 2) and not specified (n = 5). In those without surgery, a significant worsening of symptoms occurred in 12/34 (35 %). Conversely, only 7/78 (9 %) patients in the PTx group reported a worsening of symptoms (P = 0.001). Forty-four (59 %) patients claimed that they felt better after parathyroid surgery, while only one non-operated patient (3 %) reported an improvement in symptoms (P < 0.0001). During follow-up, nine patients had clinical complications: three with a kidney stone (after PTx), five with a low-trauma fracture, and one with muscle weakness. It was not possible to determine if the stones were newly formed or already present before PTx. Among patients with fractures, four of five occurred in those belonging to the non-PTx group. The rate of complications tended to be lower in the PTx group compared with the group with a medical follow-up (5 vs 15 %; P < 0.09).
Neuro-behavioral and cognitive symptoms in patients with PHPT and effect of PTx
At baseline, 183 (55 %) patients completed at least one of the neuropsychological tests (Table 1). Regarding the MMSE, 25 % displayed a pathological score with a significant cognitive impairment and a strong effect of age (ρ = −0.51; P < 0.001). Forty-seven percent had an abnormal CDT (Table 1). Similar to the MMSE, age was strongly associated with the CDT score (ρ = −0.41; P < 0.0001). To exclude cognitive impairment associated with advanced age, we limited our analysis to the 47 subjects who were younger than 60 years and had completed at least one of the neuropsychological tests. We found a negative correlation between the MMSE score and serum PTH levels (ρ = −0.13; P < 0.01) and between the CDT score and serum PTH (ρ = −0.09; P < 0.002), respectively, at baseline. No relation was found with serum calcium and 25-hydroxyvitamin D levels. When taking into consideration the whole population who had a least one HADS evaluation, no correlation was observed with serum levels of calcium or PTH.
To assess the impact of PTx on neurocognitive function, patients with PHPT were tested before and after PTx. A postsurgical improvement in the MMSE score was observed (P = 0.012). Preoperatively, 16 % of patients with PHPT scored less than 24 points for the MMSE. After PTx, only 5 % scored less than 24 points (P < 0.001) (Table 3). Those without improvement after PTx did not differ in terms of age, serum calcium or PTH levels. In 44 operated patients, anxiety (P = 0.051) and depression (P = 0.045) scores tended to improve after a median follow-up of 266 days (IQR 210–380).
In an attempt to identify those patients that would benefit from surgery in terms of cognitive and neuropsychological symptoms, we computed logistic regression models with an increase of cognitive performances or lower depression and anxiety scores as outcome. Regarding the depression score, a value before surgery of 8 or greater predicts success perfectly. Our results corroborated this measurement and all patients with an abnormal score (8 or greater) improved their score postoperatively compared with 32 % of subjects with lower values (P < 0.002). No significant changes in neuro-behavioral and cognitive symptoms were recorded in 17 non-operated patients evaluated longitudinally after a median follow-up of 287 days (IQR 229–556).
Discussion
Our study showed that the symptomatic form of PHPT was present in large proportion of the cases in Switzerland; that PHPT patients often presented elevated depression and anxiety scores and cognitive dysfunction, but rarely as isolated manifestations and these alterations may be relieved after parathyroidectomy.
It is widely recognized that the current presentation of PHPT has shifted to a mild or so-called “asymptomatic” form in relation to screening laboratory tests. It is now considered that the classic presentation of the disease with renal stones, fractures, muscle weakness or symptomatic hypercalcemia may be present in approximately 20 % of patients only. Nevertheless, it is known also that there are geographic differences in the predominant phenotypes and clinical presentations should be analyzed regionally [16]. In our study, 43 % of patients presented with the classic form. In particular, nephrolithiasis was present in 17 % of patients in our study. This frequency compares with recently published data reporting a prevalence ranging from 7 % [24] to 20 % [25, 26]. However, the number of patients was too low to evaluate whether the risk of nephrolithiasis decreases after curative PTx compared with medical observation.
In clinical practice, we usually select patients who may benefit from surgical procedures (PTx) based on NIH guidelines criteria [17–19, 27]. As these criteria do not include cognitive and psychological symptoms, there is the possibility to miss symptomatic patients with reversible manifestations. Neuropsychological symptoms may be subtle and nonspecific. Furthermore, they are not easily measurable and often not evaluated during routine clinical practice. In this study, we used sensitive screening tools for psychological or cognitive impairment that can be easily administered in daily routine. In our cohort, 83 % of the population presented either with classical symptoms (i.e., fractures, kidney stones or muscle weakness) or with at least one NIH guideline criteria. Finally, there were a very low number of patients with cognitive dysfunction or neuro-psychological symptoms without any other indication for surgery (data not shown).
Several studies have examined mood and cognitive symptoms in patients with PHPT [8, 11, 13, 14, 28–33], but only a few were performed with validated instruments, which can be used in daily routine clinical practice [34]. In this prospective cohort study, we evaluated the impact of the disease and the effect of PTx on neuro-psychological and cognitive function. We observed at baseline that a large proportion of patients presented with changes in mood and cognition. Twenty-five percent of patients had clinically significant cognitive deficits and 20–33 % were significantly depressed and/or anxious. These clinical manifestations could be related to elevated serum levels of calcium and PTH, although hypercalcemia was only mild in most patients. We found a possible association between cognitive function and serum PTH level in patients less than 60 years old. Above this age, the prevalence of age-related cognitive dysfunction is high and may have prevented any observation of a significant correlation. Depression and anxiety disorders are not related to these parameters in our study. PTx decreased both the depression score and the prevalence of depression. In addition, global cognitive functioning improved significantly after PTx, whereas no significant changes were observed in the non-operated group. To attempt to identify those patients with neuropsychological symptoms or cognitive impairment who might benefit from an improvement after PTx, we found that an abnormal depression score of 8 or greater was associated with a significant improvement postoperatively.
Regarding mood disorders, our results are similar to two recently published studies showing an increased prevalence of depression in PHPT patients and a significant improvement after surgery [13, 34]. Of note, Weber et al. used the same HADS questionnaire [34]. A positive correlation between the level of depressive symptoms and serum calcium was evidenced in some studies [8, 14, 32], but not in others [31].
As far as cognitive dysfunction is concerned, cognitive function was correlated with preoperative serum calcium levels in a limited number of studies [35, 36]. Data showing a beneficial effect of PTx are relatively scarce [13, 36–43]. Few studies examined the effect of PTx on global cognitive function [44]. This issue remains controversial as the effect of PTx has been considered insignificant in some studies [12, 45, 46]. The various studies describing PHPT-related neuro-cognitive symptoms and their response to PTx are summarized in Table 4. It should be underlined that the sample sizes of these studies were relatively limited and the tests used were heterogeneous and not easy enough to be routinely implemented in clinical setting. This table contains also three randomized studies examining the effect of PTx on quality of life [11, 29, 33]. Even though quality of life was not studied in our population, number of domains evaluated by generic instruments like SF-36 for example contain psychological well-being. Two of three randomized trial reported significant improvement following PTx [11, 33].
The positive effects of surgical intervention raise the question about the mechanism of the association between PHPT and neuro-behavioral and cognitive symptoms. Parathyroid hormone may act on the brain, through the action on PTH receptors which are found in the brain [47]. Through an ionophoretic effect it may induce cellular apoptosis by a intracellular calcium overload [48]. Serum parathyroid hormone may reduce regional cerebral blood flow [49]. Finally, the increased in serum calcium may mediate the effect, acting as a confounder. [44].
Our study has several strengths, including a large sample size at baseline and a multicentre design. The study population was not restricted to patients referred for surgery and thus represents a large spectrum of disease severity. There are also some potential limitations. The size of the included population represents approximately only 9 % of cases with an expected hospital discharge diagnosis of PHPT during the same period in Switzerland [50]. Thus, the followed sample may not be representative because of a selection bias and the participation rate was low, particularly in those who were not operated. Another limitation is the lack of randomization and/or surgical control group without parathyroid disease. Thus, we cannot exclude a placebo effect of surgery on neuropsychiatric symptoms. A further limitation was the variable intervals between evaluations. Patients who underwent surgery were evaluated 3–6 months after PTx, while those not operated were evaluated yearly using the cognitive and neuropsychological tests. The lack of the intra and inter-assay coefficient of variation between the different centers is also a limit. Finally, we did not measure the level of education, which may importantly influence the score on the cognitive test and may act as a potential confounder.
In conclusion, we observed that a large proportion of PHPT patients have elevated depression and anxiety scores and cognitive dysfunction. These symptoms are thus often missed as they are not routinely evaluated in clinical practice. Most symptoms, particularly mood changes, can be attenuated by PTx. Although the mild form of the disease is common, the classic presentation represents more than 40 % of cases. Altogether, there were a very low number of patients with cognitive dysfunction or neuro-psychological symptoms and without any other indication for surgery. Thus, neuro-psychological testing should be an additional diagnostic tool which could improve the selection of patients with PHPT to be evaluated for PTx.
References
Farr HW, Fahey TJ Jr, Nash AG, Farr CM (1973) Primary hyperparathyroidism and cancer. Am J Surg 126:539–543
Kaplan L, Katz AD, Ben-Isaac C, Massry SG (1971) Malignant neoplasms and parathyroid adenoma. Cancer 28:401–407
Palmer M, Adami HO, Bergstrom R, Jakobsson S, Akerstrom G, Ljunghall S (1987) Survival and renal function in untreated hypercalcaemia. Population-based cohort study with 14 years of follow-up. Lancet 1:59–62
Wermers RA, Khosla S, Atkinson EJ, Grant CS, Hodgson SF, O’Fallon WM et al (1998) Survival after the diagnosis of hyperparathyroidism: a population-based study. Am J Med 104:115–122
Fitz TE, Hallman BL (1952) Mental changes associated with hyperparathyroidism. Arch Intern Med 89:547–551
Reinfrank RF (1961) Primary hyperparathyroidism with depression. Arch Intern Med 108:606–610
Agras S, Oliveau DC (1964) Primary hyperparathyroidism and psychosis. Can Med Ass J. 91:1366–1367
Weber T, Keller M, Hense I, Pietsch A, Hinz U, Schilling T et al (2007) Effect of parathyroidectomy on quality of life and neuropsychological symptoms in primary hyperparathyroidism. World J Surg 31:1202–1209. doi:10.1007/s00268-007-9006-6
Talpos GB, Bone HG 3rd, Kleerekoper M, Phillips ER, Alam M, Honasoge M et al (2000) Randomized trial of parathyroidectomy in mild asymptomatic primary hyperparathyroidism: patient description and effects on the SF-36 health survey. Surgery 128:1013–1020. doi:10.1067/msy.2000.110844 (discussion 20-1)
Sheldon DG, Lee FT, Neil NJ, Ryan JA Jr (2002) Surgical treatment of hyperparathyroidism improves health-related quality of life. Arch Surg. 137:1022–1026 (discussion 6-8)
Rao DS, Phillips ER, Divine GW, Talpos GB (2004) Randomized controlled clinical trial of surgery versus no surgery in patients with mild asymptomatic primary hyperparathyroidism. J Clin Endocrinol Metab. 89:5415–5422. doi:10.1210/jc.2004-0028
Chiang CY, Andrewes DG, Anderson D, Devere M, Schweitzer I, Zajac JD (2005) A controlled, prospective study of neuropsychological outcomes post parathyroidectomy in primary hyperparathyroid patients. Clin Endocrinol (Oxf) 62:99–104. doi:10.1111/j.1365-2265.2004.02180.x
Roman SA, Sosa JA, Pietrzak RH, Snyder PJ, Thomas DC, Udelsman R et al (2011) The effects of serum calcium and parathyroid hormone changes on psychological and cognitive function in patients undergoing parathyroidectomy for primary hyperparathyroidism. Ann Surg 253:131–137. doi:10.1097/SLA.0b013e3181f66720
Joborn C, Hetta J, Lind L, Rastad J, Akerstrom G, Ljunghall S (1989) Self-rated psychiatric symptoms in patients operated on because of primary hyperparathyroidism and in patients with long-standing mild hypercalcemia. Surgery 105:72–78
White RE, Pickering A, Spathis GS (1996) Mood disorder and chronic hypercalcemia. J Psychosom Res 41:343–347
Silverberg SJ, Clarke BL, Peacock M, Bandeira F, Boutroy S, Cusano NE et al (2014) Current issues in the presentation of asymptomatic primary hyperparathyroidism: proceedings of the fourth international workshop. J Clin Endocrinol Metab 99:3580–3594. doi:10.1210/jc.2014-1415
Bilezikian JP, Khan AA, Potts JT Jr (2009) Guidelines for the management of asymptomatic primary hyperparathyroidism: summary statement from the third international workshop. J Clin Endocrinol Metab 94:335–339. doi:10.1210/jc.2008-1763
Bilezikian JP, Potts JTJ, Gel-H Fuleihan, Kleerkoper M, Neer R, Peacock M et al (2002) Summary statement from a workshop on asymptomatic primary hyperparathyroidism: a perspective for the 21st century. J Clin Endocrinol Metab 87:5353–5361
Marcocci C, Brandi ML, Scillitani A, Corbetta S, Faggiano A, Gianotti L et al (2015) Italian Society of Endocrinology Consensus Statement: definition, evaluation and management of patients with mild primary hyperparathyroidism. J Endocrinol Invest 38:577–593. doi:10.1007/s40618-015-0261-3
Folstein M, Folstein S, McHugh PR (1975) “Mini-Mental State”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Thalmann B, Spiegel R, Stähelin HB, Brubacher D, Ermini-Fünfschilling D, Bläsi S et al (2002) Dementia screening in general practice: optimized scoring for the clock drawing test. Brain Aging 2:36–43
Zigmond AS, Snaith RP (1983) The hospital anxiety and depression scale. Acta Psychiatr Scand 67:361–370
Herrmann C (1997) International experiences with the Hospital Anxiety and Depression Scale–a review of validation data and clinical results. J Psychosom Res 42:17–41 (pii: S0022399996002164)
Suh JM, Cronan JJ, Monchik JM (2008) Primary hyperparathyroidism: is there an increased prevalence of renal stone disease? AJR Am J Roentgenol 191:908–911. doi:10.2214/AJR.07.3160
Berger AD, Wu W, Eisner BH, Cooperberg MR, Duh QY, Stoller ML (2009) Patients with primary hyperparathyroidism–why do some form stones? J Urol 181:2141–2145. doi:10.1016/j.juro.2009.01.028
Rejnmark L, Vestergaard P, Mosekilde L (2011) Nephrolithiasis and renal calcifications in primary hyperparathyroidism. J Clin Endocrinol Metab 96:2377–2385. doi:10.1210/jc.2011-0569
Bilezikian JP, Brandi ML, Eastell R, Silverberg SJ, Udelsman R, Marcocci C et al (2014) Guidelines for the management of asymptomatic primary hyperparathyroidism: summary statement from the fourth international workshop. J Clin Endocrinol Metab. doi:10.1210/jc.2014-1413
Chan AK, Duh QY, Katz MH, Siperstein AE, Clark OH (1995) Clinical manifestations of primary hyperparathyroidism before and after parathyroidectomy. A case-control study. Ann Surg 222:402–412 (discussion 12-4)
Bollerslev J, Jansson S, Mollerup CL, Nordenstrom J, Lundgren E, Torring O et al (2007) Medical observation, compared with parathyroidectomy, for asymptomatic primary hyperparathyroidism: a prospective, randomized trial. J Clin Endocrinol Metab 92:1687–1692. doi:10.1210/jc.2006-1836
Pasieka JL, Parsons LL (1998) Prospective surgical outcome study of relief of symptoms following surgery in patients with primary hyperparathyroidism. World J Surg. 22:513–518 (discussion 8-9)
Dotzenrath CM, Kaetsch AK, Pfingsten H, Cupisti K, Weyerbrock N, Vossough A et al (2006) Neuropsychiatric and cognitive changes after surgery for primary hyperparathyroidism. World J Surg 30:680–685. doi:10.1007/s00268-005-0444-8
Espiritu RP, Kearns AE, Vickers KS, Grant C, Ryu E, Wermers RA (2011) Depression in primary hyperparathyroidism: prevalence and benefit of surgery. J Clin Endocrinol Metab 96:1737–1745. doi:10.1210/jc.2011-1486
Ambrogini E, Cetani F, Cianferotti L, Vignali E, Banti C, Viccica G et al (2007) Surgery or surveillance for mild asymptomatic primary hyperparathyroidism: a prospective, randomized clinical trial. J Clin Endocrinol Metab 92:3114–3121. doi:10.1210/jc.2007-0219
Weber T, Eberle J, Messelhauser U, Schiffmann L, Nies C, Schabram J et al (2013) Parathyroidectomy, elevated depression scores, and suicidal ideation in patients with primary hyperparathyroidism: results of a prospective multicenter study. JAMA Surg 148:109–115. doi:10.1001/2013.jamasurg.316
Brown GG, Preisman RC, Kleerekoper M (1987) Neurobehavioral symptoms in mild primary hyperparathyroidism: related to hypercalcemia but not improved by parathyroidectomy. Henry Ford Hosp Med J 35:211–215
Prager G, Kalaschek A, Kaczirek K, Passler C, Scheuba C, Sonneck G et al (2002) Parathyroidectomy improves concentration and retentiveness in patients with primary hyperparathyroidism. Surgery 132:930–935. doi:10.1067/msy.2002.128606 (discussion 5-6)
Numann PJ, Torppa AJ, Blumetti AE (1984) Neuropsychologic deficits associated with primary hyperparathyroidism. Surgery. 96:1119–1123
Roman SA, Sosa JA, Mayes L, Desmond E, Boudourakis L, Lin R et al (2005) Parathyroidectomy improves neurocognitive deficits in patients with primary hyperparathyroidism. Surgery 138:1121–1128. doi:10.1016/j.surg.2005.08.033 (discussion 8-9)
Walker MD, McMahon DJ, Inabnet WB, Lazar RM, Brown I, Vardy S et al (2009) Neuropsychological features in primary hyperparathyroidism: a prospective study. J Clin Endocrinol Metab 94:1951–1958. doi:10.1210/jc.2008-2574
Perrier ND, Balachandran D, Wefel JS, Jimenez C, Busaidy N, Morris GS et al (2009) Prospective, randomized, controlled trial of parathyroidectomy versus observation in patients with “asymptomatic” primary hyperparathyroidism. Surgery 146:1116–1122. doi:10.1016/j.surg.2009.09.034
Benge JF, Perrier ND, Massman PJ, Meyers CA, Kayl AE, Wefel JS (2009) Cognitive and affective sequelae of primary hyperparathyroidism and early response to parathyroidectomy. J Int Neuropsychol Soc 15:1002–1011. doi:10.1017/S1355617709990695
Babinska D, Barczynski M, Stefaniak T, Oseka T, Babinska A, Babinski D et al (2012) Evaluation of selected cognitive functions before and after surgery for primary hyperparathyroidism. Langenbecks Arch Surg 397:825–831. doi:10.1007/s00423-011-0885-5
Kahal H, Aye M, Rigby AS, Sathyapalan T, England RJ, Atkin SL (2012) The effect of parathyroidectomy on neuropsychological symptoms and biochemical parameters in patients with asymptomatic primary hyperparathyroidism. Clin Endocrinol (Oxf) 76:196–200. doi:10.1111/j.1365-2265.2011.04197.x
Lourida I, Thompson-Coon J, Dickens CM, Soni M, Kuzma E, Kos K et al (2015) Parathyroid hormone, cognitive function and dementia: a systematic review. PLoS One 10:e0127574. doi:10.1371/journal.pone.0127574
Goyal A, Chumber S, Tandon N, Lal R, Srivastava A, Gupta S (2001) Neuropsychiatric manifestations in patients of primary hyperparathyroidism and outcome following surgery. Indian J Med Sci 55:677–686
Cogan MG, Covey CM, Arieff AI, Wisniewski A, Clark OH, Lazarowitz V et al (1978) Central nervous system manifestations of hyperparathyroidism. Am J Med 65:963–970
Usdin TB, Gruber C, Bonner TI (1995) Identification and functional expression of a receptor selectively recognizing parathyroid hormone, the PTH2 receptor. J Biol Chem 270:15455–15458
Toescu EC, Vreugdenhil M (2010) Calcium and normal brain ageing. Cell Calcium 47:158–164. doi:10.1016/j.ceca.2009.11.013
Cermik TF, Kaya M, Ugur-Altun B, Bedel D, Berkarda S, Yigitbasi ON (2007) Regional cerebral blood flow abnormalities in patients with primary hyperparathyroidism. Neuroradiology 49:379–385. doi:10.1007/s00234-006-0198-5
Richert L, Trombetti A, Herrmann FR, Triponez F, Meier C, Robert JH et al (2009) Age and gender distribution of primary hyperparathyroidism and incidence of surgical treatment in a European country with a particularly high life expectancy. Swiss Med Wkly. 139:400–404 (doi: smw-12635)
Kahal H, Atkin SL, Sathyapalan T (2011) Pharmacological treatment of obesity in patients with polycystic ovary syndrome. J Obes 2011:402052. doi:10.1155/2011/402052
Acknowledgments
The authors thank Rosemary Sudan and Katy Giroux for invaluable editorial assistance. This project was supported by Amgen Switzerland through an unrestricted educational grant. The study has been endorsed by the Swiss Society of Endocrinology and Diabetology and the Swiss Association against Osteoporosis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest in relation to this study.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
All subjects gave informed written consent before study inclusion.
Rights and permissions
About this article
Cite this article
Trombetti, A., Christ, E.R., Henzen, C. et al. Clinical presentation and management of patients with primary hyperparathyroidism of the Swiss Primary Hyperparathyroidism Cohort: a focus on neuro-behavioral and cognitive symptoms. J Endocrinol Invest 39, 567–576 (2016). https://doi.org/10.1007/s40618-015-0423-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-015-0423-3