Abstract
Lignocellulosic biomass (LCB) is plentifully and naturally available carbon source produced mostly from agro, food and food processing industries with a global estimate of 1.3 billion tonnes per year. Since LCB is inexpensive and considered as waste, it opens an avenue for cost-effective alternate source of energy. Lignocellulosic biomass can be effectively and efficiently converted to biofuels (e.g. bioethanol, biodiesel and biogas) and value-added products like organic acids, enzymes, biopolymers, biochar etc. However, the bottleneck in using lignocellulosic biomass on industrial scale is its structural complexity and recalcitrance nature. Thus, pretreatment of biomass is an essential step for efficient delignification of biomass. This process separates cellulose and hemicellulose from lignin of the complex polymer matrix. Thereby, reduces the size of the matrix and increases the surface area of cellulose and hemicelluloses to be accessible for enzymes and microbes for hydrolysis and fermentation respectively. The pretreatment process includes physical, chemical, physicochemical or biological. Mechanical milling, ultrasound and microwave radiation as physical; Acid/alkaline hydrolysis, organosolv, ionic liquids and ozonolysis as chemical; ammonia fiber explosion, CO2 explosion steam explosion, liquid hot water treatment as physicochemical methods are established. The use of certain species of bacteria, fungus and yeast in biological methods of pretreatment is yet to establish on large scale.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The global energy crisis due to increased use of non-renewable fossil fuel to meet the industrial development threatens human society. The other factors which may cause the energy crisis are unexplored renewable energy options, delay in commissioning of power plants, poor distribution system, overconsumption and natural calamities. Current world population of 7.3 billion is expected to reach 8.5 billion in 2030 and 9.7 billion in 2050 (Julie Baruah et al. 2018).To commensurate with the growing population, the global energy consumption is expected to increase by 28% from 2015 (nearly 600 kilojoules) to 2040 (nearly 768 kilojoules) (EIA. 2017). These estimates are alarming for existing oil reserves, fuel prices, emission of greenhouse gases and pollution. In addition, the concerns about global warming, climate change, instability and issues on national security have led to rebirth in the invention of energy from renewable energy sources. Renewable energy (Sahu 2016) often referred to as clean energy has drawn attention as a potential alternative. Latest trend and government concern in energy policies support the greater use of renewable energy sources (i.e. wind, solar, tidal, geothermal, and biomass) with the goal of minimising greenhouse gas emissions and increasing energy efficiency. Biomass offers enormous possibilities for the production of biofuels which can reduce dependency on fossil fuel (Naiket al. 2010) without compromising the food security. This review focuses on various pretreatment methods used to produce biofuels as well as value-added products. Also, it elaborately explains recent advancement in each method and their limitations.
Lignocellulogic biomass
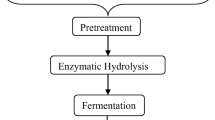
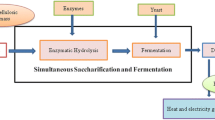
LCB is the most abundant and renewable natural bioresource derived from plants, food and agro-based industries as waste byproduct (Pérez et al., 2002a, b; Fatma et al., 2018). But, large quantity of LCB is either remain unused or disposed of by burning which aids pollution to the environment. LCB is composed of cellulose 40–60%, hemicellulose 25–35% and lignin 15–40% (Ragauskas et al., 2014; Chandel et al., 2018; Sharma et al., 2019). The restricted use of LCB is due to its structural complexity and recalcitrance nature, which reduces its accessibility for enzymatic hydrolysis (Horn et al. 2012; Zhang et al. 2007; Zavrel et al. 2009). The viable option for the large-scale use of LCB to produce biofuel (Preetha et al. 2020a) and value-added products is pretreatment, which improves and increases the surface accessibility for enzymatic hydrolysis (Preetha et al. 2020b). The pretreatment of LCB results in the disaggregation of cellulose and hemicelluloses from lignin (Fig. 1). The residues left after bioethanol production could be used as soil conditioner which reduces the need for materials manures (Singla and Inubushi 2014; Singla et al. 2013, 2014a, b).
LCB has some contributory roles in wastewater treatment (Preetha et al., 2020a). Nanocellulose, a simpler form of cellulose derived from LCB can be used as transparent paper, nano-composite and surface modified materials (Patchiya et al., 2018) (Table 1).
Biofuels and value-added products
Biofuels, an alternative source of fossil fuel which covers nearly 1.5% of global needs are cheap and eco-friendly. Release of greenhouse gases from biofuel is relatively very low in comparison to fossil fuel (Dolly et al., 2018). Biofuel comprises bioethanol, biomethanol, biobutanol, biodiesel and biogas. It has been categorised as 1st, 2nd and 3rd generations based on their source of origin (Dragone et al. 2010; Sekoai and Gueguim 2013). The first generation biofuel involves the edible parts of plants such as sugars, grains and seeds which are produced on agricultural land. It represents a step towards energy autonomy and weaning off fossil fuels for energy demands. The production of this is opposed by both economist and ecologist; as they create a threat to food security. Hence, the production of first-generation biofuels is controversial (Schenk et al. 2008). The second-generation biofuel source includes the non-edible parts of plants such as agricultural residue, non-food energy crops, woody crops and LCB. It has higher potential than first-generation biofuel since there is no competition for getting the raw materials and direct use of agricultural lands (Ohman et al. 2006). Bioethanol production from sugarcane, rice straw, corn straw etc. contributes about 65% of total biofuel. Limited reports are there for sustainable production of biomethanol from LCB (Mohanty et al. 2021) and is attracting researchers for its application in petrochemical industries, synthetic hydrocarbon products and blending gasoline (Chakrabortty et al. 2020). However, production of bioethanol from indigenous source of biomass by using auxotrophic strain is yet to explore. Bio-methane production by anaerobic fermentation of LCB seems economical and environmental friendly. It is an alternative and cost-competitive to natural gas used as energy source in various industries (Xu et al. 2019). Bio-butanol produced from LCB is equally important as bioethanol and is used as internal combusting as well as blending agent with gasoline (Iyyappan et al. 2021). Also, it has emerging demand in detergent formulations, pharmaceutical drugs and cosmetic industry. Biodiesel, an alternative source of petroleum diesel has potential to be used for transportation, energy generation, clean oil spills, paint removal and a lubricating agent (Chintagunta et al. 2021). Use of bio-hydrogen in engine and fuel cells produce high energy (nearly 122 kJ/g) (Sekoai and Gueguim 2013) and releases water as by-product which is safe for the environment. Algal fuel constitute the third generation of biofuels and may give rise to a good amount of bioethanol, biodiesel, butanol, propanol and gasoline, (Nigam and Singh 2010; Maia et al. 2020). The novel second-generation biofuels such as bioethanol, biobutanol, biomethanol, biogas and biohydrogen derived from LCB from agricultural residues and energy crops show much potential than 1st and 3rd generation biofuels because there remains no competition between food and fuel as they do not involve agricultural lands directly (Ohman et al. 2006). LCB is the most affordable, profoundly available, inexhaustible source of alternative fuel. Also, LCB has high potential to give rise various value-added products including but not limited to organic acids, biopolymers, rodenticide, animal feed, biofertilizer, fish feed, oil, facial toner, fragrance, soil conditioner (Table 2).
Pretreatment methods
The pretreatment of LCB is a necessary and vital step for the resourceful and viable conversion of cellulose/hemicelluloses into biofuel and other associated value-added products. Underlying objective of this process is to disintegrate lignin, hemicelluloses and cellulose into smaller fragments and to enhance the accessible surface area for enzymatic hydrolysis. The different pretreatment methods used are physical, chemical, physicochemical and biological. However, each pretreatment method has some merits and demerits. The physical method includes mechanical milling/grinding, ultrasound and microwave radiation are used for the breakdown of biomass but these processes involve high energy demand and hence are not economical. Whereas in chemical methods, the application of chemicals like acids, alkalis, ozone and organic solvent makes the process faster and efficient but the chemicals must be weeded out before hydrolysis which is quite expensive and the processes are also coupled with the production of harmful inhibitors. Nevertheless, the physicochemical methods such as ammonia fiber explosion, CO2 explosion steam explosion and liquid hot water treatment are currently used and are comparatively cost-effective techniques (Mosier et al. 2005). On the other hand, biological pretreatment (bacterial, fungal) or their combination offers a cost effective, eco-friendly with minuscule energy demand (Wan and Li 2012; Shi et al. 2011). It has been reported that physiochemical and biological methods of treatment are being used in textile industry. Physiochemical methods are well established, but biological method has better hope and looks promising not only for mitigation of waste effluent but also for lignocellulosic degradation and formation of value-added products (Meerambika et al. 2021). The key pretreatment methods used for the LCB are summarized (Fig. 2).
Following points may be considered before employing a particular pretreatment method: (1) the method should not reduce the particle size of biomass, (2) the structural integrity of hemicelluloses must not be affected, (3) generation of inhibitory products must be minimum, (4) low energy requirement and (5) should use low-cost pretreatment catalyst for regeneration of high-value lignin by-product. The biomass after pretreatment enables the downstream process of reducing operating cost, capital cost and biomass cost. Different types of pretreatment methods, their merits and demerits are discussed (Table 1).
Physical methods
Physical pretreatment of LCB is a prerequisite prior to enzymatic hydrolysis; it is basically carried out to decrease the degree of polymerization thereby reduce the particle size which further results the increased surface area and accessibility for enzymatic hydrolysis (Rajendran et al., 2017). As a result, the process becomes more effective and easier (Chen et al., 2017). The pretreatment method includes mechanical milling, ultrasound, microwave, pyrolysis and pulse electric field, which are eco-friendly and rarely produce any toxic compounds (Shirkavand et al., 2016). Physical pretreatment of LCB involves high energy consumption and the overall process affected by processing temperature, residence time and pressure. But, it shows synergetic effect when it applied with chemical pretreatment and gives better yield (Tu and Hallett 2019).
Mechanical pretreatment
The Mechanical pretreatment of LCB uses unique methods like milling, grinding, chipping and extrusion, however, the regularly used are milling, and grinding. Moreover, the desire of the approach is relying upon the moisture content of the biomass (Neshat et al. 2017). The mechanical pretreatment is a fundamental step for enhancing the bioconversion efficiency, particle densification, enzymatic accessibility and the overall conversion of LCB into biofuel without formation of harmful side streams. This pretreatment is not only increases the bulk density and porosity but also improves the surface area and flow property of the cellulosic matter. However, milling technique has been found to diminish the cellulose crystallinity and consequently enhance the digestibility of the lignocellulosic biomaterial. Various milling processes such as hammer milling, ball milling, colloid milling and two-roll milling and are used to improve the enzymatic hydrolysis of lignocelullosic materials (Taherzadeh and Karimi 2008).Moreover, the reduction of particle size and crystallinity is depends on the type of biomass used, type of milling method adopted and the processing time (Kumar and Sharma 2017). According to Bai et al. (2018), the pretreatment of wheat straw using rod milling has resulted significant size reduction and decrease in crystallinity at an optimum duration of 60 min over other milling methods. The power requirement for mechanical pulverisation of agricultural resources depends on the quality of plant materials and dimension of the final particle (Sun and Tomkinson 2002; Mosier et al. 2005; Hendriks et al. 2009). The process of comminution is highly energy-intensive. The mechanical properties of the biomass must be known for the design and optimisation of biomass grinding equipment, since the initial particle size, feed rate of the material, moisture content, and machine variables significantly influence the energy requirements for reducing the size of lignocellulosic material (Mayer-Laigle et al. 2018a, b). The milling technique does not generate any toxic inhibitors, like hydroxyl methyl furfural (HMF) and furfural and hence it is an appropriate method for production of both methane and ethanol. However, due to high-energy demand for milling and skyrocketing energy costs, milling is not positioned to be an economically viable option (Mayer-Laigle et al. 2018a, b).
Ultrasound
The application of ultrasound waves is an alternative process for the pretreatment of lignocellulosic material. It is based on the principle of cavitation through the application of ultrasonic radiation, in which cavitation produces shear forces that breaks the complex structure of LCB and promotes the extraction of desired component like cellulose, hemicelluloses and or lignin (Ravindran and Jaiswal 2016). This process offers lesser processing periods, low operating temperature, squeezed fermentation time and low chemical usage. The efficiency of this method relies upon the selection of ultrasonic frequency, time, process temperature and type of material used (Gómez et al. 2011; Liu et al. 2003; Lin et al. 2010), solvent used and also reactor design (Bussemaker et al. 2013). By the application of ultrasound frequency of 20–40 kHz, the internal hydrogen bonds of the biopolymers get broken and results swelling, loosening and rupturing of fibers (Bussemaker et al. 2013). The best delignification results were noticed for ultrasound frequency of 40 kHz, with respect to fragmented biomass (Bussemaker et al. 2013). According to El Achkar et al. 2018, Production of biomethane from grape pomace has significantly improved using ultrasound frequency of 50 kHz and temperature of less than 25 °C with a residence time of 40–70 min. Luo et al. 2014 reported that the use of ultrasound can reduce the hydrolysis time of biomass by up 80% aiding profit the biofuel production. However, the process is energy-intensive which needs a detailed study to optimize the process parameters for large scale application.
Microwave pretreatment
Microwave or electromagnetic radiation of frequency range between infrared to radio waves of energy 300–700 W/m3 is normally applied for the pretreatment of lignocellulosic material. By which, the material absorbs the radiation energy, gets excited and shows different modes of molecular vibrations, but the energy is not sufficient to break the chemical bonds. Moreover, this process generates thermal energy which minimizes the recalcitrance of biomass thus improving the efficiency of hydrolysis of biomass (Urbaniec and Grabarczyk 2009). The main benefits of microwave heating over traditional heating are lower power consumption, shorter reaction time and no direct contact with the feedstock (Kostas et al. 2017). Microwave treatment for longer time also increases rate of polysaccharide degradation (Bhutto et al. 2017). Use of microwave radiation before chemical hydrolysis of lignocellulosic material allows the cellulose content to attain around 56% in the hydrolysate (Nomanbhay et al. 2013). When the microwave radiation (400 W for 20 min) is applied in conjunction with either alkaline or acid pretreatment (1%, 2% or 3% sodium hydroxide or sulfuric acid), then the content of reducing sugar in hydrolysate can be accelerated fourfold (Diaz et al. 2013). The results of microwave treatment recommend the use of this technique for the production of fermentable sugars from LCB.
Pyrolysis
Pyrolysis is a thermal degradation process involves thermo-chemical decomposition of LCB at above 300 °C, as a result cellulose is disintegrated into residual char and gaseous products. According to Shafizadeh and Bradbury 1979 and Fan et al. 1987 dilute acid hydrolysis of residual char resulted in more than 80% decomposition of cellulose to reducing sugars. Moreover, the efficiency of the method can be enhanced under oxygen-rich conditions (Zwart et al. 2006), with the aid of some specific catalysts such as sodium carbonate or zinc chloride (Sun and Tomkinson 2002; Kumar et al. 2009; Nomanbhay et al. 2013). Pyrolysis is a less energy-consuming endothermic process which further classified into slow, intermediate, fast and flash pyrolysis based on the required temperature and residence time (Case et al. 2015).
Extrusion
Extrusion is the newest and upcoming physical technique used for pretreatment of lignocellulosic material. In the process of extraction, the biomass is subjected to blending, heating and shearing which actuates chemical and physical modifications during transition through the extruder under high temperature (> 3000C). The combined effect of barrel temperature and screw speed are found to disrupt the lignocellulose structure resulting shortening and defibrillation of the fiber, which in turn increases carbohydrate accessibility for enzymatic hydrolysis (Kumar and Sharma 2017). The effectiveness of the technique can be improved by taking various parameters of the reactor into account (Han et al. 2020).
Pulsed-electric-field pretreatment
Pulsed electric field (PEF) pretreatment technique applies elevated electrical energy to plant materials or biomass placed between two electrodes. A rapid electrical breakdown and structural changes in cell wall and membrane occur when critical electric potential is actuated in a cell membrane during application of a high intensity of external electric field. The electric field produces remarkable rise in mass permeability and in certain cases, it causes mechanical rupture of the plant tissue. In biomass-to-fuel conversion, pretreatment of biomass with PEFs can expose the cellulose in the plant fibers. Plant cells can be significantly ruptured by the use of high field strengths in the range of 5–20 kV/cm (Nomanbhay et al. 2013). PEF pretreatment can produce permanent pores in the cell membrane by applying electric pulses of high field strengths and thereby enabling the ingress of enzymes or acids for breaking down the cellulose into its constituent sugars (Kumar and Sharma 2017). In the chemical modification of plant material, primarily in lignocellulose hydrolysis, suitable chemical agent needed to be introduced into the tissue to help in breakdown and digestion of cell wall. The main advantages of PEF pretreatment are, processing at ambient conditions, lower energy demand (due to very short pulse time 100 μs), does not use any mechanical parts and no complex equipment design is necessary. According to Kumar et al. (2009) and Nomanbhay et al. (2013), PEF pretreatment of switchgrass has shown considerable enhancement in the enzymatic digestibility of the substrate.
Chemical methods
Acid hydrolysis
In this process, the lignocellulosic material is dried and subjected to submergence in acidic solution under certain temperature for a particular time and further, the treated material is filtrated to isolate the liquid from unhydrolysed solid substrate. The acid hydrolysis basically involves the addition of either concentrated or dilute acids of 0.2 w/w % to 2.5 w/w % to the biomass with continuous mixing from 130 to 201 °C. The acids such as hydrochloric acid (HCL), sulphuric acid (H2SO4) or nitric acid (HNO3) used as powerful agents for hydrolysis of cellulose. The dilute acid hydrolysis is performed in two different environments, such as high temperature (T > 160 °C), continuous flow process for low solid load (5–10%) and low temperature (T ≤ 160 °C), batch process for high solid load (10–40%) (Naseeruddin et al. 2013). In strong acid hydrolysis, concentrated acids like hydrochloric acid (HCL), sulphuric acid (H2SO4) are used without subsequent enzymatic hydrolysis. The pretreatment by concentrated mineral acids is more toxic, produce toxic inhibitors and corrosive and hence the process requires corrosion-resistant reactor. The recovery of concentrated acids makes the process expensive (Sivers and Zacchi 1995; Torget et al. 2000). However, hydrolysis using dilute acids found to be effectual for the pretreatment of lignocellulosic material. Hydrolysis by dilute sulphuric acid not only increases reaction rate but also enhances cellulose hydrolysis (Gupta et al. 2011). The main advantage of this method is high solubility of hemicellulose and lignin in acids which enhances the yield of glucose without the need for subsequent enzymatic hydrolysis which proves the beneficial effect of this method (Kumar et al. 2009; Peinemann and Pleissner 2020). Moreover, a high concentration of hydroxyl methyl furfural (a toxic inhibitor) is produced which essentially reduces the efficacy of this method (Sun and Cheng 2005).
Alkaline hydrolysis
The alkaline hdrolysis is carried out by soaking LCB in alkaline solutions such as potassium, calcium, ammonium hydroxide and sodium at appropriate temperature and incubated for a specific time period. This causes enlargement of lignocellulosic material, leading to increased internal surface area and results in the fractional decrystalization of cellulose, incomplete solvation of hemicelluloses and change in the organization of lignin (McMillan 1994; Lorenci et al. 2020). Alkaline process uses less harsh conditions as compared to other methods of pretreatment and showed promising results on corn stover, switchgrass, wheat, bagasse, softwood, rice straw and hardwood. However, the effectiveness of this process largely depends on the lignin content of the biomass (Shafizadeh and Bradbury 1979; McMillan 1994; Sun and Cheng 2002; Mosier et al. 2005; Kumar et al. 2009; McIntosh and Vancov 2010; Gupta et al. 2011a). The main demerits of this methods are, long residence time and neutralization of the pre-treated slurry (Wan et al. 2011).
Ionic liquids
In ionic liquid pretreatment the biomass is subjected to solubilisation in ionic liquids at 90°C–130°C under normal pressure and allowed to precipitate by adding water to the biomass and finally the precipitate is washed. The structure of lignin and hemicelluloses is not affected after treatment of the biomass which allows selective extraction of lignin, since, lignin is highly soluble in ionic solvents whereas cellulose is less soluble. This property helps in lignin separation and also enhances the accessibility of cellulose under ambient pressure and temperature without formation of toxic inhibitors, acid or alkaline agents. The ionic liquids are salts that possess heterogeneous structure comprised of both organic cations and inorganic anions. These are now increasingly used as novel solvents for the pretreatment of LCB. These ionic liquids have high thermal stability, high polarity and low vapour pressure. But, some properties of ionic liquids are not conducive of pretreatment, for example, chloride-based ILs such as 1-butyl-3methylimidazolium chloride are lethal, corrosive and hygroscopic, while others such as 1-allyl-3-3methylimidazolium chloride is viscous with reactive side chains (Xie et al. 2012). Thus, it is essential to leverage the properties of solvents for biomass pretreatment by desirable change in its structure and preparation method (Zhang et al. 2017).In fact phosphate-based solvents have higher thermal stability, lower viscosity and toxicity than chloride-based solvents (Mora-Pale et al. 2011). Though the solvents are costly but the recovery cost is not high due to its low vapour pressure. However, in ionic solvents, the enzyme cellulase is permanently inactivated (Lynd et al. 1996), which significantly brings down the efficiency of biomass conversion and escalates the cost as well (Haldar and Purkait 2021). This drawback may be addressed by developing suitable ionic solvents in which microorganisms and cellulase remain unaffected.
Organosolv process
The organosolv method employs various solvents which include organic solvents like acetone, methanol, ethylene glycol, ethanol, triethylene glycol or their combination with water and organic acids such as salicylic acid, acetylsalicylic acid and oxalic acid (Sun and Cheng 2002; Itoh et al. 2003; Xu et al. 2003; Kumar et al. 2009; Zhao et al. 2009; Gupta et al. 2011a). The pretreatment of biomass is carried out at room temperature and pressure with or without catalysts to remove lignin and hydrolyze hemicellulose for enhanced degradability of cellulose (Mosier et al. 2005; Sun et al. 2021). The solvents used in this method should be removed from the reactor before fermentation, though it is very expensive, it is required since the solvents are inhibitory for the growth of microorganisms, enzymatic hydrolysis and subsequently fermentation (Itoh et al. 2003; Xu et al. 2003; Zhao et al. 2009; Gupta et al. 2011a). This process provides a better quality lignin that is useful in producing different value-added products.
Oxidative delignification
The pretreatment of LCB using oxidizing agents such as oxygen, ozone or hydrogen peroxide significantly enhances its susceptibility to enzymatic hydrolysis. In this method, the lignin is extracted from the biomass for the enhancement of cellulose accessibility (Hendriks and Zeeman 2009). Lignin extraction is often associated with the loss of hemicellulose and cellulose. Moreover, the oxidising agents with aromatic rings convert lignin polymer to carboxylic acids, which inhibits the growth of microorganism. Unlike other methods, this method has incited less interest among industry and researchers because of the use of expensive oxidizing agents.
Ozonolysis
Ozone pretreatment method offers a great choice for the reduction of lignin in LCB. In this process, ozone acts as a strong oxidant to degrade lignin and is mostly confined to lignin, whereas hemicellulose is partially impacted but the cellulose stays majorly unaffected which results in an increased digestibility of cellulosic biomass (Kumar et al. 2009). This process accounts 60% lignin removal from wheat straw and it further enhances the enzymatic saccharification rate by almost 5 times (Vidal and Molinier 1988) The key advantages of this method are significant removal of lignin, no formation of toxic inhibitors, processing at ambient temperature and pressure (García-Cubero et al. 2009; Kumari and Singh 2018). However, this method requires huge quantities of ozone thus rendering it economically unviable.
Physicochemical methods
Physicochemical pretreatment is a combined approach of physical and chemical methods applied for the processing of LCB. This includes carbon dioxide explosion, steam explosion, ammonia fibre explosion, and liquid hot water treatment.
Carbon dioxide explosion
CO2 explosion is an efficient method which uses the combination of steam and supercritical CO2 for the pretreatment of LCB. The CO2 molecule has similar size property as water so it is easily introduced into the biomass under high pressure through a nozzle by which carbon dioxide molecules enter into the tiny pores of LCB because and rupture the structure of LCB (Puri and Mamers 1983; Lü et al. 2013). By the release of high-pressure CO2, the cellulosic structure gets disrupted which eventually increases the substrate accessibility to cellulolytic enzymes for hydrolysis (Haghighi et al. 2013; Kumar et al. 2009; Kumar and Sharma 2017). Since carbon dioxide pretreatment is carried out at low temperature; it prevents sugar decomposition by acid. Dale and Moreira (1982) concluded with experiments that CO2 pretreatment on alfalfa enabled 75% release of glucose. This method is significantly cost-effective, non-toxic, non-flammable, higher conversion yield and is known to decrease greenhouse effect. The main demerit of this method is high processing cost (Julie Baruah et al. 2018).
Steam explosion
In this method of pretreatment, the LCB is exposed to high-pressure saturated steam at temperature between 160 °C -240 °C and with corresponding pressure range of 0.7–4.8 Mpa (Agbor et al. 2011) for few minutes followed by abrupt release of pressure, which causes explosive decomposition of material (Hendriks and Zeeman 2009) resulting in fractional hydrolysis of hemicellulose with release of acetic acid. Several parameters such as particle size, moisture content, residence time and temperature determine the effectiveness of steam explosion (Talebnia et al. 2010). The particle size and initial composition of the biomass decide the time–temperature correlation of this process (Viola et al. 2008). According to Duff and Murrayh 1996 hydrolysis and hemicellulose solubilisation can be consummated by either low temperature and long residence time (190 °C, 10 min) or high temperature and short residence time (270 °C, 1 min). These two parameters ultimately influence physical accessibility, type of raw material as well as the pretreatment strategy.
The key advantages of this method are shorter residence period, lower energy consumption and no use of chemicals but the method is also associated with some limitations like degradation of xylan into hemicellulose, inefficient lignin removal and production of toxic inhibitors at high temperature (Bhutto et al. 2017).
Ammonia fiber explosion
The ammonia fiber explosion procedure is quite analogous to steam explosion method. In this method, LCB is exposed to liquid ammonia at temperature 90 °C with pressure around 0.7–2.7Mpa for about 30 min of residence time (Menon and Rao 2012). The presence of ammonia in these induced conditions results biomass swelling, change in the lignin structure, increase in accessible surface area and degradation of hemicellulose to oligomeric sugars. The increased accessible surface area and improvements in lignin structure increase the water retention potential and promote the digestibility of feedstock. Before pyrolysis, ammonia fibre explosion process is widely used to pretreat switchgrass, prairie cordgrass, and corn stover. However, the effect of the pretreatment was found to be marginal and did not significantly improve the properties of the treated material. Parameters such as moisture content, ash content, volatile matter and heating values are more or less similar before and after treatment. In the ammonia fibre explosion method, the process is carried out in a corrosive reaction environment like acid pretreatment, for which it requires suitable reactors and hence the efficiency of the process is also low (Bhutto et al. 2017). Moreover, the major drawback associated with this method is use of excessive ammonia, need for corrosive resistant reactor and its high recovery cost which consequently aggravate the cost of pretreatment thereby rendering it economically unviable.
Liquid hot water (LHW)
The liquid hot water Pretreatment of lignocellulosic material is the hydrothermal process causes degradation and dissolution of hemicelluloses resulting removal of lignin and increasing the accessibility of cellulose for hydrolysis (Agbor et al. 2011). Based on the process temperature the hydrothermal process is classified into liquid hot water pretreatment (LHW), pressurized hot water extraction (PHWE), hydrothermal carbonization (HTC), hot water extraction (HWE) and hydrothermal liquefaction (HTL). The applied temperature 140–230 °C in the LHW process is slightly higher than in PHWE which results in partial dissolution of hemicellulose, lignin and reduction of the structure. Some of the extractable water-soluble biomass components, like pectin and tannin are obtained during the low-temperature process where the process temperature is kept below the boiling point of water (100 °C). The PHWE is carried out at 150–180 °C range and is used for pretreatment of pulp dissolving process of biomass resulting in reduction of hemicellulose content of forest biorefinery products. The HTC (180–250 °C ), on the other hand, is used to transform biomass to modern carbon materials with specific physicochemical properties. The HTC process involves hydrolysis, dehydration, decarboxylation, polymerisation, aromatisation, and condensation reactions. Biocrude, an oil-like product is produced during HTL process at temperature above 280 °C. The liquid hot water treatment is also an effective method for the pretreatment of lignocellulosic material. The process uses hot compressed water with temperature (160–260 °C) and pressure (upto 5Mpa) without the aid of any chemicals and catalysts. On the basis of flow direction of biomass and flow direction of water into the reactor, liquid hot water pretreatment can be carried out in three different approaches. The first one is co-current pretreatment, in which the biomass slurry is heated in water at high temperature under-regulated residence time finally exposed to cold environment. The second approach uses the counter-current pretreatment in which hot water pumped against biomass under regulated conditions. The third approach is flow-through pretreatment, which uses flowing hot water by passing it through LCB thereby acting as a stationary bed. The efficiency of LHW pretreatment is based on pH of the reaction which should be maintained between 4 and 7. A slight change in pH that is acidic or neutral environment can convert hemicellulose into the form of oligomers which minimizes the formation of mono sugars and other additional products due to their degradation (Hendriks and Zeeman 2009). The advantages of these methods are, process is carried out at low temperature which results in lesser energy demand and no use of chemical agents. On the other hand, the limitation of this method is generation of various products with relatively low concentration due to dilution in water (Bhutto et al. 2017).
Biological method
The pretreatment of biomass using biological agents, typically microorganisms such as bacteria, fungi or an effectual combination. These microorganisms commonly produce the enzymes that degrade hemicelluloses, cellulose and lignin. The fungus mainly includes white, brown-rot fungi, actinomycetes and basidomycetes. Some white-rot fungi like P.chryosporium, Ceriporia lacerate, Cyathus stroller, Pycnoporous annarbarinus, Ceriporia subvermispora and Pleurotus ostreaus have shown significant effectiveness of delignification on various LCB (Zhou and Ingram 2000; Pérez et al. 2000; Béguin and Aubert 1994). Similarly, the bacteria such as Cellulomonas fimi and Thermoonospora fusca have been used for cellulase production and some anaerobic bacteria like Clostridium thermo cellulun and Bacteroids cellulosolvens have shown high cellulose activity but the enzymatic concentration is low (Paudel and Qin 2015). The biological pretreatment is mostly influenced by physical (temperature, size of the particles), chemical (pH) and biological (bacteria or fungi) conditions of the process (Sharma et al. 2019). The biological methods sought much attention in recent years because of their potential advantages over physical and chemical pretreatment techniques such as minimal energy consumption, high yield of desired product, no formation of toxic inhibitors and environmental friendly (Picart et al. 2015; Mandels and Reese 1960). But it is also associated with a few drawbacks such as partial hydrolysis of hemicelluloses, slower processing, microbial consumption of not only lignin but also the end product cellulose and hemicelluloses (Sukumaran et al. 2005; Ljungdahl 2008) setting up of commodious treatment areas (De Gonzalo et al. 2016). Thus, this method suffers from techno-economic challenges that render it commercially less attractive. However, the process of biomass treatment at this juncture demands more environmental friendly techniques. Therefore, more attention needs to be focused in this direction and a study showing the growing interest towards this ecofriendly biological process is encouraging (Arantes et al. 2007; Shary et al. 2008; Dashtban et al. 2009).
Conclusion
Lignocellulosic biomass is considered as major source of renewable energy. However, it is highly recalcitrant in nature due to unique arrangement of cellulose with hemicellulose-lignin matrix. Production of bioethanol and value-added products from LCB require delignification of biomass through pretreatment. Researcher throughout the globe has investigated to develop efficient physical, chemical, physiochemical and biological pretreatment methods. The chemical and physicochemical are found to be more effective in terms of cost and conversion. Analysis of each pretreatment method suggests us to the conclusion that each method has its merits and demerits depending on features and properties of biomass used. Hence, result of an individual pretreatment method is not reliable for maximum yield. To date, a benign and cost-effective pretreatment method that can disaggregate cellulose and hemicellulose from lignin is yet to be established. Therefore, it is crucial to understand biomass structure, composition and reaction mechanism of pretreatment methods. This may help to develop a sustainable and cost effective technology to minimize energy consumption and maximize productivity. On the basis of this review, we can suggest the right applications of LCB for production of optimum quantity of bioethanol and value-added products which can be achieved by the development of an economically viable and sustainable pretreatment method.
References:
Agbor VB, Cicek N, Sparling R, Berlin A, Levin DB (2011) Biomass pretreatment: fundamentals toward application. Biotechnol Adv 29:675–685. https://doi.org/10.1016/j.biotechadv.2011.05.005
Arantes V, Milagres MA, F, (2007) The synergistic action of ligninolytic enzymes (Mn P and laccase) and Fe3+ -reducing activity from white-rot fungi for degradation of Azure B. Enzyme MicrobTechnol 42:17–22. https://doi.org/10.1016/j.enzmictec.2007.07.017
Bai X, Wang G, Yu Y, Wang D, Wang Z (2018) Changes in the physicochemical structure and pyrolysis characteristics of wheat straw after rod-milling pretreatment. Bioresour Technol 250:770–776. https://doi.org/10.1016/j.biortech.2017.11.085
Baruah J, Nath BikashKar, Sharma R, Kumar S, Deka RC, Baruah DC, Kalita E (2018) Recent trends in the pretreatment of lignocellulosic biomass for value-added products. Front Energy Res. https://doi.org/10.3389/fenrg.2018.00141
Béguin P, Aubert JP (1994) The biological degradation of cellulose. FEMS Microbiol Rev 13:25–58. https://doi.org/10.1111/j.1574-6976.tb00033
Bhutto AW, Qureshi K, Harijan K, Abro R, Abbas T, Bazmi AA et al (2017) Insight into progress in pretreatment of lignocellulosic biomass. Energy 122:724–745. https://doi.org/10.1016/j.energy.2017.01.005
Bussemaker MJ, Xu F, Zhang D (2013) Manipulation of ultrasonic effects on lignocellulose by varying the frequency, particle size, loading and stirring. Bioresour Technol 148:15–23
Case PA, Truong C, Wheeler MC, De Sisto WJ (2015) Calcium-catalyzed pyrolysis of lignocellulosic biomass components. Bioresour Technol 192:247–252
Chakrabortty S, Nayak J, Ruj B, Pal P, Kumar R, Banerjee S, Sardar M, Chakraborty P (2020) Photocatalytic conversion of CO2 to methanol using membrane-integrated green approach: a review on capture, conversion and purification. J Environ Chem Eng. https://doi.org/10.1016/j.jece.2020.103935
Chandel AK, Garlapati VK, Singh AK, Antunes FAF, da Silva SS (2018) The path forward for lignocellulose biorefineries: bottlenecks, solutions, and perspective on commercialization. Bioresour Technol 264:370–381
Chen H, Liu J, Chang X, Chen D, Xue Y, Liu P et al (2017) A review on the pretreatment of lignocellulose for high-value chemicals. Fuel Process Technol 160:196–206
Chen WH, Nižetić S, Sirohi R, Huang Z, Luque R, M Papadopoulos A, Sakthivel R, Phuong Nguyen X, Tuan Hoang A. (2021) Liquid hot water as sustainable biomass pretreatment technique for bioenergy production: a review. Bioresour Technol. 26; 344(6):126207https://doi.org/10.1016/j.biortech.2021.126207
Chintagunta AD, Zuccaro G, Kumar M, Kumar SPJ, Garlapati VK, Postemsky PD, Kumar NSS, Chandel AK, Simal-Gandara J (2021) Biodiesel production from lignocellulosic biomass using oleaginous microbes: prospects for integrated biofuel production. Front Microbiol 12(12):658284. https://doi.org/10.3389/fmicb.2021.658284
Chundawat SPS, Pal RK, Zhao C, Campbell T, Teymouri F, Videto J, Nielson C, Wieferich B, Sousa L, Dale B. E, Balan, V, Chipkar S, Aguado J, Burke E, Ong R. G (2020) Ammonia fiber expansion (AFEX) Pretreatment of lignocellulosic biomass. https://doi.org/10.3791/57488
Dale BE, Moreira MJ (1982) Freezeexplosion technique for increasing cellulose hydrolysis. Biotechnol Bioeng Symp 12:31–43
Dashtban M, Qin SH, W, (2009) Fungal bioconversion of lignocellulosic residues; opportunities and perspectives. Int J Biol Sci 5:578–595. https://doi.org/10.7150/ijbs.5.578
Dawid M, Grzegorz K (2021) Microwave-assisted hydrotropic pretreatment as a new and highly efficient way to cellulosic ethanol production from maize distillery stillage. Appl Microbiol Biotechnol 105(8):3381–3392. https://doi.org/10.1007/s00253-021-11258-2
De Gonzalo G, Colpa DIH, MHM, Fraaije MW, (2016) Bacterial enzymes involved in lignin degradation. J Biotechnol 236:110–119. https://doi.org/10.1016/j.jbiotec.2016.08.011
De Paola MG, Mazza I, Paletta R, Lopresto CG, Calabrò V (2021) Small-scale biodiesel production plants. An Overview Energies 14:1901. https://doi.org/10.3390/en14071901
Den W, Sharma VK, Lee M, Nadadur G, Varma RS (2018) Lignocellulosic biomass transformations via greener oxidative pretreatment processes: access to energy and value-added chemicals. Front Chem 27(6):141. https://doi.org/10.3389/fchem.2018.00141
Diaz A, Le Toullec J, Blandino A, De Ory I, Caro I (2013) Pretreatment of rice hulls with alkaline peroxide to enhance enzyme hydrolysis for ethanol production. ChemEng Trans 32:949–954
Dragone D, Fernandes B, Vicente AA, Teixeira JA (2010) Third generation biofuels from microalgae. Current research, technology and education topics. In: A. Mendez-Vilas (ed) Applied microbiology and microbial biotechnology
Duff SJB, Murrayh WD (1996) Bioconversion of forest products industry waste cellulosic to fuel ethanol: a review. Bioresour Technol 55:1–33
Dussán K.J, Justo O.R, Perez V.H. et al. (2019) Bioethanol production from sugarcane bagasse hemicellulose hydrolysate by immobilized S. Shehatae in a Fluidized bed fermenter under magneticfield. Bioenerg. Res. 12, 338346 https://doi.org/10.1007/s12155-019-09971-y
EIA (2017) International energy outlook. US Department of Energy, Energy Information administration, Washington, DC
El Achkar JH, Lendormi T, Salameh D, Louka N, Maroun RG, Lanoisellé JL, Hobaika Z (2018) Influence of pretreatment conditions on lignocellulosic fractions and methane production from grape pomace. BioresourTechnol 247:881–889
Fatma S, Hameed A, Noman M, Ahmed T, Shahid M, Tariq M, Sohail I, Tabassum R (2018) Lignocellulosic biomass: a sustainable bioenergy source for the future. Protein Pept Lett 25(2):148–163. https://doi.org/10.2174/0929866525666180122144504
Flores EMM, Cravotto G, Bizzi CA, Santos D, Iop GD (2021) Ultrasound-assisted biomass valorization to industrial interesting products: state-of-the-art, perspectives and challenges. Ultrason Sonochem 2:105455. https://doi.org/10.1016/j.ultsonch.2020.105455
García-Cubero MT, González-Benito G, Indacoechea I, Coca M, Bolado S (2009) Effect of ozonolysispretreatment on enzymatic digestibility of wheat and rye straw. BioresourTechnol 100:1608–1613
Gasser E et al (2014) Microbial production of biopolymers from the renewable resource wheat straw. J Appl Microbiol 117(4):1035–1044
Gómez X, Fernández C, Fierro J, Sánchez ME, Escapa A, Morán A (2011) Hydrogen production: two stage processes for waste degradation. BioresourTechnol 102:8621–8627
Gupta R, Khasa YP, Kuhad RC (2011) Evaluation of pretreatment methods in improving the enzymatic saccharification of cellulosic materials. Carbohypolym 84:1103–1109
Haghighi MS, Hossein Golfeshan A, Tabatabaei M, SalehiJouzani G, Najafi GH, Gholami M et al (2013) Lignocellulosic biomass to bioethanol, a comprehensive review with a focus on pretreatment. Renew Sustain Ener Rev 27:77–93
Haldar D, Purkait MK (2021) A review on the environment-friendly emerging techniques for pretreatment of lignocellulosic biomass: mechanistic insight and advancements. Chemosphere 264(Pt2):128523. https://doi.org/10.1016/j.chemosphere.2020.128523
Han SY, Park CW, Endo T et al (2020) Extrusion process to enhance the pretreatment effect of ionic liquid for improving enzymatic hydrolysis of lignocellulosic biomass. Wood SciTechnol 54:599–613. https://doi.org/10.1007/s00226-020-01170-9
Hendriks ATWM, Zeeman G (2009) Pretreatments to enhance the digestibility of lignocellulosic biomass. BioresourTechnol 100(1):10–18. https://doi.org/10.1016/j.biortech.2008.05.027
Horn S, Vaaje-Kolstad G, Westereng B, Eijsink VG (2012) Novel enzymes for the degradation of cellulose. Biotechnol Biofuels 5:45. https://doi.org/10.1186/1754-6834-5-45
Itoh H, Wada M, Honda Y, Kuwahara M, Watanabe T (2003) Bioorganosolve pretreatments for simultaneous saccharification and fermentation of beech wood by ethanolysis and white rot fungi. J Biotech 103:273–280
Iyyappan J, Bharathiraja B, Vaishnavi A, Prathiba S (2021) Overview of current developments in biobutanol production methods and future perspectives. Methods Mol Biol 2290:3–21. https://doi.org/10.1007/978-1-0716-1323-8_1
Jennita JP, Shenbaga Muthuraman V, Karthick C, Alaswad A, Velvizhi G, Nanthagopal K (2021) Catalytic Microwave Preheated Co-pyrolysis of lignocellulosic biomasses: a study on biofuel production and its characterization. Bioresour Technol 19:126382. https://doi.org/10.1016/j.biortech.2021.126382
Kogo T, Yoshida Y, Koganei K, Matsumoto H, Watanabe T, Ogihara J, Kasumi T (2017) Production of rice straw hydrolysis enzymes by the fungi Trichoderma reesei and Humicolainsolens using rice straw as a carbon source. Bioresour Technol 233:67–73. https://doi.org/10.1016/j.biortech.2017.01.075
Kostas ET, Beneroso D, Robinson JP (2017) The application of microwave heating in bioenergy: a review on the microwave pretreatment and upgrading technologies for biomass. Renew Sustain Energy Rev 77:12–27. https://doi.org/10.1016/j.rser.2017.03.135
Kumar AK, Sharma S (2017a) Recent updates on different methods of pretreatment of lignocellulosic feed stocks: a review. Bioresour Bioprocess 4:7. https://doi.org/10.1186/s40643-017-0137-9
Kumar P, Barrett DM, Delwiche MJ, Stroeve P (2009) Methods for pretreatment of lignocellulosic biomass for efficient hydrolysis and biofuel production. IndEngChem Res 48:3713–3729
Kumari D, Singh R (2018) Pretreatment of lignocellulosic wastes for biofuel production: a critical review. Renew Sust Energy Rev 90:877–891. https://doi.org/10.1016/j.rser.2018.03.111
Li P, He C, Li G, Ding P, Lan M, Gao Z, Jiao Y (2020) Biological pretreatment of corn straw for enhancing degradation efficiency and biogas production. Bioengineered 11(1):251–260. https://doi.org/10.1080/21655979.2020.1733733
Lin Z, Huang H, Zhang H, Zhang L, Yan L, Chen J (2010) Ball milling pretreatment of corn stover for enhancing the efficiency of enzymatic hydrolysis. ApplBiochemBiotechnol 162:1872–1880
Liu H, Zhang T, Fang HHP (2003) Thermophilic. H2 production from a cellulose-containing wastewater. Biotechnol Lett 25:365–369
Ljungdahl LG (2008) Thecellulase/hemicellulase system of the anaerobic fungus Orpinomyces PC-2 and aspects of its applied use. Ann NY Acad Sci 1125:308–321. https://doi.org/10.1196/annals.1419.030
Lorenci Woiciechowski A, Dalmas Neto CJ, de Souza P, Vandenberghe L, de CarvalhoNeto DP, Novak Sydney AC, Letti LAJ, Karp SG, Zevallos Torres LA, Soccol CR (2020) Lignocellulosic biomass: acid and alkaline pretreatments and their effects on biomass recalcitrance—conventional processing and recent advances. Bioresour Technol 304:122848. https://doi.org/10.1016/j.biortech.2020.122848
Lü H, Ren M, Zhang M, Chen Y (2013) Pretreatment of corn stover using supercritical CO2 with water-ethanol as cosolvent. Chin J Chem Eng 21(5):551–557
Luo J, Fang Z, Smith RL Jr (2014) Ultrasound-enhanced conversion of biomass to biofuels. Prog Energy Combust Sci 41:56–93. https://doi.org/10.1016/j.pecs.2013.11.001
Maia JLD, Cardoso JS, Mastrantonio DJDS, Bierhals CK, Moreira JB, Costa JAV, Morais MG (2020) Microalgae starch: a promising raw material for the bioethanol production. Int J BiolMacromol. 15(165(Pt B)):2739–2749. https://doi.org/10.1016/j.ijbiomac.2020.10.159
Mandels M, Reese ET (1960) Induction of cellulase in fungi by cellobiose. J Bacteriol 79:816–826
Mayer-Laigle C, Blanc N, Rajaonarivony RK, Rouau, X (2018a) Comminution of dry lignocellulosic biomass, a review: part I. From fundamental mechanisms to milling behaviour. Bioengineering (Basel). 2;5(2):41. https://doi.org/10.3390/bioengineering5020041
Mayer-Laigle C, Rajaonarivony RK, Blanc N, Rouau X (2018b) Comminution of dry lignocellulosic biomass: Part II. technologies improvement of milling performances, and security issues. Bioengineering (Basel). 22; 5(3):50. https://doi.org/10.3390/bioengineering5030050.
McIntosh S, Vancov T (2010) Enhanced enzyme saccharification of Sorghum bicolour straw using dilute alkali pretreatment. Biores Technol. https://doi.org/10.1016/j.biortech.2010.03.116
McMillan JD (1994) Pretreatment of lignocellulosic biomass. ACS Symp Ser 566:292–324
Meneses DB, Montes De Oca-Vásquez G, Roberto Vega-Baudrit J, Rojas-Álvarez M, Corrales-Castillo J, Murillo-Araya LC (2020) Pretreatment methods of lignocellulosic wastes into value-added products: recent advances and possibilities. Biomass Convers Biorefin
Menon V, Rao M (2012) Trends in bioconversion of lignocellulose: biofuels, platform chemicals and biorefinery concept. Prog Energy Combust 38(4):522–550
Mohanty P, Singh PK, Adhya TK et al (2021) (2021) A critical review on prospects and challenges in production of biomethanol from lignocellulose biomass. Biomass Conv Bioref. https://doi.org/10.1007/s13399-02101815-0
Mora-Pale M, Meli L, Doherty TV, Linhardt RJ, Dordick JS (2011) Room temperature ionic liquids as emerging solvents for the pretreatment of lignocellulosic biomass. Biotech Bioengg 108(6):1229–2124
Mosier N, Wyman C, Dale B, Elander R, Lee YY, Holtzapple M, Ladisch M (2005) Features of promising technologies for pretreatment of lignocellulosic biomass. BioresourTechnol 96:673–686. https://doi.org/10.1016/j.biortech.2004.06.025
Naik SN, Goud VV, Rout PK, Dalai AK (2010) Production of first and second generation biofuels: a comprehensive review. Renew Sustain Ener Rev 14(2):578–597
Naseeruddin S, Yadav KS, Sateesh L, Manikyam, (2013) A. Selection of the best chemical pretreatment for lignocellulosic substrate ProsopisJuliflora. Biores Technol 136:542–549
Neshat SA, Mohammadi M, Najafpour GD, Lahijani P (2017) Anaerobic co-digestion of animal manures and lignocellulosic residues as a potent approach for sustainable biogas production. Renew Sus Energy Rev 79:308–322
Nigam PS, Singh A (2010) Production of liquid biofuels from renewable resources. Prog Energy Combust Sci 37(1):52–68
Ning P, Yang G, Hu L, Sun J, Shi L, Zhou Y, Wang Z, Yang J (2021) Recent advances in the valorization of plant biomass. Bioethanol Biofuels. 23;14(1):102. https://doi.org/10.1186/s13068-021-01949-3
Nomanbhay SM, Hussain R, Palanisamy K (2013) Microwave-assisted alkaline pretreatment and microwave assisted enzymatic saccharification of oil palm empty fruit bunch fiber for enhanced fermentable sugar yield. J Sustain BioenerSyst 3:7–17
Ohman M, Boman C, Hedman H, Eklund R (2006) Residential combustion performance of pelletized hydrolysis residue from lignocellulosic ethanol production. Ener and Fuels 20(3):1298–1304
Panda SK, Ray RC, Mishra SS, Kayitesi E (2018) Microbial processing of fruit and vegetable wastes into potential biocommodities: a review. Crit Rev Biotechnol 38(1):1–16. https://doi.org/10.1080/07388551.2017.1311295
Paudel YP, Qin W (2015) Characterization of ovelellulase-producing bacteria isolated from rotting wood samples. ApplBiochemBiotechnol 177:1186–1198. https://doi.org/10.1007/s12010-015-1806-9
Peinemann JC, Pleissner D (2020) Continuous pretreatment, hydrolysis, and fermentation of organic residues for the production of biochemicals. Bioresour Technol 295:122256. https://doi.org/10.1016/j.biortech.2019.122256
Pérez J et al (2002a) Biodegradation and biological treatments of cellulose, hemicellulose and lignin: an overview. Int Microbiol 5:53–63
Pérez J, Muñoz-Dorado J, de la Rubia T, Martínez J (2002b) Biodegradation and biological treatments of cellulose, hemicellulose and lignin: an overview. IntMicrobiol 5:53–63. https://doi.org/10.1007/s10123-002-0062-3
Picart P, de María PD, Schallmey A (2015) From gene to biorefinery: microbial β-etherases as promising biocatalysts for lignin valorization. Front Microbiol 6:916. https://doi.org/10.3389/fmicb.2015.00916
Prasad RK, Chatterjee S, Mazumder PB, Gupta SK, Sharma S, Vairale MG, Datta S, Dwivedi SK, Gupta DK (2019) Bioethanol production from waste lignocelluloses: a review on microbial degradation potential. Chemosphere 31:588–606. https://doi.org/10.1016/j.chemosphere.2019.05.142
Puri VP, Mamers H (1983) Explosive pretreatment of lignocellulosic residues with high-pressure carbon dioxide for the production of fermentation substrates. BiotechnolBioeng 25(12):3149–3161
Ragauskas AJ, Beckham GT, Biddy MJ, Chandra R, Chen F, Davis MF et al (2014) Lignin valorization: improving lignin processing in the biorefinery. Science 344:1246843
Rajendran K, Drielak E, Varma VS, Muthusamy S, Kumar G (2017) Updates on the pretreatment of lignocellulosic feedstocks for bioenergy production–a review. Biomass Conver Biorefin 8:471–483
Ravindran R, Jaiswal AK (2016) A comprehensive review on pretreatment strategy for lignocellulosic food industry waste: challenges and opportunities. Bioresour Technol 199:92–102. https://doi.org/10.1016/j.biortech.2015.07.106
Razaghi A, Karthikeyan OP, Hao HT, Heimann K (2016) Hydrolysis treatments of fruit and vegetable waste for production of biofuel precursors. Bioresour Technol 217:100–103. https://doi.org/10.1016/j.biortech.2016.03.041
Sahu S (2016) Conversion of cotton gin waste to bioethanol: pretreatment, hydrolysis and fermentation (Doctoral dissertation). National Institute of Technology Rourkela
Schenk P, Thomas-Hall S, Stephens E, Marx U, Mussgnug J, Posten C, Kruse O, Hankamer B (2008) Second generation biofuels: high-efficiency microalgae for biodiesel production. BioEner Res 1:20–43
Sekoai P, Gueguim KE (2013) A two-stage modelling and optimization of biohydrogen production from a mixture of agro-municipal waste. Int J Hydrog Energy 38(21):8657–8663
Shafizadeh F, Bradbury AGW (1979) Thermal degradation of cellulose in air and nitrogen at low temperatures. J Appl Poly Sci 23:1431–1442
Sharma HK, Xu C, Qin W (2019) Biological pretreatment of lignocellulosic biomass for biofuels and bioproducts: an overview. Waste Biomass Valori 10:235–251. https://doi.org/10.1007/s12649-017-0059-y
Sharma S, Jha PK, Panwar A (2021) Production of bioethanol from wheat straw via optimization of co-culture conditions of Bacillusli cheniformis and Saccharomy cescerevisie. Discov Energy 1:5. https://doi.org/10.1007/s43937-021-000044
Shary S, Kapich AN, Panisko EA, Magnuson JK, Cullen D, Hammel KE (2008) Differential expression in Phanerochaete chrysosporium of membrane-associated proteins relevant to lignin degradation. Appl EnvironMicrobiol 74:7252–7257. https://doi.org/10.1128/AEM.01997-08
Shi J, Qing Q, Zhang T, Wyman C, Lloyd T (2011) Biofuels from cellulosic biomass via aqueous processing. In: Ginley DS, Cahen D (eds) Fundamentals of materials for energy and environmental sustainability. Cambridge University Press, Cambridge
Shirkavand E, Baroutian S, Gapes DJ, Young BR (2016) Combination of fungal and physicochemical processes for lignocellulosic biomass pretreatment–a review. Renew Sust Energ Rev 54:217–234
Singla A, Inubushi K (2014) Effect of biochar on CH4 and N2O emission from soils vegetated with paddy. Paddy Water Environ 12:239–243
Singla A, Dubey SK, Iwasa H, Inubushi K (2013) Nitrous oxide flux from komatsuna (Brassica rapa) vegetated soil: a comparison between biogas digested liquid and chemical fertilizer. BiolFertil Soils 49:971–976
Singla A, Dubey SK, Singh A, Inubushi K (2014a) Effect of biogas digested slurry-based biochar on methane flux and methanogenic archaeal diversity in paddy soil. AgricEcosyst Environ 197:278–287
Singla A, Iwasa H, Inubushi K (2014b) Effect of biogas digested slurry based-biochar and digested liquid on N2O, CO2 flux and crop yield for three continuous cropping cycles of komatsuna (Brassica rapa var. perviridis). BiolFertil Soils 50:1201–1209
Sivers MV, Zacchi G (1995) A techno-economical comparison of three processes for the production of ethanol from pine. BioresouTechn 51:43–52
Sukumaran RK, Singhania RR, Pandey A (2005) Microbial cellulases—production, applications and challenges. J SciInd Res 64:832–844
Sun YE, Cheng JJ (2005) Dilute acid pretreatment of rye straw and Bermuda grass. BioresoTechnol 96:1599–1606
Sun RC, Tomkinson RC (2002) Characterization of hemicelluloses obtained by classical and ultrasonically assisted extractions from wheat straw. CarbohyPolym 50:263–271
Sun C, Ren H, Sun F, Hu Y, Liu Q, Song G, Abdulkhani A, LokeShow P (2021) Glycerol organosolv pretreatment can unlock lignocellulosic biomass for production of fermentable sugars: Present situation and challenges. Bioresour Technol 2:344(Pt B):126264. https://doi.org/10.1016/j.biortech.2021.126264
Suriyachai N, Weerasai K, Upajak S, Khongchamnan P, Wanmolee W, Laosiripojana N, Champreda V, Suwannahong K, Imman S (2020) Efficiency of Catalytic Liquid Hot Water Pretreatment for Conversion of Corn Stover to Bioethanol. 11;5(46):29872–29881. https://doi.org/10.1021/acsomega.0c04054
Taherzadeh MJ, Karimi K (2008) Pretreatment of lignocellulosic wastes to improve ethanol and biogas production: a review. Int J MolSci 9:1621–1651
Takano M, Hoshino K (2018) Bioethanol production from rice straw by simultaneous saccharification and fermentation with statistical optimized cellulase cocktail and fermenting fungus. BioresourBioprocess. https://doi.org/10.1186/s40643-018-0203-y
Talebnia F, Karakashev D, Angelidaki I (2010) Production of bioethanol from wheat straw: an overview on pretreatment, hydrolysis and fermentation. Bioresour Technol 101:4744–4753. https://doi.org/10.1016/j.biortech.2009.11.080
Torget RW, Kim JS, Lee YY (2000) Fundamental aspects of dilute acid hydrolysis/fractionation kinetics of hardwood carbohydrates I. Cellulose Hydrolysis Indus EnggChem Res 39:2817–2825
Tu WC, Hallett JP (2019) Recent advances in the pretreatment of lignocellulosic biomass. Curr Opin Green Sustain Chem. https://doi.org/10.1016/j.cogsc.2019.07.004
Ullah S, Noor RS, Sanaullah, et al (2021) Analysis of biofuel (briquette) production from forest biomass: a socioeconomic incentive towards deforestation. Biorefin Biomass Conv. https://doi.org/10.1007/s13399-021-01311-5
Urbaniec K, Grabarczyk R (2009) Raw materials for fermentative hydrogen production. J Clean Prod 17:959–962
Uyan M, Alptekin FM, Bastabak B et al (2020) Combined biofuel production from cotton stalk and seed with a biorefinery approach. Biomass Conv Biorefin 10:393–400. https://doi.org/10.1007/s13399-019-00427-z
Vidal PF, Molinier J (1988) Ozonolysis of lignin – improvement of in vitro digestibility of poplar sawdust. Biomass 16:1–17
Viola E, Cardinale M, Santarcangelo R, Villone A, Zimbardi F (2008) Ethanol from eel grass via steam explosion and enzymatic hydrolysis. Biomass Bioenergy 32:613–618. https://doi.org/10.1016/j.biombioe.2007.12.009
Wan C, Li Y (2012) Fungalpretreatment of lignocellulosic biomass. Biotechnol Adv 30:1447–1457. https://doi.org/10.1016/j.biotechadv.03.003
Wan C, Zhou Y, Li Y (2011) Liquid hot water and alkaline pretreatment of soybean straw for improving cellulose digestibility. Bioresour Technol 102:6254–6259. https://doi.org/10.1016/j.biortech.2011.02.075
Xie RQ, Li XY, Zhang YF (2012) Cellulose pretreatmentwith1-methyl-3-ethylimidazolium dimethyl phosphate for enzymatic hydrolysis. Cell ChemTechn 46(5–6):349–356
Xu F, Sun JX, Liu CF, Sun RC (2003) Comparative study of alkali and acidic organic solvent-soluble hemicellulosic polysaccharides from sugarcane bagasse. Carbohy Res 341:253–261
Xu N, Liu S, Xin F, Zhou J, Jia H, Xu J, Jiang M, Dong W (2019) Biomethane production from lignocellulose: biomass recalcitrance and its impacts on anaerobic digestion. Front Bioeng Biotechnol 8(7):191. https://doi.org/10.3389/fbioe.2019.00191.PMID:31440504;PMCID:PMC6694284
Zavrel M, Bross D, Funke M, Büchs J, Spiess AC (2009) High throughput screening for ionic liquids dissolving (ligno-) cellulose. BioresourTechnol 100:2580–2587. https://doi.org/10.1016/j.biortech.2008.11.052
Zhang YHP, Ding SY, Mielenz JR, Cui JB, Elander RT, Laser M, Himmel ME, McMillan JR, Lynd LR (2007) Fractionating recalcitrant lignocellulose at modest reaction conditions. BiotechnolBioeng 97:214–223. https://doi.org/10.1002/bit.21386
Zhang Q, Hu J, Lee DJ (2017) Pretreatment of biomass using ionic liquids: research updates. Renew Ener 111:77–84. https://doi.org/10.1016/j.renene.2017.03.093
Zhao X, Cheng K, Liu D (2009) Organosolvpretreatment of 260 lignocellulosic biomass for enzymatic hydrolysis. ApplMicrobio Biotech 82:815–827
Zhaoa Y, Damgaardb A, Christensenb TH (2018) Bioethanol from corn stover a review and technical assessment of alternative biotechnologies. Progress Energy Combust Sci 67:275–291
Zhou S, Ingram LO (2000) Synergistic hydrolysis of carboxymethyl cellulose and acid-swollen cellulose by two endoglucanases (CelZ and CelY) from Erwiniachrysanthemi. J Bacteriol 182:5676–5682. https://doi.org/10.1128/JB.182.20.5676-5682.2000
Zwart RWR, Boerrigter H, Drift A (2006) The impact of biomass pretreatment on the feasibility of overseas biomass conversion to Fisher–Tropsch. Prod Ener Fuels 20:2192–2197
Acknowledgements
We are greatly thankful to the management of GIET University for providing facilities. Also, we are thankful to Dr.Sushil Kumar Sahu, Asst.Professor, Dept. of Biotechnology, GIET University for valuable discussion during revision of the manuscript.
Funding
We did not use any funds in preparing this review article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors have declared that no competing interests exist.
Ethics approval
Not applicable as we have not used any animal model for any experiment. This is a review article.
Additional information
Editorial responsibility: M. Abbaspour.
Rights and permissions
About this article
Cite this article
Prasad, B.R., Padhi, R.K. & Ghosh, G. A review on key pretreatment approaches for lignocellulosic biomass to produce biofuel and value-added products. Int. J. Environ. Sci. Technol. 20, 6929–6944 (2023). https://doi.org/10.1007/s13762-022-04252-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-022-04252-2