Abstract
This study aimed to introduce clinical manifestations of patients in northeast Iran with HTLV-1-associated myelopathy/tropical spastic paraparesis (HAM/TSP) and describe the epidemiological features, as well as risk factors for HTLV-1 infection. This is a cross-sectional study of HTLV-1 infected cases and HAM/TSP patients referred by outpatient neurology clinics as well as Mashhad Blood Transfusion Center from 2005 to 2010. The study comprises 513 cases, including 358 healthy carriers (HCs) and 145 HAM/TSP patients. The majority of carriers were male (73.5 %), whereas 67.6 % of HAM/TSP sufferers were female (P < 0.001). The mean age of HAM/TSP patients and HCs was 45.9 ± 13.6 and 39.5 ± 11.58 years, respectively (P < 0.001). The history of transfusion, surgery, hospitalization and cupping was observed in a significant greater number of HAM/TSP patients than the HCs (P < 0.001, P < 0.001, P < 0.001 and P = 0.029, respectively). Gait disturbance was the most common complaint in HAM/TSP patients (72.4 %). This research develops an HTLV-1 data registry in an endemic area such as Mashhad which can serve useful purposes, including evaluation of clinical and laboratory characteristics of HAM/TSP patients and epidemiological data of HTLV-1-infected cases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human T-cell leukemia virus type 1 (HTLV-1), a type C retrovirus, is the first retrovirus known to be pathogenic in humans [1]. Although most infected individuals remain asymptomatic healthy carriers (HCs) throughout their lives [2, 3], HTLV-1 remains a known cause of a variety of diseases including neurological conditions such as HTLV-1-associated myelopathy/tropical spastic paraparesis (HAM/TSP) [4, 5] and non-neurological disorders such as adult T-cell leukemia/lymphoma (ATLL) [3], dermatitis [6], rheumatologic and pulmonary disorders, thyroid diseases and uveitis [7, 8]. It is estimated 10–20 million people worldwide are infected with the virus [2, 9] with major endemic areas in southwest Japan [10, 11], the Caribbean, Central and South America, some parts of Africa [11, 12], the Melanesian and Solomon islands in Pacific Ocean, and Eastern Europe [8, 11]. The prevalence of HTLV-1 in the Middle East had been underestimated until 1990s. The presence of HTLV-1 in the Middle East was first reported by Popovic et al. [13] in 1983. For the first time, the high prevalence of the infection was reported in a cluster of Israeli immigrants, born in Mashhad in the northeast Iran [14–16]. This finding resulted in a serological survey of HTLV-1 in Mashhad. A total of 35 out of 1,511 (2.3 %) serum samples were positive for HTLV-1, and the polymerase chain reaction study showed that the virus is the classical form of HTLV-1 belonging to the cosmopolitan molecular subtype [17, 18]. Three years later, Safai et al. [18] showed a slightly higher rate of HTLV-1 infection (21/694, 3 %) in this region. The high prevalence of HTLV-1 was also reported among blood donors in Mashhad. In 1996, Rezvan et al. [19] reported a prevalence of 2 % for HTLV-1 infection among blood donors. Other studies in 1999 and 2004 showed that the prevalence of this infection is 0.77 % [20] and 0.45 % [21], respectively, among blood bank donors.
In 2011, the overall prevalence of HTLV-1 infection in the city of Mashhad was reported as 2.12 %, suggesting this part of Iran as an endemic area for HTLV-1 infection [22]. The peculiar characteristics associated with this infection (such as the prevalence discrepancy among neighboring regions [23–27] even other Iranian provinces [28], the clustering distribution of HTLV-1 [29] and the complex immunogenic aspects of the virus) has made this infection a hot topic for researchers in the world.
Regarding the high prevalence of the HTLV-1 in Mashhad and its socioeconomic burden, a cross-sectional study was designed to introduce a data registry, describe the epidemiological features and risk factors for the infection in this endemic area and define the neurologic presentations of the HAM/TSP in Iranian patients.
Methods
Study design
A cross-sectional study was defined and approved by the Ethics Committee of the Mashhad University of Medical Sciences. Informed consent was obtained from each participant before enrolling into the study.
Setting and participants
Since 2005, subjects in this study have been recruited from two major sources: blood donors in Mashhad and the HAM/TSP patients visited the neurology department of Mashhad University of the Medical Sciences (a tertiary referral center for HTLV-1 infection in Iran). HTLV-1 infection in blood donors was screened by enzyme-linked immunosorbent assay (ELISA) and confirmed using the Western blot or polymerase chain reaction as previously described [23]. The infected individuals were referred to the study neurologists (MRA, AS or MF) for neurological consultations and were classified into the HAM/TSP or HCs according to the World Health Organization criteria (Belem Criteria) [30, 31].
Data measurements
The participants were asked to complete a questionnaire on the demographic data (age, gender, race and marital status) as well as a history of breast-feeding in infancy, blood transfusion, dialysis, surgery, dental procedures, cupping, tattooing and unsafe sexual contact (common known risk factors for virus transmission). All patients underwent full neurological examination including muscle strength examination using the Medical Research Council (MRC) scale [32], deep tendon reflexes examination, spasticity evaluation based on the modified Ashworth scale [33], motor disability measurement employing the Osame Motor Disability Score (OMDS) [34], sensory examination, cerebellar function examination and urinary complaint evaluation using Urinary Disturbance Score (UDS) [35].
Statistical methods
Categorical data were presented as numbers (percentages) and non-categorical data as mean ± standard deviation. Data were analyzed through χ 2 and McNemar’s tests using SPSS 11.5. A P value <0.05 was considered statistically significant.
Results
Participants
This registry consists of 513 HTLV-1-infected individuals including 358 cases determined as HCs, 145 patients with clinical diagnosis of HAM/TSP and 10 cases who were probable or possible in the diagnosis of HAM/TSP. The two latter groups were excluded from the study.
Descriptive data
Demographical data
The mean age of participants was 41.3 ± 12.5 years (range 13–82 years). The majorities of our cases belonged to Fars ethnic group (93.6 %) and were married (84 %). A total of 61.6 % of all cases (73.5 % of HCs and 32.4 % of HAM/TSP patients) were male. The gender difference between HAM/TSP group and HCs group was statically significant (P < 0.001) (Table 1).
Transmission
Analysis of the potential risk factors for HTLV-1 infection demonstrated that 385 out of 492 cases (88.3 %) had more than one known risk factor for virus transmission in their history. Majority of patients (95.9 %) had been breast-fed in the infancy period. The most common identifiable risk factors other than breast-feeding among HTLV-1-positive individuals were history of dental procedures (52.2 %), hospitalization (49.4 %) and previous surgery (41.9 %). A total of 27 % of our cases had experienced cupping. Other less common risk factors were records of transfusion (10.2), unsafe sexual contact (7.1 %), tattooing (3.7 %) and dialysis (0.4 %). Transfusion, surgery, hospitalization and cupping were significantly more evident in history of HAM/TSP patients compared to that of HCs (P < 0.001, P < 0.001, P < 0.001 and P = 0.029, respectively), with dental procedures more frequent in HCs (P = 0.007) (Table 1).
Clinical features of HAM/TSP patients
The mean age for HAM/TSP patients was 45.9 ± 13.6 years (ranging between 14 and 82 years). The interval between the first presentation and disease diagnosis was more than 2 months in 93.8 % of patients and more than 2 years in 68.3 %. In this study, HAM/TSP presented with gait disturbances in most cases (72.4 %), second by urinary and sensory complaints. Other frequent presentations included unexplained back pain (23.4 %), constipation (26.8 %) and erectile dysfunction (6.7 %).
Motor function
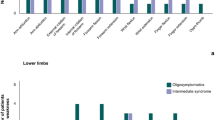
Hypertonicity and weakness was observed more commonly in lower limbs of patients than upper ones (P < 0.001) (Table 2). Most patients had brisk or exaggerated deep tendon reflexes in all extremities. However, some cases had decreased responses. Babinski’s sign was detected in 113 cases (77.9 %). Clonus and Hoffmann’s signs were determined in 35 (24.1 %) and 65 cases (44.2 %), respectively, but exaggerated jaw jerk was only seen in 3 cases (2.1 %).
Urinary disturbances
Clinical assessment of patients based on UDS revealed that 97 patients (67.8 %) complained of urinary frequency and 91 patients (63.6 %) suffered from incontinence. Furthermore, feeling of inappropriate micturition with sense of residual urine was reported in 77 cases (53.8 %) (Table 3).
Sensory functions
Foot paresthesia and tingling were the most common sensory complaints in patients, and the main sensory finding in neurologic examination was the impairment of vibration in both upper (23 cases; 16 %) and lower limbs (64 cases; 44.4 %) (P < 0.001). Light touch sensation was impaired in lower limbs in 26 cases (18.1 %) and in upper limbs in 24 cases (16.7 %). Thermal sensation was intact in nearly all patients. Twenty-nine (20.1 %) patients had proprioception abnormality in their upper limbs and 68 cases (47.2 %) had similar problems in their lower limbs. Proprioception abnormalities, including abnormal joint position and kinesthetic sensation impairment, were significantly greater in lower limbs than in upper ones (P = 0.02 and P = 0.01, respectively).
Other neurologic manifestations
Constipation (26.8 %) and back pain (23.4 %) were also common in our patients. Less frequent symptoms were palpitation, sweating and flushing, orthostatic hypotension and diminished libido (Table 3). Sense of disequilibrium was observed in 8 cases (5.5 %). No evidence of cerebellar dysfunction was detected in examination. Cranial nerve functions were normal in all patients except one who had optic nerve atrophy on neurologic examination. Other neurologic findings in our patients were parkinsonism (one case), choreoathetosis (one case) and findings resembling those of amyotrophic lateral sclerosis (2 cases).
Discussion
Mashhad is a known endemic area for HTLV-1 infection [19–22]. There is, however, a lack of strategy regarding the relevant epidemiological data and neurological findings, which highlights the importance of establishing a clinical registry as a basis for further research. In contrast to earlier studies which reported that HTLV-1 infection was more prevalent among women [36, 37], the present study established higher prevalence among males, particularly among the carriers. This finding can be explained by the fact that the female population in Iran has been traditionally reluctant to blood donation. Furthermore, a population-based study in Mashhad reported no significant difference between male and female in terms of HTLV-1 infection [22]. Consistent with previous researches [36, 38–40], this study indicates that HAM/TSP is more frequent among women. The incidence of HAM/TSP seems to increase with age, possibly due to the nature of HTLV-1(as a retrovirus) and the increased risk of exposure to different transmission risk factors [22, 36, 37, 41]. History of dental and other surgical procedures, hospitalization, traditional cupping and blood transfusion was positive in a significant percent of our cases, confirming the blood as a route for virus transmission.
There are some reports explaining other routes of viral transmission including unsafe sexual intercourse, maternal child (vertical) transmission and sharing of needles [22, 38, 41]. Unsafe sexual intercourse appears to be a major route of HTLV-1 transmission [41–43]. None of our cases had a history of homosexual contacts, but this may be a false result because of cultural issues and the fact that in our culture and religion, having homosexual contacts is inadmissible. Breast-feeding is a well known risk factor for HTLV-1. In the present study, 94.9 % of cases had been breast-fed in their childhood. Similarly, Gotuzzo et al. [40] suggested that breast-feeding is probably the main route of HTLV-1 transmission in Peru.
The neurologic manifestations of HAM/TSP in Mashhad are similar; to a great extent, two reports found in other literature. In a Brazilian cohort of HAM/TSP patients, the most prevalent complaint was walking difficulties due to gait disturbances [44–46]. Similar findings have been reported in Peru [40] and UK [47]. Among the 21 HAM/TSP patients who were born in Caribbean and immigrated to UK, sensory symptoms in lower limbs along with urinary disturbances or limb weakness were the most common initial presentations [48]. However, Martin et al. [47] reported gait disturbances as the most common first presentation in a cohort of HAM/TSP patients in UK. Low back and leg pain, urinary disturbances and constipation were also the other common findings. Like our study, in a Peruvian cohort, urinary complaints and diminished vibratory sensations were common findings in HAM/TSP patients. Yet, the prevalence of constipation and lumbar pain was higher than that of our population [40].
Even though the involvement of cerebellum and cranial nerves in HAM/TSP patients has been reported previously [49–51], no evidence of cerebellar dysfunction was observed and only one case of optic neuropathy was detected in our study. In this case, brain MRI was normal and CSF analysis showed no oligoclonal bands. Then, we concluded that optic neuropathy is not related to multiple sclerosis.
In conclusion, it seems that Iran is a perfect place for conducting international clinical and biomarker trials/studies of HTLV-1, given the high prevalence in the area. A great achievement of the current study is the creation of a dynamic databank consisting of the clinical data and blood samples of a large population of HTLV-1 patients. It is hoped that this registry will provide a valuable resource for future basic science and clinical studies. The databank also offers a suitable platform that can be utilized for international research in similar areas.
References
Lepoutre V, Jain P, Quann K, Wigdahl B, Khan ZK (2009) Role of resident CNS cell populations in HTLV-1-associated neuroinflammatory disease. Front Biosci 14:1152–1168
Barmak K, Harhaj E, Grant C, Alefantis T, Wigdahl B (2003) Human T cell leukemia virus type I-induced disease: pathways to cancer and neurodegeneration. Virology 308(1):1–12
Taylor JM, Nicot C (2008) HTLV-1 and apoptosis: role in cellular transformation and recent advances in therapeutic approaches. Apoptosis 13(6):733–747
Araujo AQ, Leite AC, Lima MA, Silva MT (2009) HTLV-1 and neurological conditions: when to suspect and when to order a diagnostic test for HTLV-1 infection? Arq Neuropsiquiatr 67(1):132–138
Osame M, Usuku K, Izumo S, Ijichi N, Amitani H, Igata A, Matsumoto M, Tara M (1986) HTLV-I associated myelopathy, a new clinical entity. Lancet 1(8488):1031–1032
La GL, Manns A, Fletcher V, Derm D, Carberry C, Hanchard B, Maloney EM, Cranston B, Williams NP, Wilks R, Kang EC, Blattner WA (1998) Clinical, pathologic, and immunologic features of human T-lymphotrophic virus type I-associated infective dermatitis in children. Arch Dermatol 134(4):439–444
Buggage RR (2003) Ocular manifestations of human T-cell lymphotropic virus type 1 infection. Curr Opin Ophthalmol 14(6):420–425
Verdonck K, Gonzalez E, Van DS, Vandamme AM, Vanham G, Gotuzzo E (2007) Human T-lymphotropic virus 1: recent knowledge about an ancient infection. Lancet Infect Dis 7:266–281
Leite AC, Mendonca GA, Serpa MJ, Nascimento OJ, Araujo AQ (2003) Neurological manifestations in HTLV-I-infected blood donors. J Neurol Sci 214(1–2):49–56
Cooper SA, van der Loeff MS, Taylor GP (2009) The neurology of HTLV-1 infection. Pract Neurol 9(1):16–26
Gessain A, Mahieux R (2000) Epidemiology, origin and genetic diversity of HTLV-1 retrovirus and STLV-1 simian affiliated retrovirus. Bull Soc Pathol Exot 93:163–171
Nakamura T (2009) HTLV-I-associated myelopathy/tropical spastic paraparesis (HAM/TSP): the role of HTLV-I-infected Th1 cells in the pathogenesis, and therapeutic strategy. Folia Neuropathol 47(2):182–194
Popovic M, Sarin PS, Robert-Gurroff M, Kalyanaraman VS, Mann D, Minowada J, Gallo RC (1983) Isolation and transmission of human retrovirus (human t-cell leukemia virus). Science 219(4586):856–859
Achiron A, Pinhas-Hamiel O, Doll L, Djaldetti R, Chen A, Ziv I, Avni A, Frankel G, Melamed E, Shohat B (1993) Spastic paraparesis associated with human T-lymphotropic virus type I: a clinical, serological, and genomic study in Iranian-born Mashhadi Jews. Ann Neurol 34:670–675
Meytes D, Schochat B, Lee H, Nadel G, Sidi Y, Cerney M, Swanson P, Shaklai M, Kilim Y, Elgat M (1990) Serological and molecular survey for HTLV-I infection in a high-risk Middle Eastern group. Lancet 336:1533–1535
Sidi Y, Meytes D, Shohat B, Fenig E, Weisbort Y, Lee H, Pinkhas J, Rosenblatt JD (1990) Adult T-cell lymphoma in Israeli patients of Iranian origin. Cancer 65:590–593
Farid R, Etemadi M, Baradaran H, Nikbin B (1993) Seroepidemiology and virology of HTLV-1 in the city of Mashhad, northeastern Iran. Serodiagn Immunother Infect Disease 5:251–252
Safai B, Huang JL, Boeri E, Farid R, Raafat J, Schutzer P, Ahkami R, Franchini G (1996) Prevalence of HTLV type I infection in Iran: a serological and genetic study. AIDS Res Hum Retroviruses 12:1185–1190
Rezvan H, Ahmadi J, Farhadi M (1996) A cluster of HTLV-I infection in northeastern of Iran. Transfus Today 27:8–9
Abbaszadegan MR, Gholamin M, Tabatabaee A, Farid R, Houshmand M, Abbaszadegan M (2003) Prevalence of human T-lymphotropic virus type 1 among blood donors from Mashhad, Iran. J Clin Microbiol 41:2593–2595
Tarhini M, Kchour G, Zanjani DS, Rafatpanah H, Otrock ZK, Bazarbachi A, Farid R (2009) Declining tendency of human T-cell leukaemia virus type I carrier rates among blood donors in Mashhad, Iran. Pathology 41:498–499
Rafatpanah H, Hedayati-Moghaddam, Fathimoghadam F, Bidkhori HR, Shamsian SK, Ahmadi S, Sohgandi L, Azarpazhooh, Rezaee SA, Farid R, Bazarbachi A (2011) High prevalence of HTLV-I infection in Mashhad, Northeast Iran: a population-based seroepidemiology survey. J Clin Virol 52:172–176
Proietti FA, Carneiro-Proietti AB, Catalan-Soares BC, Murphy EL (2005) Global epidemiology of HTLV-I infection and associated diseases. Oncogene 24:6058–6068
Voevodin A, al-Mufti S, Farah S, Khan R, Miura T (1995) Molecular characterization of human T-lymphotropic virus, type 1 (HTLV-1) found in Kuwait: close similarity with HTLV-1 isolates originating from Mashhad, Iran. AIDS Res Hum Retroviruses 11:1255–1259
Denic S, Nolan P, Doherty J, Garson J, Tuke P, Tedder R (1990) HTLV-I infection in Iraq. Lancet 336:1135–1136
Voevodin A, Gessain A (1997) Common origin of human T-lymphotropic virus type-I from Iran, Kuwait, Israel, and La Reunion Island. J Med Virol 52:77–82
al-Mufti S, Voevodin A, Ahmed S, Al-Hamdan S, Al-Bisher AA (1997) Prevalence of human T-cell lymphotropic virus type I infection among volunteer blood donors in Kuwait. J Acquir Immune Defic Syndr Hum Retrovirol 15:88–90
Miller M, Achiron A, Shaklai M, Stark P, Maayan S, Hannig H, Hunsmann G, Bodemer W, Shohat B (1998) Ethnic cluster of HTLV-I infection in Israel among the Mashhadi Jewish population. J Med Virol 56:269–274
Farid R, Shirdel A, Etemadi M, Rafatpanah H, Baradaran H, Farid R, Nikbin B (1999) Phylogenetic of Human T-cell Lymphotropic Virus Type I in Iranians Born in Mashhad. AIM 2:20–24
Osame M (1990) Review of WHO Kagoshima meeting and diagnostic guidelines for HAM/TSP. In: Blattner WA (ed) Human retrovirology: HTLV. Raven Press, New York, pp 191–197
De Castro-Costa CM, Araujo AQ, Barreto MM, Takayanagui OM, Sohler MP, da Silva EL, de Paula SM, Ishak R, Ribas JG, Rovirosa LC, Carton H, Gotuzzo E, Hall WW, Montano S, Murphy EL, Oger J, Remondegui C, Taylor GP (2006) Proposal for diagnostic criteria of tropical spastic paraparesis/HTLV-I-associated myelopathy (TSP/HAM). AIDS Res Hum Retroviruses 22:931–935
Paternostro-Sluga T, Grim-Stieger M, Posch M, Schuhfried O, Vacariu G, Mittermaier C, Bittner C, Fialka-Moser V (2008) Reliability and validity of the Medical Research Council (MRC) scale and a modified scale for testing muscle strength in patients with radial palsy. J Rehabil Med 40:665–671
Bohannon RW, Smith MB (1987) Interrater reliability of a modified Ashworth scale of muscle spasticity. Phys Ther 67:206–207
Izumo S, Goto I, Itoyama Y, Okajima T, Watanabe S, Kuroda Y, Araki S, Mori M, Nagataki S, Matsukura S, Akamine T, Nakagawa M, Yamamoto I, Osame M (1996) Interferon-alpha is effective in HTLV-I-associated myelopathy: a multicenter, randomized, double-blind, controlled trial. Neurology 46:1016–1021
Izumo S, Usuku K, Osame M, Arimura K, Igata A (1988) Effect of alpha-interferon treatment on HTLV-1 associated myelopathy (HAM). Neurology 39:242
Dourado I, Alcantara LC, Barreto ML, da Gloria TM, Galvao-Castro B (2003) HTLV-I in the general population of Salvador, Brazil: a city with African ethnic and sociodemographic characteristics. J Acquir Immune Defic Syndr 34:527–531
Sanchez-Palacios C, Gotuzzo E, Vandamme AM, Maldonado Y (2003) Seroprevalence and risk factors for human T-cell lymphotropic virus (HTLV-I) infection among ethnically and geographically diverse Peruvian women. Int J Infect Dis 7:132–137
Dourado I, Andrade T, Galvao-Castro B (1998) HTLV-I in Northeast Brazil: differences for male and female injecting drug users. J Acquir Immune Defic Syndr Hum Retrovirol 19:426–429
Rouet F, Foucher C, Rabier M, Gawronski I, Taverne D, Chancerel B, Casman O, Strobel M (1999) Human T-lymphotropic virus type I among blood donors from Guadeloupe: donation, demographic, and biologic characteristics. Transfusion 39:639–644
Gotuzzo E, Cabrera J, Deza L, Verdonck K, Vandamme AM, Cairampoma R, Vizcarra D, Cabada M, Narvarte G, De las CC (2004) Clinical characteristics of patients in Peru with human T cell lymphotropic virus type 1-associated tropical spastic paraparesis. Clin Infect Dis 39:939–944
Plancoulaine S, Buigues RP, Murphy EL, van Beveren M, Pouliquen JF, Joubert M, Remy F, Tuppin P, Tortevoye P, de The G, Moreau JP, Gessain A (1998) Demographic and familial characteristics of HTLV-1 infection among an isolated, highly endemic population of African origin in French Guiana. Int J Cancer 76:331–336
Kendall EA, Gonzalez E, Espinoza I, Tipismana M, Verdonck K, Clark D, Vermund SH, Gotuzzo E (2009) Early neurologic abnormalities associated with human T-cell lymphotropic virus type 1 infection in a cohort of Peruvian children. J Pediatr 155:700–706
Kramer A, Maloney EM, Morgan OS, Rodgers-Johnson P, Manns A, Murphy EL, Larsen S, Cranston B, Murphy J, Benichou J (1995) Risk factors and cofactors for human T-cell lymphotropic virus type I (HTLV-I)-associated myelopathy/tropical spastic paraparesis (HAM/TSP) in Jamaica. Am J Epidemiol 142:1212–1220
Champs AP, Passos VM, Barreto SM, Vaz LS, Ribas JG (2010) HTLV-1 associated myelopathy: clinical and epidemiological profile in a 10-year case series study. Rev Soc Bras Med Trop 43:668–672
Franzoi AC, Araujo AQ (2007) Disability and determinants of gait performance in tropical spastic paraparesis/HTLV-I associated myelopathy (HAM/TSP). Spinal Cord 45:64–68
Carod-Artal FJ, Mesquita HM, Ribeiro LS (2008) Neurological symptoms and disability in HTLV-1 associated myelopathy. Neurologia 23:78–84
Martin F, Fedina A, Youshya S, Taylor GP (2010) A 15-year prospective longitudinal study of disease progression in patients with HTLV-1 associated myelopathy in the UK. J Neurol Neurosurg Psychiatry 81:1336–1340
Cruickshank JK, Rudge P, Dalgleish AG, Newton M, McLean BN, Barnard RO, Kendall BE, Miller DH (1989) Tropical spastic paraparesis and human T cell lymphotropic virus type 1 in the United Kingdom. Brain 112(Pt 4):1057–1090
Waragai M, Takaya Y, Hayashi M (1995) HTLV-I associated myelopathy (HAM) with cerebellar atrophy presenting a down-beat nystagmus. Rinsho Shinkeigaku 35:549–552
Yoshida Y, Saiga T, Takahashi H, Hara A (1998) Optic neuritis and human T-lymphotropic virus type 1-associated myelopathy: a case report. Ophthalmologica 212:73–76
Taki M, Nin F, Hasegawa T, Sakaguchi H, Suzuki T, Hisa Y, Azuma Y, Nakagawa M (2011) A case report of HTLV-I associated myelopathy presenting with cerebellar ataxia and nystagmus. Auris Nasus Larynx 38:411–414
Acknowledgments
The authors thank the personnel of the blood banks of Mashhad for helping us in this study. This study was supported by the Mashhad University of Medical Sciences.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shoeibi, A., Rafatpanah, H., Azarpazhooh, A. et al. Clinical features of HTLV-1-associated myelopathy/tropical spastic paraparesis (HAM/TSP) in northeast Iran. Acta Neurol Belg 113, 427–433 (2013). https://doi.org/10.1007/s13760-013-0194-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-013-0194-6