Abstract
Morphogenetic potential of cotyledon and hypocotyl explants of cabbage (Brassica oleracea L. var. capitata) was studied to develop a reliable plant regeneration protocol. Nine days old aseptically grown seedlings of cabbage were used as source of explant for plant regeneration. Out of 36 combinations of growth regulators used in MS medium, high frequency shoot regeneration from cotyledon explant was obtained on MS medium supplemented with 1.5 mg/l BAP and 0.50 mg/l NAA and 2 mg/l BAP and 0.25 mg/l IAA respectively. High percentage root regeneration (90 %) in in vitro developed shoots were obtained on MS medium supplemented with 0.10 mg/l IBA. The regenerated complete plantlets were transferred to pots containing a mixture of sand and soil and acclimatized. A reproducible and efficient plant regeneration protocol has been standardized in cabbage cv. Pride of India.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cabbage (Brassica oleracea L. var. capitata) is one of the most popular and widely grown cole crops in the world, belonging to family Brassicaceae. This crop is a native of Asia Minor. It is rich in minerals (phosphorus, potassium, calcium and iron); protein and vitamin-C and is consumed in various form like salad and boiled vegetable. Cabbage also possesses many medicinal properties [1]. The ‘American Cancer Society’ and ‘National Research Council’ have recommended increasing the consumption of cabbage as a means of decreasing the risk of certain types of cancer [2]. Cabbage is cultivated extensively in several parts of our country including Himachal Pradesh. In Himachal Pradesh, cabbage it is a major off season vegetable crop covering an area 2,150 ha with an annual production of 28,663 kg/ha (FAO Abstract, 2002), bulk of which is exported to the markets located in plains. However, cabbage is severely affected by the insect pests such as cabbage butterfly, diamondback moth and cabbage semilooper. Being an economically important crop, application of plant tissue culture and genetic engineering in cabbage cultivation is of special value to obtain improved or desirable traits like disease and insect resistance in this vegetable crop.
Development of high frequency plant regeneration protocol is a prerequisite to carry out genetic transformation studies. Plant regeneration studies have been carried in cabbage using various explants such as cotyledons [3, 4], hypocotyl [3, 5], leaf [6, 7], petiole [8, 9], protoplast [10, 11] and anther culture [12]. In the present investigation, we examined the high frequency shoot regeneration ability of cotyledon and hypocotyls explants by using different concentration of Kn + IAA, Kn + NAA, BAP + IAA and BAP + NAA in MS medium. We are currently using this plant regeneration system for the genetic transformation of Cabbage by genetically engineered Agrobacterium tumefaciens strain containing insect resistance gene.
Materials and Methods
Plant Material and Culture Medium
The certified seeds of cabbage (B. oleracea var. capitata cv. Pride of India) were procured from the Department of vegetable crops, of our university. The seeds were surface sterilized and then inoculated on the MS half strength basal medium [13] containing 0.5 % sucrose for seed germination. Nine days old seedlings were used as a source of explants (cotyledon and hypocotyl) for plant regeneration studies. The explants were cultured on MS salt (macro and micro), vitamins supplemented with 100 mg/l meso-inositol, 3 % sucrose and 0.8 % agar agar was also used as basal medium in shoot regeneration experiments. Different concentrations and combinations of cytokinins and auxins [Kn–IAA, Kn–NAA, BAP–IAA, BAP–NAA] were used in the MS basal medium for shoot regeneration studies. The pH of the medium was adjusted to 5.8 before adding agar agar. The medium was poured in culture vessels and sterilized at 1.08 kg/cm2 for 15 min in an autoclave. All the aseptic manipulations were carried out under laminar air flow chamber.
Plant Regeneration from Cotyledon and Hypocotyl Explants
To optimize the culture medium for high frequency shoot regeneration, cotyledon (0.5–1.0 cm) and hypocotyls (0.5–1 cm) explants were excised from 9 days old aseptically grown seedlings. Both the explants were inoculated on MS medium supplemented with various combinations and concentrations of plant growth regulators such as Kn and IAA, Kn and NAA, BAP and IAA, BAP and NAA (mg/l), respectively. The explants were evaluated for average number of shoots per explants. After inoculation, the culture vessels were kept in the culture room at 26 ± 2 °C under 16 h photoperiod with cool fluorescent lamps (40 mmol/m2/s) having 70–80 % humidity. The regenerated shoots (2–3 cm), obtained from both the explants were separated and individual shoot was transferred to the MS medium containing various concentrations of different auxins IAA, NAA, IBA and 2,4-D for root induction to get a complete plantlet. These were then evaluated for percentage of root regeneration after 4 weeks of culturing.
Hardening of Regenerated Plantlets
The regenerated plantlets were taken out of the tubes and flasks carefully and the roots were washed gently under running tap water to remove adhering medium. After removal of the medium, the plantlets were kept in running tap water for a few minutes so that they do not wilt after transfer to soil. After sterilization of sand and soil, the pots were filled half with soil and sand mixture in equal proportion and half with sand. The plantlets with washed roots were transferred to the pots. The root portion was placed inside the soil and sand mixture gently and was covered with sand. The plants were watered and covered with jam jars to maintain high relative humidity. These were then transferred to the culture room in which temperature and light conditions were controlled. Water was sprayed twice a day to maintain high relative humidity. After a week when plants showed initial signs of establishment in pots with appearance of new leaves, the jars were temporarily removed daily for few hours. The plants were finally transferred to earthen pots and were acclimatized further in glass house. The percentage survival of the hardened plants was recorded 5 weeks after transfer.
Data Analysis
Each treatment consisted of at least 30 explants and each experiment was repeated thrice. The data recorded for the different parameters were subjected to “Complete Randomized Design”. The statistical analysis based on mean values per treatment was made using analysis of variance of CRD.
Results and Discussion
The cotyledon segments on different medium expanded in size after 3–4 days of inoculation. The colour of explants changed from green to violet green. However, there was no change in the colour of the medium. After 20 days in culture, shoot buds started originating from the tissue around the cut edges of cotyledon segments after little callusing on different media, whereas, in case of hypocotyls, the size of the hypocotyls explants increased after about 1 week of inoculation. Callus formation was seen round the cut ends of the explants. Shoot bud development was seen after 15 days in culture from the explants. The shoot elongation occurred on the same media.
Shoot Regeneration from Cotyledon Explants
Effect of Kn and IAA on Shoot Regeneration
Different concentrations and combination of Kinetin and IAA mg/l (in MS medium) were used for shoot induction from cotyledon explants. Out of the nine combinations of Kinetin and IAA tried, the maximum number (4.667) of shoots per explants were found in MS medium supplemented with 1 mg/l Kn + 0.25 mg/l IAA closely followed by (4.334) MS medium containing 1 mg/l Kn + 0.50 mg/l IAA which were statistically at par with each other. MS medium containing 1 mg/l Kn + 0.75 mg/l IAA (1.334) gave the least number of shoot (Table 1).
Effect of Kn and NAA on Adventitious Shoot Bud Development
Nine different combinations of Kinetin and NAA were used for shoot induction from cotyledon explants. The maximum number of shoots per explants were found in MS medium supplemented with 2 mg/l Kn + 0.25 mg/l NAA (4.000). The least performers were observed in MS medium containing 1 mg/l Kn + 0.50 mg/l NAA (1.334) (Table 2; Fig. 1b).
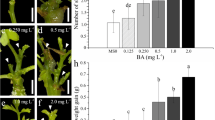
Plant regeneration from cotyledon and hypocotyl explants of Cabbage (Brassica oleracea L. var. capitata cv. Pride of India). a Regenerated shoots obtained from cotyledon explant on medium supplemented with (MS + 1.5 mg/l BAP + 0.75 mg/l IAA). b Shoots regeneration from cotyledon explants on medium supplemented with (MS + 1.5 mg/l Kn + 0.5 mg/l NAA). c Regenerated shoots obtained from hypocotyl explants on medium supplemented with (MS + 2 mg/l BAP + 0.25 mg/l IAA). d Shoots regeneration from hypocotyl explants on medium supplemented with (MS + 1 mg/l Kn + 0.25 mg/l NAA). e Root regeneration from the in vitro developed shoot regenerated from cotyledon explants on medium supplemented with (MS + 0.10 mg/l IAA). f Root regeneration from the in vitro developed shoots regenerated from hypocotyl explants on medium supplemented with (MS + 0.05 mg/l NAA). g Regenerated plantlets transferred to pots containing mixture of sterilized sand and soil for hardening. h Growth of the regenerated plant after about 20 days of its transfer in pot for hardening
Effect of BAP and IAA on Shoot Induction
BAP and IAA were used in different concentrations and combinations in MS medium for shoot induction. High frequency shoot regeneration (maximum average number of shoots per explant) was found in MS medium containing 2 mg/l BAP + 0.25 mg/l IAA (5.000) which was superior to all other combinations under study with whereas MS medium supplemented with 1 mg/l BAP + 0.50 mg/l IAA (1.000) gave the lowest shoot regeneration (Table 3; Fig. 1a).
Effect of BAP and NAA on Adventitious Shoot Bud Formation
Nine combination of BAP and NAA (mg/l) were used in the MS medium for shoot induction and best results were observed in MS medium containing 1.5 mg/l BAP + 0.50 mg/l NAA (5.334) which was statistically at par with MS medium containing 2 mg/l BAP + 0.25 mg/l NAA. The lowest number of shoots were found in MS medium containing 2 mg/l BAP + 0.50 mg/l NAA and MS medium + 1 mg/l BAP + 0.75 mg/l NAA (1.667). These shoots were excised and further subcultured on root regeneration media to obtain complete plantlets (Table 4).
Shoot Regeneration from Hypocotyls Explants
Effect of Kn and IAA on Adventitious Shoot Bud Formation
Different concentrations and combinations of Kn and IAA (mg/l) were used in order to get high frequency shoot regeneration from hypocotyls explants on MS medium supplemented with 1.5 mg/l Kn + 0.75 mg/l IAA (5.667) was found to be the best for shoot induction from hypocotyl explants followed by MS medium containing 2 mg/l Kn + 0.25 mg/l IAA (5.000) with non-significant difference. While in MS medium containing 2 mg/l Kn + 0.50 mg/l IAA (1.667) recorded the lowest number of shoots with non-significant differences with (2.334) MS medium + 2 mg/l Kn + 0.75 mg/l IAA (Table 1).
Effect of Kn and NAA on Shoot Regeneration
Nine different combinations of Kn and NAA (mg/l) were used for shoot induction from hypocotyls explants. The maximum number of shoots formed per explants were recorded in MS medium containing 2 mg/l Kn + 0.75 mg/l NAA (5.000) followed by MS medium containing 1.5 mg/l Kn + 0.25 mg/l NAA and MS medium + 2 mg/l Kn + 0.75 mg/l NAA while the poorest performers were recorded in MS medium + 1 mg/l Kn + 0.50 mg/l NAA and MS medium + 1.5 mg/l Kn + 0.50 mg/l NAA (2.334) (Table 2; Fig. 1d).
Effect of BAP and IAA on Adventitious Shoot Bud Formation
BAP and IAA were used in nine different combinations and concentrations to get high frequency shoot bud formation from hypocotyl explants. The MS medium supplemented with 2 mg/l BAP + 0.25 mg/l IAA (6.000) was the best for shoot regeneration with non-significant difference with MS medium containing 2 mg/l BAP + 0.75 mg/l IAA (5.000) whereas MS basal medium containing 1 mg/l BAP + 0.50 mg/l IAA (1.000) was the poorest of all (Table 3; Fig. 1c).
Effect of BAP and NAA on Shoot Induction
Nine different combinations and concentrations of BAP and NAA were used for shoot regeneration from hypocotyl explants. High frequency shoot regeneration (maximum average number of shoots per explant) was found in MS basal medium + 1 mg/l BAP + 0.50 mg/l NAA and MS basal medium + 2 mg/l BAP + 0.25 mg/l NAA (4.667). Culture media, MS basal + 2 mg/l BAP + 0.50 mg/l NAA and MS basal + 2 mg/l BAP + 0.75 mg/l NAA were the least performer (1.334) (Table 4).
Cotyledons from 9 days old seedlings, were almost fully expanded and turned completely green whereas, hypocotyls were greenish in colour and turgid nature. The effect of age of donor seedlings (source of explant) on shoot induction from cotyledon, hypocotyl or leaf have also been studied in different species [14–17] and in Brassica species also [18–21]. Dong and Jia [22] have reported that shoot differentiation frequencies of cotyledonary explants from seedlings older than 7 days dropped sharply, while about 5 days old cotyledons were the most sensitive to shoot induction. A possible explanation is that the young cotyledon and hypocotyl explants are physiologically and biochemically more active and they have less rigid cell wall and are easily affected by the environmental factors, such as exogenous plant growth regulators. Two types of cytokinins i.e. BAP and Kinetin were used in the present studies, which were effective in shoot induction from cotyledon and hypocotyls explants. In hypocotyls explants the maximum average number of shoots per explants was obtained from combination of BAP–IAA where as in cotyledon explants, the maximum average number of shoots per explants was obtained from combination of BAP–NAA. Guo et al. [23], has reported the effects of plant growth regulators on shoot regeneration from cotyledon and leaf segments of stem mustard (Brassica juncea var. tsatsai), in which NAA in combination with various cytokinins increased frequency of shoot regeneration. Arora et al. [24] found that MS medium fortified BAP 1.0 mg/l with IAA 0.1 mg/l in combination was best for shoot regeneration. Similarly Rafat et al. [25] reported use of 1 mg/l BAP alone for the shoot regeneration. Similar result was reported by Jin et al. [26] that BAP–NAA combination showed best result in case of hypocotyls explants. The results indicate that BAP in the medium enhanced the average number of shoots per explants in case of cotyledon explants whereas, Kinetin enhanced the number of shoots from hypocotyls explants. The similar results were reported by Yang et al. [27] in broccoli.
Root Regeneration from In Vitro Developed Shoots
Elongated shoots (about 2–3 cm in length) obtained from the cotyledon and hypocotyl explants were excised and cultured separately on MS medium supplemented with various concentration of different auxins i.e. IAA, IBA, NAA and 2,4-D. The period for the induction of roots were variable among various concentrations of different auxins but it was generally after 9–10 days in culture in cotyledon where as 7–8 days in culture in hypocotyls. When 2,4-D was used in the medium, callus formation was observed at the base of shoots in both cases. Shoots continued to grow after root regeneration and formed new leaves. It was clear from the data that the growth regulator IBA resulted in highest percentage of root regeneration (89.63 %) which was statistically at par with IAA (88.85 %) in cotyledon. Whereas in hypocotyl the highest percentage roots regeneration observed in IAA (89.63 %) which was statistically at par with IBA (87.57 %) and NAA (83.61 %). In cotyledon, NAA and 2,4-D were comparatively less effective in percentage root formation with values (71.11%) and (61.66%) whereas, in case of hypocotyl 2,4-D was the less effective (70.36 %) as it resulted in callusing at the base of the shoots. (Tables 5, 6; Fig. 1e, f). Bhalla and Weerd [28] used 0.2 mg/l IBA and obtained maximum rooting and Sretenovic et al. [29] used 4.0 mg/dm3 IBA for root regeneration from in vitro developed shoots. The concentration of different cytokinins and auxins in combination or alone required for shoot regeneration and root regeneration varies with the type of explants or cultivar used [30–37].
Hardening of Regenerated Plantlets of Cabbage
After proper in vitro development, the plantlets were taken out of the culture tubes and transferred to the pots fot hardening. Acclimatized plants were obtained after 5–6 weeks of transfer to the plastic pots with 70 % survival (Fig. 1g, h).
The primary aim of our work to develop reliable and high frequency plant regeneration protocol for the introduction of a desirable gene, in cabbage, was successfully achieved. These results clearly suggest that high frequency plant regeneration was possible in the cultivar “Pride of India” (cabbage) and it can be favourably exploited for genetic engineering purposes. The regenerated plantlets were transferred to pots containing a mixture of sand and soil for hardening. The acclimatized plantlets were obtained within 2–3 months.
Abbreviations
- BAP:
-
N 6-Benzylaminopurine
- NAA:
-
Naphthalene acetic acid
- IAA:
-
Indole-3-acetic acid
- IBA:
-
Indole-3-butyric acid
- MS:
-
Murashige and Skoog
References
Munger HM (1988) Adaptation and breeding of vegetable crops for improved human nutrition. In: Quebedeaux B, Pedrick AB (eds) Horticulture and human health
Zhang Y, Talaly P, Cho CG, Posner GH (1992) A major inducer of anticarcinogenic protective enzymes from broccoli isolation and elucidation of structure. Proc Natl Acad Sci USA 89:2399–2403
Deng-Xia, Lei C, Yu-Mei L, Mu Z, Yang-Yong Z, Zhi-Yuan F, Li-Mei Y (2011) Transformation of cabbage (Brassica oleracea L. var.capitata) with Bt cry1Ba3 gene for control of diamondback moth. Agric Sci China 10(11):1693–1700
Fan MQ, Zhu YL, Zhu LH (2005) Studies on highly efficient in vitro shoot regeneration from cotyledon of Chinese cabbage. J Nanjing Agric Univ 28(1):20–23
Zhao J, Liang A, Zhu Z, Tang Y (2006) Regeneration of Chinese transgenic plants expressing antibacterial peptide gene and cowpea trypsin inhibitor gene. Euphytica 150:397–406
Memon SA, Hou Xilin, Zhu Bo, Wolukau Joseph N (2008) High-frequency adventitious shoots regeneration from leaf of non-heading Chinese cabbage (Brassica campestris ssp.chinensis) cultured in vitro. Acta Physiol Plant 31:1191–1196
Xue-Yun Z, Shi-Jie C, Li-Ping C, Ming-Fang Z, Jing-Quan Y (2010) Induction and origin of adventitious roots from chimeras of Brassica juncea and Brassica oleracea. Plant Cell Tissue Organ Cult 101:287–294
Cao J, Anthony MS, Elizabeth DE (2008) Sequential transformation to pyramid two Bt genes in Brassica juncea L. and its potential for control of diamondback moth larvae. Plant Cell Rep 27(3):479–487
Yu ZD, He QW, Mu JH, Kang J, Wang PW (2005) Study on the tissue culture and plant regeneration of Chinese cabbage. J Jitin Agric Univ 27(4):391–395
Chen LP, Zhang MF, Xiao QB, Wu JG, Hirata Y (2004) Plant regeneration from hypocotyls protoplasts of red cabbage (Brassica oleracea) by using nurse cultures. Plant Cell Tissue Organ Cult 77(2):133–138
Kirti PB, Bhat SR, Kumar VD, Prakash S, Chopra VL (2001) A simple protoplast for regenerating mesophyll protoplasts of vegetable brassicas. J Plant Biochem Biotechnol 10(1):49–51
Ferrie AMR, Mollers C (2010) Haploids and doubled haploids in Brassica species for genetic and genomic research. Plant Cell Tissue Organ Cult 14:531–541
Murashsige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–479
Srivastava DK, Andrianova VM, Piruzian ES (1991) Regeneration and genetic transformation studies in watermelon (Citrullus vulgaris L. cv. Melitopolski). In: Prakash J, Pierik R L M, (Eds) Horticulture—new technologies and applications, pp 127–130
Srivastava V, Reddy AS, Guha Mukherjee S (1988) Transformation and regeneration of Brassica oleracea mediated by an oncogenic Agrobacterium tumefaciens. Plant Cell Rep 7:504–507
Srivastava DK, Andrianov VM, Priuzian ES (1989) Tissue culture and plant regeneration of watermelon (Citrullus vulgaris schrad cv. Meletopolski). Plant Cell Rep 8:300–302
Srivastava DK, Kolgonova TV, Mett VL, Piruzian ES (1991) Genetic transformation of cotton (Gossypium hirsutum L. Cv. 108-F). Acta Hortic 289:263–264
Baroda A, Madanpotra S, Singh ND and Jaiwal PK (2003) Stable transformation of Brassica juncea using Agrobacterium tumefaciens. In: Proceedings of 2nd international congress of plant physiology, Abstract no. S11-P8, p 462
Chakrabarty R, Viswakarma N, Bhat SR, Kirti PB, Singh BD, Chopra VL (2002) Agrobacterium mediated transformation of cauliflower: optimization of protocol and development of Bt-transgenic cauliflower. J Biosci 27(25):495–502
Dixit S, Srivastava DK, Sharma DR (1998) Genetic transformation in cauliflower (Brassica oleracea L. var. botrytis cv. Pusa Snow Ball). In: Proceedings of national symposium on biotechnology in agriculture and environment, Abstract p 2
Sharma S, Srivastava DK, Nath AK, Sharma DK (1996) Regeneration and genetic transformation studies in cauliflower (Brassica oleracea L. var. botrytis cv. Pusa Snow Ball). In: Proceedings of national symposium on horticulture biotechnology, Abstract No. V-120
Dong JZ, Jia SR (1991) High efficiency plant regeneration from cotyledons of watermelon (Citrullus vulgaris Schard). Plant Cell Rep 9:858–863
Guo DP, Zhu ZJ, Hu XX, Zheng SJ (2005) Effect of cytokinins on shoot regeneration from cotyledon and leaf segment of stem mustard (Brassica juncea var. tsatsai). Plant Cell Tissue Organ Cult 83:123–127
Arora N, Yadav NR, Yadav RC, Chowdhury JB (1997) Role of IAA and BAP on plant regeneration in cultured cotyledons of cauliflower. Crucif Newslett 19:41–42
Rafat A, Aziz MA, Rhashid AA, Abdullah SNA, Kamaladini H, Sirchi MHT, Javadi MB (2010) Optimization of Agrobacterium tumefaciens-mediated transformation and shoot regeneration after co-cultivation of cabbage (Brassica oleracea subsp. capitata) cv. KY cross with At HSP101 gene. Sci Hortic 124:1–8
Jin RG, Liu YB, Tabashnik BE, Borthakur D (2000) Development of transgenic cabbage (Brassica oleracea var. capitata) for insect resistance by Agrobacterium tumefaciens-mediated transformation. In Vitro Cell Dev Biol Plant 36:231–237
Yang JL, Seong ES, Kim MJ, Ghimire BK, Kang WH, Chang YY, Cheng HL (2010) Direct somatic embryogenesis from pericycle cell of broccoli (Brassica oleracea L. var. italica) root explants. Plant Cell Tissue Organ Cult 100:49–58
Bhalla PL, Weerd ND (1999) In vitro propagation of cauliflower, Brassica oleracea var. botrytis for hybrid seed production. Plant Cell Tissue Organ Cult 56:89–95
Sretenovic RT, Ninkovic S, Vinterhalter B, Milijus DJ, Vinterhalter D (2004) Introduction of resistance to herbicide Basta Reg. in Savoy cabbage. Biol Plant 48(3):431–436
Dai SY, Jai S, Dong G, Merg LY (1994) In vitro culture and plant regeneration of cotyledon and hypocotyls of cabbage. Adv Hortic 12:286–289
Eisner GI, Mar Yakhina I, Shenyakin MF (1992) Optimization of conditions for in vitro plant regeneration for cabbage transformations. Sov Agric Sci 11(12):15–19
Petrova S, Antonova G (1996) Plant regeneration from seedling explants of cabbage. Crucif Newslett 18:31
Zhang S, Wen FJ, Wei YT, Fu LH (1997) Optimization of growth regulators in medium for efficient plant regeneration of Chinese cabbage. Crucif Newslett 19:51–52
Cao MQ, Charlot F, Dore C (1990) Embryogenesis and plant regeneration in sauerkraut cabbage (Brassica oleracea L. subsp. capitata) by in vitro culture of isolated microspores. Comptes Rendud de l’Academie des Science 310(5):203–209
Lillo C, Shahin EA (1986) Rapid regeneration of plants from hypocotyl protoplasts and root segments of cabbage. HortScience 21(3):315–317
Koda T, Ichi T, Yamagishi H, Yoshikawa H (1988) Effect of phytohormones and gelling agents on plant regeneration from protoplasts of red cabbage. Agric Biol Chem 52(9):2337–2340
Hegazi HH, Matsubara S (1991) Plant regeneration from hypocotyl callus of inter-generic and interspecific hybrids between radish (Raphanus sativus L. daikon and radicula gropus), cabbage (Brassica oleracea L. capitata group) and Chinese cabbage (Brassica campestris L. pekinsis group). J Jpn Soc Hortic Sci 60(2):369–377
Acknowledgments
Authors thank to Professor and Head, Dept. of Vegetable Science of Dr. Y.S. Parmar University of Horticulture and Forestry, Solan, India for providing germplasm of cabbage cv. Pride of India.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharma, S., Gambhir, G. & Srivastava, D.K. High Frequency Organogenesis in Cotyledon and Hypocotyl Explants of Cabbage (Brassica oleracea L. var. capitata). Natl. Acad. Sci. Lett. 37, 5–12 (2014). https://doi.org/10.1007/s40009-013-0191-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40009-013-0191-6