Abstract
Salinity is an abiotic stress which affects the morphological and physiological characteristics of plants. In order to evaluate the effects of different concentrations (0, 40, 80, 120 and 160 mM) of NaCl on some morphological and physiological parameters in Roselle, an experiment was conducted using a simple randomized design with three replications. The results showed that salt stress decreased plant height, number of leaves and shoot fresh weight. In addition, there were no differences between treatments in the fresh and dry weight of flowers, significantly, but the number of flowers decreased in the treatments of 120 and 160 mM NaCl. The total phenolic content showed a significant reduction between the control and other concentrations except for 40 mM NaCl, but the total anthocyanin content showed a significant increase in 40 mM NaCl. The results of this study indicated the possible effect of the low saline concentration on the increase of anthocyanin content, with no change on flower yield.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Roselle (Hibiscus sabdariffa L.) as a tropical wild plant with secondary metabolites, such as organic acid, anthocyanins, flavonoids and polyphenols, has some useful properties for human health. Most studies on the calyces of Roselle in human and animal trials have indicated several biological effects such as antioxidant effects (Kaur and Kapoor 2002; McKay et al. 2009), hepatoprotective effects (Amin and Hamza 2005), nephroprotective effects (Lee et al. 2009), cytotoxic (Chang et al. 2005; Lin et al. 2005; Liu et al. 2006), hypolipidemic effects (Hopkins et al. 2013), anti-obesity effects (Herranz-López et al. 2012; Chang et al. 2014), anti-diabetic effects (Peng et al. 2011) and anti-hypertensive effects (Mozaffari-Khosravi et al. 2009; Seck et al. 2017). Furthermore, these calyces are consumed as jam, syrup, hot or cold drinks and also as natural food colorants.
The phenolic and anthocyanin compounds found in Roselle are the most frequent metabolites (Borrás-Linares et al. 2015) with medicinal properties. Phenolic compounds with antioxidant activity and free radical scavenging are among the most important compounds in medicinal plants (Waśkiewicz et al. 2013; Cai et al. 2004). In this plant, anthocyanins also have antioxidant bioactivity (Wang et al. 2000; Wu et al. 2016).
Environmental stresses have been caused a significant enhancement in total phenol and anthocyanin contents by several reports (Chalker-Scott 1999; Parida and Das 2005; Eryılmaz 2006). Some studies have shown that salt stress in Roselle changed the physiological, morphological and phytochemical characteristics. Trivellini et al. (2014) reported an increase in electrolyte leakage, abscisic acid content and a reduction in flower fresh weight, anthocyanin content and chlorophyll fluorescence parameters. Galal (2017) and Gadwal and Naik (2014) showed that NaCl affected the germination rate, vegetative growth of seedlings, length of seedlings, fresh weight, dry matter of seedlings, seed viability index, chloroplast pigments as well as chlorophyll and carotenoid biosynthesis. The objective of the present research was to evaluate the effect of different salinity levels on the content of total anthocyanin and phenol, as well as on some morphological characteristics in Roselle.
Materials and methods
Plant material and cultivation conditions
Roselle seeds were immersed in 1% sodium hypochlorite for 10 min and then drained and washed with distilled water until the neutral pH was reached. The seeds were cultivated in pots containing a soil/humus mixture (4:1, v/v) and grown in a greenhouse located in Sari, Iran (latitude, 36°44′N; longitude, 53°07′E), under natural environmental conditions (≃ 25 °C). The pots were planned in a simple randomized design with 3 replicates and 6 samples per replication. Thirty days after sowing the seeds, salt stress was applied and continued for 10 days after flowering. The treatments included: 0 mM NaCl (control), 40 mM NaCl, 80 mM NaCl, 120 mM NaCl and 160 mM NaCl. Five plants were sampled randomly from each treatment at 60 days after sowing and were measured growth and phytochemical characteristics such as plant height, shoot fresh and dry weight, flower fresh and dry weight, number of flowers per plant, number of leaves, total anthocyanin content and total phenolic content.
Preparation of samples for spectrophotometric assays
Dried flowers of H. sabdariffa were powdered and then extracted with 80% ethanol by maceration at room temperature. The extracts were concentrated using a rotary for evaporation and dried using a freeze dryer.
Determination of total phenolic and total anthocyanin contents
The total anthocyanin content was specified according to the pH differential method (Sutharut and Sudarat 2012; Lee et al. 2005). The total phenolic content of the hydroalcoholic extracts was determined by the Folin Ciocalteu method at 765 nm (Nićiforović et al. 2010). The calibration curve was plotted using different amounts of gallic acid (6.25, 12.5, 25, 50, 100 µg/ml). The total phenolic and anthocyanin content was determined respectively as milligrams of gallic acid equivalents per gram of the extract and milligrams of cyanidin 3-glucoside equivalents per 100 g of extract.
Statistical analyses
For the statistical analysis was used the SPSS package program version 23.0. Data analysis was done by one-way ANOVA and compared by Duncan’s multiple range test (DMRT) at P > 0.05%.
Results and discussion
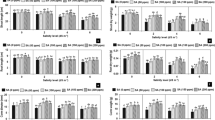
In this study, the analysis of variance showed differential responses to salinity levels (Table 1). The mean comparison of different characteristics was presented in Table 2. Salt stress caused a significant decrease in plant height in 160 mM NaCl, which was about 32% after 30 days of treatment. The number of leaves indicated a significant decrease of 26% between the control treatment and 120 and 160 mM NaCl. The shoot fresh weight showed significant differences between treatments of 0 and 40 mM NaCl with 120 and 160 mM NaCl, whereas there was no difference between treatments in shoot dry weight under salt stress. During different treatments, salt stress did not have any effect on flower fresh and dry weights. Exposure to 120 and 160 mm NaCl also resulted in 34% and 30% significant decreased respectively, compared to the control condition in the number of flowers. On the basis of the previous studies, photosynthesis (Yang et al. 2011), biomass and flower parameters (Trivellini et al. 2014) are affected by salt stress in the Hibiscus genus. The reduction rate of plant growth is a usual response to salinity, especially on leaf parameters and plant height, because of the decrease in some factors such as protein synthesis, energy and lipid metabolism and photosynthesis rate, as well as less available carbon to growth for osmotic adjustment within the cells (Parida and Das 2005; Cheeseman 1988). While studying the gene expression profiles of H. tiliaceus under salt stress condition, Yang et al. (2011) found that this plant was protected through coordination of the genes associated with the gene transcription, signaling, detoxification pathways and downstream cell transport.
The total phenolic content and total anthocyanin content were investigated as phytochemical characteristics. There was a significant decreasing rate in phenolic content between the control and other concentrations, except 40 mM NaCl. Telesiñski et al. (2008) also reported a similar response in bean leaves as a result of the accumulation of Cl-ions. However, some studies in couple and Salvia mirzayanii leaves indicate that the increase in phenolic content was as a result of the increasing phenol biosynthesis in low or moderate saline conditions and then it had a decreasing rate in high concentrations (El-Mashad and Mohamed 2012; Valifard et al. 2014). The difference in phenolic content under salt stress could be related to plant tissue, species, salinity levels, duration of salinity and the primary levels of phenol content (Waśkiewicz et al. 2013).
The total anthocyanin content showed a significant increase at 40 mM NaCl and then it reduced significantly with increasing salt concentration. Concerning the different individual anthocyanin compounds which constitute total anthocyanin, Trivellini et al. (2014) reported that the decrease in total anthocyanin was mainly due to the reduction of cyanidin 3-sophoroside in Roselle flowers under 200 mM NaCl. In this study, an increase in the rate of anthocyanin synthesis in Roselle flowers in low salt concentration could be important because it serves as a major source of antioxidant properties (Tsai et al. 2002). Anthocyanins, as a strong antioxidant molecule protecting plants in oxidative stress (Nagata et al. 2003), have been induced by different concentrations of salinity in some plants including rice (Daiponmak et al. 2010), tomato and red cabbage (Eryılmaz 2006) and the halophyte species Sesuvium portulacastrum (Slama et al. 2017).
The results of correlation coefficients between the studied characteristics in Roselle are shown in Table 3. These results showed that there was a positive and significant relationship between shoot fresh weight, shoot dry weight and number of leaves. There was a significant relevance between shoot fresh weight and anthocyanin content. Also, the phenolic content had a positive and significant relationship with plant height, the number of leaves, shoot fresh weight and number of flowers. In addition, there was no significant relevance between the phenolic and anthocyanin contents in this study. This result is the same as those reported by Borrás-Linares et al. (2015). Despite of that anthocyanins have been traced in different tissues of the plant includes flowers, fruits, leaves, roots and branches (Chalker-Scott 1999), but due to the fact that its amounts are affected in the environmental conditions and different stages of development (Steyn et al. 2002), so there is no significant correlation between anthocyanin content and other studied traits except the fresh weight of the plant. The may be due to linkage and pleiotropy (Adams 1967) or environment (Aastveit and Aastveit 1993) could justify the correlation between the traits.
Conclusion
In brief, salinity promoted several physiological and biochemical changes in Roselle including the reduction of plant height, the number of leaves, shoot fresh weight, the number of flowers per plant and total phenolic content, and also an increase of total anthocyanin content in 40 Mm NaCl. In this study, the salinity sensibility of Roselle in high salt concentration could be closely related to the decrease in total phenol and anthocyanin content as protective factors in oxidative stress. Due to the special importance of anthocyanins and phenolic compounds, as the main metabolites in the therapeutic effects of this plant and the high relationship of these values with the amount of antioxidant activity, the significant enhancement in the amount of anthocyanin in low salinity levels (40 mM NaCl) without significant reduction in the flower yield, as well as the amount of phenolic content, could be considered in studying the culture of Roselle in slightly saline soil for increasing the anthocyanin content. Further studies are necessary to investigate the molecular mechanism of the anthocyanin biosynthesis in the reaction of different saline levels in Roselle.
References
Aastveit, A., & Aastveit, K. (1993). Effects of genotype-environment interactions on genetic correlations. Theoretical and Applied Genetics, 86(8), 1007–1013.
Adams, M. (1967). Basis of yield component compensation in crop plants with special reference to the field bean, Phaseolus vulgaris 1. Crop Science, 7(5), 505–510.
Amin, A., & Hamza, A. (2005). Hepatoprotective effects of Hibiscus, Rosmarinus and Salvia on azathioprine-induced toxicity in rats. Life Sciences, 77(3), 266–278.
Borrás-Linares, I., Fernández-Arroyo, S., Arráez-Roman, D., Palmeros-Suárez, P., Del Val-Díaz, R., Andrade-Gonzáles, I., et al. (2015). Characterization of phenolic compounds, anthocyanidin, antioxidant and antimicrobial activity of 25 varieties of Mexican Roselle (Hibiscus sabdariffa). Industrial Crops and Products, 69, 385–394.
Cai, Y., Luo, Q., Sun, M., & Corke, H. (2004). Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sciences, 74(17), 2157–2184.
Chalker-Scott, L. (1999). Environmental significance of anthocyanins in plant stress responses. Photochemistry and Photobiology, 70(1), 1–9.
Chang, Y.-C., Huang, H.-P., Hsu, J.-D., Yang, S.-F., & Wang, C.-J. (2005). Hibiscus anthocyanins rich extract-induced apoptotic cell death in human promyelocytic leukemia cells. Toxicology and Applied Pharmacology, 205(3), 201–212.
Chang, H.-C., Peng, C.-H., Yeh, D.-M., Kao, E.-S., & Wang, C.-J. (2014). Hibiscus sabdariffa extract inhibits obesity and fat accumulation and improves liver steatosis in humans. Food & Function, 5(4), 734–739.
Cheeseman, J. M. (1988). Mechanisms of salinity tolerance in plants. Plant Physiology, 87(3), 547–550.
Daiponmak, W., Theerakulpisut, P., Thanonkao, P., Vanavichit, A., & Prathepha, P. (2010). Changes of anthocyanin cyanidin-3-glucoside content and antioxidant activity in Thai rice varieties under salinity stress. Science Asia, 36, 286–291.
El-Mashad, A. A. A., & Mohamed, H. I. (2012). Brassinolide alleviates salt stress and increases antioxidant activity of cowpea plants (Vigna sinensis). Protoplasma, 249(3), 625–635.
Eryılmaz, F. (2006). The relationships between salt stress and anthocyanin content in higher plants. Biotechnology and Biotechnological Equipment, 20(1), 47–52.
Gadwal, R., & Naik, G. (2014). A comparative study on the effect of salt stress on seed germination and early seedling growth of two Hibiscus species. IOSR Journal of Agriculture and Veterinary Science (IOSR-JAVS), 7(3), 90–96.
Galal, A. (2017). Physico-chemical changes in karkade (Hibiscus sabdariffa L.) seedlings responding to salt stress. Acta Biologica Hungarica, 68(1), 73–87.
Herranz-López, M., Fernández-Arroyo, S., Pérez-Sanchez, A., Barrajón-Catalán, E., Beltrán-Debón, R., Menéndez, J. A., et al. (2012). Synergism of plant-derived polyphenols in adipogenesis: Perspectives and implications. Phytomedicine, 19(3–4), 253–261.
Hopkins, A. L., Lamm, M. G., Funk, J. L., & Ritenbaugh, C. (2013). Hibiscus sabdariffa L. in the treatment of hypertension and hyperlipidemia: A comprehensive review of animal and human studies. Fitoterapia, 85, 84–94.
Kaur, C., & Kapoor, H. C. (2002). Antioxidant activity and total phenolic content of some Asian vegetables. International Journal of Food Science & Technology, 37(2), 153–161.
Lee, J., Durst, R. W., & Wrolstad, R. E. (2005). Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative study. Journal of AOAC International, 88(5), 1269–1278.
Lee, W.-C., Wang, C.-J., Chen, Y.-H., Hsu, J.-D., Cheng, S.-Y., Chen, H.-C., et al. (2009). Polyphenol extracts from Hibiscus sabdariffa Linnaeus attenuate nephropathy in experimental type 1 diabetes. Journal of Agricultural and Food Chemistry, 57(6), 2206–2210.
Lin, H. H., Huang, H. P., Huang, C. C., Chen, J. H., & Wang, C. J. (2005). Hibiscus polyphenol-rich extract induces apoptosis in human gastric carcinoma cells via p53 phosphorylation and p38 MAPK/FasL cascade pathway. Molecular Carcinogenesis, 43(2), 86–99.
Liu, J.-Y., Chen, C.-C., Wang, W.-H., Hsu, J.-D., Yang, M.-Y., & Wang, C.-J. (2006). The protective effects of Hibiscus sabdariffa extract on CCl4-induced liver fibrosis in rats. Food and Chemical Toxicology, 44(3), 336–343.
McKay, D. L., Chen, C. O., Saltzman, E., & Blumberg, J. B. (2009). Hibiscus Sabdariffa L. Tea (Tisane) lowers blood pressure in prehypertensive and mildly hypertensive adults—4. The Journal of Nutrition, 140(2), 298–303.
Mozaffari-Khosravi, H., Jalali-Khanabadi, B., Afkhami-Ardekani, M., Fatehi, F., & Noori-Shadkam, M. (2009). The effects of sour tea (Hibiscus sabdariffa) on hypertension in patients with type II diabetes. Journal of Human Hypertension, 23(1), 48–54.
Nagata, T., Todoriki, S., Masumizu, T., Suda, I., Furuta, S., Du, Z., et al. (2003). Levels of active oxygen species are controlled by ascorbic acid and anthocyanin in Arabidopsis. Journal of Agricultural and Food Chemistry, 51(10), 2992–2999.
Nićiforović, N., Mihailović, V., Mašković, P., Solujić, S., Stojković, A., & Muratspahić, D. P. (2010). Antioxidant activity of selected plant species; potential new sources of natural antioxidants. Food and Chemical Toxicology, 48(11), 3125–3130.
Parida, A. K., & Das, A. B. (2005). Salt tolerance and salinity effects on plants: A review. Ecotoxicology and Environmental Safety, 60(3), 324–349.
Peng, C.-H., Chyau, C.-C., Chan, K.-C., Chan, T.-H., Wang, C.-J., & Huang, C.-N. (2011). Hibiscus sabdariffa polyphenolic extract inhibits hyperglycemia, hyperlipidemia, and glycation-oxidative stress while improving insulin resistance. Journal of Agricultural and Food Chemistry, 59(18), 9901–9909.
Seck, S. M., Doupa, D., Dia, D. G., Diop, E. A., Ardiet, D.-L., Nogueira, R. C., et al. (2017). Clinical efficacy of African traditional medicines in hypertension: A randomized controlled trial with Combretum micranthum and Hibiscus sabdariffa. Journal of Human Hypertension, 32, 75–81.
Slama, I., M’Rabet, R., Ksouri, R., Talbi, O., Debez, A., & Abdelly, C. (2017). Effects of salt treatment on growth, lipid membrane peroxidation, polyphenol content, and antioxidant activities in leaves of Sesuvium portulacastrum L. Arid Land Research and Management, 31(4), 404–417.
Steyn, W., Wand, S., Holcroft, D., & Jacobs, G. (2002). Anthocyanins in vegetative tissues: A proposed unified function in photoprotection. New Phytologist, 155(3), 349–361.
Sutharut, J., & Sudarat, J. (2012). Total anthocyanin content and antioxidant activity of germinated colored rice. International Food Research Journal, 19(1), 215–221.
Telesiñski, A., Nowak, J., Smolik, B., Dubowska, A., & Skrzypiec, N. (2008). Effect of soil salinity on activity of antioxidant enzymes and content of ascorbic acid and phenols in bean [Phaseolus vulgaris L.] plants. Journal of Elementology, 13(3), 401–409.
Trivellini, A., Gordillo, B. N., Rodríguez-Pulido, F. J., Borghesi, E., Ferrante, A., Vernier, P., et al. (2014). Effect of salt stress in the regulation of anthocyanins and color of Hibiscus flowers by digital image analysis. Journal of Agricultural and Food Chemistry, 62(29), 6966–6974.
Tsai, P.-J., McIntosh, J., Pearce, P., Camden, B., & Jordan, B. R. (2002). Anthocyanin and antioxidant capacity in Roselle (Hibiscus sabdariffa L.) extract. Food Research International, 35(4), 351–356.
Valifard, M., Mohsenzadeh, S., Kholdebarin, B., & Rowshan, V. (2014). Effects of salt stress on volatile compounds, total phenolic content and antioxidant activities of Salvia mirzayanii. South African Journal of Botany, 93, 92–97.
Wang, C.-J., Wang, J.-M., Lin, W.-L., Chu, C.-Y., Chou, F.-P., & Tseng, T.-H. (2000). Protective effect of Hibiscus anthocyanins against tert-butyl hydroperoxide-induced hepatic toxicity in rats. Food and Chemical Toxicology, 38(5), 411–416.
Waśkiewicz, A., Muzolf-Panek, M., & Goliński, P. (2013). Phenolic content changes in plants under salt stress. Ecophysiology and responses of plants under salt stress (pp. 283–314). Berlin: Springer.
Wu, C.-H., Huang, C.-C., Hung, C.-H., Yao, F.-Y., Wang, C.-J., & Chang, Y.-C. (2016). Delphinidin-rich extracts of Hibiscus sabdariffa L. trigger mitochondria-derived autophagy and necrosis through reactive oxygen species in human breast cancer cells. Journal of Functional Foods, 25, 279–290.
Yang, G., Zhou, R., Tang, T., Chen, X., Ouyang, J., He, L., et al. (2011). Gene expression profiles in response to salt stress in Hibiscus tiliaceus. Plant Molecular Biology Reporter, 29(3), 609–617.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hashemi, A., Shahani, A. Effects of salt stress on the morphological characteristics, total phenol and total anthocyanin contents of Roselle (Hibiscus sabdariffa L.). Plant Physiol. Rep. 24, 210–214 (2019). https://doi.org/10.1007/s40502-019-00446-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40502-019-00446-y