Opinion statement
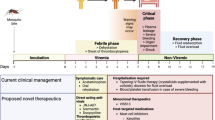
Dengue is an important mosquito-borne human viral disease globally. Dynamic clinical presentations in affected subjects have been well documented. Majority of affected people are asymptomatic, and to some extent, mild dengue fever. Only small percentage of individuals progress to life-threatening dengue, dengue hemorrhagic fever, and dengue-shock syndrome, featured with increase vascular permeability and plasma leakage. Although the death rate is low, the consequence of the public burden is always underestimated. Currently, mosquito control and supportive care are the most common practice for dengue measure and patient care, respectively. Severe form of dengue is a time-dependent febrile illness since hemorrhagic or shock manifested at the time of defervescence period, and the virus has been cleared off from the circulation of affected hosts. Severe dengue is therefore considered to be a consequence of imbalance of host response to the infection. Therapeutic modalities that can inhibit or mitigate the progression of the disease have been investigated for many decades. However, these intensive efforts on the antiviral drugs to counter the dengue virus (DENV) infection as well as recent clinical trials with repurposing drugs or with natural products from herbal plants to prevent the plasma leakage have not yet produced exciting and effective outcomes. Knowledge gained during recent years demonstrates that the DENV may have a much more complicated life cycle than researchers initially thought of. Furthermore, traditionally, dengue has been viewed to be an acute febrile illness, but recent evidence suggests that asymptomatic DENV infections in healthy subjects are a likely event. This rekindles new avenue of developing therapeutic antiviral drugs to treat dengue. However, in order to advance our view on developing antiviral drugs to dengue, an unconventional wisdom deviated from classical textbook knowledge is an innovative step to build and guide through the process.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dengue is the most important mosquito-borne human viral disease globally [1•]. The disease is induced by the infection of dengue virus (DENV) that can gain access to human through many routes, for example mosquito bites [2•], blood transfusion [3•], accidental needle injection [4,5,6], bone marrow and stem cells [7, 8], and organ transplantations [9••]. The well-known facts on dengue are wide spectrums of clinical manifestations, ranging from asymptomatic, undifferentiated fever, mild dengue fever to potential life-threatening dengue hemorrhagic fever (DHF), and dengue shock syndrome (DSS). As for the preventive measures, although there is a front runner tetravalent vaccine commercially available [10••], because of the limited efficacy and potential severe side effects [11], the recommendation for vaccination has its boundary [12•]. Currently, mosquito control measure, though limited effects, is widely adopted to reduce the spread of the dengue. As for the patient care, there is currently no specific treatment other than supportive care [2•].
With the lack of suitable animal models displaying the cardinal features of human dengue [13, 14], the development of antiviral drugs to DENV has been hindered significantly [15••, 16••]. Nevertheless, antiviral drugs or therapeutic modalities to DENV infection have been investigated for more than six decades utilizing mainly in vitro tissue culture system [17••]. Even though these antiviral drugs demonstrate an inhibition effect to DENV infection in tissue culture, the transient excitement is readily put out due to limited efficacy in small or large animal trials [17••, 18]. There are tons of literature reports on the subjects, shown in Table 1 with listed search terms in PubMed. Recently, a newer approach with natural products or repurposing drugs to tackle on DENV infection has been investigated [17••, 19,20,21]. At the end, similar outcomes have been documented [17••]. In general, as of today, over 90% of these antiviral drugs developed in vitro system go to human trials, and for those drugs that have gone through the trials, dismal efficacy are documented in phase I trials thwarting progress to further trials. PubMed search with terms indicated in Fig. 1 shows the efforts and difficulty in antiviral drugs or therapeutic modalities’ development to DENV infection.
Numerous researches revealed the difficulty of developing antiviral drugs to dengue for human usage. The search terms and the numbers of literature in PubMed were listed in each corresponding bar chart. The positive sign indicates that the term was affiliated with the initiated search term in each category.
Nevertheless, a highlighted summary of limited efficacy in recent antiviral drug trials to a common host pathway during dengue virus infection is shown in Table 2.
In addition, a brief discussion on the history and conditions for the limited efficacy in clinical trials for some of these promising anti-dengue drugs in vitro and in small animals are listed below.
Steroids
Severe forms of dengue (DHF and DSS) are considered to be a consequence of imbalance of immune response to the DENV infection in patients [37]. With the capacity of corticosteroids as potent modulators of the immune system, despite not a recommended treatment in the WHO dengue guidelines [38], the drugs have been empirically utilized to treat the complications of dengue [33, 39, 40]. However, after many small and randomized placebo-controlled trials [33, 41], the amounts of dose varying from 0.5 mg/kg/day to 30 mg every 4–6 h, their efficacy in treatment of DHF and DSS patients has been deemed to be of no benefit effect [35, 36, 39, 40, 42, 43]. However, all previous studies have been mainly in children; the effect of corticosteroid treatment in adults with DENV infection has not been evaluated. Hence, advocacy scholars believe that the benefits in reduction mortality in DSS and no major adverse effects should be paid attention to, and hence urge a need for high quality evidence and carefully designed randomized controlled trial of high-dose corticosteroids in the treatment of patients with DSS [44].
Statins
Cholesterol has been demonstrated involving in the replication cycle of DENV [45]. Statins have been shown to play a role in inhibition of the cholesterol synthesis and have been demonstrated in vitro to engage with the life cycle of viral replication by reduction of the release of infectious viral progeny [22]. In small animals infected with DENV, lovastatins treatment could increase the survival rate of infected rodents [23]. With the good safety record [46] and beneficial effects on the function of endothelial cells [47, 48], statins have been given green light to initiate the clinical trial to evaluate the efficacy effect on DENV infection [49]. However, although the initial pilot trial with 40 mg lovastatin in 30 dengue subjects showed a satisfactory effect; a randomized, double-blind, placebo-controlled trial with 80 mg lovastatin for 5 days in adult dengue subjects did not demonstrate to have an efficacy effect on any of the clinical manifestations or on dengue viremia despite the drug to be safe and well tolerated in these dengue patients [24].
Chloroquine
Upon virus infection, initiation of replication required precision steps to complete the cycle of the DENV replication. One of such critical steps is decoding and releasing viral materials to cytosol after entry. The step required engaging with acidic organelles of the cell. Alteration of the acidic PH-mediated fusion process would hinder the maturation of the virus [50]. Chloroquine is a rather poor base but is capable of neutralizing the acidic environment of intracellular organelles [51]. Empirical investigations in vitro, inhibition of DENV replication by chloroquine is not only cell type dependent but also at a dose-dependent manner [25, 52]. In addition, chloroquine appears to possess anti-inflammatory properties by modulating the production of cytokines in infected cells [53]. However, a double-blind, randomized, placebo-controlled trial of chloroquine for dengue patients, no favorable effects on viremia, NS1 antigenemia or fever duration, nor on the cytokine or T cell response were noticed [27, 54].
Iminosugars (celgosivir)
The mature virions of DENV contain envelope glycoproteins that rely on host glycosylation machinery to modify by adding oligosaccharide to specific asparagine residues on the structural membrane (prM) and envelope (E) proteins [55]. Assembling of DENV in endoplasmic reticulum (ER) is a necessity step for the cycle of viral replication. ER contains many glucosidase enzymes that are participating in the modification of the attachment of carbohydrate to the viral protein [29, 56]. As such, inhibiting host glycosylation pathways can lead to the aberration of viral glycoproteins resulting in the interference of viral assembly, secretion, and infectivity of the virus [57,58,59,60]. Iminosugars are monosaccharide that can mimic the functions of glucosidases and therefore are a competitor agent for glycoprotein processing [59, 61]. As of today, iminosugars have been demonstrated to have an inhibition effect on a number of viruses including DENV in vitro [29, 62] and in animal model [63, 64]. With the general safety profile, this line of compound was recommended to a dengue trial [65]. However, in a well-designed and conducted clinical trial to test the efficacy of celgosivir in DENV patients with initial dose at 400 mg within 6 h, followed by 200 mg every 12 h for a total of nine doses, no efficacy was found since the viral load and fever burden in patients with dengue did not apparently reduce [31].
There are several reasons contributing to the failure of the antiviral drugs development to DENV infection. Firstly, researchers adopt concept that works efficiently in the same family of flaviviruses, despite the clinical presentations are completely different within the family, such as hepatitis C virus (HCV), to investigate the antiviral drugs to DENV infection [66]. Initially, these drugs showing efficacy in HCV demonstrate an inhibition effect in DENV infection in tissue culture [67]. Gradually, disappointing outcomes cumulated in small and in large animal trials. Secondly, all these antiviral drugs are based upon in vitro tissue culture system, which is mainly based upon the cells with cancer properties. Almost all the family of flaviviruses can infect and propagate very well in cancer cells due to its high proliferation properties [68]. And yet, in natural setting, normal and healthy subjects are easily succumbed to DENV infection, suggesting that there are fundamental differences in biological and physiological properties in primary cells and tissue culture. Thirdly, there are no suitable animal models recapturing the cardinal features of human dengue [14]. However, researchers adopted immune deficiency small animals for the purpose of further screening the effectiveness of the antiviral drugs to DENV infection [13]. Although satisfactory results may be obtained in this setting, further trials in large animals often show a limited effect. Fourthly, the biology of DENV life cycle is mainly derived from in vitro system [50]. Despite detailed information about the productive cycle of DENV in vitro system has been panned out exclusively, the corresponding life cycle in humans remains unknown. Recent evidence shows that at least two different forms are seen in primary cells or in infected human beings [69••], suggesting a right system to screen and test the antiviral article is far more critical than original thought of. Fifthly, the cells that are permissive to DENV infection in vivo remain to be elucidated [70]. Generally speaking, researchers assume that the target cells for DENV to infect and amplify the infectious progeny are phagocytic cells [1•]. Due to the nature of these cells, it is difficult to differentiate between whether these cells engulf the cells infected by virus or are directly infected by DENV [71, 72••]. Since it is very difficult to identify these cells infected by DENV in vivo, more and more evidence suggests that the role of these phagocytic cells is for clearance of DENV in infected subjects rather than serving as a reservoir for the virus [71,72,73]. Recently, stem and progenitor cells, especially those megakaryocytic lineage cells, have been shown to play a major role for the DENV infection in vivo [74,75,76], providing a new avenue for development of antiviral drug to dengue. Lastly, but not the least, the receptors for the DENV infect stem and progenitor permissive cell remains at large. Although there are many candidate receptors for DENV infection in tissue culture system [77], antiviral drugs targeting on these receptors do not demonstrate an effect on inhibition.
One of the intricate developments in dengue field is that traditionally dengue is viewed as acute febrile illness, but evidence indicates that asymptomatic DENV infections assist viral dissemination and spread [78, 79••]. The later event can be a potential risk to public health and economic burden since these asymptomatic carriers may donate blood, bone marrow, or organs, and recipients may succumb to DENV infection [80••]. It is therefore imperative to establish a local regulation to guard the safety of the blood and organ supply. However, if a data base in local blood center or medical center can be shared with researchers and the health care workers, a potential drug candidate can be tested or administered to these subjects for the purpose of dengue control.
As of today, despite many antiviral drugs or therapeutic modalities have been in trails, limited effects are the common observation. Hence, an effective antiviral drug to DENV infection with newly developed knowledge urgently warrants further investigation.
Treatment
As aforementioned, as of today, there is no suitable and available treatment for dengue in spite of many decades of intensive research efforts. At the meantime, in recent years, researchers switch gear on the short cut by integration of the repurposing drugs to tackle on DENV infection. The dismal efficacy in recent clinical trials with repurposing drugs alarms researchers and clinicians that in order to have an effective drugs to treat dengue, applying the right setting and system to screen the repurposing drugs is an urgent activity. As such, the purpose of the current manuscript is to provide an innovative and alternative thought on the development of antiviral drugs to dengue. By saying this, we are therefore unable to provide any treatment options for the antiviral drugs to dengue within the manuscript. Currently, one of the common practices to mitigate the transmission of the DENV is by reduction of the density of mosquito populations, even though the measure is pricy, labor intensive, and of limited efficacy. As for the dengue patients, the standard practice is supporting care with closed monitoring.
Conclusions
Escalating evidence indicates that asymptomatic DENV infections and to some extent become persistent in healthy subjects. These individuals may carry the DENV and spread the diseases through many routes, including bitten by mosquitoes, blood transfusion, bone marrow, and organ transplantations, threatening public health and economic burden. Although a preventive vaccine is commercially available, the limited protective efficacy with potential severe side effects disallows most of the public officers to take on this measure. As of today, preventive measure is heavily relying upon, though costly with limited efficacy, mosquito population control. In order to effectively mitigate or eliminate the threat of the DENV in human society, one of the best strategies is to have an effective antiviral drug available. If there is one, in conjunction with set up on blood screening policy to uncover asymptomatic DENV infected individuals, the goal to achieve controlling or elimination of dengue is within days.
References and Recommended Reading
papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
• Guzman MG, et al. Dengue infection. Nat Rev Dis Primers. 2016;2:16055. This article describes a current preventive and control measure, and the likely challenge issues.
• WHO, GLOBAL STRATEGY FOR DENGUE PREVENTION AND CONTROL 2012–2020. 2012. This articles provides information on the epidemiology and the guidances for dengue treatment.
• Stramer SL. The potential threat to blood transfusion safety of emerging infectious disease agents. Clin Adv Hematol Oncol. 2015;13(7):420–2. The article provides evidence on an alternative route of dengue virus transmission.
Melnick JL, Curnen EC, Sabin AB. Accidental laboratory infection with human dengue virus. Proc Soc Exp Biol Med. 1948;68(1):198–200.
Nemes Z, et al. Nosocomial transmission of dengue. Emerg Infect Dis. 2004;10(10):1880–1.
Langgartner J, et al. Dengue virus infection transmitted by needle stick injury. J Inf Secur. 2002;44(4):269–70.
Punzel M, et al. Dengue virus transmission by blood stem cell donor after travel to Sri Lanka; Germany, 2013. Emerg Infect Dis. 2014;20(8):1366–9.
Rigau-Perez JG, Clark GG. Dengue activity in Puerto Rico, 1990. P R Health Sci J. 1992;11(2):65–8.
•• Perng GC. Role of bone marrow in pathogenesis of viral infections. J Bone Marrow Res. 2012;1:104. The article addresses the important role of human bone marrow in dengue pathogenesis.
•• Villar L, et al. Efficacy of a tetravalent dengue vaccine in children in Latin America. N Engl J Med. 2015;372(2):113–23. This article delivers information on the potential benefits of tetravalent dengue vaccine in trials subjects.
Halstead SB, Aguiar M. Dengue vaccines: are they safe for travelers? Travel Med Infect Dis. 2016;14(4):378–83.
• Halstead SB, Russell PK. Protective and immunological behavior of chimeric yellow fever dengue vaccine. Vaccine. 2016;34(14):1643–7. The article delivers a concern on possible adverse effects in dengue tetravalent vaccine.
Chan KW, et al. Animal models for studying dengue pathogenesis and therapy. Antivir Res. 2015;123:5–14.
Clark KB, et al. Can non-human primates serve as models for investigating dengue disease pathogenesis? Front Microbiol. 2013;4:305.
•• Lo YC, Perng GC. Novel concept on antiviral strategies to dengue. Curr Opin Virol. 2016;18:97–108. The article provides information on specificity of msoquito-borne viral biology in infected cells and new strategy in developing of mosquito-borne human viral diseases.
•• Lim SP, et al. Ten years of dengue drug discovery: progress and prospects. Antivir Res. 2013;100(2):500–19. The article updates on the lessen learned from experiences of developing antiviral drugs to dengue.
•• Kaptein SJ, Neyts J. Towards antiviral therapies for treating dengue virus infections. Curr Opin Pharmacol. 2016;30:1–7. The article reviews current status of antiviral therapies for dengue and discusses a new strategy in drug development.
Chen YL, Yokokawa F, Shi PY. The search for nucleoside/nucleotide analog inhibitors of dengue virus. Antivir Res. 2015;122:12–9.
Zandi K, et al. Novel antiviral activity of baicalein against dengue virus. BMC Complement Altern Med. 2012;12:214.
Gadhwal AK, et al. Effect of Carica papaya leaf extract capsule on platelet count in patients of dengue fever with thrombocytopenia. J Assoc Physicians India. 2016;64(6):22–6.
Faddy HM, et al. Riboflavin and ultraviolet light: impact on dengue virus infectivity. Vox Sang. 2016;111(3):235–41.
Martinez-Gutierrez M, Castellanos JE, Gallego-Gomez JC. Statins reduce dengue virus production via decreased virion assembly. Intervirology. 2011;54(4):202–16.
Martinez-Gutierrez M, et al. Lovastatin delays infection and increases survival rates in AG129 mice infected with dengue virus serotype 2. PLoS One. 2014;9(2):e87412.
Whitehorn J, et al. Lovastatin for the treatment of adult patients with dengue: a randomized, double-blind placebo-controlled trial. Clin Infect Dis. 2016;62(4):468–76.
Farias KJ, et al. Chloroquine interferes with dengue-2 virus replication in U937 cells. Microbiol Immunol. 2014;58(6):318–26.
Farias KJ, et al. Antiviral activity of chloroquine against dengue virus type 2 replication in Aotus monkeys. Viral Immunol. 2015;28(3):161–9.
Tricou V, et al. A randomized controlled trial of chloroquine for the treatment of dengue in Vietnamese adults. PLoS Negl Trop Dis. 2010;4(8):e785.
Wu SF, et al. Antiviral effects of an iminosugar derivative on flavivirus infections. J Virol. 2002;76(8):3596–604.
Sayce AC, et al. Iminosugars inhibit dengue virus production via inhibition of ER alpha-glucosidases—not glycolipid processing enzymes. PLoS Negl Trop Dis. 2016;10(3):e0004524.
Perry ST, et al. An iminosugar with potent inhibition of dengue virus infection in vivo. Antivir Res. 2013;98(1):35–43.
Low JG, et al. Efficacy and safety of celgosivir in patients with dengue fever (CELADEN): a phase 1b, randomised, double-blind, placebo-controlled, proof-of-concept trial. Lancet Infect Dis. 2014;14(8):706–15.
Sayce AC, Miller JL, Zitzmann N. Glucocorticosteroids as dengue therapeutics: resolving clinical observations with a primary human macrophage model. Clin Infect Dis. 2013;56(6):901–3.
Tam DT, et al. Effects of short-course oral corticosteroid therapy in early dengue infection in Vietnamese patients: a randomized, placebo-controlled trial. Clin Infect Dis. 2012;55(9):1216–24.
Kularatne SA, et al. Efficacy of low dose dexamethasone in severe thrombocytopenia caused by dengue fever: a placebo controlled study. Postgrad Med J. 2009;85(1008):525–9.
Tassniyom S, et al. Failure of high-dose methylprednisolone in established dengue shock syndrome: a placebo-controlled, double-blind study. Pediatrics. 1993;92(1):111–5.
Sumarmo, et al. Failure of hydrocortisone to affect outcome in dengue shock syndrome. Pediatrics. 1982;69(1):45–9.
WHO, Dengue and Dengue Hemorrhagic Fever, 2009.
WHO, Dengue: guidelines for diagnosis, treatment, prevention and control. 2009.
Min M, et al. Hydrocortisone in the management of dengue shock syndrome. Southeast Asian J Trop Med Public Health. 1975;6(4):573–9.
Pongpanich B, et al. Studies on dengue hemorrhagic fever. Clinical study: an evaluation of steroids as a treatment. J Med Assoc Thail. 1973;56(1):6–14.
Panpanich R, Sornchai P, Kanjanaratanakorn K. Corticosteroids for treating dengue shock syndrome. Cochrane Database Syst Rev. 2006;3:CD003488.
Futrakul P, et al. Hemodynamic response to high-dose methyl prednisolone and mannitol in severe dengue-shock patients unresponsive to fluid replacement. Southeast Asian J Trop Med Public Health. 1987;18(3):373–9.
Rodrigo C, et al. High dose corticosteroids in severe leptospirosis: a systematic review. Trans R Soc Trop Med Hyg. 2014;108(12):743–50.
Rajapakse S, et al. Corticosteroids in the treatment of dengue shock syndrome. Infect Drug Resist. 2014;7:137–43.
Rothwell C, et al. Cholesterol biosynthesis modulation regulates dengue viral replication. Virology. 2009;389(1–2):8–19.
Russo MW, et al. Spectrum of statin hepatotoxicity: experience of the drug-induced liver injury network. Hepatology. 2014;60(2):679–86.
Ii M, Losordo DW. Statins and the endothelium. Vasc Pharmacol. 2007;46(1):1–9.
Tong XK, Hamel E. Simvastatin restored vascular reactivity, endothelial function and reduced string vessel pathology in a mouse model of cerebrovascular disease. J Cereb Blood Flow Metab. 2015;35(3):512–20.
Whitehorn J, et al. Lovastatin for adult patients with dengue: protocol for a randomised controlled trial. Trials. 2012;13:203.
Gubler, D.J.a.G.K., ed. Dengue And Dengue Hemorrhagic Fever. First ed. 1997, CABI: Wallingford, UK.
Rolain JM, Colson P, Raoult D. Recycling of chloroquine and its hydroxyl analogue to face bacterial, fungal and viral infections in the 21st century. Int J Antimicrob Agents. 2007;30(4):297–308.
Farias KJ, Machado PR, da Fonseca BA. Chloroquine inhibits dengue virus type 2 replication in Vero cells but not in C6/36 cells. ScientificWorldJournal. 2013;2013:282734.
Gandini M, et al. Dengue virus activates membrane TRAIL relocalization and IFN-alpha production by human plasmacytoid dendritic cells in vitro and in vivo. PLoS Negl Trop Dis. 2013;7(6):e2257.
Tricou V, et al. Kinetics of viremia and NS1 antigenemia are shaped by immune status and virus serotype in adults with dengue. PLoS Negl Trop Dis. 2011;5(9):e1309.
Whitby K, et al. Castanospermine, a potent inhibitor of dengue virus infection in vitro and in vivo. J Virol. 2005;79(14):8698–706.
Sayce AC, Miller JL, Zitzmann N. Targeting a host process as an antiviral approach against dengue virus. Trends Microbiol. 2010;18(7):323–30.
Block TM, Jordan R. Iminosugars as possible broad spectrum anti hepatitis virus agents: the glucovirs and alkovirs. Antivir Chem Chemother. 2001;12(6):317–25.
Chapel C, et al. Reduction of the infectivity of hepatitis C virus pseudoparticles by incorporation of misfolded glycoproteins induced by glucosidase inhibitors. J Gen Virol. 2007;88(Pt 4):1133–43.
Chang J, et al. Combination of alpha-glucosidase inhibitor and ribavirin for the treatment of dengue virus infection in vitro and in vivo. Antivir Res. 2011;89(1):26–34.
Chang J, et al. Novel imino sugar derivatives demonstrate potent antiviral activity against flaviviruses. Antimicrob Agents Chemother. 2009;53(4):1501–8.
Qu X, et al. Inhibitors of endoplasmic reticulum alpha-glucosidases potently suppress hepatitis C virus virion assembly and release. Antimicrob Agents Chemother. 2011;55(3):1036–44.
Courageot MP, et al. Alpha-glucosidase inhibitors reduce dengue virus production by affecting the initial steps of virion morphogenesis in the endoplasmic reticulum. J Virol. 2000;74(1):564–72.
Rathore AP, et al. Celgosivir treatment misfolds dengue virus NS1 protein, induces cellular pro-survival genes and protects against lethal challenge mouse model. Antivir Res. 2011;92(3):453–60.
Schul W, et al. A dengue fever viremia model in mice shows reduction in viral replication and suppression of the inflammatory response after treatment with antiviral drugs. J Infect Dis. 2007;195(5):665–74.
Simmons CP, et al. Therapeutics for dengue: recommendations for design and conduct of early-phase clinical trials. PLoS Negl Trop Dis. 2012;6(9):e1752.
Potisopon, S., et al., The methyltransferase domain of dengue virus protein NS5 ensures efficient RNA synthesis initiation and elongation by the polymerase domain. Nucleic Acids Res, 2015.
Lescar J, et al. Towards the design of antiviral inhibitors against flaviviruses: the case for the multifunctional NS3 protein from dengue virus as a target. Antivir Res. 2008;80(2):94–101.
Kurane I, et al. Dengue-2 virus infection of human mononuclear cell lines and establishment of persistent infections. Arch Virol. 1990;110(1–2):91–101.
•• Hsu AY, et al. Infectious dengue vesicles derived from CD61+ cells in acute patient plasma exhibited a diaphanous appearance. Sci Rep. 2015;5:17990. The article addresses a distinctive dengue viral morphology in vivo as opposed to viral morphology observed in traditional tissue culture system.
Noisakran, S., et al., Cells in Dengue Virus Infection In Vivo. Advances in Virology, 2010. 2010.
Fink K, et al. Depletion of macrophages in mice results in higher dengue virus titers and highlights the role of macrophages for virus control. Eur J Immunol. 2009;39(10):2809–21.
•• Tsai JJ, C. J., Chang K, Chen PC, Liu LT, Ho TC, Tan SS, Chien YW, Lo YC, and Perng GC, Transient monocytosis subjugates low platelet count in adult dengue patients. Biomedicine Hub, 2017. The article provides information on the role of phagocytic cells in dengue pathogenesis.
Mosquera JA, et al. Ultrastructural studies on dengue virus type 2 infection of cultured human monocytes. Virol J. 2005;2:26.
Clark KB, et al. Multiploid CD61+ cells are the pre-dominant cell lineage infected during acute dengue virus infection in bone marrow. PLoS One. 2012;7(12):e52902.
Noisakran S, et al. Infection of bone marrow cells by dengue virus in vivo. Exp Hematol. 2012;40(3):250–9. e4
Noisakran, S., et al., Role of CD61(+) cells in thrombocytopenia of dengue patients. Int J Hematol, 2012.
Rothman AL. Immunity to dengue virus: a tale of original antigenic sin and tropical cytokine storms. Nat Rev Immunol. 2011;11(8):532–43.
Chastel C. Eventual role of asymptomatic cases of dengue for the introduction and spread of dengue viruses in non-endemic regions. Front Physiol. 2012;3:70.
•• Duong V, et al. Asymptomatic humans transmit dengue virus to mosquitoes. Proc Natl Acad Sci U S A. 2015;112(47):14688–93. This article provides evidence on the importance of asymptomatic in dengue outbreak.
•• Perng, G., Dengue Human Challenge Model: Is There A Risk Caveat? Persistent Infection of Dengue Virus. J Virol Emerg Dis, 2015. 1(2). The article addresses a scenario of persistent dengue virus infection in general populations.
Acknowledgments
This research was, in part, supported by the Ministry of Health and Welfare (MOHW104-CDC-C-114-114901) and Ministry of Education, Taiwan. The Aim for the Top University Project to the National Cheng Kung University (NCKU), and Ministry of Science and Technology Grant (NSC 102-2320-B-006–049 and MOST 103-2320-B-006–030-MY3) (GCP).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Yu-Chih Lo, I-Hsiu Huang, Tzu-Chuan Ho, Yu-Wen Chien, and Guey Chuen Perng declare that they have no conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Viral Infections
Rights and permissions
About this article
Cite this article
Lo, YC., Huang, IH., Ho, TC. et al. Antiviral Drugs and Other Therapeutic Options for Dengue Virus Infection. Curr Treat Options Infect Dis 9, 185–193 (2017). https://doi.org/10.1007/s40506-017-0122-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40506-017-0122-z