Abstract
The formation of blood capillaries to sustain development and growth of new tissues is referred to as angiogenesis. Angiogenesis is pivotal in both carcinogenesis and metastasis since capillaries are the sole source of supplying nutrients and oxygen to the proliferating tumor cells; therefore, this dependency of tumor growth on angiogenesis challenges researchers to halt tumor growth by targeting angiogenesis with the help of either synthetic or natural inhibitors. Many synthetic inhibitors of angiogenesis have not only come into force but also resulted in some severe adverse effects. Natural compounds may effectively fit into this condition and possibly decrease the time of treatment. In the recent past, literature is replete with evidences advocating the usefulness of natural compounds that target multiple biochemical pathways. The additional advantage of natural compounds is that their active principles interact with one another and work synergistically to give more meaningful and reliable effects than individual principle. Hence, if we are somehow able to combine more than two natural compounds, then it may be possible to enhance their potential by many folds, which shall prove to be very effective in combating tumor angiogenesis. This review shall discuss the concept of angiogenesis, molecular pathways, and angiogenic inhibitors and their specific targets and potential of natural compounds to greatly enhance the current knowledge of angiogenesis-inhibiting factors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Angiogenesis is a composite phenomenon that refers to the building of new blood vessels that supply oxygen and nutrients to the developing tissues. Angiogenesis is an essential biological process in wound healing, in sustaining pregnancy, and in menstruation [1, 2]. Angiogenesis is responsible for the onset of serious medical conditions like atherosclerosis, arthritis, and psoriasis [3]. The biological processes such as cell proliferation, differentiation, and cell-to-cell communication are well coordinated during angiogenesis. Angiogenesis is a hallmark of growth of tumors as it plays a crucial role of supplying the oxygen and nutrient required for the development and progression of tumor [4]. Cancer cells without oxygen and nutrient supply are not able to grow beyond a size of 2 mm, but they grow beyond 2 mm in an environment where angiogenesis is possible. However, angiogenesis is possible only in situations where there are sustainable regulations of signaling molecules acting as both activators and inhibitors. There is a need of downregulation of inhibitor molecules and upregulation of signaling activators in order to sustain angiogenesis. Hypoxia and stress are the two major driving forces responsible for the remodeling of tissues through the tightly regulated process of angiogenesis [4, 5]. The high proliferative rate of vascular cells is a key component of angiogenesis which otherwise divides in approximately once in 1000 days. In tumors, diabetic retinopathy, inflammatory, and other infections, angiogenesis can be activated abnormally [5–7]. Since angiogenesis is pivotal in tumor expansion, it can be extrapolated as a promising therapeutic target in cancer treatment. Literature is replete with studies advocating antiangiogenic potential of various synthetic compounds [8–12]. Angiogenesis involves complex signaling pathways, and the inhibition of one pathway by synthetic drugs (specific for specific pathway) may be compensated by another pathway; so, various synthetic drugs fail to provide effective results. On the other hand, natural products that contain more than one organic principle may possibly have different targets in multiple angiogenic pathways and therefore could prove a better and effective candidate for reducing growth and metastasis of cancer [13, 14]. The inhibitory effects of natural compounds by interacting with multiple biochemical pathways may affect angiogenesis effectively. Consideration of the synergistic role of natural compounds might be useful in deciding which combination of different natural compounds can be used to design a better therapeutic strategy to combat angiogenesis [15, 16]. This review would be helpful in providing insight into the mechanisms of angiogenesis in tumor and both the role of common antiangiogenic drugs and the antiangiogenic potential of some natural compounds.
Angiogenesis: a hallmark of cancer
Tumors are formed from a normal population of cells that become immortal after losing control over their growth. The cell population without proper regulation and high proliferation rate is also prone to have high mutation rate, leading to the development of other phenotypes [17, 18]. This tendency of gaining different phenotypes further helps to attain more favorable characteristics for growth and proliferation of cells. Tumors exploit this tendency of gaining new phenotypes to initiate angiogenesis in order to sustain the demand of nutrients and oxygen supply. This suggests that angiogenesis has an important role to play for the growth and metastasis of tumor [19]. Without angiogenesis, tumors can grow up to a certain level (1 to 2 mm in diameter) but beyond that, they must be supplied with oxygen and other nutrients by means of angiogenesis [20]. Hypoxia, endogenous angiogenesis promoters, and inhibitors play a crucial role in conversion of dormant lesion to vascularized tumor [21, 22]. There are four main steps involved in tumor angiogenesis, which include secretion of angiogenic factors, dissolution of basement membrane around a mature capillary, and proliferation and migration of endothelial cells toward angiogenic stimulus thereby leading to the formation of new capillaries (Fig. 1). The enhancement of signaling activators pushes the endothelial cells to proliferate and migrate [21]. On the other hand, augmentation of negative regulators keeps the endothelial cells in quiescent state [23]. Therefore, homeostasis between promoting and inhibiting endogenous signaling molecules controls the angiogenesis. In many medical conditions, the process of angiogenesis is tightly regulated and is allowed to fulfill the requirement of tissue repair. But, in certain angiogenesis-dependent diseases, disruption in regulatory mechanism leads to the promotion of diseases [5, 24]. The positive regulators of angiogenesis include the basic fibroblast growth factor (FGF) and vascular endothelial growth factor (VEGF) as primary angiogenic factors as they play a major role in angiogenesis. The other factors such as platelet-activating factor (PAF); platelet-derived growth factor (PDGF); epidermal growth factor (EGF), interleukin-1, interleukin-6, and interleukin-8; and transforming growth factor-alpha and beta (TGF α and β) do play a complementary role [18, 25]. The various molecular mechanisms for angiogenesis are described in Fig. 2. Diseases with insufficient angiogenesis like cutaneous ulcer and ischemic and myocardial brain disease can be treated with therapeutic angiogenesis [19, 27]. On the other hand, diseases with excessive angiogenesis like tumor can be treated with angiogenic therapy where angiogenic inhibitors are used to suppress the hyperactivity of angiogenic factors [5, 28].
Stages of angiogenesis. Step 1, secretion of angiogenic factor. Step 2; disruption of basement membrane. Step 3, proliferation and migration of endothelial cells toward angiogenic stimuli. Step 4 (early), sprout formation. Step 4 (late), union of sprout from opposite side and formation of new capillary
Schematic representation of molecular pathways responsible for cell proliferation, migration, and vascular permeability leading to initiation of angiogenesis (source modified and edited from book) [26]
Angiogenic factors (Table 1)
Biochemical species for the onset and progression of angiogenesis are called angiogenic factors. Majority of these are growth factors like vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) [29, 30]. The other angiogenic factors that play a complementary role like stimulation of angiogenesis are kinin, PDGF [31–33], and tumor growth factors α and β (TGF-α and TGF-β) [31]. VEGF and bFGF are crucial for cancer angiogenesis, inflammation, and wound healing [41]. The expression of VEGF is induced by hypoxia and other factors like EGF, TGF-α and TGF-β, FGF, and PDGF [42]. PDGF is the key biomolecule for the maturation of new vessels. FGF is essential for cell proliferation, migration, and differentiation [43]. TGF-β has a dual role to play, as low level enhances the angiogenesis through angiogenic factors whereas high levels of TGF-β inhibit the growth and proliferation of cells [43]. Macrophages and mast cells are associated with the secretion of tumor necrosis factor (TNF) [36], which further activates macrophages to release angiogenic factors. IL-6 is a well-known proangiogenic cytokine which plays an important role in angiogenesis. The exact mechanism of IL-6 is not fully understood as some studies advocate VEGF induction through IL-6 [44, 45] whereas Shao and Seng have suggested that prostaglandin E2 (PGE2) is stimulated by IL-6 through cyclooxygenase-2 (COX-2). This suggests that IL-6/PGE2/VEGF may play an important role in initiating tumor angiogenesis [46]. Further, it has also been documented that cytokine and various growth factors behave as second messenger molecules like reactive oxygen species (ROS) and hydrogen peroxide (H2O2) and cause their induction that leads to stimulation of VEGF. Another factor, angiopoietin too, has an interesting role in tumor angiogenesis as its members angiopoietin-1 (AP-1) and angiopoietin-2 (AP-2) [37] are antagonists to each other. Angiopoietin-1 is responsible for declining endothelial cell permeability and enhances vascular stability. On the other hand, angiopoietin-2 is associated with augmentation of sprouting of capillaries [47]. Enzyme COX-2 is another crucial mediator of angiogenesis and carcinogenesis as high expression of COX-2 is observed in a variety of cancers [38, 48–51]. Similarly, 12-lipoxygenase (12-LOX) is overexpressed in various carcinomas and significantly enhances angiogenesis [52–54]. Mechanistically, 12-LOX leads to secretion of eicosanoid generation which promotes carcinogenesis and metastasis [39].
NF-κB, one of the key regulators of inflammation, has a critical role in tumor angiogenesis as it is affected in different diseases including cancer [55–58]. NF-κB activation occurs through the upregulation of antiapoptotic genes to increase the chances of cell survival to withstand stress during inflammatory response [59]. Moreover, NF-κB also contributes to metastasis by regulating epithelial mesenchymal transition [60]. It inhibits the migration of endothelial cells by upregulation of metalloproteinase-1 (TIMP-1) which degrades the different components of extracellular matrix (ECM) [61]. Further increase in the VEGF expression through hypoxia-inducible factor (HIF) leads to promotion of angiogenesis. It has also been reported that NF-κB activation through TNF-α enhances the expression of PAI-1 and inhibits tPA expression [62], thereby suggesting that NF-κB activation could impair angiogenesis through decrease in ECM degradation [3]. The pathways and targets for the various angiogenic factors are described in Fig. 2.
Antiangiogenic drugs: targets and complications
Preclinical and clinical trials have demonstrated antiangiogenic action of chemotherapeutic agents which retard cancerous growth and metastasis [63]. The VEGF/VEGFR pathway is regarded as a key step to regulate angiogenesis in tumor either by inhibiting the expression of VEGF and its receptors or by downregulating signaling pathways [64]. Angiogenesis is stimulated by expressing the response of the VEGF family and signal transduction from VEGF to VEGFR2 which results in the activation of the tyrosine kinase activity that causes the dimerization of various receptors thereby triggering various specific signaling molecules [65]. The higher sensitivity of VEGF signaling in tumors than in normal tissue in response to inhibitory action of angiogenic inhibitors challenges us to extend our knowledge for antiangiogenic therapy. Extensive research in this area and the outcome are highly encouraging as numerous chemotherapeutic agents have been identified with potential antiangiogenic activity. One such antiangiogenic agent is bevacizumab (Avastin®; Genentech, Inc., South San Francisco, CA), the first FDA-approved drug that contains monoclonal IgG1 antibody against VEGF and is effective in controlling metastatic growth of colorectal, lung, and breast cancers when given in conjunction with other chemotherapeutic agents [11, 66–68]. Bevacizumab has been shown to inhibit angiogenesis via binding to VEGF-A, thereby blocking the VEGFa-VEFRs pathway [69]. Beside, drugs like sunitinib and sorafenib exert protective effects by inhibiting receptor tyrosine kinase (RTK) and Raf kinase [70, 71] and thereby affecting the signaling cascade of VEGF, PDGFR, MAPK, and Raf/MEK/ERK [72] (Fig. 3). Tipifarnib and lonafarnib have been shown to inhibit MAPK signaling [73, 74]. The other significant contributor of angiogenesis is the PI3K/AKT/m-TOR pathway, which is inhibited by temsirolimus and everolimus [75] (Fig. 3). Evidences from a number of clinical trials suggest the importance of angiogenic therapy in the treatment of cancer and that shall prove to be more promising when combined with other drugs [5, 28]. This can be evident from the studies where a combination therapy of bevacizumab with intravenous 5-fluorouracil (5-FU) showed positive results in colon and rectum tumors [11, 76]. Moreover, the treatment of cancer with tyrosine inhibitors like sunitinib and sorafenib gives promising results in renal carcinoma and gastrointestinal cancer [8, 12].
The normal activities of growth factors and their receptors are essential for normal physiology, but their activities get disturbed due to reaction of angiogenic inhibitors that block transduction of their signaling [77]. Inhibition or change in expressions of growth factors and their receptors by angiogenic inhibitors is associated with complications like disruption in vascular integrity, bleeding, and gastrointestinal perforation [78, 79]. When angiogenic inhibitors are used in combination with other drugs, as in the case of colorectal patients, serious bleeding problem may occur [80, 81]. Similarly, when bevacizumab is given along with carboplatin and paclitaxel in lung cancer patients, it may cause death due to pulmonary hemorrhage [82].
Since synthetic drugs mainly block the receptors of growth factors and are often associated with side effects, it is worthwhile to find agents that might block the synthesis of angiogenic factors rather than targeting their receptors [9]. Moreover, a combination of more than two compounds could prove to be more effective as it may improve the chances of inhibiting more than one pathway during angiogenesis. In this scenario, the plant products may prove to be useful, as many natural compounds have been identified that block the overproduction of angiogenic growth factors and their activities. Boswellic acid, curcumin, and resveratrol are some of the natural compounds that inhibit the action of angiogenic factors [83, 84]. Moreover, such compounds also improve vascular permeability and inflammation. The real advantage of natural plant products is that they show synergistic action which enhances the efficacy of these products to many folds. Presently, efforts are being made to formulate the combination of more than two natural compounds so that their cumulative effect is greater as compared to their individual effects.
Natural compounds as angiogenic inhibitors
Tumor vasculature is poorly developed and immature and hence can be targeted with antiangiogenic compounds. Natural compounds position themselves in the front row as they are safe and effective in low doses for a longer duration than are synthetic compounds which work well in single high dose [13, 14]. To explore and validate the potential of natural compounds against tumor angiogenesis, a number of clinical trials have documented positive results and provide ample hope in targeting tumor angiogenesis. Therefore, high doses of cytotoxic drug followed by side effects [85] inculcate interest in natural compounds as they have the potential of acting synergistically to block angiogenesis by targeting more than one pathway simultaneously without any side effects. This led to the discovery of many compounds (Table 2) having natural origin with great potential to inhibit angiogenesis both in vitro and in vivo experiments, and some of them some are discussed below.
EGCG (Camellia sinensis) (Table 2)
Tea, the most consumed beverage worldwide derived from Camellia sinensis, contains polyphenols which are associated with inhibition of tumor growth in different experimental models [116]. Studies have demonstrated that green tea extract inhibits tumor angiogenesis [86] by suppressing the VEGF production and expression of VEGF receptor [87–89, 117]. The most promising active component [87] of green tea and white tea is epigallocatechin-3-gallate (EGCG) [118]. EGCG is reported to inhibit signal transduction pathways that suppress the growth, proliferation, angiogenesis, and metastasis of tumors [116]. EGCG has been reported to suppress angiogenesis in breast cancer in female mice [90], gastric cancer in nude mice, and proliferation of cancer cell lines [119, 120]. Furthermore, EGCG also inhibits tumor xenograft growth in rodents [121]. This inhibitory action of EGCG is associated with the diminution in VEGF expression by inhibiting HIF-1α, NFκB activation, and expression of IL-6. Moreover, EGCG has also been reported to inhibit phosphorylation in protein kinase C (PKC). Studies have also advocated the inhibitory action of EGCG on EGFR signaling pathways [35, 92, 94]. EGCG has potential to inhibit precisely EGFR phosphorylation [93] and prevent binding of EGF to EGFR [94]. EGCG has been observed to block the phosphorylation of hepatocyte growth factor receptor (HGFR) in MDA-MB-2321 cells which prevent the binding of HGF to HGFR [122]. This suggests that EGCG affects multiple targets, which include receptors, growth factors, kinases, and enzymes, in tumor angiogenesis. So, it is undoubtedly clear from various studies that EGCG appears to be the most promising candidate for developing antiangiogenic therapy as it affects more than one target during angiogenesis. On the basis of evidences acquired from laboratory studies, EGCG has been evaluated in cancer patients so as to ascertain its protective/therapeutic efficacy with a safe dose. In chronic lymphocytic leukemia (CLL), EGCG given to patients at a dose level of 2 g/day is well tolerated for up to 6 months and has been shown to decrease the count of absolute lymphocytes [123, 124]. Similarly, Zhao et al. have seen the adjunctive role of EGCG with cisplatin and etoposide in the treatment of non-small cell lung carcinoma (NSCLC) patients [125]. They recommended a safe dose of 440 μM/L as a starting dose for a phase II trial [125]. Zhao et al. further demonstrated that EGCG solution (40–660 μM/L) in conjunction with radiotherapy reduced the radiation dermatitis in breast cancer patients [126]. The outcome of phase III clinical trials will reveal the true efficacy of EGCG in various cancer patients.
Curcumin (Curcuma longa) (Table 2)
Curcumin, a yellow pigment derived from Curcuma longa, has a long history of proven herbal medication in a variety of medical conditions. The health benefits of curcumin are due to its anticoagulant, antiparasitic, antioxidant, antiarthritic, and analgesic properties [15, 16, 127, 128]. Curcumin has also been shown to inhibit the enzyme activity of hyaluronidase and β-amyloids and is advocated for the treatment of Alzheimer’s, asthma, and HIV [129]. Moreover, dietary intake of curcumin has been reported to be associated with prevention of stomach, duodenum, and colon cancer in mice as well as progression of cancer in tongue, mammary glands, and sebaceous glands in rats [130, 131]. This antitumor activity of curcumin is considered to be associated with inhibition of angiogenesis [132]. Various in vitro and in vivo studies advocate the antiangiogenic effects of curcumin [9, 95, 96, 133]. Studies have also demonstrated antiangiogenic effects of curcumin through MAPK and COX-2 inhibition in human endothelial cells [97]. Moreover, curcumin is shown to decrease VEGF production in tumor cells by downregulating the HIF-1α [98, 99] and angiopoietin-1 and angiopoietin-2 in human umbilical vein endothelial cells (HUVECs) [100, 101]. Close interaction of curcumin with VEGF and nitric oxide in tumor leads to blockage of CD13 enzyme activity which otherwise is often higher in tumor-promoting angiogenesis [100]. The reduction of CD13 enzyme activity in tumor by curcumin results in reduction of angiogenesis and metastatic growth of tumor cells [102]. Another way to inhibit angiogenesis is by suppressing the release of angiogenic factors in to ECM. Curcumin has been reported to downregulate the expression of the matrix metalloproteinase-9 (MMP-9) and MMP-2 [103] and also block the EGFR and VEGF tyrosine kinase signaling pathways [104]. Recent studies have also demonstrated inhibitory effects of curcumin analog (GO-Y078) through actin disorganization [105]. Huang et al. have revealed the antiangiogenic potential of three curcumin pigments by downregulating the expression of VEGF and adhesion molecules [106]. Curcumin has also been shown to inhibit angiogenesis by peroxisome proliferator-activated receptor-γ (PPAR-γ) activation-dependent mechanism in hepatic fibrosis which could be another strategy to inhibit angiogenesis [107]. Studies with human patients suffering from various cancers have been conducted to evaluate the safety and efficacy of curcumin alone and in combination with other therapies. Curcumin alone (2 g/day) and in combination with FOLFOX chemotherapy reduced the spheroid number and number of cells with high aldehyde dehydrogenase activity in cancer stem cells (colorectal liver metastases) [134]. In another clinical trial, treatment of colorectal neoplasia, with 4 g of curcumin, decreased the ACF number [135]. Besides, 8 g of curcumin is found to be well tolerated and effective in the chemoprevention of cancer [136–138]. In pancreatic cancer, 8 g of curcumin per day downregulates the expressions of NFκB, COX-2, and various activators of transcription factor III in peripheral blood mononuclear cells (PBMCs) [137]. In patients with monoclonal gammopathy, there was a 12–30 % decrease in paraprotein while taking 4 g/day of curcumin [138]. One hundred eighty milligrams per day of curcuminoids, when given to patients with solid tumor undergoing chemotherapy, suppressed the systemic inflammation via decreasing the expressions of IL-6 and IL-8 and TNF-α and hs-CRP [139]. In breast cancer, 2 g/thrice a day of curcumin reduced the severity of radiation dermatitis [140]. Recently, a clinical trial (CUFOX trial) has been registered by the European drug regulatory authority to see the dose escalation of curcumin daily for colorectal cancer patients undergoing chemotherapy (oxaliplatin) [141]. In conclusion, it is evident from laboratory studies that curcumin inhibits angiogenesis by various mechanisms and clinical trials with cancer patients confer safety and efficacy of curcumin (4–8 g/day) alone and in combination with chemotherapy.
Resveratrol (Vaccinium spp.) (Table 2)
Resveratrol, a phytoalexin obtained from a variety of plants such as peanut (Vaccinium spp.), pine (Pinus spp.), and eucalyptus, especially red wine, is an important constituent of dietary intake [142, 143]. The important health benefits of resveratrol have been shown in cancer, diabetes, coronary heart diseases, and neurodegeneration [144–146]. These health benefits are attributed to the anti-inflammatory and redox-balancing properties of resveratrol [147]. Studies have demonstrated that resveratrol is quite effective in delaying the onset of molecular events leading to chemically induced carcinogenesis [148, 149]. The anticancer effects of resveratrol are due to its potential to act on cell proliferation, angiogenesis, protection against genotoxicity, promoting apoptosis, and autophagy [149]. In humans, antiangiogenesis potential has been shown to be due to downregulation of angiogenic factors by inhibiting VEGF expression through HIF-1α [108]. In vitro studies in HepG2 cells also suggested inhibition of VEGF expression due to resveratrol [109, 110].Moreover, resveratrol has also been shown to inhibit VEGF and EGF receptor-mediated MAPK activation and hence blocks the growth of new vessels in animals [150]. Kimura et al. reported the inhibition in the growth of colon cancer cell xenograft in mice by suppressing VEGF and MMP-9 [111]. Srivastava et al. demonstrated antiangiogenic effects of resveratrol by inhibiting the PI3/AKT and Ras/MEK/ERK pathways [112]. Besides, various studies have also been carried out to see the synergistic effects of different principle components (resveratrol with curcumin or with EGCG) of varying plant products [15]. In mice model, curcumin in conjunction with resveratrol has been shown to inhibit the VEGF expression and its type 2 receptor [101]. Antiangiogenic effects of resveratrol/curcumin involve multitargeting of key steps as inhibition of growth factor, growth factor receptor, and signaling pathways contributing to angiogenesis.
Clinical trials of patients with hepatic metastases showed an increase of caspase-3 activity in 39 % of malignant hepatic tissue following 5 g/day treatment of resveratrol for 14 days [151]. In colorectal cell lines, resveratrol was able to inhibit the Wnt/β-catenin pathway by downregulating the MALAT-1, MMP-7, and c-myc expression [152]. In another study, 39 women with high-risk breast cancer were randomized to receive daily dose of 5 or 50 mg of trans-resveratrol treatment, and the results indicated a decrease in methylation of tumor suppressor gene (RASSF-1α) with an increase in serum concentration of resveratrol [153]. However, more laboratory studies as well as clinical studies are required to investigate the angiogenesis inhibition pathways in order to prove the potential of resveratrol in cancer.
Artemisinin ( Artemisia annua) (Table 2)
Artemisinin is a potential drug used in malaria control and is derived from Artemisia annua (A. anna) plant [154]. Studies have revealed the cytotoxic potential of artemisinin through apoptosis induction in cancer cells [155]. In human umbilical vein endothelial model, the artesunate (derivative of artemisinin) is reported to have a dose-dependent inhibition of angiogenesis [103]. Further studies have advocated that artemisinin has a greater potential to inhibit angiogenesis rather than induct cytotoxicity [85]. HO-891 cells in nude mice, and then treated with artemisinin, revealed low expression of VEGF and VEGF receptor in endothelial cells. Moreover, it also inhibits the NF-κB which plays a significant role in the development and progression of cancer [113]. The modulation of gene expression of angiogenic factor could be the possible molecular mechanism, studies have reported the downregulation of VEGF, FGF [91], HIF-α, MMP-9, and MMP-11 [156] In addition, the upregulation of angiogenic inhibitor has also been documented [156]. One of the phase I trials in human patients evaluated the ototoxicity of artesunate (ARTICM33/2) as add-on therapy for 4 weeks with varying doses (100, 150, or 200 mg), and the trial was found to be safe and effective [157]. One hundred twenty-one patients with NSCLC were randomized to receive artesunate along with chemotherapy (vinorelbine and cisplatin), and it was observed that the artesunate-treated group had delayed the time of progression and also increased the short-time survival rate [158]. A pilot study with a dose of 100 mg of artenimol-R for 7 days followed by 200 mg/day of artenimol-R for another 21 days in treatment of advance cervical cancer was found to be safe and has resolved clinical symptom by decreasing the expression of EGFR, antiKi-67, and CD31 and increasing the expression of CD71 cells [159], hence proving its role in cancer treatment.
Boswellic acid (Boswellia serrata)
Boswellic acid (BA) is an emerging Indian frankincense obtained from the plant Boswellia serrata (B. serrata). It has the potential to cure inflammatory diseases which include bronchial asthma, osteoarthritis, chronic colitis, ulcerative colitis, and Crohn’s disease. The plant extract B. serrata has mainly four constituents which include pentacyclic triterpene acids: 3-acetyl-11-keto-beta-boswellic acid, 11-keto-beta-boswellic acid, beta-boswellic acid, and 3 acetyl-beta-boswellic acid [160]. Acetyl-11-keto-beta-boswellic acid (AKBA) is the main constituent which directly interacts with IκB kinases and inhibits nuclear factor-κB-regulated gene expression [161, 162]. AKBA has also been shown to inhibit non-competitively 5-LOX which induces angiogenesis by enhancing VEGF expression. AKBA has been shown to inhibit tumor growth and angiogenesis by targeting VEGFR2 activation and mTOR signaling pathways [163]. AKBA has been shown to suppress the constitutive activation of STAT3 in human MM cells by IL-6 which upregulates the VEGF expression. Furthermore, AKBA also downregulates the STAT3 pathway by inhibiting the phosphorylation of both Jak2 and Src, which are constituents of the STAT3 pathway. AKBA-induced inhibition is followed by the suppression of gene products such as cyclin D1, Bcl-2, Bcl-xL, Mcl-1, and VEGF which are involved in proliferation, survival, and angiogenesis. These effects correlate with the inhibition of proliferation and apoptosis in MM cells. Unswerving with these facts, overexpression of STAT3 significantly decreases the AKBA-induced apoptosis [164]. AKBA treatment in a sponge implant model has also been shown to inhibit proinflammatory cytokine TNF-α and profibrogenic cytokine TGF-β in the implants at different doses that indicate reduction in inflammation [160]. In the retina, AKBA induces the Src homology region 2 domain-containing phosphatase 1 expression and decreases phosphorylation of transcription factor signal transducer and also induces the activator of transcription 3 (STAT3) as well as VEGF expression and VEGF receptor (VEGFR)-2 phosphorylation. AKBA also affects the SHP-1/STAT3/VEGF axis which leads to inhibition of VEGFR-2 phosphorylation [165]. Antiangiogenic effects of AKBA in different models provide the scientific basis for future exploration of AKBA as a potential therapeutic factor to reduce the impact of angiogenesis in tumor. Patients with primary or secondary malignant cerebral edema undergoing radiotherapy were administered with BS4 200 mg/day or placebo group shown to significantly reduce the cerebral edema detected by magnetic resonance imaging (MRI) [166]. In another randomized double-blind trial, efficacy, tolerability, and safety of base cream containing 0.5 % BA for the treatment of photoaging was found to be well tolerated without adverse effects [167]. Clinical evaluation has been conducted with Boswellia-based cream for radiotherapy-induced skin damage in mammary carcinoma and was found to be effective for breast cancer patients who undergo radiation therapy [168]. Further, the true potential of boswellic acids as an emerging natural compound needs to be investigated by conducting laboratory experiments and clinical trials.
Promising natural plant products
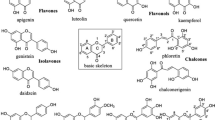
There are several other plant products that inhibit angiogenesis in tumors and among them is Ginkgo biloba (G. biloba). Although much literature is not available to advocate the antiangiogenic potential of G. biloba, some isolated studies have shown in skin inflammation that G. biloba leaf extract inhibits the VEGF and CXCL8/IL-8 release in normal human keratinocytes [169]. In contrast to this study on Wistar rats with subarachnoid hemorrhage, G. biloba extract was shown to increase the expression of VEGF [170]. The possible mechanisms for G. biloba extract involve the activation of protein tyrosine phosphatase which leads to inhibition of the Raf-MEK-ERK pathways [171]. Other than EGCG, flavonoids of fruits like silibinin, quercetin, baicalin, and baicalein are useful in tumor inhibition. Silibinin induced HIF-α and VEGF downregulation by the PI3K/mTOR pathways [171]. In basal cell carcinoma, silibinin blocks the mitogenic signaling by inhibiting EGFR, ERK, Akt, and STAT3 phosphorylation [172, 173]. Another study on a xenograft model also advocates the suppression of COX-2, VEGF, and MMP-9 expression by silibinin [174]. Viscum album extracts and its formulations induce apoptosis and have also been shown to downregulate VEGF, have cytotoxic and immunomodulatory effects, and inhibit metastasis in cell line studies [175, 176]. Baicalin and baicalein are two major flavonoids derived from Scutellaria baicalensis (Chinese skullcap herb) and have been used traditionally in China and Japan [115], and they have been found to inhibit angiogenesis in chick chorioallantoic membrane assay in a dose-dependent manner. Moreover, in HUVECs, these two flavonoids show dual effects: antiproliferative at low dose and apoptotic at high doses [115]. In human prostate cancer cells, they have also been shown to exert growth inhibitory effects by reducing VEGF and bFGF activity [177].
Conclusion and future prospective
Angiogenesis plays an essential role in the growth and metastasis of cancer cells, and angiogenic therapy can halt this growth. Synthetic drugs specific for only one target may not be functionally sufficient to retard the process of angiogenesis and even result in the manifestation of side effects that are detrimental to human health. The active principles of natural plants exhibit synergistic activity while interacting with various molecular targets of multiple pathways that include mTOR, P13K/AKT, MEK/ERK, and MAPK. There are two major challenges to exploit the full potential of these natural compound principles. The first one involves identification of natural compounds that interact synergistically to give an effective outcome, and the second is to unravel effective dose by combining more than two active principles to target more than one biochemical pathway simultaneously. Clinical studies have ascertained that these natural compounds are safe and effective to use alone and in combination with chemotherapy. Hence, future studies are warranted to ascertain meaningful outcome when more than two natural compounds are synergistically taken into consideration to target multiple pathways of angiogenesis so as to develop effective angiogenic therapy.
References
Polverini PJ. Angiogenesis in health and disease: insights into basic mechanisms and therapeutic opportunities. J Dent Educ. 2002;66:962–75.
Ribatti D, Djonov V. Intussusceptive microvascular growth in tumors. Cancer Lett. 2012;316:126–31.
Bisht M, Dhasmana DC, Bist SS. Angiogenesis: future of pharmacological modulation. Indian J Pharmacol. 2010;42:2–8.
Folkman J. Angiogenesis: an organizing principle for drug discovery? Nat Rev Drug Discov. 2007;6:273–86.
Carmeliet P. Angiogenesis in health and disease. Nat Med. 2003;9:653–60.
Ali TK, El-Remessy AB. Diabetic retinopathy: current management and experimental therapeutic targets. Pharmacotherapy. 2009;29:182–92.
Willis LM, El-Remessy AB, Somanath PR, Deremer DL, Fagan SC. Angiotensin receptor blockers and angiogenesis: clinical and experimental evidence. Clin Sci (Lond). 2011;120:307–19.
Motzer RJ, Rini BI, Bukowski RM, Curti BD, George DJ, Hudes GR, et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA. 2006;295:2516–24.
Wang Z, Dabrosin C, Yin X, Fuster MM, Arreola A, Rathmell WK, et al. Broad targeting of angiogenesis for cancer prevention and therapy. Semin Cancer Biol. 2015;35(Suppl):S224–43.
Fayette J, Soria JC, Armand JP. Use of angiogenesis inhibitors in tumour treatment. Eur J Cancer. 2005;41:1109–16.
Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350:2335–42.
Wilhelm S, Carter C, Lynch M, Lowinger T, Dumas J, Smith RA, et al. Discovery and development of sorafenib: a multikinase inhibitor for treating cancer. Nat Rev Drug Discov. 2006;5:835–44.
Eberhard A, Kahlert S, Goede V, Hemmerlein B, Plate KH, Augustin HG. Heterogeneity of angiogenesis and blood vessel maturation in human tumors: implications for antiangiogenic tumor therapies. Cancer Res. 2000;60:1388–93.
Benjamin LE, Golijanin D, Itin A, Pode D, Keshet E. Selective ablation of immature blood vessels in established human tumors follows vascular endothelial growth factor withdrawal. J Clin Invest. 1999;103:159–65.
Malhotra A, Nair P, Dhawan DK. Study to evaluate molecular mechanics behind synergistic chemo-preventive effects of curcumin and resveratrol during lung carcinogenesis. PLoS One. 2014;9:e93820.
Aggarwal BB, Prasad S, Reuter S, Kannappan R, Yadev VR, Park B, et al. Identification of novel anti-inflammatory agents from Ayurvedic medicine for prevention of chronic diseases: “reverse pharmacology” and “bedside to bench” approach. Curr Drug Targets. 2011;12:1595–653.
Papetti M, Herman IM. Mechanisms of normal and tumor-derived angiogenesis. Am J Physiol Cell Physiol. 2002;282:C947–70.
Norrby K. Angiogenesis: new aspects relating to its initiation and control. APMIS. 1997;105:417–37.
Pandya NM, Dhalla NS, Santani DD. Angiogenesis—a new target for future therapy. Vasc Pharmacol. 2006;44:265–74.
Kerbel RS. Tumor angiogenesis: past, present and the near future. Carcinogenesis. 2000;21:505–15.
Hanahan D, Folkman J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell. 1996;86:353–64.
Baeriswyl V, Christofori G. The angiogenic switch in carcinogenesis. Semin Cancer Biol. 2009;19:329–37.
Pepper MS. Role of the matrix metalloproteinase and plasminogen activator-plasmin systems in angiogenesis. Arterioscler Thromb Vasc Biol. 2001;21:1104–17.
Folkman J, Shing Y. Angiogenesis. J Biol Chem. 1992;267:10931–4.
Denekamp J. Review article: angiogenesis, neovascular proliferation and vascular pathophysiology as targets for cancer therapy. Br J Radiol. 1993;66:181–96.
Adachi S, Nagao T, To S, Joe AK, Shimizu M, Matsushima-Nishiwaki R, et al. (−)-Epigallocatechin gallate causes internalization of the epidermal growth factor receptor in human colon cancer cells. Carcinogenesis. 2008;29:1986–93.
Losordo DW, Dimmeler S. Therapeutic angiogenesis and vasculogenesis for ischemic disease. Part I: angiogenic cytokines. Circulation. 2004;109:2487–91.
Gasparini G, Longo R, Fanelli M, Teicher BA. Combination of antiangiogenic therapy with other anticancer therapies: results, challenges, and open questions. J Clin Oncol. 2005;23:1295–311.
Leung DW, Cachianes G, Kuang WJ, Goeddel DV, Ferrara N. Vascular endothelial growth factor is a secreted angiogenic mitogen. Science. 1989;246:1306–9.
Kolch W, Martiny-Baron G, Kieser A, Marme D. Regulation of the expression of the VEGF/VPS and its receptors: role in tumor angiogenesis. Breast Cancer Res Treat. 1995;36:139–55.
Sunderkotter C, Steinbrink K, Goebeler M, Bhardwaj R, Sorg C. Macrophages and angiogenesis. J Leukoc Biol. 1994;55:410–22.
Motro B, Itin A, Sachs L, Keshet E. Pattern of interleukin 6 gene expression in vivo suggests a role for this cytokine in angiogenesis. Proc Natl Acad Sci U S A. 1990;87:3092–6.
Van Meir EG. Cytokines and tumors of the central nervous system. Glia. 1995;15:264–88.
Zhang L, Rui YC, Yang PY, Qiu Y, Li TJ, Liu HC. Inhibitory effects of Ginkgo biloba extract on vascular endothelial growth factor in rat aortic endothelial cells. Acta Pharmacol Sin. 2002;23:919–23.
Hou Z, Sang S, You H, Lee MJ, Hong J, Chin KV, et al. Mechanism of action of (−)-epigallocatechin-3-gallate: auto-oxidation-dependent inactivation of epidermal growth factor receptor and direct effects on growth inhibition in human esophageal cancer KYSE 150 cells. Cancer Res. 2005;65:8049–56.
Fajardo LF, Kwan HH, Kowalski J, Prionas SD, Allison AC. Dual role of tumor necrosis factor-alpha in angiogenesis. Am J Pathol. 1992;140:539–44.
Jendraschak E, Sage EH. Regulation of angiogenesis by SPARC and angiostatin: implications for tumor cell biology. Semin Cancer Biol. 1996;7:139–46.
Gately S, Li WW. Multiple roles of COX-2 in tumor angiogenesis: a target for antiangiogenic therapy. Semin Oncol. 2004;31:2–11.
Nie D, Krishnamoorthy S, Jin R, Tang K, Chen Y, Qiao Y, et al. Mechanisms regulating tumor angiogenesis by 12-lipoxygenase in prostate cancer cells. J Biol Chem. 2006;281:18601–9.
Ma ZS, Huynh TH, Ng CP, Do PT, Nguyen TH, Huynh H. Reduction of CWR22 prostate tumor xenograft growth by combined tamoxifen-quercetin treatment is associated with inhibition of angiogenesis and cellular proliferation. Int J Oncol. 2004;24:1297–304.
Yadav L, Puri N, Rastogi V, Satpute P, Sharma V. Tumour angiogenesis and angiogenic inhibitors: a review. J Clin Diagn Res. 2015;9:XE01–XE5.
Neufeld G, Cohen T, Gengrinovitch S, Poltorak Z. Vascular endothelial growth factor (VEGF) and its receptors. FASEB J. 1999;13:9–22.
Gupta MK, Qin RY. Mechanism and its regulation of tumor-induced angiogenesis. World J Gastroenterol. 2003;9:1144–55.
Huang YH, Yang HY, Hsu YF, Chiu PT, Ou G, Hsu MJ. Src contributes to IL6-induced vascular endothelial growth factor-C expression in lymphatic endothelial cells. Angiogenesis. 2014;17:407–18.
Nagasaki T, Hara M, Nakanishi H, Takahashi H, Sato M, Takeyama H. Interleukin-6 released by colon cancer-associated fibroblasts is critical for tumour angiogenesis: anti-interleukin-6 receptor antibody suppressed angiogenesis and inhibited tumour-stroma interaction. Br J Cancer. 2014;110:469–78.
Shao J, Sheng GG, Mifflin RC, Powell DW, Sheng H. Roles of myofibroblasts in prostaglandin E2-stimulated intestinal epithelial proliferation and angiogenesis. Cancer Res. 2006;66:846–55.
Fagiani E, Lorentz P, Kopfstein L, Christofori G. Angiopoietin-1 and -2 exert antagonistic functions in tumor angiogenesis, yet both induce lymphangiogenesis. Cancer Res. 2011;71:5717–27.
Harris RE, Casto BC, Harris ZM. Cyclooxygenase-2 and the inflammogenesis of breast cancer. World J Clin Oncol. 2014;5:677–92.
Vosooghi M, Amini M. The discovery and development of cyclooxygenase-2 inhibitors as potential anticancer therapies. Expert Opin Drug Discov. 2014;9:255–67.
Knab LM, Grippo PJ, Bentrem DJ. Involvement of eicosanoids in the pathogenesis of pancreatic cancer: the roles of cyclooxygenase-2 and 5-lipoxygenase. World J Gastroenterol. 2014;20:10729–39.
Liu B, Qu L, Yan S. Cyclooxygenase-2 promotes tumor growth and suppresses tumor immunity. Cancer Cell Int. 2015.
Pidgeon GP, Lysaght J, Krishnamoorthy S, Reynolds JV, O’Byrne K, Nie D, et al. Lipoxygenase metabolism: roles in tumor progression and survival. Cancer Metastasis Rev. 2007;26:503–24.
Nassar A, Radhakrishnan A, Cabrero IA, Cotsonis G, Cohen C. COX-2 expression in invasive breast cancer: correlation with prognostic parameters and outcome. Appl Immunohistochem Mol Morphol. 2007;15:255–9.
Nie D, Nemeth J, Qiao Y, Zacharek A, Li L, Hanna K, et al. Increased metastatic potential in human prostate carcinoma cells by overexpression of arachidonate 12-lipoxygenase. Clin Exp Metastasis. 2003;20:657–63.
Weng CJ, Chou CP, Ho CT, Yen GC. Molecular mechanism inhibiting human hepatocarcinoma cell invasion by 6-shogaol and 6-gingerol. Mol Nutr Food Res. 2012;56:1304–14.
Sethi G, Shanmugam MK, Ramachandran L, Kumar AP, Tergaonkar V. Multifaceted link between cancer and inflammation. Biosci Rep. 2012;32:1–15.
Vaccaro V, Melisi D, Bria E, Cuppone F, Ciuffreda L, Pino MS, et al. Emerging pathways and future targets for the molecular therapy of pancreatic cancer. Expert Opin Ther Targets. 2011;15:1183–96.
Batra S, Balamayooran G, Sahoo MK. Nuclear factor-kappaB: a key regulator in health and disease of lungs. Arch Immunol Ther Exp. 2011;59:335–51.
Guttridge DC, Albanese C, Reuther JY, Pestell RG, Baldwin Jr AS. NF-kappaB controls cell growth and differentiation through transcriptional regulation of cyclin D1. Mol Cell Biol. 1999;19:5785–99.
Huber MA, Azoitei N, Baumann B, Grunert S, Sommer A, Pehamberger H, et al. NF-kappaB is essential for epithelial-mesenchymal transition and metastasis in a model of breast cancer progression. J Clin Invest. 2004;114:569–81.
Wilczynska KM, Gopalan SM, Bugno M, Kasza A, Konik BS, Bryan L, et al. A novel mechanism of tissue inhibitor of metalloproteinases-1 activation by interleukin-1 in primary human astrocytes. J Biol Chem. 2006;281:34955–64.
Liu RM. Oxidative stress, plasminogen activator inhibitor 1, and lung fibrosis. Antioxid Redox Signal. 2008;10:303–19.
Davis ME, Chen ZG, Shin DM. Nanoparticle therapeutics: an emerging treatment modality for cancer. Nat Rev Drug Discov. 2008;7:771–82.
Dvorak HF. Vascular permeability factor/vascular endothelial growth factor: a critical cytokine in tumor angiogenesis and a potential target for diagnosis and therapy. J Clin Oncol. 2002;20:4368–80.
Olsson AK, Dimberg A, Kreuger J, Claesson-Welsh L. VEGF Receptor signalling - in control of vascular function. Nat Rev Mol Cell Biol. 2006;7:359–71.
Sandler A, Gray R, Perry MC, Brahmer J, Schiller JH, Dowlati A, et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med. 2006;355:2542–50.
Miller K, Wang M, Gralow J, Dickler M, Cobleigh M, Perez EA, et al. Paclitaxel plus bevacizumab versus paclitaxel alone for metastatic breast cancer. N Engl J Med. 2007;357:2666–76.
Penson RT, Huang HQ, Wenzel LB, Monk BJ, Stockman S, Long 3rd HJ, et al. Bevacizumab for advanced cervical cancer: patient-reported outcomes of a randomised, phase 3 trial (NRG Oncology-Gynecologic Oncology Group protocol 240. Lancet Oncol. 2015;16:301–11.
Niu G, Chen X. Vascular endothelial growth factor as an anti-angiogenic target for cancer therapy. Curr Drug Targets. 2010;11:1000–17.
Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med. 2007;356:125–34.
Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–90.
Wilhelm SM, Adnane L, Newell P, Villanueva A, Llovet JM, Lynch M. Preclinical overview of sorafenib, a multikinase inhibitor that targets both Raf and VEGF and PDGF receptor tyrosine kinase signaling. Mol Cancer Ther. 2008;7:3129–40.
Mesa RA. Tipifarnib: farnesyl transferase inhibition at a crossroads. Expert Rev Anticancer Ther. 2006;6:313–9.
Liu M, Bryant MS, Chen J, Lee S, Yaremko B, Lipari P, et al. Antitumor activity of SCH 66336, an orally bioavailable tricyclic inhibitor of farnesyl protein transferase, in human tumor xenograft models and wap-ras transgenic mice. Cancer Res. 1998;58:4947–56.
Faivre S, Kroemer G, Raymond E. Current development of mTOR inhibitors as anticancer agents. Nat Rev Drug Discov. 2006;5:671–88.
Hurwitz H, Saini S. Bevacizumab in the treatment of metastatic colorectal cancer: safety profile and management of adverse events. Semin Oncol. 2006;33:S26–34.
Elice F, Rodeghiero F. Bleeding complications of antiangiogenic therapy: pathogenetic mechanisms and clinical impact. Thromb Res. 2010;125(Suppl 2):S55–7.
Verheul HM, Pinedo HM. Possible molecular mechanisms involved in the toxicity of angiogenesis inhibition. Nat Rev Cancer. 2007;7:475–85.
Los M, Roodhart JM, Voest EE. Target practice: lessons from phase III trials with bevacizumab and vatalanib in the treatment of advanced colorectal cancer. Oncologist. 2007;12:443–50.
Kabbinavar F, Hurwitz HI, Fehrenbacher L, Meropol NJ, Novotny WF, Lieberman G, et al. Phase II, randomized trial comparing bevacizumab plus fluorouracil (FU)/leucovorin (LV) with FU/LV alone in patients with metastatic colorectal cancer. J Clin Oncol. 2003;21:60–5.
Giantonio BJ, Catalano PJ, Meropol NJ, O’Dwyer PJ, Mitchell EP, Alberts SR, et al. Bevacizumab in combination with oxaliplatin, fluorouracil, and leucovorin (FOLFOX4) for previously treated metastatic colorectal cancer: results from the Eastern Cooperative Oncology Group Study E3200. J Clin Oncol. 2007;25:1539–44.
Johnson DH, Fehrenbacher L, Novotny WF, Herbst RS, Nemunaitis JJ, Jablons DM, et al. Randomized phase II trial comparing bevacizumab plus carboplatin and paclitaxel with carboplatin and paclitaxel alone in previously untreated locally advanced or metastatic non-small-cell lung cancer. J Clin Oncol. 2004;22:2184–91.
Kimura Y, Okuda H. Resveratrol isolated from Polygonum cuspidatum root prevents tumor growth and metastasis to lung and tumor-induced neovascularization in Lewis lung carcinoma-bearing mice. J Nutr. 2001;131:1844–9.
Banerjee S, Bueso-Ramos C, Aggarwal BB. Suppression of 7,12-dimethylbenz(a)anthracene-induced mammary carcinogenesis in rats by resveratrol: role of nuclear factor-kappaB, cyclooxygenase 2, and matrix metalloprotease 9. Cancer Res. 2002;62:4945–54.
Sagar SM, Yance D, Wong RK. Natural health products that inhibit angiogenesis: a potential source for investigational new agents to treat cancer-part 1. Curr Oncol. 2006;13:14–26.
Fassina G, Vene R, Morini M, Minghelli S, Benelli R, Noonan DM, et al. Mechanisms of inhibition of tumor angiogenesis and vascular tumor growth by epigallocatechin-3-gallate. Clin Cancer Res. 2004;10:4865–73.
Kondo T, Ohta T, Igura K, Hara Y, Kaji K. Tea catechins inhibit angiogenesis in vitro, measured by human endothelial cell growth, migration and tube formation, through inhibition of VEGF receptor binding. Cancer Lett. 2002;180:139–44.
Kojima-Yuasa A, Hua JJ, Kennedy DO, Matsui-Yuasa I. Green tea extract inhibits angiogenesis of human umbilical vein endothelial cells through reduction of expression of VEGF receptors. Life Sci. 2003;73:1299–313.
Lamy S, Gingras D, Beliveau R. Green tea catechins inhibit vascular endothelial growth factor receptor phosphorylation. Cancer Res. 2002;62:381–5.
Gu JW, Makey KL, Tucker KB, Chinchar E, Mao X, Pei I, et al. EGCG, a major green tea catechin suppresses breast tumor angiogenesis and growth via inhibiting the activation of HIF-1alpha and NFkappaB, and VEGF expression. Vasc Cell. 2013;5:9.
Zhou HJ, Wang WQ, Wu GD, Lee J, Li A. Artesunate inhibits angiogenesis and downregulates vascular endothelial growth factor expression in chronic myeloid leukemia K562 cells. Vasc Pharmacol. 2007;47:131–8.
Rodriguez SK, Guo W, Liu L, Band MA, Paulson EK, Meydani M. Green tea catechin, epigallocatechin-3-gallate, inhibits vascular endothelial growth factor angiogenic signaling by disrupting the formation of a receptor complex. Int J Cancer. 2006;118:1635–44.
Liang YC, Lin-shiau SY, Chen CF, Lin JK. Suppression of extracellular signals and cell proliferation through EGF receptor binding by (−)-epigallocatechin gallate in human A431 epidermoid carcinoma cells. J Cell Biochem. 1997;67:55–65.
Adachi S, Nagao T, Ingolfsson HI, Maxfield FR, Andersen OS, Kopelovich L, et al. The inhibitory effect of (−)-epigallocatechin gallate on activation of the epidermal growth factor receptor is associated with altered lipid order in HT29 colon cancer cells. Cancer Res. 2007;67:6493–501.
Arbiser JL, Klauber N, Rohan R, van Leeuwen R, Huang MT, Fisher C, et al. Curcumin is an in vivo inhibitor of angiogenesis. Mol Med. 1998;4:376–83.
Mohan R, Sivak J, Ashton P, Russo LA, Pham BQ, Kasahara N, et al. Curcuminoids inhibit the angiogenic response stimulated by fibroblast growth factor-2, including expression of matrix metalloproteinase gelatinase B. J Biol Chem. 2000;275:10405–12.
Binion DG, Otterson MF, Rafiee P. Curcumin inhibits VEGF-mediated angiogenesis in human intestinal microvascular endothelial cells through COX-2 and MAPK inhibition. Gut. 2008;57:1509–17.
Shan B, Schaaf C, Schmidt A, Lucia K, Buchfelder M, Losa M, et al. Curcumin suppresses HIF1A synthesis and VEGFA release in pituitary adenomas. J Endocrinol. 2012;214:389–98.
Bae MK, Kim SH, Jeong JW, Lee YM, Kim HS, Kim SR, et al. Curcumin inhibits hypoxia-induced angiogenesis via down-regulation of HIF-1. Oncol Rep. 2006;15:1557–62.
Gururaj AE, Belakavadi M, Venkatesh DA, Marme D, Salimath BP. Molecular mechanisms of anti-angiogenic effect of curcumin. Biochem Biophys Res Commun. 2002;297:934–42.
El-Azab M, Hishe H, Moustafa Y, El-Awadyel S. Anti-angiogenic effect of resveratrol or curcumin in Ehrlich ascites carcinoma-bearing mice. Eur J Pharmacol. 2011;652:7–14.
Hahm ER, Gho YS, Park S, Park C, Kim KW, Yang CH. Synthetic curcumin analogs inhibit activator protein-1 transcription and tumor-induced angiogenesis. Biochem Biophys Res Commun. 2004;321:337–44.
Chen HW, Yu SL, Chen JJ, Li HN, Lin YC, Yao PL, et al. Anti-invasive gene expression profile of curcumin in lung adenocarcinoma based on a high throughput microarray analysis. Mol Pharmacol. 2004;65:99–110.
Leu TH, Su SL, Chuang YC, Maa MC. Direct inhibitory effect of curcumin on Src and focal adhesion kinase activity. Biochem Pharmacol. 2003;66:2323–31.
Sugiyama S, Yoshino Y, Kuriyama S, Inoue M, Komine K, Otsuka K, et al. A curcumin analog, GO-Y078, effectively inhibits angiogenesis through actin disorganization. Anti Cancer Agents Med Chem. 2016;16:633–47.
Huang YF, Zhu XX, Ding ZS, Lv GY. Study on anti-angiogenesis effect of three curcumin pigments and expression of their relevant factors. Zhongguo Zhong Yao Za Zhi. 2015;40:324–9.
Zhang F, Zhang Z, Chen L, Kong D, Zhang X, Lu C, et al. Curcumin attenuates angiogenesis in liver fibrosis and inhibits angiogenic properties of hepatic stellate cells. J Cell Mol Med. 2014;18:1392–406.
Cao Z, Fang J, Xia C, Shi X, Jiang BH. Trans-3,4,5′-Trihydroxystibene inhibits hypoxia-inducible factor 1alpha and vascular endothelial growth factor expression in human ovarian cancer cells. Clin Cancer Res. 2004;10:5253–63.
Zhang H, Yang R. Resveratrol inhibits VEGF gene expression and proliferation of hepatocarcinoma cells. Hepato-Gastroenterology. 2014;61:410–2.
Kim DH, Hossain MA, Kim MY, Kim JA, Yoon JH, Suh HS, et al. A novel resveratrol analogue, HS-1793, inhibits hypoxia-induced HIF-1alpha and VEGF expression, and migration in human prostate cancer cells. Int J Oncol. 2013;43:1915–24.
Kimura Y, Sumiyoshi M, Baba K. Antitumor activities of synthetic and natural stilbenes through antiangiogenic action. Cancer Sci. 2008;99:2083–96.
Srivastava RK, Unterman TG, Shankar SFOXO. Transcription factors and VEGF neutralizing antibody enhance antiangiogenic effects of resveratrol. Mol Cell Biochem. 2010;337:201–12.
Aldieri E, Atragene D, Bergandi L, Riganti C, Costamagna C, Bosia A, et al. Artemisinin inhibits inducible nitric oxide synthase and nuclear factor NF-κB activation. FEBS Lett. 2003;552:141–4.
Harvey Lodish A B, S Lawrence Zipursky, Paul Matsudaira, David Baltimore, James Darnell., Molecular Cell Biology. 5 ed, ed. H. Lodish. 2003: Freeman, W. H. & Company. 973.
Liu JJ, Huang TS, Cheng WF, Lu FJ. Baicalein and baicalin are potent inhibitors of angiogenesis: inhibition of endothelial cell proliferation, migration and differentiation. Int J Cancer. 2003;106:559–65.
Yang CS, Wang X, Lu G, Picinich SC. Cancer prevention by tea: animal studies, molecular mechanisms and human relevance. Nat Rev Cancer. 2009;9:429–39.
Jung YD, Kim MS, Shin BA, Chay KO, Ahn BW, Liu W, et al. EGCG, a major component of green tea, inhibits tumour growth by inhibiting VEGF induction in human colon carcinoma cells. Br J Cancer. 2001;84:844–50.
Lee MJ, Maliakal P, Chen L, Meng X, Bondoc FY, Prabhu S, et al. Pharmacokinetics of tea catechins after ingestion of green tea and (−)-epigallocatechin-3-gallate by humans: formation of different metabolites and individual variability. Cancer Epidemiol Biomark Prev. 2002;11:1025–32.
Sartippour MR, Shao ZM, Heber D, Beatty P, Zhang L, Liu C, et al. Green tea inhibits vascular endothelial growth factor (VEGF) induction in human breast cancer cells. J Nutr. 2002;132:2307–11.
Zhu BH, Zhan WH, Li ZR, Wang Z, He YL, Peng JS, et al. (−)-Epigallocatechin-3-gallate inhibits growth of gastric cancer by reducing VEGF production and angiogenesis. World J Gastroenterol. 2007;13:1162–9.
Cao Y, Cao R. Angiogenesis inhibited by drinking tea. Nature. 1999;398:381.
Peschard P, Park M. From Tpr-Met to Met, tumorigenesis and tubes. Oncogene. 2007;26:1276–85.
Shanafelt TD, Call TG, Zent CS, LaPlant B, Bowen DA, Roos M, et al. Phase I trial of daily oral polyphenon E in patients with asymptomatic Rai stage 0 to II chronic lymphocytic leukemia. J Clin Oncol. 2009;27:3808–14.
Shanafelt TD, Call TG, Zent CS, Leis JF, LaPlant B, Bowen DA, et al. Phase 2 trial of daily, oral Polyphenon E in patients with asymptomatic, Rai stage 0 to II chronic lymphocytic leukemia. Cancer. 2013;119:363–70.
Zhao H, Zhu W, Xie P, Li H, Zhang X, Sun X, et al. A phase I study of concurrent chemotherapy and thoracic radiotherapy with oral epigallocatechin-3-gallate protection in patients with locally advanced stage III non-small-cell lung cancer. Radiother Oncol. 2014;110:132–6.
Zhao H, Zhu W, Jia L, Sun X, Chen G, Zhao X, et al. Phase I study of topical epigallocatechin-3-gallate (EGCG) in patients with breast cancer receiving adjuvant radiotherapy. Br J Radiol. 2016;89:20150665.
Narayan S. Curcumin, a multi-functional chemopreventive agent, blocks growth of colon cancer cells by targeting beta-catenin-mediated transactivation and cell-cell adhesion pathways. J Mol Histol. 2004;35:301–7.
Nair P, Malhotra A, Dhawan DK. Curcumin and quercetin trigger apoptosis during benzo(a)pyrene-induced lung carcinogenesis. Mol Cell Biochem. 2015;400:51–6.
Aggarwal BB, Kumar A, Bharti AC. Anticancer potential of curcumin: preclinical and clinical studies. Anticancer Res. 2003;23:363–98.
Kelloff GJ, Crowell JA, Hawk ET, Steele VE, Lubet RA, Boone CW, et al. Strategy and planning for chemopreventive drug development: clinical development plans II. J Cell Biochem Suppl. 1996;26:54–71.
Jain K, Dhawan DK. Regulation of biokinetics of (65)Zn by curcumin and zinc in experimentally induced colon carcinogenesis in rats. Cancer Biother Radiopharm. 2014;29:310–6.
Bhandarkar SS, Arbiser JL. Curcumin as an inhibitor of angiogenesis. Adv Exp Med Biol. 2007;595:185–95.
Mohankumar K, Sridharan S, Pajaniradje S, Singh VK, Ronsard L, Banerjea AC, et al. BDMC-A, an analog of curcumin, inhibits markers of invasion, angiogenesis, and metastasis in breast cancer cells via NF-kappaB pathway--a comparative study with curcumin. Biomed Pharmacother. 2015;74:178–86.
James MI, Iwuji C, Irving G, Karmokar A, Higgins JA, Griffin-Teal N, et al. Curcumin inhibits cancer stem cell phenotypes in ex vivo models of colorectal liver metastases, and is clinically safe and tolerable in combination with FOLFOX chemotherapy. Cancer Lett. 2015;364:135–41.
Carroll RE, Benya RV, Turgeon DK, Vareed S, Neuman M, Rodriguez L, et al. Phase IIa clinical trial of curcumin for the prevention of colorectal neoplasia. Cancer Prev Res (Phila). 2011;4:354–64.
Cheng AL, Hsu CH, Lin JK, Hsu MM, Ho YF, Shen TS, et al. Phase I clinical trial of curcumin, a chemopreventive agent, in patients with high-risk or pre-malignant lesions. Anticancer Res. 2001;21:2895–900.
Dhillon N, Aggarwal BB, Newman RA, Wolff RA, Kunnumakkara AB, Abbruzzese JL, et al. Phase II trial of curcumin in patients with advanced pancreatic cancer. Clin Cancer Res. 2008;14:4491–9.
Golombick T, Diamond TH, Badmaev V, Manoharan A, Ramakrishna R. The potential role of curcumin in patients with monoclonal gammopathy of undefined significance—its effect on paraproteinemia and the urinary N-telopeptide of type I collagen bone turnover marker. Clin Cancer Res. 2009;15:5917–22.
Panahi Y, Saadat A, Beiraghdar F, Sahebkar A. Adjuvant therapy with bioavailability-boosted curcuminoids suppresses systemic inflammation and improves quality of life in patients with solid tumors: a randomized double-blind placebo-controlled trial. Phytother Res. 2014;28:1461–7.
Ryan JL, Heckler CE, Ling M, Katz A, Williams JP, Pentland AP, et al. Curcumin for radiation dermatitis: a randomized, double-blind, placebo-controlled clinical trial of thirty breast cancer patients. Radiat Res. 2013;180:34–43.
Irving GR, Iwuji CO, Morgan B, Berry DP, Steward WP, Thomas A, et al. Combining curcumin (C3-complex, Sabinsa) with standard care FOLFOX chemotherapy in patients with inoperable colorectal cancer (CUFOX): study protocol for a randomised control trial. Trials. 2015;16:110.
Huang SS, Tsai MC, Chih CL, Hung LM, Tsai SK. Resveratrol reduction of infarct size in Long-Evans rats subjected to focal cerebral ischemia. Life Sci. 2001;69:1057–65.
Sebastia N, Montoro A, Manes J, Soriano JM. A preliminary study of presence of resveratrol in skins and pulps of European and Japanese plum cultivars. J Sci Food Agric. 2012;92:3091–4.
Sun AY, Wang Q, Simonyi A, Sun GY. Resveratrol as a therapeutic agent for neurodegenerative diseases. Mol Neurobiol. 2010;41:375–83.
Sonnett TE, Levien TL, Gates BJ, Robinson JD, Campbell RK. Diabetes mellitus, inflammation, obesity: proposed treatment pathways for current and future therapies. Ann Pharmacother. 2010;44:701–11.
Speakman JR, Mitchell SE. Caloric restriction. Mol Asp Med. 2011;32:159–221.
Malhotra A, Nair P, Dhawan DK. Modulatory effects of curcumin and resveratrol on lung carcinogenesis in mice. Phytother Res. 2010;24:1271–7.
Malhotra A, Nair P, Dhawan DK. Curcumin and resveratrol in combination modulates benzo(a)pyrene-induced genotoxicity during lung carcinogenesis. Hum Exp Toxicol. 2012;31:1199–206.
Borriello A, Bencivenga D, Caldarelli I, Tramontano A, Borgia A, Zappia V, et al. Resveratrol: from basic studies to bedside. Cancer Treat Res. 2014;159:167–84.
Brakenhielm E, Cao R, Cao Y. Suppression of angiogenesis, tumor growth, and wound healing by resveratrol, a natural compound in red wine and grapes. FASEB J. 2001;15:1798–800.
Howells LM, Berry DP, Elliott PJ, Jacobson EW, Hoffmann E, Hegarty B, et al. Phase I randomized, double-blind pilot study of micronized resveratrol (SRT501) in patients with hepatic metastases—safety, pharmacokinetics, and pharmacodynamics. Cancer Prev Res (Phila). 2011;4:1419–25.
Ji Q, Liu X, Fu X, Zhang L, Sui H, Zhou L, et al. Resveratrol inhibits invasion and metastasis of colorectal cancer cells via MALAT1 mediated Wnt/beta-catenin signal pathway. PLoS One. 2013;8:e78700.
Zhu W, Qin W, Zhang K, Rottinghaus GE, Chen YC, Kliethermes B, et al. Trans-resveratrol alters mammary promoter hypermethylation in women at increased risk for breast cancer. Nutr Cancer. 2012;64:393–400.
Mueller MS, Runyambo N, Wagner I, Borrmann S, Dietz K, Heide L. Randomized controlled trial of a traditional preparation of Artemisia annua L. (Annual Wormwood) in the treatment of malaria. Trans R Soc Trop Med Hyg. 2004;98:318–21.
Singh NP, Lai HC. Artemisinin induces apoptosis in human cancer cells. Anticancer Res. 2004;24:2277–80.
Anfosso L, Efferth T, Albini A, Pfeffer U. Microarray expression profiles of angiogenesis-related genes predict tumor cell response to artemisinins. Pharmacogenomics J. 2006;6:269–78.
Konig M, von Hagens C, Hoth S, Baumann I, Walter-Sack I, Edler L, et al. Investigation of ototoxicity of artesunate as add-on therapy in patients with metastatic or locally advanced breast cancer: new audiological results from a prospective, open, uncontrolled, monocentric phase I study. Cancer Chemother Pharmacol. 2016;77:413–27.
Zhang ZY, Yu SQ, Miao LY, Huang XY, Zhang XP, Zhu YP, et al. Artesunate combined with vinorelbine plus cisplatin in treatment of advanced non-small cell lung cancer: a randomized controlled trial. Zhong Xi Yi Jie He Xue Bao. 2008;6:134–8.
Jansen FH, Adoubi IJCK, DE Cnodder T, Jansen N, Tschulakow A, et al. First study of oral Artenimol-R in advanced cervical cancer: clinical benefit, tolerability and tumor markers. Anticancer Res. 2011;31:4417–22.
Saraswati S, Pandey M, Mathur R, Agrawal SS. Boswellic acid inhibits inflammatory angiogenesis in a murine sponge model. Microvasc Res. 2011;82:263–8.
Takada Y, Ichikawa H, Badmaev V, Aggarwal BB. Acetyl-11-keto-beta-boswellic acid potentiates apoptosis, inhibits invasion, and abolishes osteoclastogenesis by suppressing NF-kappa B and NF-kappa B-regulated gene expression. J Immunol. 2006;176:3127–40.
Catanzaro D, Rancan S, Orso G, Dall’Acqua S, Brun P, Giron MC, et al. Boswellia serrata preserves intestinal epithelial barrier from oxidative and inflammatory damage. PLoS One. 2015;10:e0125375.
Pang X, Yi Z, Zhang X, Sung B, Qu W, Lian X, et al. Acetyl-11-keto-beta-boswellic acid inhibits prostate tumor growth by suppressing vascular endothelial growth factor receptor 2-mediated angiogenesis. Cancer Res. 2009;69:5893–900.
Kunnumakkara AB, Nair AS, Sung B, Pandey MK, Aggarwal BB. Boswellic acid blocks signal transducers and activators of transcription 3 signaling, proliferation, and survival of multiple myeloma via the protein tyrosine phosphatase SHP-1. Mol Cancer Res. 2009;7:118–28.
Lulli M, Cammalleri M, Fornaciari I, Casini G, Dal Monte M. Acetyl-11-keto-beta-boswellic acid reduces retinal angiogenesis in a mouse model of oxygen-induced retinopathy. Exp Eye Res. 2015;135:67–80.
Kirste S, Treier M, Wehrle SJ, Becker G, Abdel-Tawab M, Gerbeth K, et al. Boswellia serrata acts on cerebral edema in patients irradiated for brain tumors: a prospective, randomized, placebo-controlled, double-blind pilot trial. Cancer. 2011;117:3788–95.
Calzavara-Pinton P, Zane C, Facchinetti E, Capezzera R, Pedretti A. Topical boswellic acids for treatment of photoaged skin. Dermatol Ther. 2010;23(Suppl 1):S28–32.
Togni S, Maramaldi G, Bonetta A, Giacomelli L, Di Pierro F. Clinical evaluation of safety and efficacy of Boswellia-based cream for prevention of adjuvant radiotherapy skin damage in mammary carcinoma: a randomized placebo controlled trial. Eur Rev Med Pharmacol Sci. 2015;19:1338–44.
Trompezinski S, Bonneville M, Pernet I, Denis A, Schmitt D, Viac J. Gingko biloba extract reduces VEGF and CXCL-8/IL-8 levels in keratinocytes with cumulative effect with epigallocatechin-3-gallate. Arch Dermatol Res. 2010;302:183–9.
Sun BL, Hu DM, Yuan H, Ye WJ, Wang XC, Xia ZL, et al. Extract of Ginkgo biloba promotes the expression of VEGF following subarachnoid hemorrhage in rats. Int J Neurosci. 2009;119:995–1005.
Koltermann A, Liebl J, Furst R, Ammer H, Vollmar AM, Zahler S. Ginkgo biloba Extract EGb 761 exerts anti-angiogenic effects via activation of tyrosine phosphatases. J Cell Mol Med. 2009;13:2122–30.
Tilley C, Deep G, Agarwal C, Wempe MF, Biedermann D, Valentova K, et al. Silibinin and its 2,3-dehydro-derivative inhibit basal cell carcinoma growth via suppression of mitogenic signaling and transcription factors activation. Mol Carcinog. 2016;55:3–14.
Kim S, Jeon M, Lee J, Han J, Oh SJ, Jung T, et al. Induction of fibronectin in response to epidermal growth factor is suppressed by silibinin through the inhibition of STAT3 in triple negative breast cancer cells. Oncol Rep. 2014;32:2230–6.
Kil WH, Kim SM, Lee JE, Park KS, Nam SJ. Anticancer effect of silibinin on the xenograft model using MDA-MB-468 breast cancer cells. Ann Surg Treat Res. 2014;87:167–73.
Harmsma M, Gromme M, Ummelen M, Dignef W, Tusenius KJ, Ramaekers FC. Differential effects of Viscum album extract IscadorQu on cell cycle progression and apoptosis in cancer cells. Int J Oncol. 2004;25:1521–9.
Park WB, Lyu SY, Kim JH, Choi SH, Chung HK, Ahn SH, et al. Inhibition of tumor growth and metastasis by Korean mistletoe lectin is associated with apoptosis and antiangiogenesis. Cancer Biother Radiopharm. 2001;16:439–47.
Miocinovic R, McCabe NP, Keck RW, Jankun J, Hampton JA, Selman SH. In vivo and in vitro effect of baicalein on human prostate cancer cells. Int J Oncol. 2005;26:241–6.
Acknowledgments
Authors are thankful to University Grant Commision (UGC), New Delhi, India for providing financial assistance in the form of Dr. DSK Postdoctoral fellowship scheme.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Rights and permissions
About this article
Cite this article
Kumar, M., Dhatwalia, S.K. & Dhawan, D.K. Role of angiogenic factors of herbal origin in regulation of molecular pathways that control tumor angiogenesis. Tumor Biol. 37, 14341–14354 (2016). https://doi.org/10.1007/s13277-016-5330-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-016-5330-5