Abstract
The corn crop biomass (CB) is widely used as a feedstock for biochemicals such as lactic acid, succinic acid, citric acid, xanthan gum, and biofuels likely bioethanol, butanol, and biogas. Since CB provides a resistive structure for enzymatic and microbial attack, ultrasonic treatment can assist to break the recalcitrance structure. Several techniques such as imaging (atomic force microscopy—AFM; scanning electron microscopy—SEM), spectroscopy (energy-dispersive X-ray spectroscopy—EDX; Fourier transform infrared spectroscopy—FTIR; Raman spectroscopy; X-ray diffraction—XRD), and thermal (TGA) were studied to characterize the ultrasonicated CB. A detailed analysis of different techniques on their potential benefits will assist the researchers to select a suitable technique to optimize the ultrasonication for various applications. The basic mechanisms behind ultrasonication, benefits, downsides, practical considerations, and factors that should be deliberated in the future studies are discussed. Sonication enhanced the hemicellulose and cellulose yield, saccharification rate, and delignification of CB. AFM, EDX, FTIR, Raman spectroscopy, SEM, TGA, and XRD described the variations in topographical features, elemental composition, molecular structure, microstructure, thermal steadiness, and degree of crystallinity, respectively, of the ultrasonicated CB. The quantitative crystallinity of CB can be analyzed through XRD and Raman spectroscopy, whereas the qualitative crystallinity and molecular structural comparisons are studied using FTIR. Imaging techniques can provide important aspects such as lignin relocalization and cell wall delamination. Integrating EDX with SEM is beneficial to determine the elemental percentage composition altered in CB due to ultrasonication.

Graphical abstract
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
One of the current global challenges is to deliver a sustainable environment by creating a bio-based economy. This is important because the application of non-renewable sources comprises inadequacies in terms of global warming and resource depletion [1]. The plant biomass-based production of biochemicals is increasing rapidly. Corn fiber, corn cob, corn husk, corn gluten meal (CGM), corn bran, corn stover, corn slurry, dried distiller’s grains with solubles (DDGS), corn tassel fibers, and corn stalk are the major corn biomass (CB) generated enormously during pre-harvesting, post-harvesting, and processing stages of CB [2,3,4,5]. CB presents prospective potential as an alternative source for ecological production of bio-products, biogas, and biofuels which helps in boosting the recent circular bioeconomy approach. Some of the potential from CB are explicitly described as follows: (1) corn fiber as a source of hemicellulose (~ 35%) which can be a potential additive in thermoplastic–starch composites to augment bio-plastic life [6]; (2) corn cob as a source of hemicellulose, xylan, and xylooligosaccharides [4]; (3) corn husk showing the capability of creating 9 million tons of natural cellulose fibers [5]; (4) CGM as a preliminary feedstock for the chemicals, food, and energy production [6]; (5) corn bran fulfilling the human nutrition demand accounting its abundance in dietary fiber [7]; and lastly corn stover as a prospective carbon source for the bio-chemicals and biofuels [8].
The pretreatments such as physical, chemical, physicochemical, and biological are indispensable for CB because CB accessibility becomes easy for the microbes/enzymes. Such pretreatments intensify CB to produce biogas, biofuels, bio-chemicals, and bioethanol and also upgrade it for animal feed and composting [9,10,11,12,13]. An effective pretreatment comprises more reactive cellulose, reduction in energy necessities, and least chemical usage and avoids the production of fermentation inhibitors likely furfural. Ultrasonication is a physical method acting as assistance with chemical pretreatment. It is considered a cost-effective process which eradicates restrictions to the mass transfer mechanisms. Moreover, the improvement in enzymatic hydrolysis due to sound waves has been examined [14,15,16].
Recently, imaging and spectroscopic techniques in combination with thermal technique have become a unique characterization tool for investigating the qualitative and quantitative process-induced variations in crystallinity, microstructure, porosity, chemical composition, elemental arrangement, and thermal behavior [17,14,15,16,21]. The characterization of ultrasonicated CB is important for comprehending the flaws for further processing. Ultrasonication being a pretreatment makes the accessibility of carbohydrates such as cellulose, hemicellulose, and lignin for the attack by enzymes or fermentative microbes. The variations in porosity, composition, surface characteristics, crystallinity, and degree of polymerization specifically in the case of CB play an important role to understand the ultrasonicated CB for sustainable conversion to bio-products. Hence, these variations need to be characterized using existing imaging, spectral, and thermal techniques. The characterization of the ultrasonicated CB would reveal the suitability of the materials for further bio-conversion. If it appears that the material is not appropriate, then further processing can be avoided that would make the process sustainable. These characterizations can be achieved using tests on a small-scale laboratory procedure. The bioethanol industries and farms generate huge amounts of by-products and agricultural wastes that can be converted into bio-products using a suitable pretreatment process such as ultrasonic processing (USP) integrated with characterization tool. In the current scenario, these by-products and wastes are an alarming sign for future environmental sustainability and global warming. Therefore, the present review is needed to help majorly to the bioethanol industries and farmers. Furthermore, this is required to disseminate an awareness about the importance of USP-treated CB and its characterization for generating an alternative income source. Literatures have been reported on the effect of pretreatments on various lignocellulosic biomass [22, 23]; however, none of them has discussed the characterization of ultrasonicated CB using imaging, spectral, and thermal techniques. Therefore, the current review focuses on the study of techniques such as X-ray diffraction (XRD), scanning electron microscopy (SEM), thermogravimetric analysis (TGA), Fourier transform infrared spectroscopy (FTIR), and Raman spectroscopy for understanding the effect of ultrasonication on CB. Furthermore, the potential of atomic force microscopy (AFM) and energy-dispersive X-ray spectroscopy (EDX) is reviewed for characterizing ultrasonicated CB considering the fact that currently, there are no studies on the application of AFM and EDX for ultrasonicated CB.
2 Ultrasonic processing
USP has been employed enormously for food processing operations in laboratory research as well as industries [15]. This incipient technology fascinated the scientific community owing to its non-thermal advantages and green nature such as process enhancements, augmented throughputs, cheapness, product superiority, and decreased energy necessity. The USP frequency range varies from 20 kHz to 10 MHz in food processing applications that are further categorized into three ranges:
-
(1)
Low frequency/power ultrasounds (20–100 kHz): In this range, huge amplitude waves are utilized which causes the variations in physicochemical characteristics and/or food structures.
-
(2)
Midway frequency/sonochemistry (100 kHz–1 MHz): In this range, chemical reactions are triggered inside food materials due to generation of free radicals [15]. For both power ultrasound as well as sonochemistry, the sound pierces and disseminates as sinusoidal waves. The medium produces acoustic cavitation through elastic vibrations. This acoustic cavitation includes concurrent expansion and contraction of gas micro- or nano-bubbles in the treated liquid medium. Consequently, pressure waves form and produce energy. The instantaneous and unanticipated bubbles collapse creates exceptionally high localized pressures and temperatures ensuing generation of free radicals and high shear forces which can be utilized to upgrade food processing operations. The induced bubbles collapse distortedly and cause the movement of fluid towards the solid surface. This phenomenon is known as the micro-jetting which alters the surface structures due to abruptness of the heat and mass transfer processes.
-
(3)
High frequency (1–10 MHz): In this range, the sound waves do not alter the food structures and is frequently used for the analytical applications such as measurement of protein, fat and moisture in foods, variations in dough uniformity, oil purity and composition, physical characteristics of batters, and honey adulteration [15].
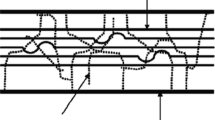
Therefore, the application of USP becomes important for CB owing to the advantages observed for other food items. The working principle of ultrasonic waves is based on cavitation phenomenon. The presence of highly complex lignin and ordered crystalline cellulose present in CB repeatedly delivers resistance to the enzymes and microbes. USP causes the structural modification and creation of oxidizing radicals which result in attacking the intricate matrices. The sound waves break the α-O-4 and β-O-4 bonds in lignin further liberating the lignin and holocellulose (hemicellulose and cellulose) fractions due to production of small cavitation bubbles [17], as illustrated in Fig. 1. The bubbles grow in size and eventually reach to a critical level resulting in severe collapse due to loss in stability. High temperatures (2000–5000 K) and pressures (1800 atm) can be obtained at collapsing point [18]. Additionally, formation of micro-jets, shear, and acoustic streaming takes place on account of ultrasonication [9].
The sound waves in USP work in two distinct ways: (1) direct mode in which a probe creates the vibrations through direct contact with the sample and (2) indirect mode in which a secondary medium such as water bath creates the vibrations imparted to the sample indirectly [19], (Fig. 2). Direct mode involves a very high centralized intensity whereas the indirect mode is applied for larger quantity of solutions at greater uniform intensity [24, 25]. The major components of an USP system are generator, transducer, and emitter, as shown in Fig. 2 a and b. The generator produces electrical energy which is transformed into sound waves by transducer at selected frequencies. The piezoelectric-type transducers convert the electrical current into physical vibrations whereas the magnetostrictive type transforms magnetic field into the physical vibrations. Emitters transfer the sound energy following transducers to the solution containing the CB.
2.1 Energy needs of USP
USP is essentially an energy exhaustive process. The techno-economic modeling is obligatory before scaling up the process on commercial level. Therefore, the optimization of process parameters must be performed. The energy demand and total cost are the two major factors influencing process suitability. The energy in sonication depends primarily on sonication time and power, and sample volume to be pretreated. The energy efficiency in ultrasonication is units of electricity truly degenerated in the CB solution. This energy further recurs in the form of heat which can be measured by calorimetry. There is a direct relation of energy efficiency and input energy during ultrasonication unless the interference among cavitation bubbles begins. The generation of interfering bubbles reduces energy transfer to the CB solution [20]. The CB characteristics and surface area of sonotrode affect the rate of power dissipation. A reduction in ultrasonication energy intake by more than 50% was relative to non-sonicated lignocellulosic biomass, accounting the equal ethanol yields [21]. Thus, USP presents a potential to be incorporated in processing of CB for further applications (Fig. 3).
2.2 Application of USP for CB
USP is commonly used as an assistance to improve yield of a target component during extraction. Most of the studies have conveyed that ultrasonication improves enzymatic hydrolysis of various CB [26,23,28]. The different factors responsible for accelerating the hydrolysis rate by sound waves have been designated as limited degradation of substrate/sample, generation of energy, and probable variation in the conformation of enzyme molecules [23]. The specific purposes of using USP are as follows:
-
(a)
Quality and yield enhancement of hemicellulose and cellulose:
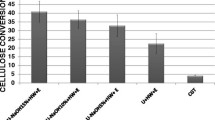
The yield of hemicelluloses such as xylan has been investigated to be higher during extraction assisted by ultrasonication [26, 29]. The xylan extracted from corn cob using ultrasonic-assisted and classical extraction methods was compared [30]. The efficiency of xylan extraction process was improved by sonication because of the reduced extraction time, temperature, and alkali concentration. Xylan with improved yield (~ 28–36%) and higher bioactivity has been extracted from corn cob using alkali-assisted USP [30]. Likewise, it was reported that ultrasonication-assisted alkali extraction can intensify the hemicellulose yield (10–40%) and lessen the corresponding extraction time [22].
-
(b)
Upgradation in saccharification rate:
Saccharification is a bio-hydrolysis process in which a carbohydrate molecule is broken down into its corresponding sugar molecules. The influence of USP on cellulose saccharification rate during enzymatic hydrolysis of corn stover was studied [23]. The results showed that saccharification rate was enhanced by 70% due to ultrasonication-assisted enzymatic hydrolysis in comparison with without ultrasonication. Likewise, mechanical impacts due to collapsing of cavitation bubbles permitted the enzymatic actions in an improved manner in the solid surfaces. Moreover, the optimum temperature was 50 °C for enzymes which provided very high enzymatic hydrolysis rate due to the increased cavitation. Another finding stated a novel ultrasonic-based pretreatment in which the corn stover mixed in sodium percarbonate (SP) was subjected to ultrasonication (US) for the efficient enzymatic saccharification of corn stover [2]. The corn stover dissolved in deionized water with application of US and dissolved in SP solution without application of US were also considered two separate pretreatments. The results indicated that the saccharification of US-SP-pretreated corn stover (2.33 mg/mL) was much higher in comparison with the US-pretreated (1.29 mg/mL) and SP-pretreated corn stover samples (1.45 mg/mL) after 24 h of enzymatic digestion. Besides, the xylose yield without furfural was greater in the case of US-SP-pretreated samples when compared with dilute acid pretreated samples. The US-SP pretreatment can be a suitable technique for future research on different biomass fractions. One of the strongest reasons is that the US-SP pretreatment was described as an antibacterial medium during enzymatic hydrolysis even after 2 days in addition to enhancement in saccharification. Generally, the microorganisms grow during long hydrolysis which is confirmed by the absence of glucose at the end of hydrolysis. In such a case, the antibacterial agents like sodium azide are added to prevent the microbial growth. Therefore, the application of US-SP can also be a cost-effective pretreatment especially in the case of CB where longer hydrolysis is needed due to complex structure. Furthermore, Khanal et al. [31] reported the application of ultrasonication to improve the saccharification and liquefaction for corn slurry. The results indicated that the rate of glucose release inclined around three times in comparison with untreated corn slurry. The enzymatic activity was higher due to ultrasonication. There was no degradation in enzymes on account of ultrasonication. Similar results were acquired in a study by Montalbo-Lomboy et al. [32] for sugary-2 maize (Zea mays L.). Additionally, Jin et al. [24] stated that multi-frequency power ultrasonication/heating-ultrasonication combination enhanced considerably the enzymolysis and degree of hydrolysis for CGM [33, 34].
-
(c)
Delignification:
Delignification of CB augments hydrolysis yields of cellulose that can be improved by USP. The success of enzymatic hydrolysis of corn cob using USP followed the most efficient delignification pretreatment. The maximum delignification and sugar recovery yield were found as 84.7 and 77.6% for corn cob at optimal conditions (sonication time 11.66 min and energy density 10 W/mL) [25]. Delignification with ultrasound was observed for CB in a variety of solvents. Specifically, delignification enhanced when ultrasonication was applied to the corn cob samples in aqueous ammonia [25]. Furthermore, lignin content is reduced in the case of corn cob pretreated by ultrasonication applied to sample bottles with sodium phosphate buffer in it [35]. Overall, the reported delignification ranged from an increase in lignin content by 8.9% in a soda solution to a decrease in lignin by 90.6% in 2% NaOH solution [36]. The vast range of delignification yield could be obtained due to ultrasonicated CB in different solvents. Additionally, carbohydrate loss is always associated with delignification. Therefore, the objective of ultrasonication must be accounted. If the purpose of USP is to augment hydrolysis yields, or comprises further usage of carbohydrates in downstream processing, carbohydrate loss might not be desirable. Some of the past research carried out in relation with ultrasonication of CB for process enhancement has been summarized in Table 1.
3 Spectroscopic, imaging, and thermal techniques
CB is a complex mixture of versatile components and comprises nanocomposites arranged in three-dimensional manner. Some of the major variations in the physical and chemical properties are alteration in lignin or cellulose location, nature of cell wall disruption, variation in elemental composition, and cellulose crystallinity, interactions among cellulose, hemicellulose and lignin, enlarged surface area, improved porosity owing to xylan isolation, decreased degree of polymerization, inclined crystallinity, melting and lignin rearrangement, etc. [2,3,4, 37]. These variations are difficult to characterize by laboratorial methods. Therefore, spectroscopic, imaging, and thermal techniques could be applied for such characterization. For instance, knowing the quantity of cellulose is not sufficient; it is also important to know the exact location of cellulose and the way it interacts with other components such as lignin and hemicellulose. On the other side, the change in cellulose location by sound waves is as important as its removal for betterment of CB hydrolysis. The feasibility of ultrasonicated CB for consequent hydrolysis can be analyzed by nanoscale and microscale scrutiny, as shown in Fig. 4. Still, there are some challenges for characterizing the CB using spectroscopic, imaging, and thermal techniques owing to complicated small sizes.
CB has a multi-scale complicated nature; therefore, smaller and larger scales provide diverse outlines. Moreover, distinct treatments and parameters can be implemented prior to the imaging to enhance the quality of the scrutiny. Some staining procedures are recommended for underlining the cell wall arrangements and their constituents. Various microscopy techniques can be applied to comprehend the variations in the cell wall structures resulted from sound waves.
The spectroscopic, imaging, and thermal techniques such as XRD, SEM, TGA, FTIR combined with Raman spectroscopy, AFM, and EDX are applied to understand the structural as well as functional changes in ultrasonicated CB. These techniques provide an insight about the qualitative and quantitative information like cell wall and microfibril structures, roughness index, crystallinity index, glass transition temperature, and elemental composition which cannot be inferred from other investigations. Earlier, SEM was the most ordinarily used imaging technique at the microscale to comprehend the difference between structures of ultrasonicated and non-ultrasonicated CB to examine their liability for further enzymatic processing; however, it still remains a challenging task.
3.1 X-ray diffraction spectroscopy
XRD is a non-destructive technique for analyzing the structure of crystalline or partially crystalline materials predominantly at the atomic or molecular level; however, it is also utilized for studying the non-crystalline constituents and phase identification of a crystalline material. The basic principle of XRD is positive intervention of single wavelength X-rays and cellulose in a CB sample. Diffraction takes place when light is scattered by a periodic array with long-range order, producing constructive interference at particular angles. The scattering of X-rays from atoms produces a diffraction pattern, which contains information about the atomic arrangement within the crystal.
The cellulose present in CB imparts crystallinity which can be reduced using ultrasonication-assisted and ultrasonication hybrid pretreatments [2, 17, 19, 38]. The cellulosic crystalline part gives strong signals in XRD peaks whereas amorphous region of cellulose represents weak and wide signals in the XRD pattern [39]. The crystallinity becomes important considering the biochemical conversion of lignocellulosic biomass. It is assumed that lower crystallinity is responsible for enhanced bio-conversion of biomass into other value-added products [40,41,42]. On the contrary, studies have stated that better digestibility was even obtained with higher crystalline biomass as well [33]. Furthermore, there are cases where no correlation has been found between digestibility and crystallinity of CB, which is associated with several aspects such as pore size, surface area, hemicellulose, and lignin content.
Du et al. [25] carried out a remarkable study where the authors examined the cellulose crystallinity of corn cob (CC), corn cob soaked in aqueous ammonia (CCSAA), and corn cob pretreated with ultrasound-assisted soaking in aqueous ammonia (CCUSAA) through XRD technique. It was noticed that crystallinity index values were 34.6, 43.7, and 42.7% for CC, CCSAA, and CCUSAA, respectively. It was concluded that CCSAA and CCUSAA pretreatment markedly amplified the crystallinity index of corn cob. Wang et al. [10] investigated the consequence of integrated process of physicochemical pretreatments and enzymatic hydrolysis to lessen the degree of crystallinity in corn straw accordingly enhancing the quantity of reducing sugar. It was found that physical methods (ultrasonic wave, extrusion + ultrasonic wave, extrusion + ultrasound + microwave) in combination with organic compounds (lime + sodium hydroxide + hydrogen peroxide) degraded hemicellulose and lignin more efficiently than untreated corn straw. The best treatments determined were extrusion + microwave + ultrasound + chemical compounds (treatment 1) and extrusion + ultrasonic wave (treatment 2) in grouping with enzymatic hydrolysis. The decomposition rates for hemicelluloses, cellulose, and lignin were upgraded by 74.51%, 61.59%, 56.19%, and reducing sugar yield by 425.98%, respectively for treatment 1. Similarly, decomposition rates for hemicelluloses, cellulose, and lignin were upgraded by 86.05%, 69.61%, 62.88%, and reducing sugar yield by 421.77%, respectively for treatment 2 which were confirmed by XRD patterns. The minimum degree of crystallinity (CrI) was computed as 23.87% for extrusion + ultrasonic wave + chemical compound ensuing processing by enzymes; however, nearly similar CrI was discerned for all the pretreatments without enzymatic processing.
Some of novel pretreatments have been developed using USP with different chemicals for various CB [2]. Nakashima et al. [2] reported a new pretreatment coalescing US and SP to improve the degradation efficiency of corn stover. It was examined that lignin removal and cellulose degradation enhanced to a large extent and crystallinity of untreated, US-pretreated, SP-treated, and combination (US-SP) pretreated samples was analyzed using XRD. A peculiar peak was perceived for untreated and pretreated samples at 22.5° due to cellulose crystallinity. CrI was computed lower for the ultrasonicated samples in comparison with non-ultrasonicated samples on account of ruptured crystalline structure of cellulose by ultrasound. SP-pretreated samples showed higher CrI than US-SP pretreated because only amorphous lignin was fractionated unchanging the crystalline cellulose which was removed during US-SP pretreatment leading to reduction in CrI. Color and swelling properties of US-SP-pretreated corn stover could be distinguished straightforwardly from the untreated one.
Xu et al. [33] investigated the effect of dilute aqueous ammonia integrated with USP on the enzymatic hydrolysis of corn stover, corn cob, and sorghum stalk. It was noticed from the XRD pattern that pretreated samples had higher CrI in comparison with fresh samples. Increase in the cellulose concentration and elimination of hemicellulose and lignin due to pretreatment were explained as the possible reasons for decrease in the crystallinity. On the contrary, Yin et al. [34] found that almost no variation in the crystallinity was marked when corn cob and corn stover were employed with a combination of ultrasound and supercritical carbon dioxide. This leads to an important conclusion that cellulose crystallinity is not the only reason accountable for the enzymatic hydrolysis. There could be other factors responsible for enzymatic hydrolysis.
Segal or peak height, deconvolution, and Ruland–Vonk or amorphous contribution subtraction are the three methods used for the computation of CrI [43,39,45]. Segal or peak height method is ordinarily employed for the CrI measurement [46] for analyzing the effect of USP on distinct CB. Still, this method has some challenges likely the accurate amount of crystal fraction of the cellulose is correlated less with peak height as compared with the peak area [47]. Moreover, it has been seen currently that the size of crystals, association, and alignment of cellulose microfibrils affected by pretreatments are the other critical parameters resulting into error in the critical results [48]. Considering crystallinity studies done on CB, it could be concluded that XRD is an unprecedented tool for ultrasonicated CB owing to its ambiguous behavior.
3.2 Scanning electron microscopy
SEM is a technique that scans the surface of a material by using a concentrated beam of electrons to generate photomicrographs. The signals are produced whenever interaction occurs between atoms and electrons. These signals consist of information about the material composition and surface topography. The frequently used mode of SEM comprises secondary electrons emitted by atoms excited using electron beam and detected through Everhart-Thornley detector. The signal intensity due to secondary electrons is governed by the topographical features of the samples.
SEM is the foremost technique used to find the surface variations in CB due to pretreatments [4, 19, 37, 38, 49]. It can be used to characterize surface in terms of superficial attritions, breakdown, and dislocation of components present in the interior of the semi permeable layer for the easy availability to the enzymes during hydrolysis [37]. However, sample destructivity and conductivity are the two major limitations of SEM. The samples can be coated by gold or carbon to make them conductive [38]. Oven drying of CB should be prevented as it causes significant surface deformation and collapse rather freeze-drying is the best alternative to all drying methods. CB should be kept for quick freezing in liquid N2 just before keeping it into the freeze dryer [37]. High resolution for better recognizing the components occupying in the cell wall could be acquired by spotting using standard stains. Lignin location in the cell wall could be identified with the help of a potassium permanganate, a lignin stain [50]. Quantitative measurements by computing the “roughness index” from SEM micrographs can be performed [40]. A direct relationship between surface roughness and enzymatic hydrolysis was investigated for pretreated corn stover in comparison with fresh samples [41].
The examination of the SEM photomicrographs of the USP-treated CB can provide distinct valuable evidence about the effects of pretreatment on the samples. Additionally, comparison between USP-treated CB and untreated CB might lead to diverse insight into the structural facets of CB. SEM photomicrographs are widely employed during ultrasonication of CB, but most of them consist of assumptions. Even though, there are few studies in which the explanations and inferences from SEM photomicrographs are supported using results of other techniques like AFM and confocal microscopy. Moreover, the studies conducted by the Biomass Surface Characterization Laboratory (BSCL), NREL, also provision the observations from SEM photomicrographs. Some of the selected studies which generate potential knowledge about USP-treated CB through SEM photomicrographs are listed in Table 2.
3.3 Thermogravimetric analysis
TGA is an analytical technique to determine a material’s thermal stability and volatile components by monitoring the weight change with temperature. The measurement is normally carried out in air or in an inert atmosphere, such as helium or argon. Sometimes, the measurement is accomplished in a lean oxygen atmosphere (1 to 5% O2 in N2 or He) to decelerate oxidation.
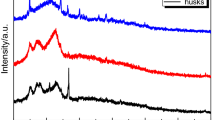
TGA is generally used to study the composition and thermal degradability of CB. Variation in weight observed with respect to temperature could be due to oxidation, decomposition, or desorption of volatile molecules. The disintegration analysis of cellulose, hemicellulose, and lignin present in CB at various temperatures leads to its further utilization for various applications such as biochemical extraction, biofuels, and nutraceutical development. Three temperature regions have been identified earlier for lignocellulosic biomass as follows: the lignin region (350–500 °C), the cellulose region (290–350 °C), and the hemicellulose region (245–290 °C) [23]. Additionally, thermal imaging can be an option to understand the temperature distribution: symmetrical or asymmetrical in ultrasonicated CB. The kind of thermal distribution can be correlated to the extraction efficiency of a nutritional or bioactive component from CB. Almost no studies have been conducted for thermal characterization of ultrasonicated CB; however, Zhang et al. [43] reported an investigation on various characteristics on corn stover during ultrasonic vibration-assisted (UVA) pelleting. The results of their study indicated that UVA formed pellets and particles were more heat resistant in comparison with untreated samples, as illustrated in Fig. 5. The ultrasonicated corn stover showed no peaks in a temperature range of 222–228 °C stipulating elimination of hemicellulose during ultrasonication. Considering lacking in literature on TGA analysis of the ultrasonicated CB, thermal characterization of alkali-pretreated maize tassel fiber has been described herewith [44]. Alkali-pretreated maize tassel fiber has been examined to be more thermally stable comparatively untreated one [44]. Ultrasonication has recently become an integral part of process enhancement for creating the value-added products from distinct biomass; yet, its application is not explored for CB. The thermal decomposition behavior of CB during ultrasonication comes into play accounting its further application; therefore, the role of TGA cannot be circumvented.
Trends in a TGA and b DTG curves for unpretreated and pretreated corn stover samples [43]
3.4 Fourier transform infrared spectroscopy and Raman spectroscopy
FTIR and Raman spectroscopy techniques use the variations in the vibrational energy state of molecules when radiations are imparted on them. Both the techniques differ energy transfer mode and complementary to each other. FTIR involves subjecting the CB sample to infrared source leading to vibrational excitation of molecules. Raman spectroscopy is based on the flexible and inflexible scattering outcome. Radiation source in Raman spectroscopy is highly active monochrome. The molecules travel to their native vibrating location for resting after reaching to a virtual location by photon re-emanation at exactly the similar frequency of incident photons in case of flexible scattering outcome. There is no absorption of energy by molecules from the incident radiations. On the other side, in inflexible or Raman scattering, the agitated molecules rest at distinguished vibrational locations.
The CrI can also be determined using FTIR through measurement of the peak areas [45]. The FTIR absorption peak region between 1420 and 1430 cm−1 is known as the crystallinity region. The CrI values computed by FTIR were compared with the XRD values and were seen in a good agreement. Also, FTIR is considered to be an innovative technique for the measurement of changes in the cellulose structure due to USP. Among the current techniques, FTIR is the simplest one with least sample groundwork and no prior particular experience is needed. Though only relative quantities are obtained from FTIR due to the presence of amorphous and crystalline portions. In addition to CrI, hydrogen linkages, cellulose II and cellulose I, cellulose Iα (3231 cm−1), and Iβ (3429 cm−1), the informative facts at the supramolecular state concerning acetyl and lignin molecules, the bio-chemical composition of the USP-treated CB can be found using FTIR. Fan et al. [45] specified FTIR as the best technique in precise scrutiny of variations caused in hemicellulose, cellulose, waxes, pectin, lignin, and mineral components of CB owing to USP. The variation in interface, bio-chemical composition, and henceforth characteristics of composites and fibers from USP-treated CB could also be efficaciously recognized by FTIR. Nevertheless, we reverently differ with such declaration for getting details about the USP-treated CB samples; as displacement of bands has arisen, the results are not at all times duplicable, and the absolute values are meaningfully dissimilar, causing this technique dubious for the designated claims, predominantly compositional investigation. Yet, so far, the qualitative examination of the crystallinity changes by FTIR in USP-treated CB continually works in a sound manner, but not by ATR-FTIR.
FTIR helps in analyzing the components present in the plant cell wall [51]. The CB was examined to see the effect of ultrasonication on the pectin, lignin, and hemicellulose content [49]. FTIR spectra showed the removal of these components due to ultrasonication. The CB comprises –OH groups due to presence of cellulose which results in the inter- and intra-molecular hydrogen linkages. These molecular level linkages lead to the formation of amorphous and crystalline portions. Moreover, these linkages are responsible for the mechanical and chemical characteristics of the CB. Hence, FTIR can be indirectly used for examining such characteristics. On the contrary, there was no significant difference between dual power ultrasonicated and non-ultrasonicated zein [52]. Similarly, there were insignificant variations in inter-molecular and intra-molecular linkages for ultrasonicated corn stover. [53]. Furthermore, another study revealed the effect of ultrasonication power by FTIR on xylan fraction of corn hull in alkaline, neutral, and aqueous solvents [54]. Their results showed that larger ultrasound power resulted in the rise in intensity at 1600 cm−1 and further shifting to 1580 cm−1 in the case of samples in alkaline solvent. This specified creation of unsaturated structures with enolic and α,β-unsaturated carbonyl groups. However, these structures were also present in the untreated samples in quantities imperceptible by FTIR spectra.
FTIR and Raman spectroscopic techniques differentiate from one another indicating that both provide in-depth information synergistically. Both are constrained up to higher and lower number of polar connections, respectively. The symmetric and non-symmetric vibrations frequently detect strong Raman and FTIR bands, correspondingly. For instance, carbon-carbon double bonds are weaker in the IR and stronger in the Raman region; carbonyl groups are stronger in the IR and weaker in the Raman region. Qu et al. [55] evaluated the yield of carbon fiber from high ash content lignin of corn stover. The results indicated that Raman spectra were able to quantify the carbon defects in the fiber. The two peaks such as G (1595 cm−1) and D (1365 cm−1) were responsible for the C-C linkages stretching in graphite and irregularity in fiber structure, respectively. The ratio of intensities at D and G (Di/Gi = 2.53 for carbon fiber) was found directly proportional to the defects in the fiber. Different peak ratios in Raman spectra have been reported in literature as a measure of crystallinity. Moreover, the relative intensity ratios of the Raman bands at 380 and 1096 cm−1 bands and those of 1481 and 1462 cm−1 in cellulose I [56] are together stated as crystallinity measures. Regrettably, FTIR and Raman techniques approach the challenges when characterizing the crystallinity in the primary cell walls of CB owing to the interfering signals from another cell wall constituents. Selected studies stated in the literature for FTIR characterization of ultrasonicated CB are summarized in Table 3.
4 Potential of atomic force microscopy
At nanoscale, AFM provides the physicochemical and topographical assets of the CB [58]. AFM comprises a cantilever with a probe (sharp tip) which scans the sample surface. Cantilever deflection occurs due to the force between the sample surface and probe once the tip comes into close proximity of the sample. Mechanical contact forces, capillary forces, van der Waals forces, electrostatic forces, and magnetic forces are the foremost which play an important role in AFM measurements. Contact, non-contact, and tapping are the three modes of operation in AFM depending upon the tip movement. Image formation is a plotting technique that creates a color mapping by varying the x-y position of the tip while recording and scanning the intensity of control signal to every x-y coordinate. The image states the intensity of a value as a hue.
AFM can be applied to analyze the texture and morphology of CB surface which alters due to alkali extraction [18, 53, 59, 60]. Kambli et al. [58] reported the application of AFM to examine the surface texture and morphology of cellulose fibers isolated from corn husks using alkali-based extraction. Their results indicated that there were maximally irregular and uneven surfaces of cellulose fibers. The computed root mean square (RMS) roughness and average roughness of corn husk fibers were 250.5 and 189.7 nm, respectively. Similarly, AFM imaging was used to detect microfibrils arrangement, cell wall boundaries, and middle lamellas, and it was found that such microstructures were similar to the natural state [38]. Zhang et al. [61] carried out a comparison among natural, dilute sulfuric acid (DSA) pretreated, and delignified corn stover (CS) samples. The results of their study revealed that the natural CS sample was covered with non-uniformly distributed lignin. The recognition signals were not observed due to the absence of cellulose networks. The cellulose microfibrils were seen slightly in case of 0.5% DSA pretreated samples which shows CS susceptibility for acid pretreatments. Furthermore, the compact structure of cellulose was discerned for delignified CS and microfibrils of crystalline cellulose were appeared barely. A little cluster possibly of lignin could be perceived; however, they were not identified in recognition images.
AFM can be combined with optical microscopy techniques for much better results. Sample preparation, metal coating, drying, and staining are not mandatory for acquiring 3D topographical AFM images. Some of the shortcomings are low scanning speeds, tip artifacts, data misunderstanding, etc. Furthermore, AFM imaging analysis is a little bit cumbersome in comparison with SEM imaging technique. Besides, ultrasonicated CB can be characterized in terms of surface roughness, porosity, interactive forces among hemicellulose, cellulose, and lignin, and identification of hydrophobic-hydrophilic regions using AFM imaging. Yet, there are no studies found related to application of AFM imaging for ultrasonicated CB.
5 Potential of energy-dispersive X-ray spectroscopy
EDX involves an accelerated electron ray imparted on the outer surface of CB samples which generates distinctive X-rays. EDX has been employed for the elemental composition of alkali-pretreated corn husk fibers and corn straw fibers [18, 62]. The intensities and energies of X-rays provide the corresponding percentage amount of elements.
Kambli et al. [58] investigated the compositional difference between bleached and unbleached corn husk fibers (CHFs). The study examined that the carbon content in CHFs extracted chemically was 57.99% (on weight), whereas it is augmented in the bleached samples (62.54%). Similarly, the oxygen present in the bleached and unbleached CHFs was 36.8% and 40.49%, respectively. Trace elements such as silicon (0.20 wt.%) and magnesium (0.13 wt.%) were discerned in the bleached corn husk fibers in comparison with their absence in unbleached samples. On the contrary, nickel was found absent in the bleached CHFs while it was present in the unbleached CHFs (0.35 wt.%). Similarly, Zang et al. [63] applied the EDX during development of superhydrophobic corn straw fibers (CSFs) using silica (SiO2) and (heptadecafluoro-1, 1, 2, 2-tetradecyl) trimethoxysilane (TES). Carbon and oxygen peaks were observed in the fresh CSFs; however, two new peaks of silicon and fluorine due to the presence of SiO2 and PTES in the developed superhydrophobic CSFs were revealed thereby providing a proof for the existence of SiO2 and PTES. Furthermore, Li et al. [64] performed a combination of ultrasonic and Fe+3 ions to improve selectivity of acid hydrolysis for preparation of microcrystalline cellulose which was analyzed by EDX. It was investigated that total amount of carbon and oxygen was 72.54% whereas a little amount of Fe+2 ions (0.02%) was also noticed. It shows showing almost no occurrence in the prepared hydrocellulose samples that leads to high purity. Overall, no variation in the elemental composition of fibers was evaluated which results in pure final hydrocellulose.
Still no studies have been researched for the elemental characterization of ultrasonicated CB using EDX. It is expected that EDX can certify itself to be a bio-spectral technique for its further utilization accounting the facts such as elemental variations, non-existence of certain elements, and detection of novel elements due to USP. Therefore, EDX shows potential in evaluation of ultrasonicated CB samples.
6 Future outlook
The value addition to the enormously generated CB is essential for the sustainability of corn industries. Therefore, characterization of USP or similarly treated CB comes as priority in determining their suitability biorefinery applications. Crystallinity is the foremost parameter creating problems in the biorefinery value chain of CB. The computed crystallinity index based on amorphous regions is ambiguous; yet, it is broadly utilized in the literature. Furthermore, the informed numerical quantities of crystallinity in the past, frequently named crystallinity index, are heavily reliant on the technique of crystallinity determination, data assessment practice, and perfectness of CB sample. Moreover, neither imaging nor compositional investigation can be utilized for exploring the crystallinity of CB. Therefore, the XRD, FTIR, or Raman spectroscopy should be explored for such purpose. The SEM, EDX, and AFM should be integrated to reveal the changes in the CB due to USP for analyzing the variations in the structure of primary and secondary cell walls, microfibril size, elemental composition, and degradation of pretreated CB.
7 Conclusions
The spectroscopic (FTIR, Raman spectroscopy, XRD, EDX), imaging (SEM, AFM), and thermal (TGA) techniques have been used to evaluate the physicochemical changes in the ultrasonicated CB. The XRD, FTIR, and Raman techniques have the potential to examine the variations in the β-D-glucan structure of cellulose, bio composition, and analyze the transformation of crystalline cellulose into amorphous cellulose due to USP in both qualitative and quantitative manner. The XRD yielded the cellulose crystallinity indices of 43.7 and 42.7% for non-USP and USP-pretreated CB, respectively. Similarly, XRD measured lesser degree of crystallinity for USP-treated CB (23.9%) as compared with untreated CB (46.9%). Furthermore, the relative intensity ratios of the Raman bands at 380 and 1096 cm−1 can represent crystallinity of cellulose. The FTIR spectra region between 1420 and 1430 cm−1 might be considered a determinant of crystallinity. However, these techniques have challenges to characterize the crystallinity in the primary cell walls of pretreated and untreated CB owing to the interfering signals from another cell wall constituent. SEM can reveal the changes like lignin relocalization, superficial attritions, breakdown, and cell wall delamination in CB due to USP. EDX spectroscopy could be integrated with SEM to provide the elemental percentage composition varied in CB due to USP. TGA can identify the weight loss of ultrasonicated CB during thermal degradation, and elimination of components such as cellulose, hemicellulose, and lignin due to USP. AFM is a potential technique for computing porosity and roughness of particles at nano level. The measurements would further provide insights into the development and optimization of ultrasonication pretreatment for CB. The characterization methods and techniques accounted for ultrasonicated CB will provide deeper perceptions as the CB industry encounters the problems with variations in feedstock quality.
Abbreviations
- AFM:
-
atomic force microscopy
- CB:
-
corn biomass
- CC:
-
corn cob
- CCSAA:
-
corn cob soaked in aqueous ammonia
- CCUSAA:
-
corn cob pretreated by ultrasound-assisted soaking in aqueous ammonia
- CGM:
-
corn gluten meal
- CHFs:
-
corn husk fibers
- CrI:
-
degree of crystallinity
- CS:
-
corn stover
- CSFs:
-
corn straw fibers
- DDGS:
-
dried distiller’s grains with solubles
- DSA:
-
dilute sulfuric acid
- EDX:
-
energy-dispersive X-ray spectroscopy
- FTIR:
-
Fourier transform infrared spectroscopy
- RMS:
-
root mean square
- SEM:
-
scanning electron microscopy
- SP:
-
sodium percarbonate
- TGA:
-
thermogravimetric analysis
- USP:
-
ultrasonic processing
- UVA:
-
ultrasonic vibration-assisted
- XRD:
-
X-ray diffraction
References
Bessou C, Ferchaud F, Gabrielle B et al (2011) Biofuels, greenhouse gases and climate change. A review. Sustain Agric 2:365–468
Nakashima K, Ebi Y, Kubo M et al (2016) Pretreatment combining ultrasound and sodium percarbonate under mild conditions for efficient degradation of corn stover. Ultrason Sonochem 29:455–460. https://doi.org/10.1016/j.ultsonch.2015.10.017
Gáspár M, Juhász T, Szengyel Z, Réczey K (2005) Fractionation and utilisation of corn fibre carbohydrates. Process Biochem 40:1183–1188. https://doi.org/10.1016/j.procbio.2004.04.004
Garrote G, Domínguez H, Parajó JC (2002) Autohydrolysis of corncob: study of non-isothermal operation for xylooligosaccharide production. J Food Eng 52:211–218. https://doi.org/10.1016/S0260-8774(01)00108-X
Sari NH, Wardana ING, Irawan YS, Siswanto E (2017) The effect of sodium hydroxide on chemical and mechanical properties of corn husk fiber. Orient J Chem 33:3037–3042. https://doi.org/10.13005/ojc/330642
Shukla R, Cheryan M (2001) Zein: The industrial protein from corn. Ind Crop Prod 13:171–192. https://doi.org/10.1016/S0926-6690(00)00064-9
Rose DJ, Inglett GE, Liu SX (2010) Utilisation of corn (Zea mays) bran and corn fiber in the production of food components. J Sci Food Agric 90:915–924. https://doi.org/10.1002/jsfa.3915
Menon V, Rao M (2012) Trends in bioconversion of lignocellulose: biofuels, platform chemicals & biorefinery concept. Prog Energy Combust Sci 38:522–550. https://doi.org/10.1016/j.pecs.2012.02.002
Subhedar PB, Gogate PR (2013) Intensification of enzymatic hydrolysis of lignocellulose using ultrasound for efficient bioethanol production: a review. Ind Eng Chem Res 52:11816–11828. https://doi.org/10.1021/ie401286z
Wang P, Liu C, Chang J et al (2019) Effect of physicochemical pretreatments plus enzymatic hydrolysis on the composition and morphologic structure of corn straw. Renew Energy 138:502–508. https://doi.org/10.1016/j.renene.2019.01.118
Karimi K (2014) Current and future ABE processes. Biofuel Res J 4:77
Salehian P, Karimi K, Zilouei H, Jeihanipour A (2013) Improvement of biogas production from pine wood by alkali pretreatment. Fuel 106:484–489. https://doi.org/10.1016/j.fuel.2012.12.092
Karimi K, Shafiei M, Kumar R (2013) Progress in physical and chemical pretreatment of lignocellulosic biomass. In: Gupta, VK, Tuohy, M G (Eds.), Biofuel Technologies. Springer, Berlin, Heidelberg, pp. 53–96.
Kumar R, Wyman CE (2013) Physical and chemical features of pretreated biomass that influence macro-/micro-accessibility and biological processing. In: Wyman CE (ed) Aqueous pretreatment of plant biomass for biological and chemical conversion to fuels and chemicals, 1st edn. Wiley, New York, pp 281–310
Tobergte DR, Curtis S (2013) Ultrasound technologies for food bioprocessing. Springer, New York
Bussemaker MJ, Zhang D (2013) Effect of ultrasound on lignocellulosic biomass as a pretreatment for biorefinery and biofuel applications. Ind Eng Chem Res 52:3563–3580. https://doi.org/10.1021/ie3022785
Kumar AK, Sharma S (2017) Recent updates on different methods of pretreatment of lignocellulosic feedstocks: a review. Bioresour Bioprocess 4:7. https://doi.org/10.1186/s40643-017-0137-9
Kunaver M, Jasiukaityte E, Čuk N (2012) Ultrasonically assisted liquefaction of lignocellulosic materials. Bioresour Technol 103:360–366. https://doi.org/10.1016/j.biortech.2011.09.051
Huezo L, Shah A, Michel FC (2019) Effects of ultrasound on fermentation of glucose to ethanol by saccharomyces cerevisiae. Fermentation 5:1–14. https://doi.org/10.3390/fermentation5010016
Gogate PR, Sutkar VS, Pandit AB (2011) Sonochemical reactors: Important design and scale up considerations with a special emphasis on heterogeneous systems. Chem Eng J 166:1066–1082. https://doi.org/10.1016/j.cej.2010.11.069
Nitayavardhana S, Shrestha P, Rasmussen ML et al (2010) Ultrasound improved ethanol fermentation from cassava chips in cassava-based ethanol plants. Bioresour Technol 101:2741–2747. https://doi.org/10.1016/j.biortech.2009.10.075
Ebringerová A, Hromádková Z (2002) Effect of ultrasound on the extractibility of corn bran hemicelluloses. Ultrason Sonochem 9:225–229. https://doi.org/10.1016/S1350-4177(01)00124-9
Zhang YQ, Fu E, Liang J (2008) Effect of ultrasonic waves on the saccharification processes of lignocellulose. Chem Eng Technol 31:1510–1515. https://doi.org/10.1002/ceat.200700407
Jin J, Ma H, Wang K et al (2015) Effects of multi-frequency power ultrasound on the enzymolysis and structural characteristics of corn gluten meal. Ultrason Sonochem 24:55–64. https://doi.org/10.1016/j.ultsonch.2014.12.013
Du R, Su R, Qi W, He Z (2018) Enhanced enzymatic hydrolysis of corncob by ultrasound-assisted soaking in aqueous ammonia pretreatment. 3. Biotech 8:166. https://doi.org/10.1007/s13205-018-1186-2
Montalbo-Lomboy M, Khanal SK, van Leeuwen J et al (2010) Ultrasonic pretreatment of corn slurry for saccharification: a comparison of batch and continuous systems. Ultrason Sonochem 17:939–946. https://doi.org/10.1016/j.ultsonch.2010.01.013
Dong C, Chen J (2019) Optimization of process parameters for anaerobic fermentation of corn stalk based on least squares support vector machine. Bioresour Technol 271:174–181. https://doi.org/10.1016/j.biortech.2018.09.085
Nikolić S, Mojović L, Rakin M et al (2010) Ultrasound-assisted production of bioethanol by simultaneous saccharification and fermentation of corn meal. Food Chem 122:216–222. https://doi.org/10.1016/j.foodchem.2010.02.063
Yachmenev V, Condon B, Klasson T, Lambert A (2009) Acceleration of the enzymatic hydrolysis of corn stover and sugar cane bagasse celluloses by low intensity uniform ultrasound. J Biobased Mater Bioenergy 3:25–31. https://doi.org/10.1166/jbmb.2009.1002
Hroma Z, Ebringerova A (1999) Study of the classical and ultrasound-assisted extraction of the corn cob xylan. Ind Crop Prod 9:101–109
Khanal SK, Montalbo M, Van Leeuwen JH et al (2007) Ultrasound enhanced glucose release from corn in ethanol plants. Biotechnol Bioeng 98:978–985. https://doi.org/10.1002/bit
Montalbo-lomboy M, Johnson L, Kumar S et al (2010) Sonication of sugary-2 corn: a potential pretreatment to enhance sugar release sugar release. Bioresour Technol 101:351–358. https://doi.org/10.1016/j.biortech.2009.07.075
Xu QQ, Zhao MJ, Yu ZZ et al (2017) Enhancing enzymatic hydrolysis of corn cob, corn stover and sorghum stalk by dilute aqueous ammonia combined with ultrasonic pretreatment. Ind Crop Prod 109:220–226. https://doi.org/10.1016/j.indcrop.2017.08.038
Yin J, Hao L, Yu W et al (2014) Enzymatic hydrolysis enhancement of corn lignocellulose by supercritical CO2 combined with ultrasound pretreatment. Chin J Catal 35:763–769. https://doi.org/10.1016/s1872-2067(14)60040-1
Pérez-Rodríguez N, García-Bernet D, Domínguez JM (2016) Effects of enzymatic hydrolysis and ultrasounds pretreatments on corn cob and vine trimming shoots for biogas production. Bioresour Technol 221:130–138. https://doi.org/10.1016/j.biortech.2016.09.013
García A, Alriols MG, Llano-Ponte R, Labidi J (2011) Ultrasound-assisted fractionation of the lignocellulosic material. Bioresour Technol 102:6326–6330. https://doi.org/10.1016/j.biortech.2011.02.045
Donohoe BS, Vinzant TB, Elander RT et al (2011) Surface and ultrastructural characterization of raw and pretreated switchgrass. Bioresour Technol 102:11097–11104. https://doi.org/10.1016/j.biortech.2011.03.092
Yarbrough JM, Himmel ME, Ding SY (2009) Plant cell wall characterization using scanning probe microscopy techniques. Biotechnol Biofuels 2:1–11. https://doi.org/10.1186/1754-6834-2-17
Terinte N, Ibbett R, Schuster KC (2011) Overview on native cellulose & microcrystalline cellulose I structures studies by XRD (WAXD): comparison between measurement techniques. Lenzinger Berichte 89:118–131
Banerjee S, Yang R, Courchene CE, Conners TE (2009) Scanning electron microscopy measurements of the surface roughness of paper. Ind Eng Chem Res 48:4322–4325. https://doi.org/10.1021/ie900029v
Ciesielski A, Samorì P (2014) Graphene via sonication assisted liquid-phase exfoliation. Chem Soc Rev 43:381–398. https://doi.org/10.1039/c3cs60217f
Ostovareh S, Karimi K, Zamani A (2015) Efficient conversion of sweet sorghum stalks to biogas and ethanol using organosolv pretreatment. Ind Crop Prod 66:170–177. https://doi.org/10.1016/j.indcrop.2014.12.023
Zhang Q, Zhang P, Pei Z, Wang D (2017) Investigation on characteristics of corn stover and sorghum stalk processed by ultrasonic vibration-assisted pelleting. Renew Energy 101:1075–1086. https://doi.org/10.1016/j.renene.2016.09.071
Maepa CE, Jayaramudu J, Okonkwo JO et al (2015) Extraction and characterization of natural cellulose fibers from maize tassel. Int J Polym Anal Charact 20:99–109. https://doi.org/10.1080/1023666X.2014.961118
Fan M, Dai D, Huang B (2012) Fourier transform infrared spectroscopy for natural fibres. Fourier Transform - Materials Analysis 3:45–68. https://doi.org/10.5772/35482
Segal L, Creely JJ, Martin AE, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the x-ray diffractometer. Text Res J 29:786–794. https://doi.org/10.1177/004051755902901003
Ju X, Bowden M, Brown EE, Zhang X (2015) An improved X-ray diffraction method for cellulose crystallinity measurement. Carbohydr Polym 123:476–481. https://doi.org/10.1016/j.carbpol.2014.12.071
French AD, Santiago Cintrón M (2013) Cellulose polymorphy, crystallite size, and the segal crystallinity index. Cellulose 20:583–588. https://doi.org/10.1007/s10570-012-9833-y
Krishnaiah P, Ratnam CT, Manickam S (2017) Enhancements in crystallinity, thermal stability, tensile modulus and strength of sisal fibres and their PP composites induced by the synergistic effects of alkali and high intensity ultrasound (HIU) treatments. Ultrason Sonochem 34:729–742. https://doi.org/10.1016/j.ultsonch.2016.07.008
Karp EM, Resch MG, Donohoe BS et al (2015) Alkaline pretreatment of switchgrass. ACS Sustain Chem Eng 3:1479–1491. https://doi.org/10.1021/acssuschemeng.5b00201
Mouille G, Robin S, Lecomte M et al (2003) Classification and identification of Arabidopsis cell wall mutants using Fourier-transform infrared (FT-IR) microspectroscopy. Plant J 35:393–404. https://doi.org/10.1046/j.1365-313X.2003.01807.x
Zhou C, Hu J, Yu X et al (2017) Heat and/or ultrasound pretreatments motivated enzymolysis of corn gluten meal: hydrolysis kinetics and protein structure. LWT Food Sci Technol 77:488–496. https://doi.org/10.1016/j.lwt.2016.06.048
Tian S, Wang Z, Fan Z, Zuo L (2012) Comparison of ultrasonic and CO2 laser pretreatment methods on enzyme digestibility of corn stover. Int J Mol Sci 13:4141–4152. https://doi.org/10.3390/ijms13044141
Ebringerová A, Hromádková Z (1997) The effect of ultrasound on the structure and properties of the water-soluble corn hull heteroxylan. Ultrason Sonochem 4(4):305–309. https://doi.org/10.1016/S1350-4177(97)00037-0
Qu W, Liu J, Xue Y et al (2018) Potential of producing carbon fiber from biorefinery corn stover lignin with high ash content. J Appl Polym Sci 135:1–11. https://doi.org/10.1002/app.45736
Agarwal UP, Reiner RS, Ralph SA (2010) Cellulose I crystallinity determination using FT–Raman spectroscopy: univariate and multivariate methods. Cellulose 17:721–733. https://doi.org/10.1007/s10570-010-9420-z
Jin J, Ma H, Wang B, Yagoub AEGA, Wang K, He R, Zhou C (2016) Effects and mechanism of dual-frequency power ultrasound on the molecular weight distribution of corn gluten meal hydrolysates. Ultrason Sonochem 30:44–51. https://doi.org/10.1016/j.ultsonch.2015.11.021
Kambli ND, Samanta KK, Basak S et al (2018) Characterization of the corn husk fibre and improvement in its thermal stability by banana pseudostem sap. Cellulose 25:5241–5257. https://doi.org/10.1007/s10570-018-1931-z
Sluiter A, Hames B, Ruiz R, Scarlata C, Sluiter J, Templeton D, Crocker D (2008) Determination of structural carbohydrates and lignin in biomass. Laboratory Analytical Procedure, NREL/TP-510-42618.
Ciolacu D, Ciolacu F, Popa VI (2011) Amorphous cellulose–structure and characterization. Cellul Chem Technol 45:13–21
Zhang M, Chen G, Kumar R, Xu B (2013) Mapping out the structural changes of natural and pretreated plant cell wall surfaces by atomic force microscopy single molecular recognition imaging. Biotechnol Biofuels 6:1–11. https://doi.org/10.1186/1754-6834-6-147
Isogai A, Atalla AH (1991) Amorphous celluloses stable in aqueous media. Regeneration from SO2-amine solvent systems. J Polym Sci Part A: Polym. Chem 29:113–119. https://doi.org/10.1002/pola.1991.080290113
Zang D, Zhang M, Liu F, Wang C (2016) Superhydrophobic/superoleophilic corn straw fibers as effective oil sorbents for the recovery of spilled oil. J Chem Technol Biotechnol 91:2449–2456. https://doi.org/10.1002/jctb.4834
Li J, Qiang D, Zhang M et al (2015) Joint action of ultrasonic and Fe3+ to improve selectivity of acid hydrolysis for microcrystalline cellulose. Carbohydr Polym 129:44–49. https://doi.org/10.1016/j.carbpol.2015.04.034
Acknowledgements
This work was supported by BioFuelNet Canada; Ontario Ministry of Agriculture, Food and Rural Affairs, Guelph Ontario, IGPC Ethanol Inc., Aylmer, Ontario, Canada.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sharma, S., Pradhan, R., Manickavasagan, A. et al. Characterization of ultrasonic-treated corn crop biomass using imaging, spectral and thermal techniques: a review. Biomass Conv. Bioref. 12, 1393–1408 (2022). https://doi.org/10.1007/s13399-020-00748-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-020-00748-4