Abstract
A crucial element in the “the landscape of fear” concept is that prey animals are aware of varying levels of predation risk at a spatial scale. This often leads to a negative spatial relationship between prey and predator in which prey avoid the most risky sites in the landscape. In this paper, we argue that our understanding of large carnivore-ungulate interactions is biased by studies from highly heterogeneous landscapes (e.g. the Yellowstone National Park). Due to a high availability of refuges and foraging sites in such landscapes, prey are able to reduce predation risk by showing habitat shifts. Besides the spatial heterogeneity at the landscape scale, the ungulate response to predation risk can be affected by the hunting mode (stalking vs. cursorial) of the predator. We propose that prey cannot easily avoid predation risk by moving to less risky habitats in more homogenous landscapes with concentrated food resources, especially where the large carnivores’ assemblage includes both stalking and cursorial species. No distinct refuges for prey may occur in such landscapes due to equally high accessibility to predators in all habitats, while concentrated resources make prey distribution more predictable. We discuss a model of a densely forested landscape based on a case study of the Białowieża Primeval Forest, Poland. Within this landscape, ungulates focus their foraging activity on small food-rich forest gaps, which turn out to be “death traps” as the gaps are primarily targeted by predators (stalking lynx and cursorial wolf) while hunting. No alternative of moving to low predation risk areas exist for prey due to risk from wolves in surrounding closed-canopy forest. As a result, the prey is exposed to constant high predation pressure in contrast to heterogeneous landscapes with less concentrated resources and more refuge areas. Future research should focus on explaining how ungulates are coping with predation risk in these landscapes that offer little choice of escaping predation by considering behavioural and physiological (e.g. metabolic, hormonal) responses.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Avoidance of predation risk by animals is a widely accepted concept of the dynamic interactions between prey and their predators (e.g. Brown 1999; Verdolin 2006; Hammond et al. 2007; Hochman and Kotler 2007; Valeix et al. 2009b; Thaker et al. 2011; Burkepile et al. 2013; Laundré et al. 2013; Venter et al. 2014). Yet as prey species do not always have the chance to directly confront their predators, perception of predation risk should often rely on indirect cues. The awareness of various potential sources of mortality, including predation, should thus induce behavioural strategies in the prey, which are based on the trade-off between the need for acquiring food and the need for safety (Brown 1999). That prey animals are altering their habitat use or behaviour by trading the decrease in forage quality or quantity over the higher security from predation is well documented (e.g. Sih 1980; Creel et al. 2005; Kauffman et al. 2007; Barnier et al. 2014). However, the mechanism behind this trade-off is not easily understood. Among others, the components of risk can be differently perceived by various prey species (Burkepile et al. 2013), can vary with the hunting mode of the predators (Thaker et al. 2011; Bouskila 1995; Verdolin 2006) and be influenced by various environmental factors (Warfe and Barmuta 2004).
Intuitively, habitat structure may play an important moderating role during interactions between predator and prey. It may either facilitate or hamper both survival of the prey and hunting success of the predator. This has been empirically demonstrated for wolf (Canis lupus)–moose (Alces alces) (Kunkel and Pletscher 2000) and wolf–elk interactions (Kauffman et al. 2007). For the prey, some habitat characteristics, such as openness, can be positive with regard to foraging while negative with regard to predation risk (Brown 1999; Creel et al. 2005; Hernández and Laundré 2005; Hebblewhite and Merrill 2009; Rieucau et al. 2009). For the predator, the same traits may imply the abundance of prey as well as its low accessibility or catchability (Hopcraft et al. 2005). Little is known on how these relationships differ between areas with contrasting landscapes, i.e. heterogeneous landscapes offering a variety of refuges and foraging sites of different quality and homogenous areas consisting of a single habitat type with only highly concentrated food resources. The consequences of the landscape heterogeneity may be particularly important when the presence of predators is highly unpredictable, especially in case of large mammalian carnivores that typically move long distances in short time (e.g. Beier et al. 1995; Valeix et al. 2011). Here, the predator has an advantage over its prey since it may quickly move from one habitat patch to another choosing the best conditions for a successful hunt. As a result, the prey has imperfect knowledge of the predator’s current whereabouts and the actual degree of risk at a certain location (Brown et al. 1999). That is likely the reason why the habitat features may constitute one of most important cues for prey species of potential predation risk (Brown et al. 1999; Laundré et al. 2001; Laundré et al. 2010). However, when the prey selects for sites with the highest escape probability, what should then be the most successful strategy for the predator? Should the predator focus on areas where the prey is more easily caught (e.g. Hopcraft et al. 2005) or on the areas with the highest chances of encountering it?
If the strategies of predators’ detection and avoidance by prey as well as searching for and acquiring prey by predators are to be evolutionarily stable (Maynard Smith and Price 1973; Kriva and Cressman 2009), ideally the prey should seek patches richest in food and lowest in predation risk, and the predator should focus on sites where prey is most abundant and/or most vulnerable to predation (Sih 2005). What emerges from these ostensibly conflicting strategies is that it often leads to a negative relationship between the prey and predators’ spatial distribution (Kunkel and Pletscher 2000; Orrock et al. 2004; Creel et al. 2005; Thaker et al. 2011). An experimental study on dragonfly–tadpole spatial interactions revealed that when predators used a high resource patch more, prey used that patch less (Hammond et al. 2007). The outcome of these interactions in the wild may, however, depend on various factors including a behavioural interplay between predator and prey (Mitchell and Lima 2002). The spatial correlation between predator and prey among the Atlantic fish species was shown to be positive at larger scales and negative at smaller scales, and it was additionally affected by the presence of refuges (Rose and Leggett 1990). Negative spatial correlation of predator and prey may thus be more likely to occur in composite, spatially heterogeneous habitats as suggested by a study of a spider–Collembola system (Birkhofer et al. 2010). We expect that in vertebrates, these correlations may be more complex. Mathematical modelling based on “shell-game” theory showed that in systems with predators having good spatial memory, prey should randomize their foraging sites in order to decrease predictability of being encountered (Mitchell and Lima 2002). Accordingly, a recent field study in a wolf–caribou–moose system revealed completely different patterns of encounters for the predators with its prey and for the prey with its predator (Courbin et al. 2013) suggesting there is an asymmetry in relative probabilities of encounters between predators and prey. While there was a high risk of encountering wolves by caribou in a given land-cover type, there could be low probability of crossing caribou’s path by wolves in the same area, relative to the time they spend there. It is thus not surprising that asymmetric encounter probabilities may work best in terrestrial heterogeneous ecosystems with clearly distinguished refuges such as the Yellowstone ecosystem where ungulate prey species showed a shift towards low-risk areas on a large scale once wolves returned to this system (Creel et al. 2005; Hernández and Laundré 2005; Fortin et al. 2005; Ripple and Beschta 2006).
Here, by analysing potential factors and conditions which may affect the observed outcome of predator–prey behavioural interactions, we present an alternative concept of responses of ungulates in relation to predation risk. First, we explore how (1) type of predator (cursorial versus stalking) and (2) spatial structure of landscape (patchy versus homogeneous distribution of resources) affect anti-predator behaviour of ungulates. Next, based on recent studies conducted in the densely forested Białowieża Primeval Forest (BPF), Poland, we present a new concept of the interactions between large carnivores and ungulates. As this area is composed primarily of forest habitat, we regard it as a relatively homogenous landscape as compared to well-studied systems in North America (Yellowstone) or African savannahs. In BPF, ungulates live under constantly high risk of predation which are present virtually everywhere. Under these conditions, we argue that prey cannot easily escape predators in space as shown by many published studies from highly heterogeneous landscapes.
Stalking versus cursorial hunting
Mammalian predators show two basic hunting modes—stalking (e.g. most of felids) and cursorial (e.g. most of canids). Each of these modes is linked to habitat structure in different ways and this may affect a recognizable landscape of fear for prey species. Predators with different hunting modes have been shown to have contrasting effects on prey (Schmitz 2005, 2008). Several recent studies have also suggested that a similar dichotomy in predator effects operate in large carnivore–ungulate ecosystems, with cursorial predators creating the weakest habitat-mediated fear effects (Kauffman et al. 2010; Thaker et al. 2011). Typical cursorial predators, such as wolves, are actively pursuing their prey and they do not require cover to surprise their target as the stalking predators. However, habitat features are linked to hunting success of both cursorial predators (Bergman et al. 2006; Kauffman et al. 2007) and for stalking predators (Hopcraft et al. 2005, Podgórski et al. 2008), but they seem to operate at different scales. The patterns of predation by cursorial predators are shaped by large-scale landscape heterogeneity (Kauffman et al. 2007). Thus, the predation risk cues they are producing are not easily predictable as they are not specifically connected to precise locations. In contrast, hunting activity of stalking predators is closely linked to fine-scale habitat features (Laundré and Hernández 2003; Podgórski et al. 2008) as they need to approach their prey at close distance undetected using habitat features such as a stalking cover. This results in more spatially predictable habitat-linked risk factors at a fine scale. Interestingly, there has been much attention for the effects of cursorial carnivores on the spatial distribution of their prey (for an overview, see Beschta and Ripple 2009), whereas the predicted much stronger indirect effects of stalking predators have been largely neglected (but see Thaker et al. 2011; Laundré et al. 2010).
If predation efficiency is driven by landscape variables, the same traits should affect the perception and response of the ungulates hunted by them when they have the possibility to learn. This concept has been supported by several studies. Elk, the major prey of wolves in the Yellowstone ecosystem, avoid open foraging areas when wolves were in close vicinity and searched for a refuge from predation in woodlands (Creel et al. 2005). Hence, ungulate prey do show large-scale movements resulting from risk effects created by cursorial predators. However, the study by Winnie et al. (2006) showed that the presence of predators may change the threshold of prey sensitivity to environmental conditions. Elk became temporarily less responsive to habitat variables when wolves were present in comparison to times when they were absent. Other studies demonstrated that elk responded to wolf presence on a short time scale and changed behaviour or movement and grouping patterns (Creel et al. 2005; Creel et al. 2008; Liley and Creel 2008). In other words, prey may decrease its selectivity towards available low-risk habitats when there is an imminent threat of a being killed. This indicates that for a typical cursorial predator, prey is more likely to react to “risky times” resulting from actual presence of the predator, rather than “risky places” related to habitat features (Creel et al. 2008).
Even fewer studies exist on the importance of habitat characteristics in mediating the interactions between prey and stalking carnivores. It can be expected that when faced with predators which require cover for ambushing (e.g. Laundré and Hernández 2003; Hopcraft et al. 2005), prey species can detect predation risk based on indirect habitat cues with higher accuracy and predictability than with a cursorial predator. Therefore, the behavioural response of both predator and prey animals should be tightly linked to fine-scale habitat structure.
Schaller (1972), in his classic study, unequivocally showed that African lions (Panthera leo) did not evoke anxiety in prey animals until they remain in sight. In contrast, he noted that “prey is particularly cautious about entering thickets” (p. 235). Therefore, the lions’ presence alone is not enough to induce anti-predatory behaviour in the prey species. This concept was recently studied in detail by Valeix et al. (2009a, b), who clearly showed that many prey species of lions avoided using risky bush-lands and woodlands. Moreover, as lions concentrated their hunting activity around waterholes (Valeix et al. 2010), ungulates responded with behavioural adjustments to reduce predation risk in these risky sites (Valeix et al. 2009a). They reduced the use of waterholes at night when there was highest predation risk and increased their vigilance while drinking. These results show a high wariness in prey animals suggesting that the chance of a successful hunt by lions should decrease at the most attractive and well distinguishable sites. In fact, it was found that lions have higher hunting success in areas with good protective cover rather than in those characterized with a higher abundance of ungulates (Hopcraft et al. 2005; Loarie et al. 2013).
Although lions are typically stalking predators while hunting, they are often exceptionally conspicuous on other occasions, particularly during their social activities. Thus, the interplay of periods when lions disappear and come back into sight may give a good cue as to which prey can predict predation risk. We would then expect that yet stronger reliance on habitat structures as a cue of predation risk occurs in ecosystems with solitary stalking carnivores, which are hardly visible to prey. In line with this, Holmes and Laundré (2006) showed that pumas (Puma concolor) focused their foraging activity within forest edges, which resulted in the most successful hunting occurring there (Laundré and Hernández 2003). This transitory habitat allowed pumas to ambush ungulates as they moved between forest and open patches. As a result, ungulates perceived higher predation risk in the edge habitat and responded by higher foraging activity in open habitat and higher vigilance at the forest edges (Altendorf et al. 2001). Similarly, it was found that leopards (Panthera pardus) were selecting habitats with sufficient vegetation cover for stalking prey, though not too dense to impede their chase, whereas their prey was most abundant in habitats characterized either by high visibility or by cover dense enough to prevent successful pursuit by leopard (Balme et al. 2007).
To conclude, one can expect that stalking, solitary predators rather than cursorial predators are more likely to generate consistent cues for predation risk which are strongly linked to fine-scale habitat characteristics and therefore clearly identifiable by prey animals. This can induce a situation in which prey show a shift in fine-scale habitat selection and increase their use of low-risk habitats. In other words, if the prey chooses sites with the lowest predation risk, the predator would be forced to hunt in areas with the lowest abundance of prey (Laundré 2010). However, the question is if such a strategy can guarantee the persistence of the predator’s population. We agree with Laundré’s (2010) hypothesis that the final outcome of these opposing strategies of the predator and prey can be notably influenced by the share of habitat types with different predation risk. Landscapes dominated by protective cover for ungulates (safe habitats) should promote a high prey–predator ratio (and high prey densities), whereas in landscapes dominated with risky habitats the opposite should be expected. Therefore, especially in ecosystems with stalking predators present, and habitat conditions favouring hunting success by predators, the prey may have little chance to avoid predation.
Effects of the landscape structure
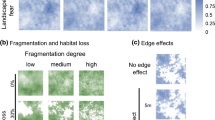
If the trade-off between the quality of a foraging patch and predator avoidance is one of the main mechanisms driving the habitat selection and spatial distribution of prey animals, one can expect that a similar strategy should be valid regardless of the type of ecosystem. However, the majority of empirical data originate from relatively heterogeneous and large North-American (e.g. Yellowstone, Zion, Banff National Parks) or African (Serengeti, Hwange NP) ecosystems. In such landscapes, the risky habitats and refuge areas for prey species are often clearly identifiable, even for researchers (Fig. 1). The low-risk patches for ungulates may consist of either places with good visibility and a long escape distance in case of stalking predators (e.g. Holmes and Laundré 2006) or with protective cover decreasing the probability of being detected by cursorial predators (Creel et al. 2005). In contrast, risky patches may be characterized by having structures which limit the possibility of the prey’s escape or the visual assessment of the predator’s presence and therefore facilitate ambush by a predator (Halofsky and Ripple 2008).
Two contrasting ecosystems with potentially different effects on creating the landscape of fear: left—Yellowstone National Park (photo courtesy of Dan Zachariah)—a heterogeneous landscape with clearly distinguishable foraging and refuge patches, USA; right—Białowieża Primeval Forest, Poland (photo courtesy of Jan Walencik)—a relatively homogeneous landscape constituting a relatively uniform body of the forest with very concentrated forging sites in small forest gaps. Both ecosystems are inhabited by large carnivores and ungulates
Heterogeneity of the environment has been proposed as the factor allowing the elk to reduce the risk of predation from wolves in the Yellowstone National Park (YNP; Kauffman et al. 2007). Surprisingly, elk are not necessarily avoiding even the riskiest locations in YNP (Fortin et al. 2005; Kauffman et al. 2007; Mao et al. 2005). Nevertheless, owing to the heterogeneity of habitats offering a range of food quality in combination with varying levels of predation risk within the park, elk are able to balance the predation risk and feeding requirements with various behavioural responses. These included either aggregation in open places, which facilitated the detection of predators and the lowering of predation risk by dilution effects (Mao et al. 2005), or switching habitat preferences at finer scale to safer habitats (Fortin et al. 2005; Thaker et al. 2011).
Although identifying qualitatively the levels of predation risk in strongly differentiated habitats does not seem to raise many difficulties, interpreting its effect on predator–prey relationships is not so obvious. Even more complex interactions may occur within more homogenous landscapes which are, as yet, virtually unexplored. In such environments, risk may be more evenly dispersed over the landscape (Fig. 1). Moreover, the indirect cues of predation risk based on habitat features might primarily act at fine spatial scale (Kuijper et al. 2013, 2014; Wikenros et al. 2015).

Studies on the habitat selection of both large carnivores and ungulates in a densely forested ecosystem were conducted in the BPF, Poland (Box 1). Despite its rich composition at the scale of forest stands (forest types, vertical and horizontal forest stand structure), the entire area can be regarded as very homogenous at the landscape scale as it is all covered with forest habitat (lacking large river valleys, open grass planes, etc.) in contrast to well-studied systems in North America and Africa. This entire area is mainly composed of lowland mixed forest, with only 0.8–3 % of it covered with open habitats (Michalczuk 2001; Kowalczyk 2010). These open areas include marsh lands and small gaps in forest stand resulting from natural gap formation (inside the protected part of BPF—the Białowieża National Park, up to 1 ha of size) or removing a few trees (≤0.5 ha) in the managed part of the forest. However, these gaps do not disrupt the general habitat homogeneity due to its small size as compared to the total available habitat (Fig. 1).
In this system, the stalking predator, Eurasian lynx (Lynx lynx), was found to hunt most successfully in the forest gaps; the lynx kills were located in these gaps 37 % more often than the random locations (Podgórski et al. 2008). Likewise, red deer killed by wolves were also found more often in open places than in closed forest (Theuerkauf and Rouys 2008). The choice of the forest gaps may be advantageous for the predator due to good view of the prey provided by the gaps while it approaches from the closed forest being concealed from the prey. At the same time, forest gaps in BPF are highly attractive foraging sites for ungulates which concentrate their foraging activity there (Kuijper et al. 2009). Gaps provide an abundance of forage due to the higher intensity of regeneration of woody browse and higher cover of herbaceous vegetation (Bobiec 2007). As a result, patches with higher ungulate visitation and expected higher hunting success are very predictable for the predators in this system. Moreover, good foraging but high risk areas for ungulate prey is likewise very predictable. Ungulates likely have to concentrate foraging in these rich forest gaps, simply because food availability is very low under the surrounding closed canopy with sparse regeneration and low or lacking herbaceous vegetation cover.
Although intuitively correct, and in accord with the optimal foraging theory (MacArthur and Pianka 1966), the equal preference for gaps by predators and prey requires more attention. Stalking predators like lynx select for forest gaps because of higher prey encounter rate and higher chance for a successful ambush of their prey. Ungulate prey may be forced to select for these sites to forage as there is little tree regeneration under a closed canopy (Bobiec 2007). Having no alternative low-risk high quality forage patches, ungulates are facing “death traps” in the forest gaps as the probability of being killed is much higher while foraging there compared to in the closed canopy forest. Although evidences exist that prey animals are often trading the risk of being killed with better food quality (e.g. Cooper 2000; Kittle et al. 2008; Wilson et al. 2012), our example turns attention to specific circumstances where foraging under high predation risk is the best choice for prey animals. Whereas in heterogeneous landscapes prey may find habitats with varying levels of forage quality and quantity and refuges even within the areas highly used by wolves (Fortin et al. 2005), such behavioural responses may not be possible in more homogenous environments, such as a closed forest ecosystem with limited and small food-abundant patches and no refuge areas. While forest gaps are most successful hunting places for both lynx and wolf, the surrounding matrix is not devoid of the risk of being killed by wolves. Despite of the preferential use of forest gaps by deer in BPF (Kuijper et al. 2009), they cannot support long-term occupation by ungulates due to their small size. This contrast to heterogeneous landscapes with both forest and large open spaces, where prey aggregates for considerable amount of time in open habitat (Courant and Fortin 2012; Harvey and Fortin 2013). In our study area, deer use the forest gaps only for short periods of time, with each patch visited less than 2 min, meaning that most of the time they are present in closed forest habitat (Kuijper et al. 2009). Thus, ungulates in homogeneous habitats may experience two sources of predation risk—while foraging in the gaps (“death traps” targeted by both lynx and wolf) as well as while staying out for extended periods of time in the surrounding closed-canopy forest (where they are vulnerable to cursorial wolves). In homogeneous habitats which lack areas free from lynx and wolves (see Box 1) and with concentrated spots of food, prey animals are exposed to chronic high predation risk with little chance to avoid it. As the model of risk allocation hypothesis assumes that maintaining high vigilance under chronically high predation risk is impossible (Lima and Bednekoff 1999), the question becomes what mechanisms allow the prey animals to reduce the predation risk under such conditions is open, particularly because the majority of empirical data on that come from heterogeneous landscapes.
Synthesis: living under chronic high predation risk in a homogenous environment
It is evident that animals have to trade-off between habitat forage quality and predation risk (Sih 1980; Hebblewhite and Merrill 2009). In various circumstances, this trade-off will result in different outcomes, from an extreme avoidance of risky situations at the cost of limited foraging opportunities to a seemingly “relaxed” intensive use of food-rich, but very risky habitats. While the occurrence of the latter strategy seems to be rare, we argue that it may characterize multi-species predator–prey communities inhabiting relatively homogenous landscapes with concentrated high quality feeding patches. We believe that the ungulate–predator system in the Białowieża Forest described above may represent a good example for this.
We suggest the negative relationship between the spatial distribution of prey and predators, which is emerging from many studies, may be limited to ecosystems with clearly heterogeneous habitat. This notion is in accordance with the conclusion of Birkhofer et al. (2010) that predators and prey may segregate spatially providing habitat heterogeneity is present. We propose that positive spatial relationships between predator and prey are most likely to evolve in relatively homogenous environments with concentrated foraging patches for prey. The system similar to the one reported here (BPF) may occur in the semi-arid savannah with waterholes serving as indispensable spots for ungulates to return to (Valeix et al. 2010; Davidson et al. 2013). The mechanism that may lead to such spatial aggregation (positive relationship) of predator and prey may involve the presence of highly concentrated resources for the prey species in combination with the lack of distinct habitats offering refuges. This relationship can be illustrated by a conceptual model (Fig. 2) predicting that along with the concentration of resources, the availability of refuges (being related with heterogeneity of habitats) should decrease, and the probability that prey is killed by the predator should increase. This concept may particularly be true in multi-predator systems, especially if there are both stalking and cursorial predators. The risk effect of one predator, which would eventually shift the prey into “safer” habitat, should increase the predation risk from another predator, as it was recently evidenced by Atwood et al. (2009) for a wolf–cougar–elk–mule deer (Odocoileus hemionus) system.
Conceptual model showing the hypothetical relationships leading to the occurrence of a death trap. With the increase of resource (food) concentration, the availability of refuges (being related to heterogeneity of habitats) decreases while the probability of being killed by the predator will increase. At highest resource concentration, prey species become highly predictable causing the death trap to occur. The model assumes the probability of finding refuge decreases at low rate within relatively wide range of resource concentrations with the highest rate occurring when resources become highly concentrated and ungulates have no alternative foraging sites. The shape of the kill probability curve is more steep than that of the refuge, indicating that the chance of successful hunt is very high only at the highest resource concentration when ungulates are highly predictable
The described case of BPF system may also clearly support such interactions due to the wolf and lynx partial overlap in dietary preferences, focusing on both red and roe deer (Jędrzejewska and Jędrzejewski 1998), as well as nearly full overlap in space use (Schmidt et al. 2009). This all contributes to a more evenly distributed predation risk both in time and space through intensive use of all available area by both predators. Under such conditions, the prey is compelled to forage in spots with the highest foraging gains irrespective of the associated lethal consequences (Fig. 3).
Conceptual spatial models of interactions between large cursorial (C) and stalking (S) predators (thick arrows) and prey (thin arrows) within heterogeneous and homogeneous landscapes (grey colour represents homogeneous habitat). In a heterogeneous habitat with only cursorial predators, the prey uses distinct refuge patches (R, with low accessibility to predators) from which they disperse to forage in surrounding open landscapes which constitute their foraging areas (F). It allows for a negative relationship between the prey and predators’ spatial distribution. Here, the predictability of both the predator and prey location is low. In homogeneous closed-forest habitats, positive spatial relationships between predator and prey should be expected to occur. Foraging is clearly focused on small food-rich forest gaps, which are highly attractive for ungulates and targeted by both types (C and S) of predators and there are no distinct refuges for prey outside the gaps, as whole available habitat is accessible to (particularly cursorial) predators. The gaps become the death traps as they are easily predicted by predators
Focusing on foraging benefits seems the best option for ungulates in these circumstances, though it leads to death traps because they become very predictable for predators. Following the opinion of Kittle et al. (2008), the potential fitness gain (i.e. higher forage availability) should be still high enough to offset the inevitable risk. However, we speculate a major indirect cost of predation for ungulates lies in feeding under constant awareness of threat rather than in losing foraging opportunities through avoidance of risk. The coexistence and familiarity of prey species with an incessant risk of predation should favor reinforcing their sensitivity to very fine-scale cues of risk, allowing them to efficiently detect and tackle the immediate threat of predators. The prey may thus respond to predation risk at a fine scale within habitats and not between habitats, as shown by recent studies in BPF (Kuijper et al. 2013). The predation risk perceived by ungulates may be yet more precisely fine-tuned based on olfactory cues as they showed unambiguous reaction to a single freshly deposited wolf scat (Kuijper et al. 2014; Wikenros et al. 2015). This ability may help them to perceive the actual vicinity or a recent visitation of a predator indicating near-imminent risk of death.
The question, however, remains what effect the constant predation risk exerts on the prey at the population level. For example, a still unsolved issue is the possibility of affecting the population reproduction rates through predation-induced stress (McArthur et al. 2014). Although its role is not yet well established in shaping ungulate populations (Creel et al. 2009), there is evidence that glucocorticoid stress hormones might play an important role in anti-predator response in prey animals (Thaker et al. 2010). The ungulate prey should utilize a wide array of responses including behavioural and hormonal reactions, of which the effect on their populations should be farther studied empirically in different types of landscapes including both heterogeneous and homogeneous environments.
References
Altendorf KB, Laundré JW, López Gonzalez CA, Brown JS (2001) Assessing effects of predation risk on foraging behavior of mule deer. J Mammal 82:430–439
Atwood TC, Gese EM, Kunkel KE (2009) Spatial partitioning of predation risk in a multiple predator–multiple prey system. J Wildl Manag 73:876–884
Balme G, Hunter L, Slotow R (2007) Feeding habitat selection by hunting leopards Panthera pardus in a woodland savanna: prey catchability versus abundance. Anim Behav 74:589–598
Barnier F, Valeix M, Duncan P, Chamaillé-Jammes S, Barre P, Loveridge AJ, Macdonald DW, Fritz H (2014) Diet quality in a wild grazer declines under the threat of an ambush predator. P R Soc B 281:20140446
Beier P, Choate DC, Barrett RH (1995) Movement patterns of mountain lions during different behaviours. J Mammal 76:1056–1070
Bergman EJ, Garrott RA, Creel S, Borkowski JJ, Jaffe R, Watson EGR (2006) Assessment of prey vulnerability through analysis of wolf movements and kill sites. Ecol Appl 16:273–284
Beschta RL, Ripple WJ (2009) Large predators and trophic cascades in terrestrial ecosystems of the western United States. Biol Conserv 142:2401–2414
Birkhofer K, Scheub S, Wiegand T (2010) Assessing spatiotemporal predator–prey patterns in heterogeneous habitats. Basic Appl Ecol 11:486–494
Bobiec A (2007) The influence of gaps on tree regeneration: a case study of the mixed lime-hornbeam (Tilio-Carpinetum Tracz. 1962) communities in the Białowieża primeval forest. Pol J Ecol 55:441–455
Bouskila A (1995) Interactions between predation risk and competition—a field-study of kangaroo rats and snakes. Ecology 76:165–178
Brown JS (1999) Vigilance, patch use and habitat selection: foraging under predation risk. Evol Ecol Res 1:49–71
Brown JS, Laundré JW, Gurung M (1999) The ecology of fear: optimal foraging, game theory, and trophic interactions. J Mammal 80:385–399
Burkepile DE, Burns CE, Tambling CJ, Amendola E, Buis GM, Govender N, Nelson V, Thompson DI, Zinn AD, Smith MD (2013) Habitat selection by large herbivores in a southern African savanna: the relative roles of bottom-up and top-down forces. Ecosphere 4: art139
Cooper WE (2000) Tradeoffs between predation risk and feeding in a lizard, the broad-headed skink (Eumeces laticeps). Behaviour 137:1175–1189
Courant S, Fortin D (2012) Time allocation of bison in meadow patches driven by potential energy gains and group size dynamics. Oikos 121:1163–1173
Courbin N, Fortin D, Dussault C, Fargeot V, Courtois R (2013) Multi-trophic resource selection function enlightens the behavioural game between wolves and their prey. J Anim Ecol 82:1061–1071
Creel S, Winnie J, Maxwell B, Hamlin K, Creel M (2005) Elk alter habitat selection as an antipredator response to wolves. Ecology 86:3387–3397
Creel S, Winnie JA Jr, Christianson D, Liley S (2008) Time and space in general models of antipredator response: tests with wolves and elk. Anim Behav 76:1139–1146
Creel S, Winnie JA, Christianson D (2009) Glucocorticoid stress hormones and the effect of predation risk on elk reproduction. Proc Natl Acad Sci U S A 106:12388–12393
Davidson Z, Valeix M, Van Kesteren F, Loveridge AJ, Hunt JE et al (2013) Seasonal diet and prey preference of the African lion in a waterhole-driven semi-arid savanna. PLoS ONE 8(2), e55182
Faliński JB (1986) Vegetation dynamics in temperate lowland primeval forest. Dr. W. Junk, Dordrecht
Fortin D, Beyer HL, Boyce MS, Smith DW, Duchesne T, Mao JS (2005) Wolves influence elk movements: behaviour shapes a trophic cascade in Yellowstone National Park. Ecology 86:1320–1331
Halofsky JS, Ripple WJ (2008) Fine-scale predation risk on elk after wolf reintroduction in Yellowstone National Park, USA. Oecologia 155:869–877
Hammond JI, Luttbeg B, Sih A (2007) Predator and prey space use: dragonflies and tadpoles in an interactive game. Ecology 88:1525–1535
Harvey L, Fortin D (2013) Spatial heterogeneity in the strength of plant-herbivore interactions under predation risk: the tale of bison foraging in wolf country. PLoS ONE 8:e73324
Hebblewhite M, Merrill EH (2009) Trade-offs between predation risk and forage differ between migrant strategies in a migratory ungulate. Ecology 90:3445–3454
Herfindal H, Linnell JDC, Odden J, Nilsen EB, Andersen R (2005) Prey density, environmental productivity and home-range size in the Eurasian lynx (Lynx lynx). J Zool Lond 265:63–71
Hernández L, Laundré JW (2005) Foraging in the ‘landscape of fear’ and its implications for habitat use and diet quality of elk Cervus elaphus and bison Bison bison. Wildl Biol 11:215–220
Hochman V, Kotler BP (2007) Patch use, apprehension, and vigilance behavior of Nubian Ibex under perceived risk of predation. Behav Ecol 18:368–374
Hopcraft JGC, Sinclair ARE, Packer C (2005) Planning for success: Serengeti lions seek prey accessibility rather than abundance. J Anim Ecol 74:559–566
Holmes BR, Laundré JW (2006) Use of open, edge and forest areas by pumas Puma concolor in winter: are pumas foraging optimally? Wildl Biol 12:201–209
Jędrzejewska B, Jędrzejewski W (1998) Predation in vertebrate communities. The Białowieża Primeval Forest as a case study. Springer, Berlin
Jędrzejewski W, Schmidt K, Theuerkauf J, Jędrzejewska B, Kowalczyk R (2007) Territory size of wolves Canis lupus: linking local (Białowieża Primeval Forest, Poland) and Holarctic-scale patterns. Ecography 30:66–76
Kauffman MJ, Smith VN, Stahler DW, MacNulty DR, Boyce MS (2007) Landscape heterogeneity shapes predation in a newly restored predator–prey system. Ecol Lett 10:690–700
Kauffman MJ, Brodie JF, Jules ES (2010) Are wolves saving Yellowstone’s aspen? A landscape-level test of a behaviorally mediated trophic cascade. Ecology 91:2742–2755
Kittle AM, Fryxell JM, Desy GE, Hamr J (2008) The scale-dependent impact of wolf predation risk on resource selection by three sympatric ungulates. Oecologia 157:163–175
Kowalczyk R (2010) European bison—the king of the forest or meadows and river valleys? In: Kowalczyk R, Ławreszuk D, Wójcik MJ (eds) European bison conservation in the Białowieża Primeval Forest. Threats and prospects of the population development. Mammal Research Institute PAS, Białowieża, pp 123–134 [in Polish]
Kriva V, Cressman R (2009) On evolutionary stability in predator–prey models with fast behavioural dynamics. Evol Ecol Res 11:227–251
Kuijper DPJ, Cromsigt JPGM, Churski M, Adam B, Jedrzejewska B, Jedrzejewski W (2009) Do ungulates preferentially feed in forest gaps in European temperate forest? For Ecol Manag 258:528–1535
Kuijper DPJ, de Kleine C, Churski M, van Hooft P, Bubnicki J (2013) Landscape of fear in Europe: wolves affect spatial patterns of ungulate browsing in Białowieża Primeval Forest, Poland. Ecography 36:1263–1275
Kuijper DPJ, Verwijmeren M, Churski M, Zbyryt A, Schmidt K, Jędrzejewska B, Smit C (2014) What cues do ungulates use to assess predation risk in dense temperate forests? PLoS ONE 9(1):e84607
Kunkel KE, Pletscher DH (2000) Habitat factors affecting vulnerability of moose to predation by wolves in southeastern British Columbia. Can J Zool 78:150–157
Laundré JW (2010) Behavioral response races, predator–prey shell games, ecology of fear, and patch use of pumas and their ungulate prey. Ecology 91:2995–3007
Laundré JW, Hernández L (2003) Winter hunting habitat of pumas Puma concolor in northwestern Utah and southern Idaho, USA. Wildl Biol 9:123–129
Laundré JW, Hernández L, Altendorf KB (2001) Wolves, elk, and bison: reestablishing the “landscape of fear” in Yellowstone National Park, USA. Can J Zool 79:1401–1409
Laundré JW, Hernández L, Ripple WF (2010) The landscape of fear: ecological implications of being afraid. Open Ecol J 3:1–7
Laundré JW, Hernández L, Medina PL, Campanella A, López-Portillo J, González-Romero A, Grajales-Tam KM, Burke AM, Gronemeyer P, Browning DM (2013) The landscape of fear: the missing link to understand top-down and bottom-up controls of prey abundance? Ecology 95:1141–1152
Liley S, Creel S (2008) What best explains vigilance in elk: characteristics of prey, predators, or the environment? Behav Ecol 19:245–254
Lima SL, Bednekoff PA (1999) Temporal variation in danger drives antipredator behavior: the predation risk allocation hypothesis. Am Nat 153:649–659
Loarie SR, Tambling CJ, Asner GP (2013) Lion hunting behaviour and vegetation structure in an African savanna. Anim Behav 85:899–906
Mao JS, Boyce MS, Smith DW, Singer FJ, Vales DJ, Vore JM, Merrill EH (2005) Habitat selection by elk before and after wolf reintroduction into Yellowstone National Park. J Wildl Manag 69:1691–1707
MacArthur RH, Pianka ER (1966) On optimal use of a patchy environment. Am Nat 100:603–609
McArthur C, Banks P, Boonstra R, Forbey J (2014) The dilemma of foraging herbivores: dealing with food and fear. Oecologia 176:677–689
Maynard Smith J, Price GR (1973) The logic of animal conflict. Nature 246:15–18
Mitchell WA, Lima SL (2002) Predator–prey shell games: large-scale movement and its implications for decision-making by prey. Oikos 99:249–259
Michalczuk C (2001) Forest habitats and tree stands of the Białowieża National Park. Phytocoenosis Suppl Cartogr Geobot 13:1–22
Orrock J, Danielson B, Brinkerhoff R (2004) Rodent foraging is affected by indirect, but not by direct, cues of predation risk. Behav Ecol 15:433–437
Podgórski T, Schmidt K, Kowalczyk R, Gulczyńska A (2008) Microhabitat selection by Eurasian lynx and its implications for species conservation. Acta Theriol 53:97–110
Rieucau G, Vickery W, Doucet G (2009) A patch use model to separate effects of foraging costs on giving-up densities: an experiment with white-tailed deer (Odocoileus virginianus). Behav Ecol Sociobiol 63:891–897
Ripple WJ, Beschta RL (2006) Linking wolves to willows via risk-sensitive foraging by ungulates in the northern Yellowstone ecosystem. For Ecol Manag 230:96–106
Rose GA, Leggett WC (1990) The importance of scale to predator-prey spatial correlations: an example of Atlantic fishes. Ecol 71:33–43
Schaller GB (1972) The Serengeti lion: a study of predator-prey relations. The University of Chicago Press, Chicago, USA
Schmidt K (2008) Behavioural and spatial adaptation of the Eurasian lynx to a decline in prey availability. Acta Theriol 53:1–16
Schmidt K, Jędrzejewski W, Okarma H (1997) Spatial organization and social relations in the Eurasian lynx population in Białowieża Primeval Forest, Poland. Acta Theriol 42:289–312
Schmidt K, Jędrzejewski W, Okarma H, Kowalczyk R (2009) Spatial interactions between grey wolves and Eurasian lynx in Białowieża Primeval Forest, Poland. Ecol Res 24:207–214
Schmitz OJ (2005) Behavior of predators and prey and links with population level processes. In: Barbosa P, Castellanos I (eds) Ecology of predator–prey interactions. Oxford University Press, Oxford, UK, pp 256–278
Schmitz OJ (2008) Effects of predator hunting mode on grassland ecosystem function. Science 319:952–954
Sih A (1980) Optimal behavior: can forages balance two conflicting demands? Science 210:1041–1043
Sih A (2005) Predator–prey space use as an emergent outcome of a behavioral response race. In: Barbosa P, Castellanos I (eds) Ecology of predator–prey interactions. Oxford University Press, Oxford, UK, pp 240–255
Thaker M, Vanak A, Lima S, Hews D (2010) Stress and aversive learning in a wild vertebrate: the role of corticosterone in mediating escape from a novel stressor. Am Nat 175:40–50
Thaker M, Vanak AT, Owen CR, Ogden MB, Niemann SM, Slotow R (2011) Minimizing predation risk in a landscape of multiple predators: effects on the spatial distribution of African ungulates. Ecology 92:398–407
Theuerkauf J, Rouys S (2008) Habitat selection by ungulates in relation to predation risk by wolves and humans in the Białowieża Forest, Poland. For Ecol Manag 256:1325–1332
Valeix M, Chamaillé-Jammes S, Loveridge AJ, Davidson Z, Hunt JE, Madzikanda H, Macdonald DW (2011) Understanding patch departure rules for large carnivores: lion movements support a patch-disturbance hypothesis. Am Nat 178:269–275
Valeix M, Fritz H, Loveridge A, Davidson Z, Hunt J, Murindagomo F, Macdonald D (2009a) Does the risk of encountering lions influence African herbivore behaviour at waterholes? Behav Ecol Sociobiol 63:1483–1494
Valeix M, Loveridge AJ, Chamaillé-Jammes S, Davidson Z, Murindagomo F, Fritz H, Macdonald DW (2009b) Behavioral adjustments of African herbivores to predation risk by lions: spatiotemporal variations influence habitat use. Ecology 90:23–30
Valeix M, Loveridge A, Davidson Z, Madzikanda H, Fritz H, Macdonald D (2010) How key habitat features influence large terrestrial carnivore movements: waterholes and African lions in a semi-arid savanna of north-western Zimbabwe. Landsc Ecol 25:337–351
Venter J, Nabe-Nielsen J, Prins HT, Slotow R (2014) Forage patch use by grazing herbivores in a South African grazing ecosystem. Acta Theriol 59:457–466
Warfe DM, Barmuta LA (2004) Habitat structural complexity mediates the foraging success of multiple predator species. Oecologia 141:171–178
Verdolin J (2006) Meta-analysis of foraging and predation risk trade-offs in terrestrial systems. Behav Ecol Sociobiol 60:457–464
Wikenros C, Kuijper DPJ, Behnke R, Schmidt K (2015) Behavioural responses of ungulates to indirect cues of presence of an ambush predator. Behaviour (in print)
Wilson TL, Rayburn AP, Edwards TC (2012) Spatial ecology of refuge selection by an herbivore under risk of predation. Ecosphere 3:6–6
Winnie J Jr, Christianson D, Creel S, Maxwell B (2006) Elk decision-making rules are simplified in the presence of wolves. Behav Ecol Sociobiol 61:277–289
Acknowledgments
We are grateful to Ms C. O’Brien-Moran for correcting the English of the first draft of this manuscript. Our special thanks are to Justin Boyles for his help in revising the English grammar of the final version. This paper was financed by the statutory budget of the MRI PAS and the National Centre of Science, Poland (grant no: 2011/01/B/NZ8/04337 and DPJK grant nr 2012/05/B/NZ8/01010).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Justin G. Boyles
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schmidt, K., Kuijper, D.P.J. A “death trap” in the landscape of fear. Mamm Res 60, 275–284 (2015). https://doi.org/10.1007/s13364-015-0229-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13364-015-0229-x