Abstract
Objective
To find the effect of zinc supplementation on the outcome of neonatal sepsis at one month of age.
Methods
This randomized controlled trial was conducted in a tertiary care neonatal unit, enrolling neonates with clinical features of sepsis and positive blood culture or positive sepsis screening tests. The treatment group received 3 mg/kg/twice a day of zinc sulfate monohydrate orally for 10 d along with standard antibiotic therapy. The control group received standard antibiotic treatment without zinc. Samples were collected from both the groups before and after the treatment. Babies were monitored till discharge and followed up as out-patients till one month of age.
Results
Demographic characteristics were similar between the cases and controls. After 10 d of treatment, the mean serum zinc level between the two groups was 737.09 ± 219.97 vs. 801.26 ± 405.56, (p = 0.20). Outcome measures like days of hospital stay (15 vs. 15; p = 0.69) and mortality rate (4.5 % vs. 13.6 %; p = 0.27) were not found to be significantly different between the groups. At one month of age, more number of control neonates had abnormal neurological findings as compared to the zinc supplemented group [(P = 0.02); RR (95%CI) = 0.28 (0.11–0.73)].
Conclusions
Zinc supplementation in neonates with sepsis improves the neurological status at one month of age although the mortality reduction was not statistically significant.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Severe infections remain the major cause of neonatal mortality and morbidity worldwide. It is reported that the incidence of neonatal sepsis varies from 7.1 to 38 per 1000 live births in Asia [1]. Neonates are more prone to infections because of immature immune function. Despite use of numerous antibiotics, newborns remain hospitalized for longer duration due to microbial infections [2]. Studies have shown the beneficial role of zinc in low birth weight infants and children with severe bacterial infections [3, 4]. Zinc also reduces bacterial infections, common cold and diarrhea [5]. It has been reported that preterm neonates require more zinc than term neonates as 60 % of fetal zinc is acquired during the third trimester of pregnancy. Preterm neonates have poor intestinal absorption due to their immature digestive system [6]. Studies have reported correlation between zinc deficiency and poor weight gain among low birth weight and preterm babies [7]. A randomized double- blind, placebo controlled trial conducted by Bhatnagar et al. showed improved outcome with supplementation of 10 mg zinc given orally in infants aged 7–120 d with serious bacterial infections [8]. In northeast Brazil, low birth weight babies who were administered 5 mg zinc orally for 8 wk showed better weight gain compared to those who received 1 mg Zn or placebo [9]. Recently, it was found that zinc supplementation leads to significant increase in length and weight of preterm infants at three and six months of age, developmental scores assessed using the Age and Stage Questionnaire (ASQ) compared to non-zinc supplemented group [10]. The authors hypothesized that zinc supplementation will reduce mortality among neonates with sepsis and improve outcome. There is insufficient data available to prove the effectiveness of zinc supplementation in reducing the mortality and improving the outcome in neonates with sepsis. Therefore the present study was conducted to find out the effect of zinc supplementation on mortality and outcome till one month of age among babies with sepsis.
Material and Methods
The study was conducted in NICU of a tertiary care hospital, from April 2013 through September 2014. The study was approved by the Institute Ethics Committee and was registered in Clinical Trials Registry of India (CTRI/2014/08/004,866). Inborn babies with clinical signs of sepsis fulfilling the inclusion criteria viz. gestational age ≥ 32 wk and < 28 d, on significant enteral feeds (>50 % feeds), with at least two screening tests positive among microESR, CRP and band cell count or positive blood culture were recruited for the study after getting written informed consent from their parents. The exclusion criteria were surgical conditions, major congenital malformations, Apgar score < 6 at 5 min, necrotizing enterocolitis and neonates already on antibiotic treatment for sepsis. Sample size was calculated using Open Epi software. Among zinc group, assuming reduction in the mortality rate of 25 %, the minimum sample size required for the study was estimated to be n = 43 in each group with alpha error of 5 % and 80 % power.
Babies with screening tests positive were given 7 d of antibiotics and those with culture tests positive were given 10 d antibiotics. Zinc at the dose of 3 mg/kg twice a day was administered for 10 d. Babies were given zinc only after the initial stabilization with oral feeding and most of the babies in the study group were <10 d of life. During the study period, care was taken to observe for any side effects. Blood samples (1.5 ml) were collected from the babies in the treatment and control groups before recruitment and after 10 d. The samples were centrifuged for 15–20 min at 10,000 rpm, sera separated and stored in polypropylene tubes at −20 °C for subsequent estimation. Serum zinc was estimated at Indian Council of Medical Research (ICMR), New Delhi by Inductively Coupled Plasma-Mass Spectrometry (ICP-MS). The babies were examined daily till discharge and followed up using Hammersmith neurological examination at one month of age by a trained therapist who was blinded from the trial. The examination was done when the neonate was in an awake and quiet state. Neurological findings in the form of posture, movements, altered tone and reflexes and abnormalities of visual, auditory orientation and behavior were assessed; neonate having any 2 of these abnormal was considered to be neurologically abnormal [11].
The data were analyzed using IBM SPSS (Version 20.0). The categorical variables such as gender, birth weight etc., were expressed as frequency and percentages. Two sample‘t’ test was used within the groups and values were expressed as Mean ± SD. Fisher’s exact test and Chi-square test were used to find out the statistical significance between the groups. P < 0.05 was considered statistically significant.
Results
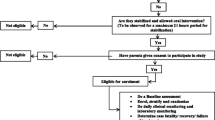
A total of 215 babies with clinical features suggestive of sepsis were eligible for the trial. Among them, 134 neonates had atleast two screening tests or blood culture positive. Forty six babies were excluded as they did not meet the inclusion criteria like being on nil per oral (n = 31), necrotizing enterocolitis (n = 6) and absence of parental consent (n = 9). Eighty eight neonates were randomized using sequential numbers kept in opaque sealed envelops (SNOSE) into two groups– treatment (n = 44) and control group (n = 44) (Fig. 1). Baseline characteristics like gender, birth weight, maturity, type of delivery, Apgar score, number of early onset/late onset sepsis, positive blood cultures were similar between cases and controls (Table 1).
The serum zinc level at baseline was (737.09 ± 219.97) and after 10 d of zinc supplementation it was (801.26 ± 405.56). The serum zinc concentration after 10 d was observed to show an increasing trend but the difference was not statistically significant compared to the baseline (p = 0.12). The serum zinc concentration in the control group before and after standard antibiotic treatment was 699 ± 248.3 vs . 692.94 ± 380.9 (p = 0.62). The mortality rate (13.6 % vs. 4.5 %), though important clinically, was not statistically significant. The hospital stay in days [15 (10–25) vs. 15 (11–28), p = 0.69] was not statistically significant between the two groups (Table 2).
At one month follow up, neonates in the control group (31.82 % vs. 9.1 %) were found to have any 2 of the abnormal neurological findings in the form of posture, movements, altered tone and reflexes and abnormalities of visual, auditory orientation and behavior. There was 70 % less chance of having abnormalities in the zinc group compared to controls [(p < 0.02), RR (95%CI) = 0.28 (0.11–0.73)] (Table 3).
Discussion
It is evident that the immune system is immature in newborns leading to several infections. Zinc plays a major role by its action on cell maturation, synthesis of DNA, RNA and protein molecules [12]. It is also involved in neurotransmission, growth of brain cells and found to improve the cognitive neurodevelopmental outcome [13]. Preterm neonates are more prone to be zinc deficient due to various reasons like endogenous losses, poor absorption, incapable of retaining the nutrients and immature development of the body organs.
In the index study, serum zinc concentration after 10 d in the treatment group increased compared to non-zinc supplemented group, although the difference was not statistically significant. Babies in the control group were found to have more neurological abnormalities compared to the zinc supplemented group (P < 0.05).The mortality rate and duration of NICU stay did not show significant difference. A double-blind, randomized placebo controlled trial conducted in a tertiary hospital in Nepal also did not reveal any significant difference between the zinc group and control group with respect to duration of hospital stay, requirement of higher lines of antibiotics and mortality [14].
Gerg et al. noted that zinc supplementation in 3rd trimester of pregnancy resulted in beneficial effects on growth and development of the fetus compared to the control group [15]. Sharma et al. reported that zinc deficiency affects mainly the pregnancy outcomes leading to low birth weight, growth retardation, abnormalities in DNA, RNA and protein synthesis [16]. Sazawal et al. randomized preterm babies (n = 100) into two groups– zinc and multivitamin in one group and only multivitamin in the second group. Neonates who received zinc in addition to multivitamins had shown significant improvement in linear growth and weight at 6 wk of life [17]. Parakh et al. noted that infants fed exclusively with breast milk and zinc supplementation showed significantly higher serum zinc ratio compared to non zinc supplemented and exclusively breast fed infants (1.55 ± 0.65 vs. 0.85 ± 0.406; p < 0.01). There was no statistically significant difference observed with regard to growth, neurodevelopmental outcome and morbidity profiles [18]. Ragab et al. noted that preterm neonates supplemented with 2 mg/kg/d zinc with breast milk and multivitamins had significant increase in weight and length at 3 and 6 mo of age compared to non-zinc supplemented group fed on breast milk and multivitamins only (p < 0.001) [10].
Infants with bacterial infection on antibiotics, aged from 7 to 120 d were randomized into zinc and placebo groups by Bhatnagar et al. The study revealed a lesser incidence of treatment failure in the zinc group (10 %) compared to the placebo group (17 %) with a relative risk reduction of 40 %, (P = 0.013) [8]. Prasad et al. have observed that zinc deficiency not only affects growth and neurodevelopmental outcome, but also suppresses immune function leading to morbidity that causes infections [19]. The present study showed that zinc supplementation at a dose of 3 mg/kg, given twice a day for a duration of 10 d along with standard antibiotic therapy significantly improves the neurological status of neonates at one month of age. The cases had fewer neurological abnormalities like altered tone and reflexes and abnormalities of visual, auditory orientation and behavior compared to the controls. Although one month of age is too early to detect any definite neurological abnormality, the findings of this study will have an impact on future studies. Babies less than 32 wk were not included in the study, since most of the smaller babies were not having significant enteral feedings.
Gulani et al. stated that three trials conducted in developing countries did not provide significant results, with regard to weight and height of babies following zinc supplementation. Though the levels of serum zinc were found to be higher in the intervention group, no significant decrease in mortality rate was observed and therefore zinc supplementation was not adviced for breastfed LBW newborns [20]. Zinc supplementation (5 mg) was administered in small for gestational age (SGA) infants aged 30 d to 9 mo of age to assess the cognitive and motor development. Developmental and behavior changes were assessed using Bayley scale of infant development II. Babies with more weight showed better improvement in all scores compared to low birth weight infants [21]. In the present study, zinc supplementation in neonatal sepsis improved the neurological status at one month of age [p = 0.02, RR (95%CI) = 0.28 (0.11–0.73)].
Based on the various observations and discussions made, it is evident that majority of the studies favored zinc supplementation for improving the survival rate and neurological outcome. There were no side effects observed during zinc supplementation. The present study has some limitations. Sample size was under powered to assess mortality in the zinc supplemented group. Moreover, zinc supplementation of 3 mg/kg twice a day could only marginally increase the blood levels of serum zinc and the levels were within the normal limits even after supplementation. Higher doses of zinc administration may have more beneficial effect.
Conclusions
It is concluded that zinc supplementation reduces the rate of mortality which is clinically relevant but not statistically significant. However it has a beneficial effect on the neurological status at one month of age in neonates treated for sepsis.
References
Singh M, Paul VK, Bhakoo ON. Neonatal nomenclature and data collection. New Delhi: National Neonatology Forum;1989. p. 67–8.
Yakoob MY, Theodoratou E, Jabeen A, et al. Preventive zinc supplementation in developing countries: impact on mortality and morbidity due to diarrhea, pneumonia and malaria. BMC Public Health. 2011;11:S3–23.
Terrin G, Canani RB, Passariello A, et al. Zinc supplementation reduces morbidity and mortality in very-low-birth-weight preterm neonates: a hospital-based randomized, placebo-controlled trial in an industrialized country. Am J Clin Nutr. 2013;98:1468–74.
Friel JK, Andrews WL, Matthew JD, et al. Zinc supplementation in very-low-birth-weight infants. J Pediatr Gastroenterol Nutr. 1993;17:97–104.
Deshpande JD, Joshi MM, Giri PA. Zinc: the trace element of major importance in human nutrition and health. Int J Med Sci Public Health. 2013;2:1–6.
Ma AG, Chen XC, Xu RX, Zheng MC, Wang Y, Li JS. Comparison of serum levels of iron, zinc and copper in anaemic and non-anaemic pregnant women in China. Asia Pacific J Clin Nutr. 2004;13:348–52.
Aydemir F, Cavdar AO, Soylemez F, Cengiz B. Plasma zinc level during pregnancy and its relationship to maternal and neonatal characteristics: a longitudinal study. Biol Trace Elem Res. 2003;91:193–202.
Bhatnagar S, Wadhwa N, Aneja S, et al. Zinc as adjunct treatment in infants aged between 7 and 120 days with probable serious bacterial infection: a randomised, double-blind, placebo-controlled trial. Lancet. 2012;379:2072–8.
Lira PI, Ashworth A, Morris SS. Effect of zinc supplementation on the morbidity, immune function, and growth of low-birth-weight, full-term infants in northeast Brazil. Am J Clin Nutr. 1998;68:418S–24S.
Ragab SM, Hegran HH, Kassem SA. The effect of zinc supplementation on growth and development in preterm neonates. Menoufia Med J. 2014;27:524–8.
Chaudhari S, Deo B. Neurodevelopmental assessment in the first year with emphasis on evolution of tone. Indian Pediatr. 2006;43:527–34.
Brown K, Wuehler S, Peerson J. The importance of zinc in human nutrition and estimation of the global prevalence of zinc deficiency. Food Nutr Bull. 2001;22:113–25.
Anjos T, Altmae S, Emmett P, et al. Nutrition and neurodevelopment in children: focus on NUTRIMENTHE project. Eur J Nutr. 2013;52:1825–42.
Mehta K, Bhatta NK, Majhi S, Shrivastava MK, Singh RR. Oral zinc supplementation for reducing mortality in probable neonatal sepsis: a double blind randomized placebo controlled trial. Indian Pediatr. 2013;50:390–3.
Garg HK, Singhal KC, Arshad Z. A study of the effect of oral zinc supplementation during pregnancy on pregnancy outcome. Indian J Physiol Pharmacol. 1993;37:276–84.
Sharma M, Mishra S. Effect of zinc deficiency on growth and morbidity in infants. IOSR J Nurs Health Sci. 2013;1:22–5.
Sazawal S, Black RE. Effect of oral zinc supplementation on the growth of preterm infants. Indian Pediatr. 2010;47:841–2.
Parakh M, Gupta BD, Bhansali S, et al. Role of zinc supplementation in growth and neuro-development of premature and small for gestational age (SGA) babies. Natl J Community Med. 2012;3:736–9.
Prasad AS. Zinc: an overview. Nutrition. 1995;11:93–9.
Gulani A, Bhatnagar S, Sachdev HPS. Neonatal zinc supplementation for prevention of mortality and morbidity in breastfed low birth weight infants: systematic review of randomized controlled trials. Indian Pediatr. 2011;48:111–7.
Black MM, Sazawal S, Black RE, Khosla S, Kumar J, Menon V. Cognitive and motor development among small-for-gestational-age infants: impact of zinc supplementation, birth weight, and caregiving practices. Pediatrics. 2004;113:1297–305.
Acknowledgments
The assistance in statistical analysis by Dr. Palanivel and Dr. Divya, Department of P&SM, is acknowledged. The authors are thankful to Dr.Toteja from Indian Council of Medical Research (ICMR), New Delhi for estimating the serum zinc levels.
Contributions
BBD and BVB designed the study. BN has done laboratory works and drafted the manuscript. NM assessed the neurological outcome of the babies. BVB, SMG and NM reviewed the manuscript. BVB will act as guarantor for the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None.
Source of Funding
Extramural Research Grant, Indian Council of Medical Research (ICMR), New Delhi.
Rights and permissions
About this article
Cite this article
Newton, B., Bhat, B.V., Dhas, B.B. et al. Effect of Zinc Supplementation on Early Outcome of Neonatal Sepsis - A Randomized Controlled Trial. Indian J Pediatr 83, 289–293 (2016). https://doi.org/10.1007/s12098-015-1939-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-015-1939-4