Abstract
Objective
To study the role of Zinc in the treatment of neonatal sepsis.
Design
Double blind, randomized, placebo controlled trial.
Setting
Tertiary Care Hospital.
Participants
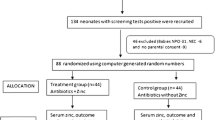
614 neonates with probable neonatal sepsis.
Intervention
The drug group (n=307) received 1mg/kg/day of elemental zinc, and placebo group (n=307) received the placebo, in addition to antibiotic therapy and supportive care, till the final outcome (discharge/death).
Outcome Measures
Decrease in mortality rates (primary outcome), duration of hospital stay and need of higher lines of antibiotic therapy (secondary outcomes) were tested.
Results
Baseline characteristics of the two groups were similar. No statistically significant differences between drug and placebo group were found in mortality rate (9.77% vs 7.81 %; P=0.393), mean duration of hospital stay (142.85±69.41 hrs, vs. 147.99±73.13 hrs; P=0.841), and requirement of higher lines of antibiotic therapy (13.35% vs 12.05%, P=0.628) after supplementation.
Conclusions
This study does not report decrease in mortality rates, duration of hospital stay and requirement of higher lines of antibiotic therapy following zinc supplementation in neonatal sepsis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Singh M, Paul VK, Bhakoo ON. Neonatal Nomenclature and Data Collection. New Delhi: National Neonatology Forum; 1989.p. 63–74.
Vergnano S, Sharland M, Kazembe P, Mwansambo C, Health PT. Neonatal sepsis: an international perspective. Arch Dis Child Fetal Neonatal. 2005;90:F220–F224.
Nepal Demographic and Health Survey 2011 — Preliminary report. MOHP, Govt. of Nepal. Available from: URL: http://www.mohp.gov.np/english/publication/NDHS%20%202011%20Preliminary%20Report.pdf. Accessed July 15, 2012.
State of World Children. UNICEF report. Available from: URL: http://www.unicef.org. Accessed July 15, 2012.
Stoll BJ, Kleigman RM. The fetus and the neonatal infant. In: Behrman RE, Kliegman RM, Jenson HB, Nelson Textbook of Pediatrics. 17ed, Philadelphia: WB Saunders Co; 2004;552;623–639.
Shankar AH, Prasad AS. Zinc and immune function: the biological basis of altered resistance to infection. Am J Clin Nut. 1998;68,447S–463S.
Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med. 2008; 14:353–357.
IZiNCG. Systematic reviews of zinc intervention strategies. International Zinc Nutrition Consultative Group Technical Document #2. Brown KH, Hess SY, editors. Food Nutr Bull. 2009;30:S1–S184.
Sazawal S, Black RE, Menon VP, Dinghra P, Caulfield LE, Dhingra U, et al. Zinc supplementation in infants born small for gestational age reduces mortality: a prospective, randomized, controlled trial. Pediatrics. 2001;108:1280–1286.
Castillo-Duran C, Rodriguez A, Venegas G, Alvarez P, Icaza G. Zinc supplementation and growth of infants born small for gestational age. J Pediatr. 1995;127:206–211.
Islam MN, Chowdhury M, Siddika M, Qurishi SB, Bhuiyan MK, Hoque MM, et al. Effect of oral zinc supplementation on the growth of preterm infants. Indian Pediatr. 2010;47:845–849.
Friel JK, Andrews WL, Matthew JD, Long DR, Cornel AM, Cox M, et al. Zinc supplementation in very-low-birthweight infants. J Pediatr Gastroenterol Nutr. 1993;17:97–104.
International Classification of Diseases (ICD), 10th revision, 1989. Geneva: World Health Organization; 1989.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mehta, K., Bhatta, N.K., Majhi, S. et al. Oral zinc supplementation for reducing mortality in probable neonatal sepsis: A double blind randomized placebo controlled trial . Indian Pediatr 50, 390–393 (2013). https://doi.org/10.1007/s13312-013-0120-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13312-013-0120-2