Abstract
The effectiveness of electron and proton transport to anode and cathode is the key criteria in microbial fuel cell technology in order to improve the electricity generation. An innovative linked anode was designed to enhance the mass transfer of protons and electrons in the scaled-up up-flow membrane-less microbial fuel cell. The common cube anode was used to compare with the linked anode. The performance of voltage output for the cube anode and the linked anode was examined by various hydraulic retention times and the electrode spacing distances. The maximum power density of the linked anode was almost identical at all electrode spacing distances. Meanwhile, this result demonstrated that the configuration of linked anode has better directional fluid flow, mass transfer of protons and electrons, and voltage output (stationary phase) than those of the cube anode at all hydraulic retention times. The finding could suggest that the different configuration of bioanode in an up-flow membrane-less microbial fuel cell is an important factor to be considered for future real application.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Most of the present wastewater treatment plants are using conventional aerobic treatment system with oxygen supply (Vázquez et al. 2006; Adav et al. 2010; Kushwaha et al. 2011). The energy consumption in the wastewater treatment plant is generally contributed by aeration. Anaerobic treatment system without aeration is getting more popular for wastewater treatment due to less energy requirement. Hence, anaerobic treatment system is more cost-effective for high-strength wastewater. An alternative anaerobic treatment system with simultaneous electricity generation has gained more attentions from many researchers (Clauwaert et al. 2007; Yoshizawa et al. 2014; Saratale et al. 2017). Microbial fuel cell (MFC) is one of the promising anaerobic treatment systems that have demonstrated its capability for simultaneous wastewater treatment and electricity generation. Wastewater can be used as the sources of energy in MFC (Liu et al. 2004; Daniel et al. 2009). MFC generates electricity by using microorganism to oxidize the biodegradable organic substances in wastewater (Bennetto et al. 1983; Habermann and Pommer 1991; Min and Logan 2004). Higher concentration of organic wastewater has higher chemical energy per unit volume. Therefore, higher concentration of organic wastewater is required in order to achieve higher electricity production (Huang et al. 2011). Many researchers have proved that MFC can treat high concentration of COD (Pant et al. 2010; Liao et al. 2014; Sangeetha et al. 2016).
In recent years, the performance of MFCs has been improved gradually by various bioreactor designs and materials that are used for fabrication (Li et al. 2013; Fernando et al. 2014; Cui et al. 2014; Rahimnejad et al. 2015; Neoh et al. 2016). MFCs typically consist of biotic anode and abiotic cathode compartments with proton exchange membrane (PEM) as separator between the two compartments. Anode chamber will undergo microbial oxidation reaction, and cathode will undergo oxygen reduction process. PEM is used to allow only proton transfer from anode to cathode compartment and subsequently avoid the oxygen diffuse back to the anode compartment (Liu and Logan 2004; Ghasemi et al. 2013). Oxygen supply for the reduction at cathode region may diffuse to anode region in a MFC without using membrane (Wang et al. 2013). Oxygen diffusion may affect the activity of anaerobic microorganism at anode region (Logan et al. 2005). Subsequently, the voltage output may reduce because of insufficient electrons and protons.
Despite PEM having several benefits to MFC, the cost of the PEM is relatively high. Besides, PEM that is used over a period will cause biofouling that may reduce the efficiency of proton transfer through the membrane (Ghasemi et al. 2013). It is necessary to reduce the operating cost by eliminate the maintenance of membrane. Hence, a scaled-up MFC without using membrane is drastically reducing the cost of fabrication. Besides, membrane-less MFC may reduce the internal resistance due to less complexity design, and thus, it is simpler for industrial scaled-up. Despite recent design efforts, reducing operating costs remains one of the important factors for commercialization of scaled-up MFC. Most of the researchers reported in the past were using platinum as a catalyst in the cathode region. Therefore, the catalyst is necessary to reduce the fabrication cost of upscaled MFC. Recently, the use of biomass as a catalyst has been investigated as a low-cost alternative to platinum (Gajda et al. 2014; Bajracharya et al. 2016). Moreover, Rossi et al. (2019) increased the cathode size with a low-cost multi-panel air cathode that contained fifteen smaller cathodes welded into a single metal sheet to improve the power output in a large-scaled MFC.
One of the challenges in MFC is the effectiveness of electron transfer between microorganism and anode. It is used to define the potential of power output in MFC. Carbon-based electrodes are commonly utilized because of low cost and the high effectiveness of microbial attachment (Crittenden et al. 2006). The microbes attached on the surface of the carbon electrode will act as biocatalyst to improve the electron transfer efficiency. Meanwhile, mass transfer from anode to the cathode regions is an important factor to produce higher voltage output. There are several operating conditions, and configuration may affect the efficiency of mass transfer in MFC and includes volumetric flow rate, directional flow pattern, operation mode, ohmic resistance and substrate concentration (Nasharudin et al. 2014; Pasupuleti et al. 2016).
Two scaled-up up-flow membrane-less MFCs that use aerobic microorganism as a catalyst at the cathode region were designed and investigated in this study. The design of this scaled-up membrane-less MFC introduces an up-flow water body from the bottom of MFC, improves the mass transfer of protons and substrates from anode to the cathode regions and reduces oxygen diffusion from cathode to the anode regions. This study mainly demonstrates the efficiency of electron and proton transfer by a different configuration of anode in up-flow membrane-less MFC. Mass transfer efficiency was examined by using different hydraulic retention times (HRTs) and electrode spacing distances. The outcome of this study may be beneficial to the commercial application of membrane-less MFC in the wastewater treatment plant in the future.
The research was carried out between January and July 2017, at the Water Research Group (WAREG), School of Environmental Engineering, University Malaysia Perlis, Malaysia.
Materials and methods
Inoculum and substrates
Synthetic wastewater was used as fuel for this study. The composition of synthetic wastewater consists of sodium acetate (3.138 g/L) as the main organic matter, nutrient and buffer solution (pH 6.55) which contains: NH4Cl (0.31 g/L), KCl (0.13 g/L), K2HPO4 (3.4 g/L), KH2PO4 (4.4 g/L), MgCl2·6H2O (0.1 g/L), CaCl2·6H2O (0.1 g/L) and NaCl (0.116 g/L). Packing material and anode electrode were immersed in mixed culture activated sludge about one month for anaerobic bacteria cultivation. The activated sludge was obtained from a wastewater treatment plant of a rubber glove industry, Shorubber (Malaysia) Sdn. Bhd.
Fabrication of SUFML MFC reactor and operation
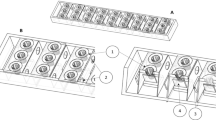
Two single-chamber scaled-up up-flow membrane-less microbial fuel cells (SUFML MFCs) were developed by using acrylic column (Fig. 1). The column of SUFML MFC has a diameter of 18 cm and height of 50 cm. The SUFML MFC was divided into a total of five sections. A total of three sections from the bottom of the reactor were anode region with three anode electrodes (A1, A2 and A3) at different distances (36, 27 and 18 cm) to the cathode region. The top section at the SUFML MFC was the cathode region. Another section was a junction region between the anode and cathode regions. The total void volume of the reactor was 6.9 L. Anode region and junction region were filled with biopacking material (1 cm diameter) for biofilm development. Aeration was supplied at the cathode region for the oxygen reduction process. A total of five sampling points (S1, S2, S3, S4 and effluent) were built in every section of the SUFML MFC. Carbon felt (SG-222, Maido Corporation, Japan) and carbon plate were used as anode and cathode electrode, respectively. Figure 2 illustrates the size of the two different configurations of anode (cube carbon felt and linked carbon felt) at the same total surface area, 328 cm2. The dimension of cathode carbon plate was 10 cm × 10 cm (L × W).
The SUFML MFC was operated in a semi-continuous mode with a peristaltic pump (BT100—100 M, Longer Precision Pump, China) at various flow rates of 2.396, 4.792 and 7.152 ml/min. A tank was placed on magnetic stirrer and connected to the influent and effluent. This allows the continuous recirculation flow of synthetic wastewater from the bottom anode region to the upper cathode region. The anode and cathode electrodes were connected with copper wire through 1000 Ω resistor. The SUFML MFCs were operated in the room temperature (28 °C).
Analytical methods
The voltage output was measured and recorded continuously at every hour by using a data logger (Graphtec GL820, USA). The polarization curve was obtained by using various resistors (50–20,000 Ω). All water samples that were collected from the anode region and cathode region were centrifuged (Cence L500, China) at 4200 rpm for 10 min before analysis. Spectrophotometer (DR 2800, Hach) was used to measure the chemical oxygen demand (COD) concentration. Dissolved oxygen (DO) was measured by using DO meter (HANNA HI 9146, USA). Ohm’s law (V = IR) was used for current (I) calculation where V and R represent the voltage and resistance, respectively. Current density (A/m2) and power density (W/m2) were calculated by the geometrical surface area of anode electrode. The Columbic efficiency (CE) was calculated by dividing the integrated total amount of experimental Coulombs of theoretical Coulombs transferred. \(CE = M\int {I{\text{d}}t} \left( {Fbv\Delta {\text{COD}}} \right)^{ - 1}\) where M is the molecular weight of substrate, I is the stable current, F is the Faraday’s constant (96,485 C/mol), b is the number of moles of electrons produced per mol of substrate, v is the volume liquid at anode and ∆COD is the difference in the influent and effluent COD (Liu and Logan 2004; Logan et al. 2006).
Results and discussion
Characteristics of scaled-up up-flow membrane-less MFC
A total of two SUFML MFCs were fabricated for this study (Fig. 1). These SUFML MFCs were configured with two different types of anode design, whereas cube anode and linked anode are shown in Fig. 2. Both anode configurations have the same total surface area but different in rigid volume. The synthetic wastewater was fed into SUFML MFC by using semi-continuous mode. The conductivity of the synthetic wastewater is important to provide stable operating condition for MFC (Feng et al. 2008). The conductivity of anode and cathode for both reactors was ranging from 11.92 to 12.50 mS/cm. DO in the cathode region was around 4.3 mgO2/L, whereas DO for anode region A1, A2 and A3 was 0, 0 and 0.62 mgO2/L, respectively. The supplied aeration was maintained with flow meter at 40 L/h.
Effect of an anode configuration on MFC performance at different HRTs
COD performance
In order to study the effect of anode configuration (cube anode and linked anode) on voltage output and COD reduction, the SUFML MFCs were operated under semi-continuously at three different HRTs (2, 1 and 0.67 d), corresponding to flow rates of 2.396, 4.792 and 7.152 ml/min. The COD concentration of the influent tank, anode region and cathode region at different HRTs for cube anode and linked anode is shown in Fig. 3. The initial COD concentration of the influent tank was 1968 ± 66 mg/L. The total COD reduction at three different HRTs for cube anode and linked anode was almost similar. The percentage of COD reduction was varied between 88 and 97%. This could be explained by the identical reactor configuration and total cross-sectional area of two different anode configurations, which provide a homogeneous environment for microbial growth. The linked anode configuration (Fig. 2b) in this study was designed to have a larger active region (258 cm2) than that of cube anode (208 cm2), but the result shows that the larger active region did not directly benefit to the COD reduction. (Area of the most active region is expected to have high microbial activity.) Rossi et al. (2019) reported that the presence of a higher number of anodes did not benefit the COD removal, although the number of anodes did impact the amount of COD converted to electricity. Moreover, the COD concentration at anode (cube and linked) and cathode (cube and linked) regions was remained fluctuate in 9 days between 36–347 and 66–333 mg/L, accordingly.
The COD performance at anode (cube and linked) and cathode (cube and linked) did not show a distinctive trend with HRT, but the COD performance at influent tank was distinguishable. The COD performance at low HRT 0.67 was better than high HRT 1.0 and HRT 2.0. The COD performance of the influent tank at HRT 2.0 took 5 days for reduction, while HRT 1.0 and HRT 0.67 were only used 4 and 3 days, respectively (Fig. 3). The COD performance order was as follows: HRT 0.67 > HRT 1.0 > HRT 2.0. This can be explained by the anaerobic microorganisms in the SUFML MFC are sufficient to degrade organic substrate in higher COD concentration. Result demostrated that the SUFML MFC has great potential in high concentration COD reduction even at low HRT 0.67.
Voltage output level
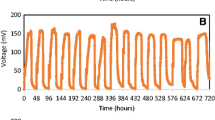
The voltage output of cube anode and linked anode operated in 216 h at different HRTs is shown in Fig. 4. The configuration of linked anode is expected to have better mass transfer from anode region to cathode region (Fig. 2b), greater directional fluid flow and higher area of most active region to enhance the voltage generation. However, the maximum voltage output of linked anode was comparable to cube anode in all three different HRTs, but the overall voltage output of linked anode was still better than cube anode. The voltage output of cube anode and linked anode at HRT 2.0 during the stationary phase was able to sustain about 122 h and maximum voltage output at 727 ± 42 and 734 ± 54 mV, respectively. However, the sustainability of voltage output for cube anode and linked anode at HRT 1.0 was reduced to 73 h, whereas the maximum voltage output for cube anode and linked anode remained around 721 ± 92 and 719 ± 84 mV, correspondingly. The maximum voltage output at HRT 0.67 was further deteriorated for cube anode (613 ± 85 mV) and linked anode (640 ± 91 mV), and the sustainability of voltage output was reduced to 56 h. Lower HRT usually delivers better in mass transfer, and lower internal resistance results in better voltage output. However, these results showed that high HRT can generate more sustainable voltage output (Fig. 4). This phenomenon could be merited by the total amount of electrons used for electricity generation. High HRT with a low flow rate in the reactor could provide better contact time, which benefits anode to obtain the electrons from substrate degradation and consequently generated more sustainable voltage output (Li et al. 2013; Sharma and Li 2010). In contrast, a fast flow rate with low HRT may cause unfavorable electron transfer to anode for electricity generation. Although the voltage output at the stationary phase of linked anode is better than cube anode, the total voltage output of cube anode for 216 h was greater than linked anode. The voltage output of cube anode during the decline phase was higher than linked anode in all different HRTs. This could be because the rigid volume of one-piece cube carbon felt anode (240 cm3) is larger than linked carbon felt anode (140 cm3), which retained more electrons in the carbon felt. The deterioration in voltage output during decline phase may be due to the insufficient electrons and protons supplied over the time.
The total charge number generated in 216 h by cube anode and linked anode at HRT 2.0 was 476.75 and 393.92 C, respectively. The charge number was diminished at HRT 1.0, 363.79 C for cube anode and 291.75 C for linked anode. The charge number produced by cube anode (280.83 C) and linked anode (203.23 C) further deteriorated at HRT 0.67. These results showed that the cube anode was clearly outstanding in the overall electricity generation, as well as the CE. The CE was calculated based on the COD reduction. As the total COD reduction for both cube anode and linked anode was identical, the overall CE of cube anode was greater than linked anode. Besides, the overall CE in HRT 2.0 was better than HRT 1 and HRT 0.67 (Fig. 4). This phenomenon could be described that the other microorganisms in SUFML MFC may be accounted for more organic carbon oxidation rather than electrochemical active microorganisms at low HRT, thus reducing electron recovery.
Effect of electrode spacing distance on power density and internal resistance at different HRTs
The plausible explanation for low voltage output is relatively high internal resistance, which may hamper the electron transfer by causing a significant decrease in voltage output due to ohmic loss (Logan et al. 2006; Rabaey and Verstraete 2005). The internal resistance can be obtained from polarization curve, where the internal resistance is equal to external resistance at maximum power output (Fan et al. 2008; Feng et al. 2014). Cube anode and linked anode were configured to study the mass transfer efficiency, while linked anode has better directional fluid flow than that of cube anode (Fig. 2). Linked anode was designed to improve the mass transfer of electrons and protons from anode region to cathode region, and subsequently reduce the internal resistance to improve the power output. The SUFML MFC was designed with total three anodes at different electrode spacing distances, which potentially reduce the mass transfer efficiency from anode to cathode regions. The electrode spacing distances between cathode and anode A1, A2 and A3 were 36, 27 and 18 cm, respectively. Figure 5 shows that the linked anode has better mass transfer efficiency from anode region to cathode region than that of cube anode. Based on the polarization curved of linked anode, the pattern of the curve was similar at any electrode spacing distance. The internal resistance and maximum power output of linked anode were almost identical at any anode spacing distance, while those of cube anode were not identical. The maximum power output of cube anode at HRT 2.0 was decreased as the electrode spacing distance reduced from 36 to 18 cm. This could be described that the high HRT with low flow rate does not provide sufficient of electrons to the cube anode A3. Nevertheless, low HRT (1.0 and 0.67) with higher flow rate significantly improved the mass transfer of electrons and provided sufficient electrons to cube anode A2 and A3. As a result, the maximum power output of low HRT was almost identical at any anode spacing distance.
Figure 5 also shows that the maximum power density of cube anode and linked anode was affected significantly at different HRTs. Maximum power density of cube anode (A1 and A2) was increased when the HRT is high. However, the maximum power density of cube anode A3 was not significantly affected by HRT. This is because the cube anode configuration was unfavorable for mass transfer. The maximum power density order for cube anode was as follows: HRT 2.0 > HRT 1.0 > HRT 0.67. As for linked anode, the maximum power density order was as follows: HRT 1.0 > HRT 2.0 > HRT 0.67. The results showed that linked anode has better overall power output potential than that of cube anode at any anode spacing distance. The maximum power density of any anode spacing distance for linked anode was almost identical. This probably can be ascribable by the linked anode configuration that has better directional fluid flow with great mass transfer of electrons and protons. However, cube anode A1 has the highest maximum power density of 30.3 mW/m2 at HRT 2.0, yet linked anode A2 has a maximum power density of 23.2 mW/m2 at HRT 1.0.
Conclusion
Linked anode generated better voltage output during the stationary phase at all different HRTs. Moreover, the maximum power density of every electrode spacing distance for linked anode was almost identical. Therefore, linked anode configuration has been successfully demonstrated as a lower internal resistance anode with better directional fluid flow and mass transfer efficiency than those of cube anode in SUFML MFC. These finding results suggest that the different configuration of bioanodes will potentially affect the MFC performance. It is an important factor for future real application in the wastewater treatment plant, especially in an up-flow membrane-less MFC.
References
Adav SS, Lee DJ, Lai JY (2010) Microbial community of acetate utilizing denitrifiers in aerobic granules. Appl Microbiol Biotechnol 85:753–762. https://doi.org/10.1007/s00253-009-2263-6
Bajracharya S, Sharma M, Mohanakrishna G et al (2016) An overview on emerging bioelectrochemical systems (BESs): technology for sustainable electricity, waste remediation, resource recovery, chemical production and beyond. Renew Energy 98:153–170. https://doi.org/10.1016/j.renene.2016.03.002
Bennetto HP, Stirling JL, Tanaka K, Vega CA (1983) Anodic reactions in microbial fuel cells. Biotechnol Bioeng 25:559–568. https://doi.org/10.1002/bit.260250219
Clauwaert P, Rabaey K, Aelterman P et al (2007) Biological denitrification in microbial fuel cells. Environ Sci Technol 41:3354–3360. https://doi.org/10.1021/es062580r
Crittenden SR, Sund CJ, Sumner JJ (2006) Mediating electron transfer from bacteria to a gold electrode via a self-assembled monolayer. Langmuir 22:9473–9476. https://doi.org/10.1021/la061869j
Cui D, Guo Y-Q, Lee H-S et al (2014) Efficient azo dye removal in bioelectrochemical system and post-aerobic bioreactor: optimization and characterization. Chem Eng J 243:355–363. https://doi.org/10.1016/j.cej.2013.10.082
Daniel DK, Das Mankidy B, Ambarish K, Manogari R (2009) Construction and operation of a microbial fuel cell for electricity generation from wastewater. Int J Hydrog Energy 34:7555–7560. https://doi.org/10.1016/j.ijhydene.2009.06.012
Fan Y, Sharbrough E, Liu H (2008) Quantification of the internal resistance distribution of microbial fuel cells quantification of the internal resistance distribution of microbial fuel cells. Environ Sci Technol 42:8101–8107. https://doi.org/10.1021/es801229j
Feng Y, Wang X, Logan BE, Lee H (2008) Brewery wastewater treatment using air-cathode microbial fuel cells. Appl Microbiol Biotechnol 78:873–880. https://doi.org/10.1007/s00253-008-1360-2
Feng Y, He W, Liu J et al (2014) A horizontal plug flow and stackable pilot microbial fuel cell for municipal wastewater treatment. Bioresour Technol 156:132–138. https://doi.org/10.1016/j.biortech.2013.12.104
Fernando E, Keshavarz T, Kyazze G (2014) Complete degradation of the azo dye Acid Orange-7 and bioelectricity generation in an integrated microbial fuel cell, aerobic two-stage bioreactor system in continuous flow mode at ambient temperature. Bioresour Technol 156:155–162. https://doi.org/10.1016/j.biortech.2014.01.036
Gajda I, Greenman J, Melhuish C et al (2014) Water formation at the cathode and sodium recovery using Microbial Fuel Cells (MFCs). Sustain Energy Technol Assess 7:187–194. https://doi.org/10.1016/j.seta.2014.05.001
Ghasemi M, Wan Daud WR, Ismail M et al (2013) Effect of pre-treatment and biofouling of proton exchange membrane on microbial fuel cell performance. Int J Hydrog Energy 38:5480–5484. https://doi.org/10.1016/j.ijhydene.2012.09.148
Habermann W, Pommer EH (1991) Biological fuel cells with sulphide storage capacity. Appl Microbiol Biotechnol 35:128–133. https://doi.org/10.1007/BF00180650
Huang J, Yang P, Guo Y, Zhang K (2011) Electricity generation during wastewater treatment: an approach using an AFB-MFC for alcohol distillery wastewater. Desalination 276:373–378. https://doi.org/10.1016/j.desal.2011.03.077
Kushwaha JP, Srivastava VC, Mall ID (2011) An overview of various technologies for the treatment of dairy wastewaters. Crit Rev Food Sci Nutr 51:442–452. https://doi.org/10.1080/10408391003663879
Li X, Zhu N, Wang Y et al (2013) Animal carcass wastewater treatment and bioelectricity generation in up-flow tubular microbial fuel cells: effects of HRT and non-precious metallic catalyst. Bioresour Technol 128:454–460. https://doi.org/10.1016/j.biortech.2012.10.053
Liao Q, Zhang J, Li J et al (2014) Electricity generation and COD removal of microbial fuel cells (MFCs) operated with alkaline substrates. Int J Hydrog Energy 39:19349–19354. https://doi.org/10.1016/j.ijhydene.2014.06.058
Liu H, Logan BE (2004) Electricity generation using an air-cathode single chamber microbial fuel cell in the presence and absence of a proton exchange membrane. Environ Sci Technol 38:4040–4046. https://doi.org/10.1021/Es0499344
Liu H, Ramnarayanan R, Logan BE (2004) Production of electricity during wastewater treatment using a single chamber microbial fuel cell. Environ Sci Technol 38:2281–2285. https://doi.org/10.1021/es034923g
Logan BE, Murano C, Scott K et al (2005) Electricity generation from cysteine in a microbial fuel cell. Water Res 39:942–952. https://doi.org/10.1016/j.watres.2004.11.019
Logan BE, Hamelers B, Rozendal R et al (2006) microbial fuel cells: methodology and technology. Environ Sci Technol 40:5181–5192. https://doi.org/10.1021/es0605016
Min B, Logan BE (2004) Continuous electricity generation from domestic wastewater and organic substrates in a flat plate microbial fuel cell. Environ Sci Technol 38:5809–5814. https://doi.org/10.1021/Es0491026
Nasharudin MN, Kamarudin SK, Hasran UA, Masdar MS (2014) Mass transfer and performance of membrane-less micro fuel cell: a review. Int J Hydrog Energy 39:1039–1055. https://doi.org/10.1016/j.ijhydene.2013.09.135
Neoh CH, Noor ZZ, Mutamim NSA, Lim CK (2016) Green technology in wastewater treatment technologies: integration of membrane bioreactor with various wastewater treatment systems. Chem Eng J 283:582–594. https://doi.org/10.1016/j.cej.2015.07.060
Pant D, Van Bogaert G, Diels L, Vanbroekhoven K (2010) A review of the substrates used in microbial fuel cells (MFCs) for sustainable energy production. Bioresour Technol 101:1533–1543. https://doi.org/10.1016/j.biortech.2009.10.017
Pasupuleti SB, Srikanth S, Dominguez-Benetton X et al (2016) Dual gas diffusion cathode design for microbial fuel cell (MFC): optimizing the suitable mode of operation in terms of bioelectrochemical and bioelectro-kinetic evaluation. J Chem Technol Biotechnol 91:624–639. https://doi.org/10.1002/jctb.4613
Rabaey K, Verstraete W (2005) Microbial fuel cells: novel biotechnology for energy generation. Trends Biotechnol 23:291–298. https://doi.org/10.1016/j.tibtech.2005.04.008
Rahimnejad M, Adhami A, Darvari S et al (2015) Microbial fuel cell as new technology for bioelectricity generation: a review. Alexandria Eng J 54:745–756. https://doi.org/10.1016/j.aej.2015.03.031
Rossi R, Jones D, Myung J et al (2019) Evaluating a multi-panel air cathode through electrochemical and biotic tests. Water Res 148:51–59. https://doi.org/10.1016/j.watres.2018.10.022
Sangeetha T, Guo Z, Liu W et al (2016) Cathode material as an influencing factor on beer wastewater treatment and methane production in a novel integrated upflow microbial electrolysis cell (Upflow-MEC). Int J Hydrog Energy 41:2189–2196. https://doi.org/10.1016/j.ijhydene.2015.11.111
Saratale GD, Saratale RG, Shahid MK et al (2017) A comprehensive overview on electro-active biofilms, role of exo-electrogens and their microbial niches in microbial fuel cells (MFCs). Chemosphere 178:534–547. https://doi.org/10.1016/j.chemosphere.2017.03.066
Sharma Y, Li B (2010) Optimizing energy harvest in wastewater treatment by combining anaerobic hydrogen producing biofermentor (HPB) and microbial fuel cell (MFC). Int J Hydrog Energy 35:3789–3797. https://doi.org/10.1016/j.ijhydene.2010.01.042
Vázquez I, Rodríguez J, Marañón E et al (2006) Study of the aerobic biodegradation of coke wastewater in a two and three-step activated sludge process. J Hazard Mater 137:1681–1688. https://doi.org/10.1016/j.jhazmat.2006.05.007
Wang H, Jiang SC, Wang Y, Xiao B (2013) Substrate removal and electricity generation in a membrane-less microbial fuel cell for biological treatment of wastewater. Bioresour Technol 138:109–116. https://doi.org/10.1016/j.biortech.2013.03.172
Yoshizawa T, Miyahara M, Kouzuma A, Watanabe K (2014) Conversion of activated-sludge reactors to microbial fuel cells for wastewater treatment coupled to electricity generation. J Biosci Bioeng 118:533–539. https://doi.org/10.1016/j.jbiosc.2014.04.009
Acknowledgements
We would like to thank Science Fund MOSTI Grant (02-01-15-SF0201) for their support on this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial responsibility: Binbin Huang.
Rights and permissions
About this article
Cite this article
Thung, WE., Ong, SA., Ho, LN. et al. Enhancement of mass and charge transport in scaled-up microbial fuel cell by using innovative configuration of bioanode. Int. J. Environ. Sci. Technol. 16, 8175–8184 (2019). https://doi.org/10.1007/s13762-019-02390-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-019-02390-8