Abstract
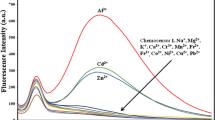
A novel coumarin derivative[7-diethylamino-2-oxo-2H-chromene-3-carboxylic acid(6-amino-pyridin-2-yl)-amide, CFe1] has been synthesized and its potential application as a chemosensor for the detection of metal ions has been further investigated. The responses of CFe1 to Fe3+ were studied by fluorescence emission spectrometry in the presence of other metal ions such as Al3+, Ba2+, Ca2+, Co3+, Cr3+, Cu2+, Fe2+, Hg2+, Mg2+, Mn2+, Na+, Ni+, Pb2+, Zn2+, K+, and Ag+. CFe1 showed a good selectivity for Fe3+ with fast response, a wide pH span of 3.3–9.18, and a large Stocks shift. CFe1 in the presence of Fe3+ and ethylene diamine tetraacetic acid(EDTA) makes the blue solution fade to colorless, which is due to the formation of CFe1-Fe3+ complex instead of any catalytic action of Fe3+. Furthermore, the imaging of Fe3+ in cultured single mice microglia cells was realized with the aid of CFe1, indicating that CFe1 has a great potential to be used as promising models for the future design of novel and robust chemosensor for metal ion detection in the field of biomedical and environmental analyses.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Meneghini R., Free Radic. Biol. Med., 1997, 23(5), 783
Rouault T. A., Nat. Chem. Biol., 2006, 2(8), 406
Lohani C. R., Lee K. H., Sens. Actuator B, 2010, 143, 649
QueirÓs C., Silva A. M. G., Lopes S. C., Ivanova G., Gameiro E., Rangel M., Dyes Pigm., 2012, 93, 1447
Cheng P. F., Xu K. X., Yao W. Y., Kong H. J., Kou L., Ma X. D., Wang C. J., Chem. Res. Chinese Universities, 2013, 29(4), 642
Yao W. Y., Xu K. X., Kong H. J., Kou L., Zhang Q., Supramol. Chem, 2013, 25, 146
Peng R., Wang F., Sha Y., Molecules, 2007, 12, 1191
Durukan İ., Şahin Ç. A., Şatlroğlu N., Bektaş S., Microchem. J., 2011, 99, 159
Liang P., Sang H. B., Sun Z. M., J. Colloid. Interface Sci., 2006, 304, 486
Yang Z., Hou X. D., Jones B. T., Sane D. C., Thomas M. J., Schwenke D. C., Microchem. J., 2002, 72, 49
Kozono S., Haraguchi H., Anal. Chim. Acta, 2003, 482(2), 189
Laly S., Nakagawa K., Arimura T., Kimijima T., Spectrochim. Acta, 1996, 51(11), 1393
Phillips C. V., Ayling K., Crowther E., Harrison M. E., Jordan M. A., Miner. Eng., 1994, 7(10), 1327
Amin A. S., Gouda A. A., Talanta, 2008, 76(5), 1241
Liu B., Yang X. H., Wang K. M., Tan W. H., Chem. Res. Chinese Universities, 2012, 28(1), 37
Zhu Y., Liu E., Yang X. H., He L. L., Li Q. Z., Wang Q., Wang K. M., Huang J., Liu J. B., Chem. J. Chinese Universities, 2012, 33(12), 2651
Tang B., Xing Y. L., Li R., Zhang N., Yu F. B., Yang G. W., J. Am. Chem. Soc., 2007, 129, 11666
Xiao Y., Li S., Huang H. M., Zhang Y., Xue Z. Z., Tang X. Y., He X. X., Wang K. M., Chem. Res. Chinese Universities, 2013, 29(5), 845
Lin W. Y., Yuan L., Feng J. B., Cao X. W., Eur. J. Org. Chem., 2008, (16), 2689
Lin W. Y., Yuan L., Cao X. W., Tetrahedron Lett., 2008, 49, 6585
Zhou Q. Y., Liu W. Z., Chang L., Chen F., Spectrochimica Acta Part A, 2012, 92, 78
She M. Y., Yang Z., Yin B., Zhang J., Gu J., Yin W. T., Li J. L., Zhao G. F., Shi Z., Dyes Pigm., 2012, 92, 1337
Katie L. C., Lynne M. H., Samir D., Katherine J. F., Inorg. Chem., 2010, 49, 6808
Jiang R. S., Feng F., Chen Z. Z., Anal. Lett., 2008, 41(12), 2203
Lim N. C., Brückner C., Chem. Commun., 2004, 1094
Suzuki Y., Komatsu H., Iked T., Satio N., Araki S., Citterio D., Hisamoto H., Kitamura Y., Kubota T., Nakagawa J., Oka K., Suzuki K., Anal. Chem., 2002, 74, 1423
Brunet E., Alonso M. T., Juannes Q., Sedano R., Rodriguez-Ubis J. C., Tetrahedron Lett., 1997, 38(25), 4459
Samankumara K. R. A., Imazu S., James T. D., Mikami M., Shinkai S., Chem. Lett., 1995, 2, 139
Plater M. J., Greig I., Helfrich M. H., Ralston S. H., J. Chem. Soc. Perkin Trans. 1, 2001, 2553
Kim G. J., Kim H. J., Tetrahedron Lett., 2010, 51(1), 185
Jung H. S., Kwon P. S., Lee J. W., Kim J. L., Hong C. S., Kim J. W., Yan S., Lee J. Y., Lee, J. H., Joo T., Kim J. S., J. Am. Chem. Soc., 2009, 131, 2008
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(No.21175085) and the Natural Science Foundation of Shanxi Province, China(No.2009011015-1).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wang, R., Wan, Q., Feng, F. et al. A novel coumarin-based fluorescence chemosensor for Fe3+ . Chem. Res. Chin. Univ. 30, 560–565 (2014). https://doi.org/10.1007/s40242-014-3192-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-014-3192-1