Abstract
Implantable cardioverter-defibrillators (ICDs) are used for patients at risk of sudden cardiac death. Patients considering ICD therapy are faced with several preference-sensitive decisions. Our aim was to explore patients’ ICD decision-making experiences from the decision to implant to the consideration of deactivation at end of life. We conducted an integrative review using Whittemore and Knafl’s five steps. MEDLINE, CINAHL, PubMed, PsycINFO, and the Cochrane library were searched from 2000 to 2013. Eligible studies focused on the patient response regarding ICD decision-making. Key themes were identified and used as a framework to discuss findings through the chronological course of decisions faced by these patients. Of 354 potential citations, 25 articles were included. The trajectory of key decision points was whether or not to initiate ICD therapy, replace the battery, and deactivate at end of life. Three common themes from patients’ perspectives were the influence of the patient–practitioner consultation on knowledge uptake, patients’ decision-making preference, and their desire to live. Patients faced with ICD-related decisions often misunderstood the functionality of their ICD, or overestimated its benefit. They also expressed mixed preferences for the desire to be involved in decisions. The decisions around ICDs are particularly difficult for patients given the life and death trade off. Clinicians involved in the care of these patients could better support them by verifying their understanding and eliciting their preferences. Using a shared decision-making approach has the potential to achieve improved patient outcomes.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
When a patient is identified as a potential candidate for an implantable cardioverter-defibrillator (ICD), they embark upon a decision-making trajectory for the tenure of their implanted device. |
To achieve informed consent, patients need to understand their options (including the option to deactivate the device) and the benefits and harms of ICD therapy. |
Given the chronic nature of the condition, patients will need to revisit their decision to maintain ICD therapy with battery changes and/or as their health state changes. |
Clinicians could better support patients facing these decisions by using a shared decision-making approach. |
1 Introduction
Clinical evidence supports the use of implantable cardioverter-defibrillators (ICDs) for patients at risk of sustained ventricular arrhythmias and sudden cardiac death [1–3]. ICD therapy used for patients who have survived a cardiac arrest or a sustained ventricular arrhythmia is referred to as a secondary prevention indication [4]. Primary prevention ICDs are indicated in patients who have been identified as high risk for sudden cardiac death based on their underlying cardiac condition, but who have not yet had a sustained ventricular arrhythmia [4]. Along with its life-saving potential, ICDs are associated with procedural risks [5–7], inappropriate shocks [8], psychological risks [9, 10], and the potential for harm and suffering at end of life [11–13]. ICD therapy is considered a preference-sensitive treatment choice, as an individual can value its benefits and burdens differently than others [14]. Moreover, a person’s value for their life-saving device may diminish as their health and contextual circumstances change [15]. The goal for preference-sensitive decisions is to achieve a higher quality decision based on current scientific evidence and consistent with patients’ informed values and preferences [16, 17].
Patients with ICDs are faced with a series of decisions, including the initial implantation, battery replacement, and deactivation. Each ICD decision point warrants careful consideration to verify that patients understand their treatment options, and to ensure that their preferences are actively elicited and honored. This can be achieved by adopting a shared decision-making approach where clinicians and patients share the best available evidence when faced with health-related decisions, and where patients are supported to consider options to achieve informed preferences [18]. Shared decision-making supports the tenets of patient-centered care [19–21]. Healthcare professionals’ ethical and legal responsibilities of informing patients prior to medical interventions are explicitly clear. To achieve informed consent, a patient must have received all relevant information about a treatment, including the treatment’s nature, benefits, risks, and alternatives, and have their related queries answered prior to making a decision [22]. However, engaging informed patients in their personal medical decisions and honoring their preferences is more challenging. An increasing number of patients have shifted from being passive bystanders to active participants in the clinical decision-making process. Yet many patients feel uncertain about the best course of action and need support to be engaged in decisions about their health [23].
The aim of our integrative review was to explore patients’ decision-making experiences regarding ICDs from the decision to implant to the consideration of deactivation at end of life, in the interest of helping healthcare professionals enhance the care and support they provide.
2 Methods
We conducted an integrative review using Whittemore and Knalf’s five-step approach [24]. The five steps include the identification of a problem, literature review, data evaluation, data analysis, and the dissemination of findings. As a theoretical framework, we used the Ottawa Decision Support Framework, which asserts that patients and their support networks’ unresolved decisional needs affect decisional quality [25].
A search strategy was designed with a reference librarian and conducted using MEDLINE, CINAHL, PubMed, PsycINFO, and the Cochrane library from January 2000 to November 2013. We restricted the search to the last 13 years given the dramatic increase in ICD utilization in the early 2000s; particularly when ICDs for primary prevention became indicated. The following terms were searched: implantable defibrillators, ventricular arrhythmia, cardiac arrhythmia, patient preference, decision-making, choice behaviour, patient participation, deactivation, and end of life. We included original quantitative and qualitative research articles that directly studied the patient response regarding ICD decision-making. Patients had to be 18 years of age or older, and ICD eligibility could be for either primary or secondary prevention. Exclusion criteria were articles that did not incorporate the patient’s perspective, if they solely focused on living with or adjusting to the ICD, or if they only included other implantable devices such as pacemakers and cardiac resynchronization therapy. Case studies, reviews, discussion papers, and those not published in English were excluded. Reference lists of included articles were reviewed.
Titles and abstracts were screened for inclusion. Full texts were reviewed for eligible articles by KBL and article inclusion was verified with DDM. Study appraisal was conducted by KBL using the Mixed Methods Appraisal Tool [26, 27], which evaluates the methodological quality of each study on criteria specific to its design. For each criteria met, one point is allocated, for a maximum of four points. Scores are divided by four then multiplied by 100 to get a percentage score. For mixed methods studies, the overall quality score cannot exceed the score of its weakest component. As this review was an attempt to capture a comprehensive account of patient preferences, the tool was not used to exclude studies but rather to appraise their methodological quality.
To identify themes, qualitative data analysis was approached using a constant comparative method. Data reduction involved the extraction, coding, and grouping of data into systematic categories. Using an iterative process, data were compared to identify patterns, relationships, and themes. These were used to interpret the data, and the final themes were verified with the original articles for accuracy and confirmability.
3 Findings
3.1 Study Demographics
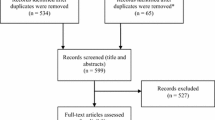
A total of 354 articles were identified. Of 68 studies reviewed as full text, 50 were excluded as they did not directly relate to the patient’s decision-making experience. Hand searching of reference lists of included articles revealed six articles. A more recent article meeting inclusion criteria was identified during the peer review process, resulting in a total of 25 included studies.
Details of the 25 included studies that addressed the patients’ perspectives surrounding ICD decision-making are described in Table 1. The number of patients with ICDs per study ranged from 8 to 3,067 patients and totaled 5,321 across all studies. There were 1,205 women, and 3,788 men. Two studies did not report sex of participants. Studies either included patients with primary prevention ICDs (seven studies), secondary prevention (one), both (six), or did not report (11). Across studies, there were various cardiac etiologies, length of time with the ICD, and total shocks received. Perspectives of family members and support systems were only sought in one retrospective study assessing end-of-life experiences for deceased patients as remembered by their next of kin.
We identified three common decision points: the decision to implant the ICD (eight studies), replace the battery (one), deactivate ICD therapies (14), and both ICD replacement and deactivation (two) (see Table 1).
3.2 Themes
Three key themes emerged from this analysis: the influence of the patient–practitioner consultation on knowledge uptake, patients’ decision-making preference, and their desire to live. Each category influenced the other, and were all mediated by factors such as the patient’s health status, social influence, and trust in their physician. These themes will be explored through the chronological course of decisions faced by patients with ICDs.
3.2.1 The Decision to Implant the Implantable Cardioverter-Defibrillators (ICDs) (Eight Studies)
The decision to implant an ICD was studied by the authors of eight studies, three of which included the perspective of patients who declined the ICD. Aside from these patients, all others had already accepted or received their ICD. None of these studies captured the process while the patient was contemplating the decision. Six studies utilized qualitative methodologies, including semi-structured interviews (three), grounded theory (two), and the combination of focus groups and individualized interviews (one). There was one mixed methods study, and one non-experimental survey study.
3.2.1.1 The Influence of the Patient–Practitioner Consultation
Hauptman et al. [28] asserted that patient–physician communication is characterized by the omission of information. Several studies have supported this belief. Some patients could not recall discussions regarding treatment alternatives to the ICD [29, 30], operative complications [31], ICD recalls [28], psychological risks [28], or prognosis [29]. The overestimation of device benefits and misunderstanding of device function were also frequently reported [32–38]. For some patients, the best source of understanding the ICD’s impact on lifestyle was the opportunity to personally experience its benefits and burdens [28, 39, 40]. In one study, some patients’ inability to recall was attributed to altered cognition, as the initial consultation occurred in the hospital, mere days or weeks following a cardiac arrest [29]. Alternatively, in a cohort of 75 patients, 79 % believed they had received sufficient information prior to implantation. In that same cohort, 83 % claimed to understand the reason for ICD implantation, yet when a subgroup of 25 were asked to describe, no patient suggested the termination of potentially life-threatening arrhythmia [31].
The length of time used to deliberate about the ICD when first offered differed amongst patients. The entire process took only minutes for some, and up to 2 years for others [30]. In particular, patients who were actively involved in decision-making required more time to reflect [41]. Another study reported that all decisions were made quickly—particularly for those in hospital—as very few subjects were offered time to contemplate the decision [28]. Some patients accepted the ICD, after reconsideration of their initial refusal [30]. This suggests that preferences can fluctuate within the same individual over time [30].
3.2.1.2 Types of Decision-Making Preferences
Patients faced with the decision to accept or decline an ICD approached their decision-making either actively or passively [30, 31, 39, 42]. ICD uptake or refusal did not appear to correlate with a person’s degree of involvement [30]. Active decision makers were engaged, sought multiple opinions and information from various sources, and assumed control [30, 39]. They carefully considered their health state, balanced their risk for sudden cardiac death against the benefits of the device, and contemplated the impact of their potential sudden death on their family [30, 42]. Despite this perceived independence, active decision makers sought reassurance from both their practitioners and family members [42]. Passive decision makers showed signs of indifference, and deferred decision-making control to others [28, 30, 31, 39, 42]. In three studies, this was actually the preferred decision-making style [29–31]. Due to the perceived complexity of the information, some patients did not believe they were adequately prepared to make the decision [29, 30, 41]. Some did not even see the decision as theirs to make [30, 42]. Reasons for this include the fact that their life had been threatened and living was the only option [29, 42], trust in God [39], and trust in their physician’s recommendation [29, 30, 39, 41].
Those who declined the ICD could also be distinguished as active or passive decision makers [30, 39, 40]. Passive decision makers either minimized their risk of sudden cardiac death [30, 39, 40], were disinterested [30], or perceived the ICD to be a purely elective option [40]. Decliners who adopted an active decision-making style carefully balanced the benefits and harms, and determined that the burdens outweighed the benefits [30, 39]. Others reported that they were pleased with the current status of their lives and preferred to avoid invasive life-prolonging procedures [30, 40]. One patient considered the benefit of a sudden death against other modes of death [39].
3.2.1.3 The Will to Live
When patients were first informed of their risk for sudden cardiac death, fear and uncertainty about the future characterized many of their responses [28, 30, 31]. The risk of sudden death and the chance for survival are powerful incentives for patients to accept an ICD. For the majority of patients faced with this reality, living was the only option [30, 39]. For many, the inconveniences of the device were worth the possibility that life could be saved. Accepting the ICD was a means to control fear and achieve a sense of security [28, 31, 42].
Four studies included patients who declined the ICD [30, 38–40]. One patient understood that an ICD would deny him the “luxury of a sudden death.” (p. 9) [38]. Another patient weighed quality versus quantity of life and preferred to live out the former. Some patients expressed that accepting the ICD equated with acceptance of their risk for sudden death, which they would rather ignore [30]. It is impossible to know the distribution of patients who accepted versus declined an ICD; however, for those with ICDs, satisfaction rates ranged from 93 to 100 % [29, 31].
3.2.2 The Decision to Replace the Battery (Three Studies)
Three studies included in this review considered patient preferences at ICD battery replacement. One survey study was concerned with the decision to replace the battery when an ICD had been recalled by the manufacturer. A semi-structured interview-based study and a survey study considered patients opinions and their hypothetical choice between replacement and non-replacement for a depleted battery.
3.2.2.1 The Influence of the Patient–Practitioner Consultation
Gibson et al. [43] focused on patients’ decision-making processes when an ICD had been recalled by the manufacturer. All 31 patients were sent a notification letter from their physician, yet only 61 % of patients remembered receiving it. The letter was supplemented by an in-clinic discussion. Although patients did not have to think back to the initial consultation to remember the implications of a recall, over 33 % of patients incorrectly recalled the risk of device failure and 71 % overestimated the rate of failure. A total of 19 % of patients chose to remove and replace their recalled device, 16 % were undecided, and 65 % chose not to replace it. The option of removal and non-replacement was not discussed. Fluur et al. [33] conducted semi-structured interviews and focused on patients’ knowledge of the ICD, and their thoughts on the battery replacement process. This study revealed similar misunderstandings of device functions and overestimation of device benefits. Some patients could not think of an occurrence when the ICD could cause harm, which could explain why the majority had never reflected on any other option but to replace the battery. In the survey study by Thylén et al. [44], 25 % of their sample had undergone battery replacement in the past. The remaining 75 % answered the survey’s battery replacement questions hypothetically. Discussions about battery replacement occurred 43 % of the time, but it was unclear if the option of non-replacement was part of their discussions. Even if ICD therapies were not needed during the first generator’s life, 79 % said they would replace the battery, 16 % could not decide, and 5 % would not. Younger patients, those with lower education levels, and those with depressive symptoms were more likely to not want replacement. If very old or seriously ill, the proportions changed slightly to 63 and 55 % wanting replacement, 27 and 34 % were unsure, and 10 and 11 %, respectively, did not want the battery replaced in these circumstances [44].
3.2.2.2 Types of Decision-Making Preferences
Little information was provided about patients’ preferred level of involvement in decision-making in these three studies. For patients affected by the recall, there was no information as to how or why that decision was made [43]. Fluur et al. [33] identified this replacement decision as “standing at crossroads,” whereby some individuals could choose an option without question, while others deliberately reflected. Active decision makers deliberately reflected about the future and their prognosis. Some reported they “had already lived on overtime” (p. 205) [33] and therefore considered the battery replacement non-imperative. Others deferred the decision to their trusted physician, reflecting a passive decision-making style [33]. These patients ignored their illness trajectory, and rather, lived 1 day at a time [33].
3.2.2.3 The Will to Live
At this decision point, patients described the ICD objectively, rather than in existential terms. Replacement of the ICD was considered a necessity, “like an auxiliary engine that required regular service”. (p. 205) [33]. Others would not live without it because they considered it their lifesaver from all causes of death [33]. Some patients understood that their health status was poor and their heart condition would continue to deteriorate, and thus deliberated replacement. Regardless of shock history, the vast majority stated they would replace the battery [33]. In some cases, patients had been advised by their physician not to replace the battery, but nevertheless proceeded with the change.
3.2.3 The Decision to Deactivate ICD Therapies (16 Studies)
Of 16 articles that explored patients’ preferences regarding the decision to deactivate their ICD, 13 measured patients’ preferences in hypothetical scenarios (such as terminal illness and decreased quality of life) and three reviewed the decisions and expressed preferences of deceased patients—or their next of kin—in the last moments before their death. There was a combination of qualitative methodologies, such as focus groups (one), interviews (one), grounded theory (one), non-experimental survey studies (11), and chart reviews (two).
3.2.3.1 The Influence of the Patient–Practitioner Consultation
Inaccuracies prevailed when patients were questioned about ICD deactivation. Raphael et al. [45] identified two barriers to deactivation discussions: the patient’s understanding of the device, and the physician’s willingness to broach the subject. Patients in four studies believed that deactivation would cause immediate death and was akin to euthanasia or assisted suicide [33, 36, 46, 47]. One of these studies found that 57 % of their patient cohort questioned the legality of ICD deactivation [47]. In five studies, the majority of participants had never discussed the possibility of deactivation with their physician, and therefore were unaware of the option [33–36, 38, 44]. However, in two recently published articles, Raphael et al. [45] and Pederson et al. [48] reported that 38 and 68 % of patients, respectively, knew that device deactivation was possible. Knowing when to broach this topic of discussion with patients has been debated. Fear of reducing hope or causing distress has often delayed this conversation. Yet, according to findings from an interview-based study, none of the 54 subjects found the topic distressing [45]. In fact, those who had previously received shocks were more likely to think deactivation should be discussed [44, 45]. Of surveyed patients, 49–52 % believed that device deactivation should be addressed prior to insertion [44, 45, 48]. Others preferred to discuss it when life expectancy decreased and when end of life neared [33, 44, 45, 48]. Thylén et al. [44] reported that 40 % of patients never wanted the physician to initiate a discussion, with 85 % stating that they would prefer to broach the topic on their own terms.
The studies concerned with patients’ preferences for deactivation at end of life revealed variations in preferred course of action. In one study of patients with malignant tumors, ICD deactivation was extensively discussed with six of eight patients [49]. None wished to abandon ICD therapy. Lewis et al. [50] conducted a post mortem chart review designed to assess the efficacy of a comprehensive care approach for 20 terminally ill patients with ICDs. All 20 patients requested deactivation of therapies, and therefore, did not experience shocks at end of life. These patients were compared with another group of deceased patients who were never classified as terminally ill. None had their shock therapy disabled, and 21 % received shocks within 30 days of their death, significantly closer to their time of death than the other group (p = 0.04). Goldstein et al. [34] reported that having a ‘do not resuscitate’ status and a higher Charlston co-morbidity score was statistically significant with having a discussion about ICD deactivation. Yet, there was no association between having a living will or healthcare proxy [34]. Of their 100-patient cohort, 27 next of kin recalled a discussion regarding ICD deactivation, six of which occurred in the last hours, and one in the last minutes prior to death.
3.2.3.2 Types of Decision-Making Preferences
In regards to deactivation decisions, some patients preferred that the physician make the decision [34, 45], while others wanted to be involved [45, 49]. For example, in a study of deceased patients with malignant tumors, all patients were relieved that they were given the option to decide for themselves [49]. However, in a focus group study, the 15 patients would only discuss hypothetical deactivation in terms of their preferred role in decision-making [34]. Patient quotes were indicative of passive decision-making styles, and expressed that the physician should make the decision. Patients who thought deactivation should not be routinely discussed feared that the information may be too confusing, and that deactivation may prematurely end life and therefore it was a decision best left to the physician [45].
3.2.3.3 The Will to Live
At initial implant, only 7–12 % of patients identified a time when they would consider the abandonment of ICD therapies [28, 51]. When ICD recipients were asked about deactivation in hypothetical terms, the majority showed great reluctance to turn off their device, even in the setting of multiple shocks [33, 37], if they were given a 1-month prognosis [37], or if dyspneic at rest [37]. Others expressed a desire to deactivate in the setting of terminal illness [32, 33, 35–37, 45, 51] or impaired quality of life [32, 48, 51], and to avoid shock-related pain [45, 48]. Thylén et al. [44] reported considerable indecisiveness, particularly from women, those with depressive symptoms, and those with worse ICD experiences, about engaging in deactivation discussions and expressing preferences.
The authors of the one study that reported the highest number of patients who would choose deactivation (71 %), had presented them with hypothetical scenarios representing various functional and cognitive limitations [32]. Another study reported that 47 % of patients with ICDs would deactivate it should their condition deteriorate [35]. Incidentally, these two groups selected their deactivation preferences immediately following an informational script of the benefits and burdens of ICD therapy, suggesting that a review of the ICD’s function preceding these discussions may be useful to enhance understanding.
When asked about deactivation at end of life, some patients chose deactivation while others did not. In a retrospective chart study of eight deceased patients with malignant tumors, none wished to abandon therapies [49], whereas another study identified that all 20 patients with terminal illness requested deactivation [50]. In a third study, out of 100 patients, there were 27 device deactivation discussions for which 21 deactivations were requested and performed in the days, hours, or minutes before death [11]. The reduction of unwanted shocks is a benefit of device deactivation at end of life, as indicated by patients who preferred ICD deactivation to be able to have a quick and natural death [33, 45, 48].
4 Discussion
The purpose of this integrative review was to explore patients’ perceptions or concerns about their decision-making experiences around ICDs in the interest of helping healthcare professionals enhance the care and support they provide. We also sought to reveal patients’ perspectives and preferences regarding ICD decision-making, and what factors influence their decision-making. The three main themes that appeared to influence patients’ decisions across studies were the influence of the patient–practitioner consultation on knowledge uptake, patients’ decision-making preferences, and a patient’s desire to live. The main findings of this review revealed a significant degree of misunderstanding and inaccurate recall of information regarding ICD function at all decision points. In terms of deactivation decisions, the majority of patients were not aware of this option. Furthermore, when they are informed, their preferences for deactivation need to be considered in the context of their health status. What a patient might opt to do when presented with a hypothetical scenario may differ what they may actually do when faced with terminal illness. Finally, our findings suggest that, for the majority of patients, their desire to live over-rules the inconveniences of the device—unless they have considered the value trade-off between a prolonged death versus a sudden one.
Patients’ perceptions and misunderstandings of the functionality of ICDs impede ICD management, particularly in discussions about decisions. In contrast to our findings, 93 % of surveyed cardiologists believe patients understand the intricacies of device functioning [52]. These rates of misunderstanding do not solely reflect patient–practitioner communication problems, but also suggest that patients have difficulty with information uptake and assimilation. This may be explained by numerous factors as outlined here. There may be significant practice variations amongst clinicians or organizational cultures that may result in patients being offered different amounts, types, or sources of information [53]. Also, patients’ decision-making preferences (whether active or passive) and health literacy levels may affect their ability to understand information. Comprehension is fundamental to informed consent [22]. Strategies are needed to verify patients’ understanding of information. Patient decision aids are effective interventions that can help patients participate in these decisions. Patient decision aids are evidence-based tools that make explicit the decision to be made, provide information on options, benefits, and harms, and help patients clarify their values in association with the options [54]. Evidence from 115 randomized controlled trials show they improve patients’ knowledge, expectations of outcomes, participation in decision-making, and improve the values–choice concordance [54]. Although decision aids are effective, none of the articles included in this review evaluated patient decision aids for patients facing decisions about ICDs. Patient decision aids for the initial decision to implant an ICD exist [55, 56]. The development and feasibility testing of a patient decision aid for primary prevention ICDs is ongoing [57].
Many patients and next of kin are unaware of the possibility to deactivate the ICD. Our findings suggest that many of these discussions only occur when the practitioner chooses to introduce the subject—if they do at all. In many cases, this introduction occurs when end of life is near, limiting the time a patient and their family have to reflect upon this complex decision. If too late, patients may be subjected to unwanted shocks. By sharing this information earlier in the trajectory, patients are granted a degree of control to re-initiate the discussion when they are prepared. The majority (84 %) of over 3,000 surveyed patients with ICDs reported that broaching the topic themselves is their preference [44]. This information will also grant them the knowledge required to establish advanced directives. Few patients with ICDs have advanced directives, but those who do are more likely to express preferences for deactivation [51]. However, only 0–8 % of these included their preference for the ICD in their advanced directives [35, 47, 51]. The use of advanced directives has been cautioned, as some individuals may dramatically revise what they want and accept as treatment when faced with the alternative of death [58]. With this in mind, when eliciting patients’ preferences for deactivation, their responses need to be considered in the context of their health status. What remains unclear is when healthy patients express their preferences for deactivation in certain scenarios, do these same preferences hold when they are terminally ill? Contextual influences are unpredictable and powerful, and so it is difficult to foresee how one may act when faced with decline and impending death.
For the most part, patients’ desire to live trumped the inconveniences of the ICD. The ICD can be described in terms of a “one-value trade off” with death. In an existential sense, the ICD is meant to save a life of quality, but when considered from a different person’s perspective or by the same person at a different time, it can be viewed with the potential for undue suffering and harm. The wide range of deactivation rates amongst the studies at end of life highlights the preference-sensitive nature of this decision. Increasingly, patients want control over their end-of-life experiences, and for ICD patients, discussing deactivation is an important and justifiable way to achieve this. The multidisciplinary comprehensive counseling approach adopted in Lewis et al. [50] may have helped terminally ill patients and their families understand and contextualize the trade-off more clearly. Practitioners need to be clear about the potential for pain and suffering from repeated firings at end of life, and that for some, a sudden death may be a better mode of dying.
There appears to be increasing interest in understanding how to integrate patient preferences into ICD decision-making, particularly given the number of articles published on this topic within the last 9 years (Fig. 1). Consideration for patient preferences emphasizes a patient-centered care approach, which is axiomatic to the delivery of quality care. Rather than idolize an increasing rate of implantation and productivity, we should begin to measure and improve the rate and quality of discussions that occur over the device’s lifetime to ensure that patients’ initial and subsequent decisions are informed and reflective of their preferences [59].
Number of articles published per decision-making point on the patients’ perspective in ICD decision-making from 2004 to 2013. This figure highlights the increase in the number of articles that have been published on the patients’ perspective on ICD decision-making in the last 10 years. ICD implantable cardioverter-defibrillator
5 Limitations
Our findings are limited by the fact that various methodologies have been included, which do not lend themselves easily to a collective analysis. However, the five-step integrative review framework [24] we have utilized facilitates such combinations, and common themes were clearly revealed. Second, given the uniqueness of ICD therapy, these findings are not transferable to other forms of implantable devices. Third, there was little emphasis on the presence of family members and support persons during consultations. Their presence is an important component of patient-centered care [60], and they can adopt an influential role in information recall and decision-making. Some limitations were inherent to the studies included in the review. Many of the studies were from single centers and retrospective in nature. A high degree of recall bias may exist as subjects may not clearly remember the details of past discussions. Only two studies provided a clear blueprint of the consent procedures, making it difficult to know which factors contributed to better understanding and recall.
6 Future Directions
The legal and ethical imperative of informed consent binds clinicians to a comprehensive discussion prior to the initial implantation. Yet, this discussion should not be limited to that sole decision-making point. Nurses, physicians, and all other healthcare professionals involved in the care of these patients need to consider ICD therapy as a trajectory of dynamic decisions; decisions that are subject to review. The decision-making points highlighted in this review are three of plenty opportune moments to reiterate the benefits and burdens, confirm understanding, and present options in a balanced and transparent manner. Strategies are needed to bridge the gap in comprehension, such as confirmation of understanding, the use of decision support tools, and the presence of support persons. A patient’s non-cardiac medical history should also be actively monitored to initiate timely deactivation discussion and allow adequate time for reflection and deliberation.
Future research should consider investigating the effect of patient decision-making styles and practitioners’ consultation approach on patients’ understanding and uptake of information. It remains unclear if a patient’s mood or mental health state has any effect on ICD decision-making, and, if so, how these factors may influence the decision. Future multicenter research could be designed to dilute the effect of practice and institutional variations. Longitudinal studies could follow patients through this decision-making journey, and assess whether initial patient perceptions can predict future decision-making. Finally, there is a paucity of information regarding decision-making processes at battery replacement. More research is required to know how best to advocate for patients and their support persons at this increasingly common decision-making point.
7 Conclusion
The decisions around ICDs are particularly difficult for patients given the life and death trade off. Whether or not patients want to make decisions related to their ICDs, informed consent requires that patients understand the benefits and harms of ICDs and are aware of alternative options. By helping patients explore their preferences, they can be better supported to participate in several elements of a shared decision-making process and achieve higher-quality decisions. Otherwise, we risk inappropriately using a treatment modality that informed patients simply do not want, or, when standing at life’s crossroads, may no longer value.
References
Bardy GH, Lee KL, Mark DB, Poole JE, Packer DL, Boineau R, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med. 2005;352:225–37.
Moss AJ, Zareba W, Hall WJ, Klein H, Wilber DJ, Cannom DS, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med. 2002;346:877–83.
Connolly SJ, Hallstrom AP, Cappato R, Schron EB, Kuck KH, Zipes DP, et al. Meta-analysis of the implantable cardioverter defibrillator secondary prevention trials. AVID, CASH and CIDS studies. Antiarrhythmics vs Implantable Defibrillator study. Cardiac Arrest Study Hamburg. Canadian Implantable Defibrillator Study. Eur Heart J. 2000;21:2071–8.
Epstein AE, Dimarco JP, Ellenbogen KA, Estes NA III, Freedman RA, Gettes LS, et al. ACC/AHA/HRS 2008 Guidelines for device-based therapy of cardiac rhythm abnormalities. Heart Rhythm. 2008;5:e1–62.
Krahn AD, Lee DS, Birnie D, Healey JS, Crystal E, Dorian P, et al. Predictors of short-term complications after implantable cardioverter-defibrillator replacement: results from the Ontario ICD Database. Circ Arrhythm Electrophysiol. 2011;4:136–42.
Nery PB, Fernandes R, Nair GM, Sumner GL, Ribas CS, Menon SM, et al. Device-related infection among patients with pacemakers and implantable defibrillators: incidence, risk factors, and consequences. J Cardiovasc Electrophysiol. 2010;21:786–90.
Poole JE, Gleva MJ, Mela T, Chung MK, Uslan DZ, Borge R, et al. Complication rates associated with pacemaker or implantable cardioverter-defibrillator generator replacements and upgrade procedures: results from the REPLACE registry. Circulation. 2010;122:1553–61.
Daubert JP, Zareba W, Cannom DS, McNitt S, Rosero SZ, Wang P, et al. Inappropriate implantable cardioverter-defibrillator shocks in MADIT II: frequency, mechanisms, predictors, and survival impact. J Am Coll Cardiol. 2008;51:1357–65.
Kapa S, Rotondi-Trevisan D, Mariano Z, Aves T, Irvine J, Dorian P, et al. Psychopathology in patients with ICDs over time: results of a prospective study. Pacing Clin Electrophysiol. 2010;33:198–208.
Sears SF Jr, Conti JB, Curtis AB, Saia TL, Foote R, Wen F. Affective distress and implantable cardioverter defibrillators: cases for psychological and behavioral interventions. Pacing Clin Electrophysiol. 1999;22:1831–4.
Goldstein NE, Lampert R, Bradley E, Lynn J, Krumholz HM. Management of implantable cardioverter defibrillators in end-of-life care. Ann Intern Med. 2004;141:835–8.
Nambisan V, Chao D. Dying and defibrillation: a shocking experience. Palliat Med. 2004;18:482–3.
Sherazi S, McNitt S, Aktas MK, Polonsky B, Shah AH, Moss AJ, et al. End-of-life care in patients with implantable cardioverter defibrillators: a MADIT-II substudy. Pacing Clin Electrophysiol. 2013;36:1273–9.
O’Connor AM, Wennberg JE, Legare F, Llewellyn-Thomas HA, Moulton BW, Sepucha KR, et al. Toward the ‘tipping point’: decision aids and informed patient choice. Health Aff (Millwood). 2007;26:716–25.
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE Jr, Drazner MH, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2013;128:e240–319.
Elwyn G, O’Connor A, Stacey D, Volk R, Edwards A, Coulter A, et al. Developing a quality criteria framework for patient decision aids: online international Delphi consensus process. BMJ. 2006;333:417.
Ratliff A, Angell M, Dow RW, Kuppermann M, Nease RF Jr, Fisher R, et al. What is a good decision? Eff Clin Pract. 1999;2:185–97.
Elwyn G, Laitner S, Coulter A, Walker E, Watson P, Thomson R. Implementing shared decision making in the NHS. BMJ. 2010;341:c5146.
Charles C, Gafni A, Whelan T. Shared decision-making in the medical encounter: what does it mean? (or it takes at least two to tango). Soc Sci Med. 1997;44:681–92.
Charles C, Gafni A, Whelan T. Decision-making in the physician-patient encounter: revisiting the shared treatment decision-making model. Soc Sci Med. 1999;49:651–61.
Laine C, Davidoff F. Patient-centered medicine. A professional evolution. JAMA. 1996;275:152–6.
Government of Ontario. Health Care Consent Act. In: S.O.1996 ed, 1996:c.2-Sched. A.
O’Connor AM, Drake ER, Wells GA, Tugwell P, Laupacis A, Elmslie T. A survey of the decision-making needs of Canadians faced with complex health decisions. Health Expect. 2003;6:97–109.
Whittemore R, Knafl K. The integrative review: updated methodology. J Adv Nurs. 2005;52:546–53.
O’Connor AM, Tugwell P, Wells GA, Elmslie T, Jolly E, Hollingworth G, et al. A decision aid for women considering hormone therapy after menopause: decision support framework and evaluation. Patient Educ Couns. 1998;33:267–79.
Pace R, Pluye P, Bartlett G, Macaulay AC, Salsberg J, Jagosh J, et al. Testing the reliability and efficiency of the pilot Mixed Methods Appraisal Tool (MMAT) for systematic mixed studies review. Int J Nurs Stud. 2012;49:47–53.
Pluye P, Gagnon MP, Griffiths F, Johnson-Lafleur J. A scoring system for appraising mixed methods research, and concomitantly appraising qualitative, quantitative and mixed methods primary studies in Mixed Studies Reviews. Int J Nurs Stud. 2009;46:529–46.
Hauptman PJ, Chibnall JT, Guild C, Armbrecht ES. Patient perceptions, physician communication, and the implantable cardioverter-defibrillator. JAMA Intern Med. 2013;173:571–7.
Agard A, Lofmark R, Edvardsson N, Ekman I. Views of patients with heart failure about their role in the decision to start implantable cardioverter defibrillator treatment: prescription rather than participation. J Med Ethics. 2007;33:514–8.
Carroll SL, Strachan PH, de Laat S, Schwartz L, Arthur HM. Patients’ decision making to accept or decline an implantable cardioverter defibrillator for primary prevention of sudden cardiac death. Health Expect. 2013;16:69–79.
Groarke J, Beirne A, Buckley U, O’Dwyer E, Sugrue D, Keelan T, et al. Deficiencies in patients’ comprehension of implantable cardioverter defibrillator therapy. Pacing Clin Electrophysiol. 2012;35:1097–102.
Dodson JA, Fried TR, Van Ness PH, Goldstein NE, Lampert R. Patient preferences for deactivation of implantable cardioverter-defibrillators. JAMA Intern Med. 2013;173:377–9.
Fluur C, Bolse K, Stromberg A, Thylén I. Patients’ experiences of the implantable cardioverter defibrillator (ICD); with a focus on battery replacement and end-of-life issues. Heart Lung. 2013;42:202–7.
Goldstein NE, Mehta D, Siddiqui S, Teitelbaum E, Zeidman J, Singson M, et al. “That’s like an act of suicide” Patients’ attitudes toward deactivation of implantable defibrillators. J Gen Intern Med. 2007;23:7–12.
Habal MV, Micevski V, Greenwood S, Delgado DH, Ross HJ. How aware of advanced care directives are heart failure patients, and are they using them? Can J Cardiol. 2011;27:376–81.
Kirkpatrick JN, Gottlieb M, Sehgal P, Patel R, Verdino RJ. Deactivation of implantable cardioverter defibrillators in terminal illness and end of life care. Am J Cardiol. 2012;109:91–4.
Stewart GC, Weintraub JR, Pratibhu PP, Semigran MJ, Camuso JM, Brooks K, et al. Patient expectations from implantable defibrillators to prevent death in heart failure. J Card Fail. 2010;16:106–13.
Strachan PH, Carroll SL, de Laat S, Schwartz L, Arthur HM. Patients’ perspectives on end-of-life issues and implantable cardioverter defibrillators. J Palliat Care. 2011;27:6–11.
Matlock DD, Nowels CT, Masoudi FA, Sauer WH, Bekelman DB, Main DS, et al. Patient and cardiologist perceptions on decision making for implantable cardioverter-defibrillators: a qualitative study. Pacing Clin Electrophysiol. 2011;34:1634–44.
Yuhas J, Mattocks K, Gravelin L, Remetz M, Foley J, Fazio R, et al. Patients’ attitudes and perceptions of implantable cardioverter-defibrillators: potential barriers to appropriate primary prophylaxis. Pacing Clin Electrophysiol. 2012;35:1179–87.
Matlock DD, Nowels CT, Bekelman DB. Patient perspectives on decision making in heart failure. J Card Fail. 2010;16:823–6.
Kantor PB, Bullinger JM, Gal CS. Patient decision-making modes and causes: a preliminary investigation. J Am Soc Inf Sci Technol. 2012;63:1339–49.
Gibson DP, Kuntz KK, Levenson JL, Ellenbogen KA. Decision-making, emotional distress, and quality of life in patients affected by the recall of their implantable cardioverter defibrillator. Europace. 2008;10:540–4.
Thylén I, Moser DK, Chung ML, Miller J, Fluur C, Stromberg A. Are ICD recipients able to foresee if they want to withdraw therapy or deactivate defibrillator shocks? IJC Heart Vessels. 2013;1:22–31.
Raphael CE, Koa-Wing M, Stain N, Wright I, Francis DP, Kanagaratnam P. Implantable cardioverter-defibrillator recipient attitudes towards device deactivation: how much do patients want to know? Pacing Clin Electrophysiol. 2011;34:1628–33.
Kapa S, Mueller PS, Hayes DL, Asirvatham SJ. Perspectives on withdrawing pacemaker and implantable cardioverter-defibrillator therapies at end of life: results of a survey of medical and legal professionals and patients. Mayo Clin Proc. 2010;85:981–90.
Kramer DB, Kesselheim AS, Salberg L, Brock DW, Maisel WH. Ethical and legal views regarding deactivation of cardiac implantable electrical devices in patients with hypertrophic cardiomyopathy. Am J Cardiol. 2011;107:1071–5.
Pederson SS, Chaitsing R, Szili-Torok T, Jordaens L, Theuns D. Patients’ perspectives on deactivation of the implantable cardioverter-defibrillator near the end of life. Am J Cardiol. 2013;111:1443–7.
Kobza R, Erne P. End-of-life decisions in ICD patients with malignant tumors. Pacing Clin Electrophysiol. 2007;30:845–9.
Lewis WR, Luebke DL, Johnson NJ, Harrington MD, Costantini O, Aulisio MP. Withdrawing implantable defibrillator shock therapy in terminally ill patients. Am J Med. 2006;119:892–6.
Berger JT, Gorski M, Cohen T. Advance health planning and treatment preferences among recipients of implantable cardioverter defibrillators: an exploratory study. J Clin Ethics. 2006;17:72–8.
Goldstein N, Bradley E, Zeidman J, Mehta D, Morrison RS. Barriers to conversations about deactivation of implantable defibrillators in seriously ill patients: results of a nationwide survey comparing cardiology specialists to primary care physicians. J Am Coll Cardiol. 2009;54:371–3.
Matlock DD, Peterson PN, Heidenreich PA, Lucas FL, Malenka DJ, Wang Y, et al. Regional variation in the use of implantable cardioverter-defibrillators for primary prevention: results from the National Cardiovascular Data Registry. Circ Cardiovasc Qual Outcomes. 2011;4:114–21.
Stacey D, Légaré F, Col NF, Bennett CL, Barry MJ, Eden KB, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2014:CD001431.
Healthwise. Heart rhythm problems: Should I get an implantable cardioverter-defibrillator (ICD)? https://www.healthwise.net/cochranedecisionaid/Content/StdDocument.aspx?DOCHWID=abk4103.
Healthwise. Heart Failure: should I get an implantable cardioverter-defibrillator? 2013. https://www.healthwise.net/cochranedecisionaid/Content/StdDocument.aspx?DOCHWID=uf9848.
Carroll SL, McGillion M, Stacey D, Healey JS, Browne G, Arthur HM, et al. Development and feasibility testing of decision support for patients who are candidates for an implantable defibrillator: a study protocol for a pilot randomized controlled trial. Trials. 2013;14:346.
Fargelin A, Schneider CE. Enough: the failure of living will. Hastings Cent Rep. 2004;34:30–42.
Bonow RO, Ganiats TG, Beam CT, Blake K, Casey DE Jr, Goodlin SJ, et al. ACCF/AHA/AMA-PCPI 2011 performance measures for adults with heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Performance Measures and the American Medical Association-Physician Consortium for Performance Improvement. Circulation. 2012;125:2382–401.
Institute of Medicine. Crossing the quality chasm: a new health system for the 21st century. National Academy Press; 2001.
Acknowledgments
The authors have no conflicts of interest to disclose. No funding was received for the completing of this study.
Disclosures
The authors have no conflicts of interest to disclose. No funding was received for the completion of this study. Dr. Matlock was supported by a career development award from the National Institutes on Aging (K23AG040696).
Author contribution
KBL—Conception and design, article selection, analysis and interpretation of data, drafting the article, final approval.
DS—Critical revision of the article for important intellectual content, final approval.
DDM—Conception and design, article selection, analysis and interpretation of data, revising the article, final approval.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lewis, K.B., Stacey, D. & Matlock, D.D. Making Decisions About Implantable Cardioverter-Defibrillators from Implantation to End of Life: An Integrative Review of Patients’ Perspectives. Patient 7, 243–260 (2014). https://doi.org/10.1007/s40271-014-0055-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40271-014-0055-2