Abstract
Broccoli (Brassica oleracea L. var. italica) is an important, nutritionally rich vegetable crop, but severely affected by environmental stresses, pests and diseases which cause massive yield and quality losses. Genetic manipulation is becoming an important method for broccoli improvement. In the present study, a reproducible and highly efficient protocol for obtaining organogenesis from hypocotyl, cotyledon, leaf and petiole explants of broccoli (Brassica oleracea L. var. italica cv. Solan green head) has been developed. Hypocotyl and cotyledon explants were used from 10 to 12 days old aseptically grown seedlings whereas leaf and petiole explants were excised from 18 to 20 days old green house grown seedlings and surface sterilized. These explants were cultured on shoot induction medium containing different concentration and combination of BAP and NAA. High efficiency shoot regeneration has been achieved in hypocotyl (83.33 %), cotyledon (90.11 %), leaf (62.96 %) and petiole (91.10 %) explants on MS medium supplemented with 3.5 mg/l BAP + 0.019 mg/l NAA 2.5 mg/l BAP + 0.5 mg/l NAA, 4.0 mg/l BAP + 0.5 mg/l NAA and 4.5 mg/l BAP + 0.019 mg/l NAA respectively. Petiole explants showed maximum shoot regeneration response as compared to other explants. MS medium supplemented with 0.10 mg/l NAA was found best for root regeneration (100 %) from in vitro developed shoots. The regenerated complete plantlets were transferred to the pots containing cocopeat and successfully acclimatized. This optimized regeneration protocol can be efficiently used for genetic transformation in broccoli. This is the first comparative report on multiple shoot induction using four different types of explants viz. hypocotyl, cotyledon, leaf and petiole.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Brassica oleracea L. var. italica, commonly known as broccoli belongs to the family Brassicaceae, which is nutritionally rich, high in vitamin A and C, soluble fiber and also contains the compound glucoraphanin, leading to an anticancer compound sulforaphane. It deserves greater breeding attention by seed companies with its increasing area of production in recent years and the anti-carcinogenic properties detected in some cultivars (Kirsh et al. 2007). However, broccoli is severely affected by the insect pest such as diamondback moth (Plutella xylostella), cabbage looper (Trichoplusia sp.), beetles (Phyllotreta cruciferae) and aphids (Brevicoryne brassicae). Being an economically important crop, application of plant tissue culture and genetic engineering in broccoli cultivation is of special value to obtain improved or desirable traits like disease and insect resistance in this vegetable crop. Development of high frequency plant regeneration protocol is a pre-requisite to carry out genetic transformation studies. Plant regeneration studies in broccoli were carried out by various workers using various explants such as peduncle (Christey and Earle 1991), hypocotyl (Puddephat et al. 2001; Kim and Botella 2002; Ravanfar et al. 2009), leaf tissue (Robertson and Earle 1986; Cao and Earle 2003) and cotyledon (Qin et al. 2006; Huang et al. 2011). In this paper, we described the high frequency shoot regeneration ability of hypocotyl, cotyledon, leaf and petiole explants by using different combination of BAP and NAA in broccoli (Brassica oleracea L. var. italica cv. Solan green head). Plant regeneration studies are genotype specific and the present genotype cv. Solan Green Head has not been worked out so far. This is the first comparative report on high frequency organogenesis using four explants hypocotyl, cotyledon, leaf and Petiole as compared to the previous studies. The aim of developing an efficient plant regeneration protocol for broccoli was to provide a useful method for producing genetically modified plants through Agrobacterium—mediated gene transfer technique.
Material and methods
Plant material and culture medium
The certified, uniform and healthy seeds of broccoli (Brassica oleracea L. var italica cv. Solan green head) were procured and surface sterilized. These seeds were inoculated on half strength MS basal medium (Murashige and Skoog 1962) containing 0.5 % sucrose for germination. Aseptically grown 10–12 days old seedlings of broccoli were used as a source of explants for hypocotyl and cotyledon whereas, leaf and petiole explants were procured from 18 to 20 days old glass house grown seedlings of broccoli for plant regeneration studies. The explants were cultured on MS salt (macro and micro), supplemented with vitamins and 100 mg/l meso-inositol, 3 % sucrose and 0.8 % agar agar was also used as basal medium in shoot regeneration experiments. Different combinations and concentrations of BAP and NAA were used in MS basal medium for shoot regeneration studies. The pH of the medium was adjusted to 5.8 before adding agar-agar to the medium. The medium was poured in culture vessels and sterilized at 15 lb per inch square for 15 min in an autoclave. All the aseptic manipulations were carried out under laminar air flow chamber.
Plant Regeneration from four different types of explants viz. hypocotyl, cotyledon, leaf and petiole
To optimize the culture medium for high frequency shoot regeneration, hypocotyl (0.5–1.0 cm) and cotyledon (0.5–1.0 cm) explants were excised from aseptically grown seedlings whereas, leaf (0.5–1.0 cm) and petiole (0.5–1.0 cm) explants were excised from in vivo grown seedling. All the explants were cultured on MS basal medium supplemented with various combinations and concentrations of BAP and NAA (mg/l). Nine different concentrations were constituted for each combination of the plant growth regulators in order to achieve the objective of high frequency plant regeneration in broccoli (Tables 1 and 2). The explants were evaluated for average number of shoots per explant and percent shoot regeneration. After inoculation, the culture vessels were kept in the culture room at 26 ± 2 °C under 16 h photoperiod with cool white fluorescent lamps (40 m mol/m2/s) having 70–80 % humidity. The regenerated shoots (2-3 cm) obtained from both the explants were separated and individual shoot was transferred to the root regeneration medium containing various concentrations of different auxins IAA, NAA and IBA for root induction to get a complete plantlet. After the regeneration of roots, the percentage of shoots forming roots was calculated.
Acclimatization of regenerated plantlets
The regenerated plantlets were taken out of the culture tubes carefully and the rooted plantlets were washed gently under running tap water in order to remove traces of adhering medium. After removal of the medium, the plantlets were kept in running tap water for a few minutes so that they do not wilt after transfer to soil. Plantlets were treated with 0.5 % bavistin solution for 5 min. The in vitro regenerated plantlets were transferred to sterilized cocopeat containing pots. After the transfer of the plantlets they were covered with the polythene bags in order to maintain optimum relative humidity. These were then transferred to the culture room in which temperature and light conditions were controlled. Water was sprayed twice a day to maintain high relative humidity. After 21 days when plants showed initial signs of establishment in pots with appearance of new leaves, the polythene bags were temporarily removed daily for few hours. The plants were finally transferred to earthen pots containing sand: soil: FYM in the ratio of 1:1:1, 30 days after the transfer of plantlets to cocopeat. The percentage survival of the hardened plants was recorded after 5 weeks of transfer to the pots.
Statistical analysis
Each treatment consisted of at least 30 explants and each experiment was repeated thrice. The data recorded for the different parameters were subjected to “Complete Randomized Design”. The statistical analysis based on mean values per treatment was made using analysis of variance of CRD (Gomez and Gomez 1984).
Results and discussion
Multiple shoot induction from hypocotyl, cotyledon, leaf and petiole explants of broccoli
Hypocotyl explants showed swelling at margins after 6–8 days of inoculation. Callus initiation at the cut ends was observed after 13–15 days and shoot initiation was observed between 30 and 35 days on different combinations of growth regulators. The cotyledon explants, on different combinations and concentrations of growth regulators showed initial expansion after 1 week. The colour of the explants turned purple green from green while no change in the colour of the media was observed. Callus initiation was observed between 18 and 20 days. Shoot initiation was observed between 35 and 40 days in case of cotyledon explants. Whereas in case of leaf explants callus initiation was observed from the cut edges between 12 and 18 days of culturing. Shoot regeneration was observed from the callus after 30–35 days of culturing. Whereas in case of petiole explants, during the initial days of culturing, swelling and expansion in the petiole explant was observed. Callus initiation was observed after 10–14 days. Adventitious shoot initiation was observed from the callus after 25–30 days of culturing.
Effect of BAP and NAA on shoot regeneration from hypocotyl explant
Nine different concentrations and combinations of BAP and NAA were used for the shoot regeneration from hypocotyl and cotyledon explants. In hypocotyl explants maximum percent shoot regeneration (83.33 %) with maximum average number of shoots (2.14) per explant was obtained on MS medium supplemented with 3.5 mg/l BAP + 0.019 mg/l NAA. (Table 1, Fig. 1a).
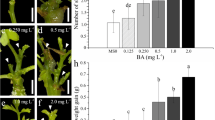
Plant regeneration studies in broccoli (Brassica oleracea L. var. italica cv. Solan green head). (a) Shoot regeneration from hypocotyls explants on MS medium supplemented with 3.5 mg/l BAP + 0.019 mg/l NAA (b) Shoot regeneration from cotyledon explants on MS medium supplemented with 2.5 mg/l BAP + 0.5 mg/l NAA (c) Shoot regeneration from leaf explants on MS medium supplemented with 4.0 mg/l BAP + 0.5 mg/l NAA (d) Shoot regeneration from petiole explants on MS medium supplemented with 4.5 mg/l BAP + 0.019 mg/l NAA (e) Root regeneration in in vitro regenerated shoots on MS medium supplemented with 0.10 mg/l NAA and development of in vitro complete plantlet (f) Hardening of in vitro developed complete and successful acclimatization in the pot after 30 days
Effect of BAP and NAA on shoot regeneration from cotyledon explant
Nine different concentrations and combinations of BAP and NAA were used for the shoot regeneration from cotyledon explants. Best shoot regeneration medium in cotyledon explants was found to be MS medium supplemented with 2.5 mg/l BAP + 0.5 mg/l NAA. Maximum percent shoot regeneration (90.11 %) and maximum average number of shoots (3.24) per explant was obtained under this condition. (Table 2, Fig. 1b).
Effect of BAP and NAA on shoot regeneration from leaf explant
Nine different concentrations and combinations of BAP and NAA were used for the shoot regeneration from leaf explants. In leaf explants maximum percent shoot regeneration (62.96 %), with maximum average number of shoots (2.48) per explant was obtained on MS medium supplemented with 4.0 mg/l BAP + 0.5 mg/l NAA. (Table 2, Fig. 1c).
Effect of BAP and NAA on shoot regeneration from petiole explant
Nine different concentrations and combinations of BAP and NAA were used for the shoot regeneration from petiole explants. In petiole explants, maximum percent shoot regeneration (91.10 %) and maximum average number of shoots (3.61) per explant was obtained on MS medium supplemented with 4.5 mg/l BAP + 0.019 mg/l NAA. This combination also showed high frequency regeneration among all the explants on different combinations and concentrations of BAP and NAA. (Table 1, Figs. 1d and 2).
Shoot multiplication and elongation
The regenerated shoots from the shoot induction medium were separated from each other and sub-cultured on the same media for shoot multiplication and elongation. Shoot multiplication and elongation of shoots from hypocotyl, cotyledon, leaf and petiole explants were observed on the same medium respectively.
The study reported here were aim to develop high frequency plant regeneration in broccoli (Brassica oleracea L. var. italica cv. Solan green head). In vitro regeneration is influenced by many factors such as culture environment, culture medium composition, explant source, plant growth regulators and genotype (Zhang et al. 1998; Rajicic et al. 2002; Bano et al. 2010; Jana and Shekhawat 2010; Dhir and Shekhawat 2014). In the present study, young tender completely green leaves and petiole explants from in vivo grown seedlings and completely green fully expanded cotyledons and hypocotyl which were greenish in colour and turgid nature from10-12 days old seedlings were used for efficient shoot regeneration studies. The effect of donar seedlings (Source of explants and age of explants) on shoot induction from cotyledon, hypocotyl and leaf have also been studied in different species. (Srivastava et al. 1988, 1989; Srivastava et al. 1991a, b; Compton and Gray 1993; Amita et al. 2012 and Sharma et al. 2014). Rani et al. (2013) have reported that in most Brassica species regeneration is dependent on the age of the explants. Young explants have been shown to give better results than older explants in most Brassica species (Chakrabarty et al. 2002; Sharma et al. 2012; Sharma et al. 2014). Dong and Jia (1991) have reported that shoot differentiation frequencies of cotyledonary explants from seedlings older than 7 days dropped sharply. A possible explanation is that young cotyledon and hypocotyl explants are physiologically and biochemically more active as they have less rigid cell wall and are easily affected by the environmental factors such as exogenous plant growth regulators. Nine different concentrations and combinations of BAP and NAA were used in the present studies, which were effective in multiple shoot induction. Petiole explants showed better shoot regeneration (91.10 %) followed by cotyledon (90.11 %), hypocotyl (83.33 %) and leaf (62.96 %) (Fig. 2). Yang et al. 2010 ; Huang et al. 2011; Sharma et al. 2012; Sharma et al. 2014 also reported better shoot regeneration response by using combinations of BAP and NAA. Qin et al. (2006) found that cotyledon was more responsive as compared to hypocotyl explants, whereas Ravanfar et al. (2009) found that hypocotyl and shoot tips were promising explants for shoot regeneration in broccoli. According to George et al. (2008), BAP promotes differentiation of cells into adventitious buds followed by the formation of shoots. The regenerated shoots elongated on the same medium. Ravanfar et al. 2009 and 2011 reported that BAP gave the highest number of shoots per hypocotyl and cotyledon segment with or without NAA on different cultivar of broccoli cv. Green Marvel. Varied responses could be due to genotypic differences of the cultivar or plants reacting differently during in vitro regeneration. Plant regeneration studies are genotype specific and the present genotype cv. Solan Green Head has not been worked out so far.
Root regeneration from in vitro developed shoots
Elongated shoots (about 2–3 cm long) obtained from four different types of explants viz. hypocotyl, cotyledon, leaf and petiole were excised and cultured separately on MS medium supplemented with various concentrations of different auxins i.e. IAA, IBA and NAA. Root initiation started after 10–14 days of inoculation. Maximum (100 %) root regeneration response was observed on MS half strength basal medium containing 0.1 mg/l NAA and well developed roots were observed after 20–22 days (Table 3, Fig. 1e). Lazzeri and Dunwell (1986) reported that high concentration of NAA was more effective for root regeneration as compared to high concentration of IAA or IBA. Ravanfar et al. 2009 and Sharma et al. 2014 reported that medium containing IBA was most suitable for root regeneration. Nevertheless, the influence of NAA, IAA or IBA on root induction was highly dependent on genotype (Arnison et al. 1990; Vandemoortele et al. 1999).
Hardening of regenerated plantlet of broccoli
After the complete in vitro development of plantlets, they were taken out of the culture tubes and transferred to pots containing sterilized cocopeat for acclimatization. Acclimatized plantlets showed the sign of establishment (emergence of new leaves) after 4–5 weeks of transfer to the pots with 75 % survival rate and all the plantlets were morphologically normal. (Fig. 1f).
The primary aim of our work to develop reliable and high frequency plant regeneration protocol for the introduction of a desirable gene, in broccoli, was successfully achieved. These results clearly suggest that high frequency plant regeneration was possible in broccoli cultivar “Solan green head” (broccoli) and it can be favorably exploited for genetic engineering purposes. To the best of our knowledge, this is the first comparative report on high frequency organogenesis using four explants hypocotyl, cotyledon, leaf and petiole from India.
Abbreviations
- MS :
-
Murashige & Skoog
- NAA :
-
Naphthalene acetic acid
- BAP- 6 :
-
Benzyl aminopurine
- IAA :
-
Indole-3-acetic acid
- IBA:
-
Indole-3-butyric acid
References
Amita G, Aggarwal G, Srivastava DK (2012) Plant regeneration, genetic transformation and expression of foreign gene cotyledon and petiole tissues of cucumber (Cucumis sativus L. cv. K-75). Adv Appl Res 4(1):46–52
Arnison PG, Donaldson P, Jackson A, Semple C, Keller W (1990) Genotype-specific response of cultured broccoli (Brassica oleracea var. italica) anthers to cytokinins. Plant Cell Tis Org Cul 20:217–228
Bano R, Khan MH, Khan RS, Rashid H, Swat ZA (2010) Development of an efficient regeneration protocol for three genotypes of Brassica juncea. Pak J Bot 42(2):963–969
Cao J, Earle ED (2003) Transgene expression in broccoli (Brassica oleracea var. italica) clones propagated in vitro via leaf explants. Plant Cell Rep 21:789–796
Chakrabarty R, Viswakarma N, Bhat SR, Kirti PB, Singh BD, Chopra VL (2002) Agrobacterium-mediated transformation of cauliflower: optimization of protocol and development of Bt-transgenic cauliflower. J Biosci 27:495–502
Christey MC, Earle ED (1991) Regeneration of Brassica oleracea from peduncle explants. Hortic Sci 26:1069–1072
Compton ME, Gray DJ (1993) Shoot organogenesis and plant regeneration from cotyledons of diploid, triploid and tetraploid watermelon. J Am Soc Hortic Sci 118:151–157
Dhir R, Shekhawat GS (2014) In vitro propagation using transverse thin cell layer culture and homogeneity assessment in Ceropegia bulbosa Roxb. J Plant Growth Regul 33(4):820–830
Dong JZ, Jia SR (1991) High efficiency plant regeneration from cotyledons of watermelon (Citrullus vulgaris Schard). Plant Cell Rep 9:858–863
George EF, Hall MA, Klerk GJD (2008) Plant propagation by tissue culture. The background, 3rd edn. Springer Publisher Dordrecht, London
Gomez KA, Gomez AA (1984) Statistical procedures for agricultural research. John Wiley and Sons, New York
Huang K, Qiuyun W, Juncleng L, Zheng J (2011) Optimization of plant regeneration from broccoli. Afr J Biotechnol 10(20):4081–4085
Jana S, Shekhawat GS (2010) Plant growth regulators, adenine sulfate and carbohydrates regulate organogenesis and in vitro flowering of Anethum graveolens. Acta Physiol Plant 33:305–311
Kim JH, Botella JR (2002) Callus induction and plant regeneration from broccoli (Brassica oleracea var. italica) for transformation. J Plant Biochem 45(3):177–181
Kirsh VA, Peters U, Mayne ST, Subar AF, Chatterjee N, Johnson CC, Hayes RB (2007) Prospective study of fruit and vegetable intake and risk of prostate cancer. J Natl Cancer Inst 99(15):1200–1209
Lazzeri PA, Dunwell JM (1986) In vitro regeneration from seedling organs of Brassica oleracea var. italica Plenck cv. Green comet. I. Effect of plant growth regulators. Ann Bot 58(5):689–697
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Puddephat IJ, Robinson HT, Fenning TM, Barbara DJ, Morton A, Pink DAC (2001) Recovery of phenotypically normal transgenic plants of Brassica oleracea L. var. italica upon Agrobacterium rhizogenes-mediated co-transformation and selection of transformed hairy roots by GUS assay. Mol Breed 7(3):229–242
Qin Y, Li HL, Guo YD (2006) High frequency embryogenesis, regeneration of broccoli (Brassica oleracea var. italica) and analysis of genetic stability by RAPD. Sci Hortic 111:203–208
Rajicic TS, D Stevanovic, R Djordjevic, M Veelickovic, Z Susic (2002) Maintenance of Prospective Cabbage Brassica oleracea var. capitata) Lines by Micropropagation. Acta Hortic. 579: II Balkan Symposium onVegetables and Potatoes.
Rani T, Yadav RC, Yadav NR, Asha R, Singh D (2013) Genetic transformation in oilseed brassicas: a review. Indian J Agric Sci 83(4):367–373
Ravanfar SA, Aziz MA, Kadir MA, Rashid AA, Haddadi (2011) In vitro shoot regeneration and acclimatization of Brassica oleracea var. italica cv. Green marvel. Afr J Biotechnol 10(29):5614–5619
Ravanfar SA, Aziz MA, Kadir MA, Rashid AA, Sirchi MHT (2009) Plant regeneration of Brassica oleracea var. italica (broccoli) cv. Green marvel was affected by plant growth regulators. Afr J Biotechnol 8(11):2523–2528
Robertson D, Earle ED (1986) Plant regeneration from leaf protoplasts of Brassica oleracea L. var. italica. Plant Cell Rep 5(1):61–64
Sharma S, Gambhir G, Srivastava DK (2014) High frequency organogenesis in cotyledon and hypocotyls explants of cabbage (Brassica oleracea L. var. capitata). Nat Acad Sci Lett 37:5–12
Sharma S, Sharma C, Srivastava DK (2012) Plant regeneration genetic transformation and gene expression in in vitro tissues of cauliflower (Brassica oleracea L. var botrytis). Bioinfolet 9(4B):760–764
Srivastava DK, Andrianov VM, Piruzian ES (1989) Tissue culture and plant regeneration of watermelon (Citrullus vulgaris Schard cv. Melitopulski). Plant Cell Rep 8:300–302
Srivastava DK, Andrianov VM, Piruzian ES (1991a) Regeneration and genetic transformation studies in watermelon (Citrullus vulgaris L. cv. Melitopolski). In: Horticulture- New Technologies and Applications, pp. 127–130 (Parkash J and Pierika RL M, eds.)
Srivastava DK, Kolgonova TV, Mett VL, Piruzian ES (1991b) Genetic transformation of cotton (Gossypium hirsutum L. cv. 108-F). Acta Hortic 289:263–264
Srivastava V, Reddy AS, Guha MS (1988) Transformation and regeneration of Brassica oleracea mediated by an oncogenic Agrobacterium tumefaciens. Plant Cell Rep 7:504–507
Vandemoortele JL, Billiard JP, Boucaud J, Gaspar T (1999) Evidence for an interaction between basal medium and plant growth regulators during adventitious or axillary shoot formation of cauliflower. In Vitro Cell Dev Biol Plant 35:13–17
Yang JL, Seong ES, Kim MJ, Ghimire BK, Kang WH, Chang YY, Cheng HL (2010) Direct somatic embryogenesis from pericycle cell of broccoli (Brassica oleracea L. var. italica) root explants. Plant Cell Tissue Organ Cult 100:49–58
Zhang FL, Takahata Y, Xu JB (1998) Medium and genotype factors influencing the regeneration from cotyledonary explants of Chinese cabbage (Brassica compestris L. ssp. Pekinensis). Plant Cell Rep 17:780–786
Acknowledgments
We are grateful to the Professor and Head, Department of Vegetable Science, Dr. Y. S. Parmar University of Horticulture and Forestry- Nauni, Solan for providing us the germplasm of broccoli cv. Solan green head. The senior author (P.K.) thankfully acknowledge the award of DST INSPIRE fellowship New Delhi.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, P., Srivastava, D.K. High frequency organogenesis in hypocotyl, cotyledon, leaf and petiole explants of broccoli (Brassica oleracea L. var. italica), an important vegetable crop. Physiol Mol Biol Plants 21, 279–285 (2015). https://doi.org/10.1007/s12298-015-0282-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-015-0282-6