Abstract
The aim of this paper was to review the cost-effectiveness studies of implantable cardioverter defibrillators (ICD) for primary or secondary prevention of sudden cardiac death (SCD). A systematic review of the literature published in English or Spanish was performed by electronically searching MEDLINE and MEDLINE in process, EMBASE, NHS-EED, and EconLit. Some keywords were implantable cardioverter defibrillator, heart failure, heart arrest, myocardial infarction, arrhythmias, syncope, sudden death. Selection criteria were the following: (1) full economic evaluations published after 1995, model-based studies or alongside clinical trials (2) that explored the cost-effectiveness of ICD with or without associated treatment compared with placebo or best medical treatment, (3) in adult patients for primary or secondary prevention of SCD because of ventricular arrhythmias. Studies that fulfilled these criteria were reviewed and data were extracted by two reviewers. The methodological quality of the studies was assessed and a narrative synthesis was prepared. In total, 24 studies were included: seven studies on secondary prevention and 18 studies on primary prevention. Seven studies were performed in Europe. For secondary prevention, the results showed that the ICD is considered cost-effective in patients with more risk. For primary prevention, the cost-effectiveness of ICD has been widely studied, but uncertainty about its cost-effectiveness remains. The cost-effectiveness ratios vary between studies depending on the patient characteristics, methodology, perspective, and national settings. Among the European studies, the conclusions are varied, where the ICD is considered cost-effective or not dependent on the study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The implantable cardioverter defibrillator (ICD) is a device implanted during surgery under skin in the pectoral region in order to detect and treat ventricular arrhythmias with electrical discharges and to prevent sudden cardiac death (SCD). It is indicated for both primary and secondary prevention in patients with a high risk of ventricular arrhythmias, ventricular tachycardia (VT), or ventricular fibrillation (VF), in several pathologies [1].
The increase in the number of ICDs is because of the increased indications as primary prevention in patients with heart failure, regardless of an underlying ischemic origin [2]. Behind this increase is the publication of two randomised clinical trials (RCT), the MADIT II study in 2002 [3] and the SCD-HeFT study in 2005 [4]. A certain inter-country variability has also been observed [5].
The most recent systematic reviews and clinical practice guidelines support the efficacy of ICD in some patient groups and reveal the need to correctly identify the patient to recommend ICD in primary prevention [6, 7]. The most commonly used criteria for the severity classification is left ventricular ejection fraction (LVEF) and the New York Heart Association (NYHA) classification (lower LVEF or higher NYHA indicate more risk or severity). Some studies suggest that the ICD can be cost-effective in patients with a medium–high risk of SCD in some countries [6, 8, 9]. After an initial search, we found three studies that had reviewed the cost-effectiveness of ICD including studies up until 2002 [6, 10] and 2005 [8]. However, the recent increase in the number of published economic evaluations makes an update of our knowledge necessary. Consequently, we performed a systematic review of ICD cost-effectiveness studies for primary and secondary prevention of SCD with the aim of informing decision-makers.
Methods
A report published in Spanish included the systematic review of the efficacy and cost-effectiveness of ICD until May 2010 [11]. In this article we report the systematic review of the cost-effectiveness of ICD and its update until January 2013. The methods were documented in a protocol developed by consensus with cardiologists and health technology assessment specialists.
Information sources and search
The electronic databases searched to identify the relevant economic studies were MEDLINE and MEDLINE in process (PubMed), EMBASE (Ovid), NHS Economic Evaluation Database (Centre for Reviews and Dissemination’s website), and EconLit (EBSCOhost). The original search was performed in May 2010 and updated by the end of January 2013 in all the aforementioned databases, except EMBASE and EconLit where the original search did not identify any relevant study. A documentalist designed the search strategy with the help of cardiologists and health technology assessment experts (F.J.G.G., A.G.Q., E.C.D., L.G.P.). This strategy combined Medical Subject Headings (MeSH) and text terms such as the following: ‘Defibrillators, Implantable’, implantable defibrillator, implantable cardioverter defibrillator, ‘Tachycardia, Ventricular’, ‘Death, Sudden, Cardiac’, heart failure, heart arrest, myocardial infarction, arrhythmias, syncope, sudden death; filters for economic evaluations were applied in PubMed and EMBASE. The original strategy was applied without any date or language limits. The update was limited to English or Spanish papers published from January 2010 to January 2013. Regular alerts were established on PubMed database to capture new studies. The reference lists of the articles included and the studies identified for the systematic review of effectiveness were also verified.
Selection, data extraction, and quality assessment
The selection of papers and the data extraction and quality assessment of the studies included were performed by one reviewer (economist) (L.G.P., P.P.D., R.L.) and then verified by a second reviewer (cardiologist) (A.G.Q., E.C.D., F.J.G.G.). Disagreements between reviewers were resolved by consensus or by consulting a third reviewer. Data were collated in spreadsheets designed ad hoc and piloted to obtain a standard format for each study. The extracted data were aim, design, time horizon, perspective, subject’s characteristics, description of arms, data sources, analysis, results including costs, outcomes and incremental cost-effectiveness ratios (ICER), and authors’ conclusions. Study quality was assessed by means of the Drummond and Jefferson [12] criteria for economic evaluations. The data were synthesized by narrative procedures, and the main characteristics and outcomes of each study were displayed in structured tables. The results of studies of ICD for primary and secondary prevention are reported separately. The original ICERs per life year (LY) and per quality adjusted life years (QALY) were converted to a common currency and price year, 2011 international dollars (US), according to the usual formulae in literature [13] that includes purchasing power parity and gross domestic product deflator [14].
Eligibility criteria
We selected papers published in peer-reviewed journals in English or Spanish that fulfilled the following selection criteria (structured according to the PICOS question):
Types of participant: adult patients with high risk of SCD because of ventricular arrhythmias with the following indications for ICD:
-
1.
Secondary prevention:
-
Cardiac arrest because of VT or VF.
-
Spontaneous sustained VT with syncope or hemodynamic compromise.
-
Sustained VT without syncope or cardiac arrest, with reduced LVEF (<0.35) and NYHA functional class between I and III.
-
-
2.
Primary prevention:
-
Myocardial infarct history and
-
Nonsustained VT during Holter monitoring for 24 h and sustained VT inducible at electrophysiological testing, or other criteria for arrhythmic risk.
-
Left ventricular dysfunction with LVEF < 0.40 and NYHA I–III.
-
-
Previous myocardial infarction (MI) and LVEF ≤ 0.30.
-
Non-ischemic dilated cardiomyopathy with/without high risk arrhythmia for SCD and LVEF ≤ 0.35.
-
We excluded studies that included patients with primary or secondary prevention criteria that did not report results separately. We also planned to exclude studies where >20 % of the patients were <21 years old in case we found studies with a mix of children and adults.
Types of interventions: ICD with or without associated treatment. Studies of ICD with cardiac resynchronisation were excluded.
Type of comparators: to be included the study had to compare the ICD with placebo or best medical treatment (BMT) such as anti-arrhythmic drugs, like amiodarone, or catheter ablation.
Types of outcomes: costs, results (LY and QALY) and ICERs reported separately.
Types of studies: full economic evaluations (cost-benefit analysis, cost-utility analysis, cost-effectiveness analysis, and cost-minimisation analysis). We excluded partial economic studies and economic evaluations published before 1995 (the date for the first RCT published about ICD). Economic evaluations published before 1995 were based on observational studies and assessed first generation devices. Economic evaluations based on the analysis of registries were also excluded.
Results
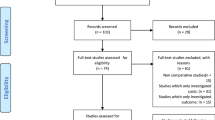
The study selection procedure (Fig. 1) identified 599 references after discarding duplicates. Their titles and abstracts were screened. Of these, 72 articles were retrieved for full review, 48 of them were excluded for different reasons (detailed reasons for exclusion are accessible upon request). Review of the list of references and the manual review yielded no further additional references, but a new publication published in January 2013 was identified due to the alert system. Subsequently, 25 papers related to 24 studies were included in the systematic review: seven papers on secondary prevention [15–21] and 19 papers on primary prevention [21–39]. One study included both types of prevention [21].
By means of the Drummond and Jefferson [12] criteria for economic evaluations, it is possible to see the evolution in methodological quality over time (see Electronic Supplementary Material). While the first study (published in 1996) did not clearly report the costs, the timeline was short (2 years) and there were no sensitivity analyses [15], the following studies have overcome most of those limitations. The research questions were generally stated clearly and the methods were suitable. However, population and BMT were generally poorly described.
Seven out of 24 studies were performed in Europe. In general, the results show that ICD is more effective and more costly than its comparators. Tables 1 and 2 describe the characteristics and results of secondary prevention; Tables 3 and 4 describe the studies for primary prevention. Given that not every study reported results in QALYs, we mainly discuss the ICERs per LY in the text. The ratios in the original currency and the ratios expressed as 2011 dollars are given in Tables 2 and 4.
Cost-effectiveness of the ICD for secondary prevention
A total of seven economic evaluations of ICD in secondary prevention, which included patients with a history of cardiac arrest because of VT or VF [15–21], were identified. Four of the studies were performed in parallel with three RCTs, and the other three studies used Markov models. One of the studies compared ICD with anti-arrhythmic treatment guided by electrophysiologic study and the remainder compared ICD with the most common medical treatment (amiodarone) (Table 1).
The first economic evaluation published was developed in parallel to an RCT and this had significant shortcomings; this study found that the anti-arrhythmic treatment guided by electrophysiologic study was a strategy dominated by the ICD; that is, it was more costly and less effective than the ICD [15]. In the first of the financial models published, Owens et al. [16] estimated ICER variables for different situations according to hypothetic reductions in total mortality. The sensitivity analyses showed that the frequency of generator replacement substantially affected the cost-effectiveness [16]. Larsen et al. [19] used data from the RCT AVID [40] whilst O’Brien et al. [17] and Sheldon et al. [18] used data from the Canadian RCT CIDS [41]. In the study by Larsen et al. [19], the ratio of patients with LVEF≤ 0.35 was slightly less than the ratio obtained for all the patients, but markedly lower than the ratio obtained for patients with LVEF >0.35. These authors found unstable estimations in the long term (20 years and lifetime) due to the uncertainty about the long-term survival of patients treated with anti-arrhythmic drugs at the time [19]. In the study by O’Brien et al. [17], the ICD was dominated by amiodarone treatment in those patients with LVEF >0.35. In the study by Sheldon et al. [18], the ICER in patients with less than two risk factors were much greater than any willingness-to-pay threshold. In this study the risk factors considered were ≥70 years, NYHA class 3, and LVEF ≤0.35. The uncertainty analysis concluded that neither the ICD cost nor the length of stay affected the results and that the probability of ICD being cost-effective (<100,000 Canadian dollars per LY) was 73 % for patients with two or more risk factors [18]. A study with a 20-year time horizon compared ICD against amiodarone in the United Kingdom [20]. This study found most favourable ICERs for patients with LVEF <0.35 and for those 70 years old and over in comparison with the younger ones [20]. The many uncertainty analyses demonstrated that the model was robust although there was uncertainty involved in modelling beyond the experience of the trials [20]. Finally, a study performed in Argentina analysed the ICD for secondary and primary prevention from three perspectives (public health, social insurance, and the private sector). This study estimated higher ICERs for secondary prevention than for primary prevention [21]. In general, the studies found that the ICD is more cost-effective in those patients with more risk factors or less LVEF (Table 2).
Cost-effectiveness of ICD for primary prevention
In 19 papers that reported 18 economic evaluations, ICD and BMT were compared in patients with heart failure [21–39]. Seven studies included only patients with ischemic cardiopathy [21–23, 25, 27, 29, 38]; the remainder included both patients with ischemic and non-ischemic cardiopathy, or they were not explicit about the type of cardiopathy. Nine of the studies adopted the social perspective according to the authors, although only one included indirect costs. Three of the studies were performed in parallel with RCTs; the remainder were financial models, and of these, 11 used Markov models. In general, the comparator selected was BMT, usually poorly reported. Except for two studies that performed a cost-benefit analysis by means of simulation of discreet events, the remainder performed cost-effectiveness analysis taking LY or QALY gained as an outcome measure. Most studies also adopted the timeline of the patient’s whole life; RCTs and cost-benefit analyses are the five studies that analysed the results of the ICD with a shorter timeline (Table 3).
The ICERs varied in these studies between US$17,000 and 294,000/LY; among the European studies the ICERs varied between US$30,511 and 75,644/LY (US$2011) (Table 4).
For the economic evaluations performed in parallel with the RCT MADIT [42] and MADIT II [3], disparate results were found. Whilst the ICER was acceptable for MADIT patients [22], the ICER was almost US$300,000/LY for MADIT II patients [29]. However, a subsequent Markov model, which considered results 8 years after patients were included in the MADIT II study, estimated an ICER of approximately US$50,000/LY [38]. The univariate sensitivity analyses in this study showed similar ICERs except for the hazard ratio of mortality for ICD therapy from year 5 to year 8 that affected notably the cost-effectiveness of the ICD [38]. An economic evaluation was also carried out in parallel with the RCT SCD-HeFT—a study that included both patients with ischemic and non-ischemic cardiopathy; a ratio <US$50,000/LY was obtained in the base-case while the bootstrap analysis concluded that the ratio was under US$100,000/LY in 99 % of samples [28].
The research group of Sanders et al. has published several versions of its Markov model on ICD [23, 26, 37]. In the first of these, published in 2001, they obtained a very high ICER in patients with LVEF between 0.31 and 0.4 [23]. In 2005 they estimated ratios for each RCT that existed to date and observed differences between them although the ratios were always below US$100,000/LY [26]. Finally, they evaluated ICD in people over 65 years and concluded that further studies are necessary for this group of patients [37]. In this last study, the 95 % confidence intervals ranged from US$20,000/LY to even ICD as a dominated alternative when data from the SCD-HeFT or DEFINITE clinical trials were used. Another model performed by independent authors in the USA compared ICD and BMT in patients with heart failure and estimated an ICER higher than US$100,000/QALY [24]. This study found that the ICER was sensitive to changes in utilities, sudden death mortality, and cost of ICD implantation, and that ICD did not prove to be cost-effective under any scenario [24].
Several financial models have also been made outside the United States. Two studies revealed very different results even though both were performed in Belgium and used similar methods [31, 32]. The reason for this difference lies both in the costs included and the effectiveness values assumed. In one, the sensitivity analyses showed that the most influential parameters were the mortality rates, the utilities and the cost of ICD replacement [31]. In the other study, the most sensitive parameter was the mortality rates, although the replacement period, age, and quality of life also had an effect on the ICERs [32]. This study found an estimated probability of 97 % that ICD was cost-effective for a willingness-to-pay of 60,000 €/QALY [32]. Ribeiro et al. [35, 36] reported their model in two papers. One only included the analysis from the perspective of the Brazilian public health system [35], whilst the other added the perspective of the private health system [36]. According to their results, the ICD, although costly, is an efficient intervention regardless of the adopted perspective. The parameters with the highest impact on results were the mortality related to the ICD, the replacement and the cost of ICD implantation [35, 36]. In Argentina, Alcaraz et al. concluded that the cost-effectiveness depends on the characteristics of patients in such a way that the ICD could be cost-effective in MADIT patients [21]. The most recent of the papers published attempts to tackle the economic evaluation of ICD in Europe assuming costs from the Netherlands [39]. According to their assumptions they found that ICD was efficient for patients with LVEF < 0.4 [39]. For a willingness to pay of 80,000 €/QALY, the ICD was cost-effective in 65 % of simulations [39].
Other analyses by risk subgroups have been performed. Two papers concurred that the implantation of ICD according to risk determined with MTWA is more cost-effective than implanting ICD for all patients [27, 34]. In one, the ICER for an ICD-for-all strategy compared with MTWA risk stratification had a 74 % probability of being below US$100,000/QALY [27]. Conversely, the other concluded that neither the ICD in all patients nor the selective ICD according to MTWA were efficient alternatives as they revealed some ICER higher than US$100,000/LY [34].
Finally, two studies performed a cost-benefit analysis considering unitary costs in France and the United Kingdom [30] and in Canada [33]. For the first two countries the health benefits gained are more than five times more valuable than the net amount that has to be invested [30]. For the Canadians, the health benefits gained are more than 20 times more valuable than the net amount to invest [33]. Both studies concluded that it is worth investing in the ICD as an alternative to amiodarone (data not included in Table 4).
Discussion
The best technologies for ICD and the tests provided by successive RCTs have added new ICD indications and have contributed to the increased number of implants [1, 2, 5], although some statistics talk of under-use and observed inter-country variability [5]. Changes in clinical practice and use of health technologies should be based on the best scientific evidence and criteria such as cost-effectiveness, and the observed variability should not occur because of differences in the indication criteria for this device. This study systematically reviewed cost-effectiveness studies in both primary and secondary prevention.
A total of seven economic evaluations for ICD published between 1996 and 2011 were identified for secondary prevention [15–21]. Clinical research for secondary prevention soon revealed that the ICD, albeit costly, is the most effective option so this was quickly incorporated into clinical practice guidelines as an effective treatment for secondary prevention [1, 2]. The ICER from studies on secondary prevention varied from US$37,000/LY to US$100,000/LY (US$ 2011); some much higher ratios were obtained in patients with LVEF > 0.35 or a few risk factors, that is, with lower risk.
The cost-effectiveness of ICD as primary prevention reveals some uncertainty. The number of economic evaluations published to date, 18 in total [21–39], reveals the interest in assessing the use of ICD in primary prevention. The ICER vary in these studies between US$17,000 and 294,000/LY (US$ 2011). This broad range could be explained by differences across studies in patient characteristics (heterogeneity of patients with heart failure of varying degrees of seriousness), methodology, perspective and national settings and costs considered for the evaluation. We could not find a unique explanation for the differences in ratios. This made the task of synthesis difficult. The case of the economic evaluations made for Belgium is paradoxical [31, 32], with similar aims but different values for the main efficacy and cost parameters and very different ICERs [43, 44]. The most recent primary prevention studies performed in Europe found different conclusions as well. While Smith et al. [39] concluded that ICD could be considered cost-effective for patients with ischaemic and non-ischaemic heart disease and LVEF ≤ 0.40 in Europe, Gandjour et al. [38] concluded that ICD cannot be considered cost-effective for primary prevention of SCD in patients with previous MI and LVEF ≤ 0.3 in Germany.
The initial studies assessed the cost-effectiveness of ICD in primary prevention in patients with ischemic cardiopathy; the most recent studies have assessed ICD in patients with heart failure without differentiating the type of cardiopathy. A recent meta-analysis concluded that the ICD reduced the relative risk of death for any reason in patients with ischemic cardiopathy and non-ischemic cardiopathy by 33 and 26 %, respectively [45]. Other studies mention the need to stratify risk and assess the implantation of ICD selectively depending on the risk and/or origin of the cardiopathy with a separate analysis of ischemic and non-ischemic cardiopathy [27, 34, 39, 43, 46]. Thus, this technology used in selected patients would lead to more efficiency within the health system.
In general, the methodological quality is acceptable although perhaps none of the studies offered sufficient information to transfer the methods to our jurisdiction [47]. Besides, most of the articles showed some uncertainty about the results. According to the sensitivity analyses reported by the authors, the elements that seemed to affect the results were the cost of the ICD implantation, frequency of replacement of the ICD, quality of life, and mortality rates, especially the rates in the long term. Of all these factors, the cost could be the most controllable. The cost of the technology and the frequency of replacement of the battery have decreased since the first devices. According to a recent study, devices implanted after 2002 have a significantly improved battery longevity as compared with devices implanted before 2002 [48]. Another study concluded that extended device longevity has an important effect on the long-term cost of device therapy [49]. So it is plausible to expect a tendency of lower prices of ICD and improved device longevity in the future. On the other hand, several studies failed to demonstrate differences in quality of life between patients with heart failure treated with ICD and patients treated with BMT [50–53]. Also, long-term studies assessing the mortality rates and studies in older patients are needed to understand the relationship between time, mortality, and cost-effectiveness [54, 55].
Previous systematic reviews [6, 8, 10] included some studies excluded from this review because they did not comply with our inclusion criteria; two of them involved studies published prior to the first clinical trials more than 20 years ago [56, 57]. Like all systematic reviews this study presents some shortcomings arising from the methodology: the possible non-inclusion of unpublished relevant studies in English or Spanish (publication and language bias), variety of populations analyzed, and the lack of direct transferability of this kind of study. Nonetheless, considerable effort has been made to locate all the relevant literature on ICD for primary and secondary prevention to present an actual study based on the best evidence.
In conclusion, the incorporation of ICD into secondary prevention appears to be accepted throughout developed countries, while the cost-effectiveness of ICD in primary prevention should be proven for different subgroups of patients and it should be used in those who can benefit from this technology, that is, high risk patients.
References
Zipes, D.P., Camm, A.J., Borggrefe, M., Buxton, M., Chaitman, B., Fromer, M., et al.: ACC/AHA/ESC 2006 guidelines for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. J. Am. Coll. Cardiol. 48, e247–e346 (2006)
Peinado, R., Torrecilla, E.G., Ormaetxe, J., Alvarez, M., Cózar, R., Alzueta, J.: Spanish Implantable Cardioverter-Defibrillator Registry. Registro Español de Desfibrilador Automático Implantable. V Informe Oficial del Grupo de Trabajo de Desfibrilador Automático Implantable de la Sociedad Española de Cardiología (2008). Rev. Esp. Cardiol. 62, 1435–1449 (2009)
Moss, A.J., Zareba, W., Hall, W.J., Klein, H., Wilber, D.J., Cannom, D.S., et al.: Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N. Engl. J. Med. 346, 877–883 (2002)
Bardy, G.H., Lee, K.L., Mark, D.B., Poole, J.E., Packer, D.L., Boineau, R., et al.: Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N. Engl. J. Med. 352, 225–237 (2005)
Camm, A.J., Nisam, S.: European utilization of the implantable defibrillator: has 10 years changed the ‘enigma’? Europace. 12, 1063–1069 (2010)
Bryant, J., Brodin, H., Loveman, E., Payne, E., Clegg, A.: The clinical and cost-effectiveness of implantable cardioverter defibrillators: a systematic review. Health Technol. Assess. 9(36), 1–150, iii (2005)
National Institute for Health and Clinical Excellence: Implantable cardioverter defibrillators for arrhythmias. Review of Technology Appraisal 11. Technology Appraisal 95. NICE. http://guidance.nice.org.uk/TA95/Guidance/pdf/English (2007). Accessed 20 Feb 2010
Ho, C., Li, H., Noorani, H., Cimon, K., Campbell, K., Tang, A., Birnie, D.: Implantable cardiac defibrillators for primary prevention of sudden cardiac death in high risk patients: a meta-analysis of clinical efficacy, and a review of cost-effectiveness and psychosocial issues [Technology report no 81]. Canadian Agency for Drugs and Technologies in Health. http://www.cadth.ca/media/pdf/332_ICD_tr_e.pdf (2007). Accessed 20 Feb 2010
García-García, F.J.: Evaluación económica de los desfibriladores automáticos implantables. Rev. Esp. Cardiol. Supl. 8, 9A–21A (2008)
Lynd, L.D., O’Brien, B.J.: Cost-effectiveness of the implantable cardioverter defibrillator: a review of current evidence. J. Cardiovasc. Electrophysiol. 14(9 Suppl.), S99–S103 (2003)
García Pérez, L., Linertová, R., Worbes Cerezo, S.M., García García, F.J., García Quintana, A., Caballero Dorta, E., Pinilla Domínguez, P., Serrano Aguilar. P.G.: Efectividad y coste-efectividad de los desfibriladores automáticos implantables en España. Plan de Calidad para el Sistema Nacional de Salud del Ministerio de Sanidad, Política Social e Igualdad. Servicio de Evaluación del Servicio Canario de la Salud; Informes de Evaluación de Tecnologías Sanitarias: SESCS Nº 2009/5 (2011)
Drummond, M.F., Jefferson, T.O.: Guidelines for authors and peer reviewers of economic submissions to the BMJ. The BMJ Economic Evaluation Working Party. BMJ 313, 275–283 (1996)
Welte, R., Feenstra, T., Jager, H., Leidl, R.: A decision chart for assessing and improving the transferability of economic evaluation results between countries. Pharmacoeconomics. 22, 857–876 (2004)
The World Economic Outlook (WEO) database [database in Internet]. [Washington?]: The International Monetary Fund. http://www.imf.org/external/pubs/ft/weo/2009/01/weodata/index.aspx (2009). Accessed 14 May 2012
Wever, E.F., Hauer, R.N., Schrijvers, G., van Capelle, F.J., Tijssen, J.G., Crijns, H.J., et al.: Cost-effectiveness of implantable defibrillator as first-choice therapy versus electrophysiologically guided, tiered strategy in postinfarct sudden death survivors. A randomized study. Circulation 93, 489–496 (1996)
Owens, D.K., Sanders, G.D., Harris, R.A., McDonald, K.M., Heidenreich, P.A., Dembitzer, A.D., et al.: Cost-effectiveness of implantable cardioverter defibrillators relative to amiodarone for prevention of sudden cardiac death. Ann. Intern. Med. 126, 1–12 (1997)
O’Brien, B., Connolly, S., Goeree, R., Blackhouse, G., Willan, A., Yee, R., et al.: Cost-effectiveness of the implantable cardioverter defibrillator study (CIDS). Circulation 103, 1416–1421 (2001)
Sheldon, R., O’Brien, B.J., Blackhouse, G., Goeree, R., Mitchell, B., Klein, G., et al.: Effect of clinical risk stratification on cost-effectiveness of the implantable cardioverter-defibrillator: the Canadian implantable defibrillator study. Circulation 104, 1622–1626 (2001)
Larsen, G., Hallstrom, A., McAnulty, J., Pinski, S., Olarte, A., Sullivan, S., et al.: Cost-effectiveness of the implantable cardioverter-defibrillator versus antiarrhythmic drugs in survivors of serious ventricular tachyarrhythmias. Results of the antiarrhythmics versus implantable defibrillators (AVID) economic analysis substudy. Circulation 105, 2049–2057 (2002)
Buxton, M., Caine, N., Chase, D., Connelly, D., Grace, A., Jackson, C., et al.: A review of the evidence on the effects and costs of implantable cardioverter defibrillator therapy in different patient groups, and modelling of cost-effectiveness and cost-utility for these groups in a UK context. Health Technol. Assess. 10(27), iii–iv, ix–xi, 1–164 (2006)
Alcaraz, A., Gonzalez-Zuelgaray, J., Augustovski, F.: Cost effectiveness of implantable cardioverter-defibrillators for patients who are at risk for sudden death in Argentina. Value Health. 14(5 Suppl. 1), S33–S38 (2011)
Mushlin, A., Hall, J., Zwanziger, J., Gajary, E., Andrews, M., Marron, R., et al.: The cost-effectiveness of automatic implantable cardiac defibrillators: results from MADIT. Circulation 97, 2129–2135 (1998)
Sanders, G.D., Hlatky, M.A., Every, N.R., McDonald, K.M., Heidenreich, P.A., Parsons, L.S., et al.: Potential cost-effectiveness of prophylactic use of the implantable cardioverter defibrillator or amiodarone after myocardial infarction. Ann. Intern. Med. 135, 870–883 (2001)
Chen, L., Hay, J.W.: Cost-effectiveness of primary implanted cardioverter defibrillator for sudden death prevention in congestive heart failure. Cardiovasc. Drugs Ther. 18, 161–170 (2004)
Al Khatib, S.M., Anstrom, K.J., Eisenstein, E.L., Peterson, E.D., Jollis, J.G., Mark, D.B., et al.: Clinical and economic implications of the Multicenter Automatic Defibrillator Implantation Trial-II. Ann. Intern. Med. 142, 593–600 (2005)
Sanders, G.D., Hlatky, M.A., Owens, D.K.: Cost-effectiveness of implantable cardioverter-defibrillators. N. Engl. J. Med. 353, 1471–1480 (2005)
Chan, P.S., Stein, K., Chow, T., Fendrick, M., Bigger, J.T., Vijan, S.: Cost-effectiveness of a microvolt T-wave alternans screening strategy for implantable cardioverter-defibrillator placement in the MADIT-II-eligible population. J. Am. Coll. Cardiol. 48, 112–121 (2006)
Mark, D., Nelson, C., Anstrom, K., Al-Khatib, S., Tsiatis, A., Cowper, P., et al.: Cost-effectiveness of defibrillator therapy or amiodarone in chronic stable heart failure: results from the sudden cardiac death in heart failure trial (SCD-HeFT). Circulation 114, 135–142 (2006)
Zwanziger, J., Hall, J., Dick, A., Zhao, H., Mushlin, A., Marron, R., et al.: The cost effectiveness of implantable cardioverter-defibrillators. Results from the multicenter automatic defibrillator implantation trial (MADIT)-II. J. Am. Coll. Cardiol. 47, 2310–2318 (2006)
Caro, J.J., Ward, A., Deniz, H.B., O’Brien, J.A., Ehreth, J.L.: Cost-benefit analysis of preventing sudden cardiac deaths with an implantable cardioverter defibrillator versus amiodarone. Value Health. 10, 13–22 (2007)
Neyt, M., Thiry, N., Ramaekers, D., Van Brabandt, H.: Cost effectiveness of implantable cardioverter-defibrillators for primary prevention in a Belgian context. Appl. Health Econ. Health Policy. 6, 67–80 (2008)
Cowie, M.R., Marshall, D., Drummond, M., Ferko, N., Maschio, M., Ekman, M., et al.: Lifetime cost-effectiveness of prophylactic implantation of a cardioverter defibrillator in patients with reduced left ventricular systolic function: results of Markov modelling in a European population. Europace. 11, 716–726 (2009)
Deniz, H.B., Ward, A., Jaime, C.J., Alvarez, P., Sadri, H.: Cost-benefit analysis of primary prevention of sudden cardiac death with an implantable cardioverter defibrillator versus amiodarone in Canada. Curr. Med. Res. Opin. 25, 617–626 (2009)
Filion, K.B., Xie, X., van der Avoort, C.J., Dendukuri, N., Brophy, J.M.: Microvolt T-wave alternans and the selective use of implantable cardioverter defibrillators for primary prevention: a cost-effectiveness study. Int. J. Technol. Assess. Health Care 25, 151–160 (2009)
Ribeiro, R.A., Stella, S.F., Camey, S.A., Zimerman, L.I., Pimentel, M., Rohde, L.E., et al.: Cost-effectiveness of implantable cardioverter-defibrillators in Brazil: primary prevention analysis in the public sector. Value Health. 13, 160–168 (2010)
Ribeiro, R.A., Stella, S.F., Zimerman, L.I., Pimentel, M., Rohde, L.E., Polanczyk, C.A.: Cost-effectiveness of implantable cardioverter defibrillators in Brazil in the public and private sectors. Arq. Bras. Cardiol. 95, 577–586 (2010)
Sanders, G.D., Kong, M.H., Al Khatib, S.M., Peterson, E.D.: Cost-effectiveness of implantable cardioverter defibrillators in patients > or = 65 years of age. Am. Heart J. 160, 122–131 (2010)
Gandjour, A., Holler, A., Adarkwah, C.C.: Cost-effectiveness of implantable defibrillators after myocardial infarction based on 8-year follow-up data (MADIT II). Value Health. 14, 812–817 (2011)
Smith, T., Jordaens, L., Theuns, D.A., van Dessel, P.F., Wilde, A.A., Hunink, M.G.: The cost-effectiveness of primary prophylactic implantable defibrillator therapy in patients with ischaemic or non-ischaemic heart disease: a European analysis. Eur. Heart J. 34, 211–219 (2013)
Antiarrhythmics Versus Implantable Defibrillators (AVID) Investigators: A comparison of antiarrhythmic-drug therapy with implantable defibrillators in patients resuscitated from near-fatal ventricular arrhythmias. N. Engl. J. Med. 337, 1576–1583 (1997)
Connolly, S.J., Gent, M., Roberts, R.S., Dorian, P., Roy, D., Sheldon, R.S., et al.: Canadian Implantable Defibrillator Study (CIDS): a randomized trial of the implantable cardioverter defibrillator against amiodarone. Circulation 101, 1297–1302 (2000)
Moss, A.J., Hall, W.J., Cannom, D.S., Daubert, J.P., Higgins, S.L., Klein, H., et al.: Multicenter Automatic Defibrillator Implantation Trial Investigators. Improved survival with an implanted defibrillator in patients with coronary disease at high risk for ventricular arrhythmia. N. Engl. J. Med. 335, 1933–1940 (1996)
Van Brabandt, H., Neyt, M.: Pitfalls in health-economic evaluations: the case of cost-effectiveness of prophylactic implantable cardioverter-defibrillator therapy in Belgium. Europace. 11, 1571–1573 (2009)
Cowie, M.R.: Response to editorial: Pitfalls in economic analysis. Europace. 12, 1044-1046; author reply 1046-1047 (2010)
Theuns, D.A., Smith, T., Hunink, M.G., Bardy, G.H., Jordaens, L.: Effectiveness of prophylactic implantation of cardioverter-defibrillators without cardiac resynchronization therapy in patients with ischaemic or non-ischaemic heart disease: a systematic review and meta-analysis. Europace. 12, 1564–1570 (2010)
Ezekowitz, J.A., Rowe, B.H., Dryden, D.M., Hooton, N., Vandermeer, B., Spooner, C., et al.: Systematic review: implantable cardioverter defibrillators for adults with left ventricular systolic dysfunction. Ann. Intern. Med. 147, 251–262 (2007)
Drummond, M., Barbieri, M., Cook, J., Glick, H.A., Lis, J., Malik, F., et al.: Transferability of economic evaluations across jurisdictions: ISPOR Good Research Practices Task Force report. Value. Health 12, 409–418 (2009)
Thijssen, J., Borleffs, C.J., van Rees, J.B., Man, S., de Bie, M.K., Venlet, J., et al.: Implantable cardioverter-defibrillator longevity under clinical circumstances: an analysis according to device type, generation, and manufacturer. Heart Rhythm. 9, 513–519 (2012)
Boriani, G., Braunschweig, F., Deharo, J.C., Leyva, F., Lubinski, A., Lazzaro, C.: Impact of extending device longevity on the long-term costs of implantable cardioverter-defibrillator therapy: a modelling study with a 15-year time horizon. Europace. 15, 1453–1462 (2013)
Huang, D.T., Sesselberg, H.W., McNitt, S., Noyes, K., Andrews, M.L., Hall, W.J., et al.: Improved survival associated with prophylactic implantable defibrillators in elderly patients with prior myocardial infarction and depressed ventricular function: a MADIT-II substudy. J. Cardiovasc. Electrophysiol. 18, 833–838 (2007)
Mark, D.B., Anstrom, K.J., Sun, J.L., Clapp-Channing, N.E., Tsiatis, A.A., Davidson-Ray, L., et al.: Quality of life with defibrillator therapy or amiodarone in heart failure. N. Engl. J. Med. 359, 999–1008 (2008)
Noyes, K., Corona, E., Zwanziger, J., Hall, W.J., Zhao, H., Wang, H., et al.: Health-related quality of life consequences of implantable cardioverter defibrillators: results from MADIT II. Med. Care 45, 377–385 (2007)
Strickberger, S.A., Hummel, J.D., Bartlett, T.G., Frumin, H.I., Schuger, C.D., Beau, S.L., et al.: Amiodarone versus implantable cardioverter-defibrillator: randomized nonischemic dilated cardiomyopathy and asymptomatic nonsustained ventricular tachycardia—AMIOVIRT. J. Am. Coll. Cardiol. 41, 1707–1712 (2003)
Santangeli, P., Di Biase, L., Dello, R.A., Casella, M., Bartoletti, S., Santarelli, P., et al.: Meta-analysis: age and effectiveness of prophylactic implantable cardioverter-defibrillators. Ann. Intern. Med. 153, 592–599 (2010)
Kong, M.H., Al-Khatib, S.M., Sanders, G.D., Hasselblad, V., Peterson, E.D.: Use of implantable cardioverter-defibrillators for primary prevention in older patients: a systematic literature review and meta-analysis. Cardiol. J. 18, 503–514 (2011)
Kuppermann, M., Luce, B.R., McGovern, B., Podrid, P.J., Bigger Jr, J.T., Ruskin, J.N.: An analysis of the cost effectiveness of the implantable defibrillator. Circulation 81, 91–100 (1990)
Kupersmith, J., Hogan, A., Guerrero, P., Gardiner, J., Mellits, E.D., Baumgardner, R., et al.: Evaluating and improving the cost-effectiveness of the implantable cardioverter-defibrillator. Am. Heart J. 130(3 Pt 1), 507–515 (1995)
Acknowledgments
The authors would like to thank Melany Worbes-Cerezo, Pedro Serrano-Aguilar, and Andrés Fernández-Ramos. The authors would also like to thank Jason Willis-Lee for copyediting support. This work was partly funded by Instituto de Salud Carlos III and Fundación Canaria de Investigación y Salud (FUNCIS), Spain.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
García-Pérez, L., Pinilla-Domínguez, P., García-Quintana, A. et al. Economic evaluations of implantable cardioverter defibrillators: a systematic review. Eur J Health Econ 16, 879–893 (2015). https://doi.org/10.1007/s10198-014-0637-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10198-014-0637-x
Keywords
- Systematic review
- Cost-effectiveness
- Implantable cardioverter defibrillator
- Prevention
- Sudden cardiac death
- Heart failure