Abstract
Background and Objective
As a β-adrenoceptor antagonist (β-blocker), esmolol can reduce cardiac output and the phosphodiesterase III inhibitor milrinone has been shown to improve heart contractility in patients with septic shock. This study was performed to assess the effects of esmolol combined with milrinone in patients with severe sepsis.
Methods
This prospective randomized study was conducted in patients with severe sepsis in the intensive care unit of the Jiangxi Provincial People’s Hospital (Nanchang, Jiangsu, China) between June 2013 and June 2014. Patients were randomly divided into control (C), milrinone (M), and milrinone–esmolol (ME) groups. The primary outcome was the rate of controlling the heart rate (HR) to achieve target levels. Secondary outcomes included the 28-day survival rate and changes in hemodynamic variables, organ function variables, myocardial injury markers, and the serum levels of proinflammatory factors.
Result
A total of 90 patients with severe sepsis were included in this study (30 per group). The HR in the ME group was lower than in the M and C groups after 12 h. The rate of successful HR control during the first 96 h was significantly higher in the ME group (60.0 vs. 33.3 % in the M group, vs. 26.7 % in the C group). Also, patients in the ME group had higher 28-day overall survival compared with the M (Log rank statistic = 5.452; P = 0.020) and C groups (Log rank statistic = 10.206; P = 0.001). Additionally, several variables showed significant improvement in the ME group 96 h after treatment compared with the M and C groups (P < 0.05).
Conclusion
Combination therapy with milrinone and esmolol could improve cardiac function and the 28-day survival rate in patients with severe sepsis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Combination therapy with milrinone and esmolol could effectively improve cardiac function in patients with severe sepsis. |
Combination therapy with milrinone and esmolol could reduce 28-day mortality in patients with severe sepsis. |
All patients tolerated the treatment of combination therapy with milrinone and esmolol well. |
1 Introduction
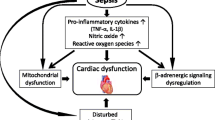
Sepsis is defined as a systemic inflammatory response syndrome (SIRS) that is caused by an infective organism with the consequence of severe systemic organ dysfunction and septic shock, eventually leading to death. Severe sepsis, septic shock, and their complications are major healthcare problems that affect millions of people around the world each year, killing one in four [1–5]. Severe sepsis is a complex syndrome, with dysfunction of one or more organ, particularly heart dysfunction, and is characterized by hemodynamic disorder [6, 7]. Besides, as reported by Vieillard-Baron [8] in 2011, the depression of intrinsic left ventricular contractility is constant in patients with septic shock, and a hyperkinetic state induced by severe sepsis is indicative of a profound and persistent vasoplegia associated with a high mortality rate.
Septic cardiomyopathy is a syndrome in patients with sepsis in which there is myocardial depression without structural changes, which is often associated with a decreased ejection fraction and increased end-diastolic volume index [6, 9, 10], a typical symptom diagnosed by doctors through echocardiography. A number of factors could induce septic cardiomyopathy, such as bacterial toxins, inflammatory factors, nitric oxide, and oxidative stress [11]. However, the possible mechanism for development of septic cardiomyopathy is still unclear. Recently, Kume et al. [12] and Wittstein et al. [13] discovered that the increased cardiac catecholamines due to the exaggerated sympathetic activation of patients with sepsis could trigger myocardium stunning, leading to the development of septic cardiomyopathy. On the other hand, patients with severe septic cardiomyopathy often suffer from tachycardia to compensate for the low cardiac output. Tachycardia would increase the myocardial oxygen demand and cardiac load as well as shorten the ventricular diastolic time, thus leading to insufficient coronary perfusion [14].
As a rapid-effect intravenous β-adrenoceptor antagonist (β-blocker), esmolol can reduce cardiac output in proportion to the percentage decreases in heart rate (HR) without adversely affecting oxygen [15], while as a phosphodiesterase III inhibitor, milrinone has been shown to improve heart contractility in patients with septic shock [16]. Therefore, we hypothesized that inhibiting the activation of sympathetic nerves by β-blockers could reduce the releasing of catecholamines and consequently improve the symptoms and prognosis of severe sepsis when combined with milrinone. In our study, we treated severe sepsis or septic shock patients with a combination therapy of milrinone and esmolol to assess whether HR could be controlled to the target range and whether the 28-day overall survival could be improved, by comparing the combination with milrinone alone and usual care. Additionally, changes in hemodynamic and blood gas measurements and organ function variables, as well as levels of proinflammatory factors and myocardial injury markers, were tested.
2 Methods
2.1 Design
This prospective, single-center, randomized, non-blinded study was conducted in the intensive care unit (ICU) of the Jiangxi Provincial People’s Hospital (Nanchang, Jiangsu, China) between June 2013 and June 2014. The hospital’s Ethics Committee approved this study and each patient or their caregivers signed a written informed consent after an adequate explanation of the study.
2.2 Patient
To enroll in this study, patients had to (1) be ≥18 years; (2) have suspected or confirmed infection, as indicated by the blood culture; (3) have at least two of the systemic inflammation response syndrome criteria, i.e., HR >90 beats/min, respiratory rate >20 breaths/min, temperature >38 or <36 °C, white blood count >12,000 or <4000 cells/mm3; and (4) have a HR ≥95 beats/min after early goal-directed therapy (EGDT). Patients were excluded if they had (1) a history of bronchial asthma history; (2) sinus bradycardia, defined as an HR below 60 beats/min with a normal sequence of cardiac activation; (3) second- or third-degree atrioventricular block; or (4) a history of allergic reaction to milrinone or esmolol.
2.3 Outcomes
The primary outcome was the reduction of HR to lower than the predefined threshold of 95 beats/min and maintenance within the target range between 75 and 94 beats/min during the first 96 h after a different intervention. The secondary outcome included the 28-day survival rate, which was recorded during hospitalization or re-admission. Other secondary outcomes included norepinephrine requirements, changes in hemodynamic variables, organ function variables, myocardial injury markers, the level of proinflammatory factors in the serum, duration in the ICU and in hospital, as well as adverse events caused by drug treatment up to 28 days after enrolment.
2.4 Sample Estimation
Power analysis and sample size (PASS) 11.0 software (NCSS Statistical Software, Kaysville, UT, USA) was used to calculate the sample size for the present study. To provide 80 % power with a two-sided 5 % significance level to detect a 20 beats/min reduction in HR, assuming a standard deviation (SD) of 25 beats/min, a minimum of 27 patients were to be enrolled in each treatment group. Given a dropout rate of 10 %, 30 patients per groups (90 total) were necessary.
2.5 Randomization
Patients were randomly assigned by a computer-based random numbers generator in a ratio of 1:1:1 to three different groups: control (C) group, milrinone (M) group, and milrinone–esmolol (ME) group. The Rv.Uniform function in SPSS® 17.0 analysis software (SPSS Inc., Chicago, IL, USA) was adapted to generate random numbers.
2.6 Procedures
Systemic hemodynamic data were monitored by pulse-indicated continuous cardiac output (PiCCO) after subclavian vein and femoral artery catheterization. EGDT was performed on all patients to keep optimal cardiac preload and hematocrit ≥0.30 according to the international guidelines for management of severe sepsis and septic shock [17]. During EGDT, patients received adequate crystalloid-based volume resuscitation or red blood cell transfusion. In addition, all patients were given norepinephrine to maintain a mean arterial pressure (MAP) ≥65 mmHg. Patients in the M group were additionally treated with a continuous intravenous infusion of milrinone that commenced with a loading dosage of 30 μg/kg and was maintained at 0.375–0.5 μg/kg/min. Besides the milrinone treatment, patients in the ME group received esmolol through continuous intravenous infusion.
2.7 Data Collection
The characteristics for all patients, including age, sex, severity of disease, and source of infection, were collected on ICU admission. Severity of disease was assessed by Acute Physiology and Chronic Health Evaluation II (APACHE II) score. The vital signs of patients before different treatments, including body temperature, HR, respiratory rate, and white blood count, were also recorded in the ICU. Mortality was recorded in the ICU and on day 28. In addition, ICU-free days, hospital-free days, as well as adverse events occurring after treatment initiation up to day 28 were also recorded.
Clinical data were recorded at baseline (before initiation of treatment) and 12, 24, 48, 72, and 96 h after different drug treatments for hemodynamic parameters including MAP, central venous pressure (CVP), HR, Cardiac Index (CI) and Stroke Volume Index (SVI); and for organ function variables including blood lactic acid, creatinine, bilirubin, aspartate aminotransferase, alanine aminotransferase (ALAT), urine output, the ratio of the arterial oxygen partial pressure to the fraction of inspired oxygen (PaO2/FiO2), and for norepinephrine dosages.
2.8 Blood Samples
Blood samples were collected in 5 mL tubes immediately at baseline and 24, 48, 72, and 96 h after treatment, centrifuged, and serum was preserved at –80 °C until analysis. The serum level of proinflammatory factors including tumor necrosis factor (TNF)-α and interleukin (IL)-6, and markers of myocardial injury including creatine kinase-MB (CK-MB), troponin I (TnI), and brain natriuretic peptide (BNP) were both detected by enzyme-linked immunosorbent assay (ELISA; Alpco, Windham, NH, USA). The type of ELISA kit used was as follows: TNF-α ELISA, 45-TNFHU-E01; IL-6 ELISA, 45-IL6HU-E01; CK-MB, 25-CKMHU-E01; TnI ELISA, 25-TR1HU-E01; and BNP ELISA, 04-BI-20852W. The serum level of high mobility group box-1 (HMGB-1) was also detected by an ELISA kit (ST51011; IBL, Toronto, ON, Canada).
2.9 Statistical Analysis
SPSS® version 17.0 was used for randomization and statistical analyses. Continuous variables were presented as mean ± standard deviation (S.D.) and non-parametric continuous variables were presented as median (minimum to maximum). Categorical variables were presented as number (%). One-way analysis of variance (ANOVA) followed by Duncan’s Multiple Range test were used to compare the differences in continuous variables among three different groups at baseline and other observation timepoints. Wilcoxon tests were performed to compare the differences in non-parametric continuous variables. Pearson’s Chi-squared (χ 2) test was used to compare enumeration data of patients among three groups (if expected values were below 5, Fisher’s exact test was used). Also, 28-day overall survival rates were compared using Kaplan–Meier survival curves followed by a Log rank test. P values <0.05 were regarded as statistically significant.
3 Results
3.1 Patients
A total of 182 patients were screened. Among them, 92 were excluded because of an HR <95 beats/min after EGDT (n = 77), β-blocker therapy prior to the present study (n = 6), sinus bradycardia or atrioventricular block (n = 5), and rejection to participate (n = 4). As a result, 90 patients were enrolled in this study and randomly assigned to the C, M, and ME groups in a 1:1:1 ratio (Fig. 1). All enrolled subjects finished the whole trial and thus were included in the analyses. Patients among the groups were matched for basic characteristics including age, sex, APACHE II score, vital signs, and source of infection (Table 1). Blood cultures were obtained from all patients in the three treatment groups; 16 (53.3 %) patients had positive blood cultures in the C group, 12 patients (40 %) had positive blood cultures in the M group, and 15 (50 %) patients had positive blood cultures in the ME group. All sepsis patients received antibiotics.
3.2 Primary Outcome
The 96-h successful HR control rates for the C, M, and ME groups were 43.3, 53.3, and 100 %, respectively. There was a significant statistical difference between the ME and M groups (P < 0.001), as well as between the M and C groups (P < 0.001).
3.3 Secondary Outcomes
3.3.1 28-Day Overall Survival
A significant difference in 28-day overall survival was detected among the C, M, and ME groups [Figs. 2, 3; Log rank statistic = 10.528; degrees of freedom (df) = 2; P = 0.005]. Patients in the M group had a similar overall survival to those in the C group (Log rank statistic = 0.976; P = 0.323). However, overall survival was higher in the ME group than in the M (Log rank statistic = 5.452; P = 0.020) and C (Log rank statistic = 10.206; P = 0.001) groups.
Comparison of rate of 28-day survival and heart rate control of patients among the three treatment groups. Pearson’s Chi-squared (χ 2) test was used to compare enumeration data of patients among the three groups (if expected values were below 5, Fisher’s exact test was used). Data are presented as number/total number (100 %). C group control group, HR heart rate, M group milrinone group, ME group milrinone–esmolol group, *P < 0.05 compared to the C group, # P < 0.05 compared to the M group
3.3.2 Hemodynamic Variables
Before treatment, patients in the three study groups had comparable hemodynamic values (Table 2). No significant difference in MAP and CVP was found after treatment. However, 12 h later, the HRs of patients in the ME group were significantly lower than those of patients in the C and M groups (P < 0.05) (Table 2). Meanwhile, the CI and SVI levels of patients in the M and ME groups were statistically higher than those of the C group at 12 h after treatment (P < 0.05) (Table 2). Moreover, there was a significant decrease in the norepinephrine dosage for patients in the ME and M groups when compared with that for patients in the C group (P < 0.05) 72 h after treatment (P < 0.05).
3.3.3 Organ Function Variables
The results indicating liver, kidney, as well as arterial blood gas functions of patients from the three treatment groups are shown in Table 3. Baseline disturbances of these functions were positively changed after 96 h in the ME group and there was a significant improvement of liver and renal function of patients in ME group when compared with that of patients in the C group after 96 h (P < 0.05 for blood lactic acid, creatinine, ALAT, and urine output). However, all variables similarly improved between M and ME group with no significant difference.
3.3.4 Proinflammatory Factors and Myocardial Injury Markers
There were no significant differences in TNF-α, IL-6, HMGB-1, CK-MB, TnI, and BNP levels among the three groups before treatment, but these measures were significantly lower in the ME group than in C and M groups after 24 h of treatment (P < 0.05) (Tables 4, 5).
3.3.5 Duration in the Intensive Care Unit and Hospital
There was no significant difference in both ICU-free days and hospital-free days among the three groups (P > 0.05; Fig. 4).
Comparison of intensive care unit-free days and hospital-free days in patients among the three treatment groups. Wilcoxon tests were performed to compare the difference. Data are presented as median (minimum–maximum). C group control group, ICU intensive care unit, M group milrinone group, ME group milrinone–esmolol group
3.3.6 Adverse Effects
Among the patients in the ME group, two patients showed asymptomatic bradycardia. No significant arrhythmia was detected in any group. There was no significant difference in the adverse effects among the three groups.
4 Discussion
Septic cardiomyopathy caused by severe sepsis is usually associated with the clinical characteristics of lower cardiac output and high mortality [18]. In 2012, Macchia et al. [19] reported that the mortality of patients with severe septic cardiomyopathy could be reduced by continuously taking β-blockers. Suzuki et al. [20] have shown that esmolol infusion in sepsis can improve oxygen utilization of the myocardium and preserve myocardial function in rats [20]. Schmittinge et al. [21] showed that low doses of enteral metoprolol in combination with milrinone were feasible in patients with septic shock and cardiac depression, and could increase SVI and pH and reduce arterial lactate significantly [21]. Moreover, for patients in septic shock, open-label use of esmolol was associated with reductions in the HR to achieve target levels and lower mortality [22]. In our study, the results showed that combination therapy with milrinone and esmolol could reduce arterial lactate, improve cardiac function, and reduce the mortality of septic cardiomyopathy patients, which was consistent with the previous studies.
CK-MB, TnI, and BNP are markers of myocardial injury dysfunction, of which significantly higher values have been observed in septic cardiomyopathy patients [23]. Previous studies have shown that CK-MB, TnI, and BNP could be reliable markers for identification of patients developing sepsis-induced myocardial depression, and could be used as a prognostic marker to identify patients with an elevated risk for an adverse outcome [24–26]. Patients with septic cardiomyopathy who have elevated markers of myocardial injury dysfunction still had a high mortality even though the cardiac output was normal, which indicated their vital impact on disease severity and the prognostic value in patients with septic cardiomyopathy [27]. The 2014 study by Papanikolaou et al. [28] showed that fast BNP decline after therapy was associated with favorable outcomes in critical sepsis [28]. We found that following combination therapy of milrinone and esmolol, patients’ cardiac function was improved, with reduced 28-day mortality, and the serum CK-MB, TnI, and BNP levels were also significantly reduced.
The monocytes were activated in patients accompanying with the initiation of the immune system in patients with sepsis, causing abundant release of proinflammatory factors such as TNF-α, IL-6, and HMGB-1 [29], which could cause significant myocardial depression and depress myocardial contractile function, eventually leading to the development of septic cardiomyopathy [30]. According to the results of Li et al. [31], modulation of the PI3K/p110α signaling pathway and inhibition of cardiac HMGB-1 may be beneficial for the prevention and/or management of septic cardiomyopathy, while Suzuki et al. [20] showed that esmolol infusion in sepsis could reduce TNF-α concentrations and improve cardiac output and cardiac efficiency in esmolol-treated rats in order to improve oxygen utilization of the myocardium and preserve myocardial function [20]. Similarly, we can conclude from our study that combination therapy with milrinone and esmolol could improve patients’ cardiac function and reduce mortality, as well as reduce the serum level of proinflammatory factors such as TNF-α, IL-6, and HMGB-1.
Patients with severe septic cardiomyopathy quite often suffer from tachycardia. However, if the tachycardia still exists after sufficient dilation of the blood volume and elimination of pain or dysphoria, excessive sympathetic nerve excitement should be taken into consideration. Tachycardia would increase the myocardial oxygen demand and cardiac load as well as shorten the ventricular diastolic time [14]. What’s more, the excessive sympathetic nerve excitement could result in left ventricular apical ballooning, myocardial stunning, apoptosis and necrosis of myocardial cells [32, 33]. However, it is controversial as to whether the tachycardia of septic cardiomyopathy patients should be controlled or not, as lowering the HR would cause decreased cardiac output and reduced tissue perfusion, although it would reduce myocardial oxygen demand and improve ventricular diastolic dysfunction and coronary perfusion. Thus, it appears necessary to choose appropriate drugs to control patients’ HR within the optimal range, which would reduce the myocardial oxygen demand without affecting cardiac output.
As a rapid-effect intravenous β-blocker, esmolol can reduce cardiac output in proportion to the percentage decreases in HR without adversely affecting oxygen [15]. Thus, in the present study, we treated patients with esmolol and controlled patients’ HR in the range of 75–94 beats/min and the results at 28 days showed that the lactate, cardiac function, and mortality were all significantly improved, which was consistent with the previous studies of Schmittinge et al. [21] and Morelli et al. [22].
However, there are some limitations to our study. First, the lack of echocardiography data limited the detailed investigation of cardiac function improvement after combination therapy with milrinone and esmolol in our analysis. Secondly, as the dosage of esmolol was adjusted according to the HR level, the study was designed as a non-blind trial, which is prone to bias. Thirdly, although the predefined HR threshold in our study was set following that of previously reported data, this threshold was set arbitrarily without being individualized according to the specific myocardial characteristics of each patient. In addition, the 28-day mortality of patients in the C group was very high. Most patients in this group died because of complications outside of the hospital setting, which was speculated to be due to insufficient care for these patients out of hospital. This phenomenon should be brought to the surgeon’s attention as adequate care is still needed when patients with stable vital signs leave the hospital.
5 Conclusion
Combination therapy with milrinone and esmolol could improve the cardiac function and 28-day survival rates in patients with severe sepsis. In addition, the combination treatment could reduce patient’s HR and inhibit the inflammatory response caused by sepsis. Future prospective controlled trials on combination therapy with milrinone and esmolol for septic cardiomyopathy and their influence on the prognosis are warranted.
References
Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001;29:1303–10.
Dellinger RP. Cardiovascular management of septic shock. Crit Care Med. 2003;31:946–55.
Martin GS, Mannino DM, Eaton S, Moss M. The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med. 2003;348:1546–54.
Linde-Zwirble WT, Angus DC. Severe sepsis epidemiology: sampling, selection, and society. Crit Care. 2004;8:222–6.
Dombrovskiy VY, Martin AA, Sunderram J, Paz HL. Rapid increase in hospitalization and mortality rates for severe sepsis in the United States: a trend analysis from 1993 to 2003. Crit Care Med. 2007;35:1244–50.
Phillips DP, Kaynar AM. Septic cardiomyopathy. Int Anesthesiol Clin. 2012;50:187–201.
Kumar A, Krieger A, Symeoneides S, Kumar A, Parrillo JE. Myocardial dysfunction in septic shock: part II. Role of cytokines and nitric oxide. J Cardiothorac Vasc Anesth. 2001;15:485–511.
Vieillard-Baron A. Septic cardiomyopathy. Ann Intensiv Care. 2011;1:6.
Hunter JD, Doddi M. Sepsis and the heart. Br J Anaesth. 2010;104:3–11.
Jozwiak M, Persichini R, Monnet X, Teboul JL. Management of myocardial dysfunction in severe sepsis. Semin Respir Crit Care Med. 2011;32:206–14.
Flynn A, Chokkalingam Mani B, Mather PJ. Sepsis-induced cardiomyopathy: a review of pathophysiologic mechanisms. Heart Fail Rev. 2010;15:605–11.
Kume T, Kawamoto T, Okura H, Toyota E, Neishi Y, Watanabe N, et al. Local release of catecholamines from the hearts of patients with tako-tsubo-like left ventricular dysfunction. Circ J. 2008;72:106–8.
Wittstein IS, Thiemann DR, Lima JA, Baughman KL, Schulman SP, Gerstenblith G, et al. Neurohumoral features of myocardial stunning due to sudden emotional stress. N Engl J Med. 2005;352:539–48.
Magder SA. The ups and downs of heart rate. Crit Care Med. 2012;40:239–45.
Gore DC, Wolfe RR. Hemodynamic and metabolic effects of selective beta 1 adrenergic blockade during sepsis. Surgery. 2006;139:686–94.
Lindsay CA, Barton P, Lawless S, Kitchen L, Zorka A, Garcia J, et al. Pharmacokinetics and pharmacodynamics of milrinone lactate in pediatric patients with septic shock. J Pediatr. 1998;132:329–34.
Dellinger R, Levy M, Rhodes A, Annane D, Gerlach H, Opal S, et al. Surviving Sepsis Campaign Guidelines Committee including the Pediatric Subgroup Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 2013;41:580–637.
Charpentier J, Luyt CE, Fulla Y, Vinsonneau C, Cariou A, Grabar S, et al. Brain natriuretic peptide: a marker of myocardial dysfunction and prognosis during severe sepsis. Crit Care Med. 2004;32:660–5.
Macchia A, Romero M, Comignani PD, Mariani J, D’ettorre A, Prini N, et al. Previous prescription of beta-blockers is associated with reduced mortality among patients hospitalized in intensive care units for sepsis. Crit Care Med. 2012;40:2768–72.
Suzuki T, Morisaki H, Serita R, Yamamoto M, Kotake Y, Ishizaka A, et al. Infusion of the beta-adrenergic blocker esmolol attenuates myocardial dysfunction in septic rats. Crit Care Med. 2005;33:2294–301.
Schmittinger CA, Dunser MW, Haller M, Ulmer H, Luckner G, Torgersen C, et al. Combined milrinone and enteral metoprolol therapy in patients with septic myocardial depression. Crit Care. 2008;12:R99.
Morelli A, Ertmer C, Westphal M, Rehberg S, Kampmeier T, Ligges S, et al. Effect of heart rate control with esmolol on hemodynamic and clinical outcomes in patients with septic shock: a randomized clinical trial. JAMA. 2013;310:1683–91.
Slack JA, Mcguirk SM, Erb HN, Lien L, Coombs D, Semrad SD, et al. Biochemical markers of cardiac injury in normal, surviving septic, or nonsurviving septic neonatal foals. J Vet Intern Med. 2005;19:577–80.
Post F, Weilemann LS, Messow CM, Sinning C, Munzel T. B-type natriuretic peptide as a marker for sepsis-induced myocardial depression in intensive care patients. Crit Care Med. 2008;36:3030–7.
Klouche K, Pommet S, Amigues L, Bargnoux AS, Dupuy AM, Machado S, et al. Plasma brain natriuretic peptide and troponin levels in severe sepsis and septic shock: relationships with systolic myocardial dysfunction and intensive care unit mortality. J Intensiv Care Med. 2013;29:229–37.
Mehta NJ, Khan IA, Gupta V, Jani K, Gowda RM, Smith PR. Cardiac troponin I predicts myocardial dysfunction and adverse outcome in septic shock. Int J Cardiol. 2004;95:13–7.
Ueda S, Nishio K, Akai Y, Fukushima H, Ueyama T, Kawai Y, et al. Prognostic value of increased plasma levels of brain natriuretic peptide in patients with septic shock. Shock. 2006;26:134–9.
Papanikolaou J, Makris D, Mpaka M, Palli E, Zygoulis P, Zakynthinos E. New insights into the mechanisms involved in B-type natriuretic peptide elevation and its prognostic value in septic patients. Crit Care. 2014;18:R94.
Cohen J. The immunopathogenesis of sepsis. Nature. 2002;420:885–91.
Pathan N, Hemingway CA, Alizadeh AA, Stephens AC, Boldrick JC, Oragui EE, et al. Role of interleukin 6 in myocardial dysfunction of meningococcal septic shock. Lancet. 2004;363:203–9.
Li C, Hua F, Ha T, Singh K, Lu C, Kalbfleisch J, et al. Activation of myocardial phosphoinositide-3-kinase p110alpha ameliorates cardiac dysfunction and improves survival in polymicrobial sepsis. Plos One. 2012;7:e44712.
Park JH, Kang SJ, Song JK, Kim HK, Lim CM, Kang DH, et al. Left ventricular apical ballooning due to severe physical stress in patients admitted to the medical ICU. Chest. 2005;128:296–302.
Schmittinger CA, Dunser MW, Torgersen C, Luckner G, Lorenz I, Schmid S, et al. Histologic pathologies of the myocardium in septic shock: a prospective observational study. Shock. 2013;39:329–35.
Acknowledgments
QW participated in the design of this study. XN performed the statistical analysis. JG carried out the study, together with CY, and collected important background information. ZW drafted the manuscript. QW conceived of this study, participated in the design, and helped to draft the manuscript. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work has no source of funding.
Conflict of interest
All authors declare that they have no conflict of interest to state.
Ethical approval
The Ethics Committee of the Jiangxi Provincial People’s Hospital (Nanchang, Jiangsu, China) approved this study.
Informed consent
Each patient or their caregivers signed a written informed consent after an adequate explanation of the study.
Additional information
Z. Wang and Q. Wu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, Z., Wu, Q., Nie, X. et al. Combination Therapy with Milrinone and Esmolol for Heart Protection in Patients with Severe Sepsis: A Prospective, Randomized Trial. Clin Drug Investig 35, 707–716 (2015). https://doi.org/10.1007/s40261-015-0325-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-015-0325-3