Abstract
With its growth characteristic and chemoresistance, glioblastoma is the most deadly brain tumor. Twenty-five core genes that influence the chemosensitivity of glioblastoma were screened in our previous experiments, and Id2, the inhibitor of DNA binding 2, an oncogene encoding a helix–loop–helix protein, was identified. The elevated expression levels of Id2 have been reported in several malignancies. The aim of this study is to investigate the effects of Id2 expression on the chemosensitivity of glioma cells. In this study, Id2 expression was investigated in a malignant glioma cell line. Then, we silenced the expression of Id2 with the highly specific posttranscriptional suppression of RNA interference (RNAi) in U87 cells. The changes in response to antitumor agents Me-CCNU, VM26, and TMZ were evaluated by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Apoptosis was measured using an annexin V–fluorescein isothiocyanate (FITC) apoptosis detection kit. The relationship between Id2 expression and caspase 3 was tested by RT-PCR and Western blot. This study demonstrated that Id2 was significantly upregulated in glioma tissues, and Id2 correlated well with the advancement of glioma grade and a worse prognosis in response to temozolomide treatment. The RNAi-mediated decrease of Id2 expression enhanced chemosensitivity to Me-CCNU, VM26, and TMZ in the U87 cell line. We further discovered that silencing of Id2 expression could promote apoptosis of glioblastoma cells, which could be attributed to the fact that Id2 affects tumor cell chemosensitivity. Downregulation of the Id2 gene by RNAi could increase the chemosensitivity of glioblastoma cells. Id2 could be a good molecular target for glioblastoma gene therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Glioblastoma multiforme (GBM) is the most common, aggressive primary brain tumor in adults. Because of its diffuse, infiltrative growth and abundant angiogenesis, it is difficult to achieve complete surgical resection in GBM. Even with the recent advances in neuroimaging, neurosurgical stereotactic navigation systems, and targeted radiotherapy, recurrence is still very common. Postdiagnosis survival is 1 year on average because of the inevitable relapse and uncontrollable progression [1, 2]. Chemotherapy is becoming an important adjuvant treatment for GBM patients to kill residual tumor cells.
Semustine (methyl-CCNU, Me-CCNU), teniposide (VM26), and temozolomide (TMZ) are antineoplastic drugs commonly used for the treatment of brain tumors. Me-CCNU, known as an alkylating agent, could cross the blood–brain barrier easily due to its lipophilic character. VM26 is a semisynthetic podophyllotoxin derivative and a topoisomerase II inhibitor, which has been used frequently in combination with Me-CCNU. The combination chemotherapy has demonstrated a survival benefit for patients with malignant glioma [3]. TMZ is also a cytotoxic alkylating agent and has been increasingly used in the treatment of GBM.
However, the benefit of using these agents is rather limited. The survival time of patients with GBM is only prolonged for 2 months with the addition of chemotherapy other than radiotherapy. These chemicals also exhibit substantial hematopoietic, pulmonary, and renal toxicity, which further limits their therapeutic utility. In addition, intrinsic or acquired drug resistance and tumor heterogeneity are responsible for the poor response to chemotherapy in malignant gliomas. Recent researches on exploring the molecular abnormalities of GBM have discovered novel therapeutic methods targeting the key factors critical for cancer initiation and progression.
In our previous experiments, the microarray data was analyzed and 25 core genes that influenced the chemosensitivity of GBM to Me-CCNU were obtained [4]. Id2 was widely expressed in gliomas.
In this study, we demonstrated that the expression of Id2 in primary glioma was upregulated and correlated with glioma grade and outcome of TMZ treatment. We further inhibited Id2 expression with RNA interference (RNAi) and tested its roles in antineoplastic drug susceptibility. These results indicated that targeting Id2 might provide a promising therapeutic approach to sensitizing GBM cells to chemotherapeutic agents.
Material and methods
Cell culture and reagents
U87, U251, and SHG44 human GBM cell lines (from the Institute of Biochemistry and Cell Biology, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences) were cultured in DMEM medium containing 10 % fetal bovine serum (HyClone, UT, USA) at 37 °C in a humidified atmosphere with 5 % CO2. Human astrocytes-cerebellar (HAc) astroglial cells were used as controls.
RNA interference
Small interfering RNA (siRNA) was designed by a siRNA Target Finder program (Ambion, TX, USA). The sequence was 5′-CGTCGCCGTCCAGCTCGACCAG-3′. The oligonucleotides were inserted into the pGCSIL-GFP lentiviral vector (Invitrogen, USA) according to the manufacturer’s protocol. The infection efficiency was evaluated and the infection rate was high for the U87 cell line. As a negative control, a scrambled sequence of siRNA was utilized. The levels of Id2 protein inhibition were determined by Western blotting.
Western blot and immunohistochemistry
Tissue section lysates or proteins were prepared from specimens and cell lines, respectively. Proteins were separated on SDS-PAGE and transferred onto PVDF membranes (PALL, NY, USA) by electroblotting. Anti-Id2 (CalBioreagents, M214) and β-actin antibody (Sigma-Aldrich, Shanghai) were incubated overnight at 4 °C, and the secondary antibody was incubated for 30 min at room temperature in PBS-T containing 2 % BSA. Signals were visualized and quantified by ImageJ software (NIH, USA).
Paraffin-embedded tissues were sectioned 4 μm thick and mounted on poly-d-lysine-coated glass slides used for immunohistochemistry as described. Polyclonal antibodies against Id2 together with an ABC Kit (Maixin Biotech, China) were used to detect Id2 proteins. The nuclei were stained with hematoxylin. The images were collected with a light microscope (Olympus) equipped with a DSC-T50 camera (Sony, Japan).
MTT assay
To determine whether Id2 RNAi affected chemosensitivity of U87 cells, we performed 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. U87 glioma cells were plated in 96-well plates at a density of 104 cells/well. The cells were treated with various concentrations of Me-CCNU, VM26, and TMZ (Sigma, USA) for 72 h. Cells were allowed to attach for 24 h and then transfected with 50 nM siRNAs. After the appropriate time interval, 0.5 mg/ml of MTT reagent (Sigma, USA) was added to each well. After 4-h incubation at 37 °C, the supernatant was aspirated gently and formazan crystals were dissolved in 100 μl of DMSO. The absorbance of each well was measured with an ELISA plate reader (Bio-Rad, USA) at 570-nm wavelength at 0, 24, 48, and 72 h after treatment, respectively.
Apoptosis analysis
Apoptosis was measured using an annexin V–fluorescein isothiocyanate (FITC) apoptosis detection kit (Bender Med System, CA). Cells cultured in 6-cm dishes were trypsinized, washed, and stained with FITC-conjugated anti-annexin V antibody in darkness for 15 min at room temperature and then analyzed with a flow cytometer (FACSCalibur, BD Biosciences).
Real-time PCR
Total RNA was isolated with TRIzol Reagent (Invitrogen) following the manufacturer’s instructions. The first-strand complementary DNA (cDNA) was synthesized with a cDNA synthesis kit (Promega, WI, USA). The gene expression levels were analyzed by quantitative real-time PCR conducted on an ABI 7500 real-time PCR system (Applied Biosystems, CA, USA). Id2 primer sequences were 5′-GACCCGATGAGCCTGCTATAC-3′ and 5′-AATAGTGGGATGCGAGTCCAG-3′. After an initial incubation for 2 min at 50 °C, cDNA was denatured at 95 °C for 10 min, followed by 45 cycles of PCR (95 °C, 15 s; 60 °C, 30 s), and melting curve (95 °C, 60 s; 55 °C, 60 s). The relative gene expression level was calculated by the 2−ΔΔCt formula. All results were obtained from at least three independent experiments.
Patients and samples
All 224 patients were diagnosed and underwent surgical treatment in the Department of Neurosurgery, Changzheng Hospital (Secondary Military Medical University, Shanghai, China) between 2007 and 2011. The selection criteria of this study were as follows: (i) the subject had a primary diagnosis of astrocytoma and no history of other tumors and (ii) the subject had complete clinical data, including age, gender, clinical manifestations, extent of resection, histological type, pathological grade, and adjuvant therapy. All human glioma tissues were also obtained from the Changzheng Hospital in compliance with the laws and institutional guidelines as approved by the Hospital Ethics Committee. Gliomas were graded by the Pathology Department of Changzheng Hospital according to the World Health Organization grading system. Eight human normal brain tissues (mostly from the cortex) were obtained from patients with physical injuries of the brain and were authorized by patients for further experiments.
Follow-up
The follow-up was carried out in all patients, with survival time being censored in January 2012. The follow-up was conducted every 6 months by telephone, and the last time of the follow-up to contact with patients was July 2012. Other treatment options, including adjuvant chemotherapy and radiotherapy, were fully discussed with the patients. Overall survival was defined as the period from the time of diagnosis to the time of death.
Statistical analysis
Statistical analysis was performed using SPSS software package (version 11.5; SPSS, Inc., Chicago, IL). The continuous data were summarized as means ± SD. Statistical analysis between two groups was performed by Student’s t test, and the comparison between three or more groups was performed by ANOVA analysis followed by Dunnett’s t test. One-way ANOVA or the Kruskal–Wallis test was used to compare the data between the groups. P values less than 0.05 were considered to be statistically significant.
Results
Expression of Id2 in GBM cell lines
We estimated the protein expression levels of Id2 in malignant glioma cells including U87, U251, and SHG44 and normal astrocytes (Fig. 1a). The results of the Western blot indicated that Id2 protein expression was increased in malignant glioma cells compared with normal astrocytes (1.25 ± 0.46 vs 0.49 ± 0.17; P ≤ 0.05) (Fig. 1b). Thus, Id2 expression is correlated with the proliferative activity of glioblastoma cells.
Expression of Id2 protein in human astrocytes, U87, U251, and SHG44 human GBM cell lines. a Western blotting showed protein expression of Id2 was higher in malignant glioma cells including U87, U251, and SHG44 cells than in normal astrocytes. b Id2 gene expression in human astrocytes and malignant gliomas cells. HAC human astrocyte cells, MGC malignant glioma cells. (*P < 0.05 vs HAC). c The expression level of Id2 proteins in different grades of glioma tissues was determined by Western blot. d The statistical results of Id2 protein expression measured by immunoblot in the normal brain (Normal, n = 8) and glioma tissues (Tumor, n = 65). ***P < 0.01 vs normal. e The statistical results of Id2 mRNA expression measured by real-time PCR in the normal brain (Normal, n = 8) and glioma tissues (Tumor, n = 65). ***P < 0.01 vs normal. f The representative image of Id2 immunostaining in normal brain and glioma tissues
Significant upregulation of Id2 in glioma tissues
To further explore the association between Id2 expression and gliomas grade, we then investigated the specimens obtained from patients diagnosed with gliomas. Expression of messenger RNA (mRNA) and protein was evaluated in 65 glioma tissues and 8 normal brain tissues by real-time PCR and Western blot, respectively. Id2 mRNA level was specifically increased in glioma tissues compared with that in normal brain tissues (3.89 ± 0.51 vs 1.40 ± 0.29; P < 0.01) (Fig. 1e). Consistently, Id2 protein level was also upregulated in glioma tissues determined by immunoblotting compared with that in normal brains (1.68 ± 0.82 vs 0.26 ± 0.07; P < 0.01) (Fig. 1c, d). To see if Id2 is expressed in tumor cells, immunohistochemistry staining of Id2 demonstrated that Id2 was overexpressed in glioma cells, but not stromal cells of tumor tissues, and the expression level of Id2 was low in neuron and normal glia cells of normal brain tissues (Fig. 1f).
The level of Id2 expression is positively correlated with glioma grades and negatively correlated with disease outcome
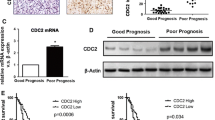
To determine the contribution of Id2 to glioma progression, we collected 65 glioma samples of different grades. Immunostaining showed that in low grades of glioma (WHO I–II) (n = 20), Id2 was increased moderately, while in high grades of glioma (WHO III–IV), Id2 was highly upregulated (Fig. 2a) (n = 45).
The level of Id2 expression in different grades of glioma and its relation with clinical outcome. a The representative image of Id2 immunostaining in the low-grade (left, a case of WHO I grade) and high-grade (right, a case of WHO IV grade) glioma tissues. b The statistical results of Id2 mRNA expression measured by real-time PCR in the normal brain (Normal, n = 8), low-grade glioma tissues (Low grade, n = 20), and high-grade glioma tissues (High grade, n = 45). ***P < 0.01 vs normal. c The statistical results of Id2 protein expression in the normal brain (Normal, n = 8), low-grade glioma tissues (Low grade, n = 20), and high-grade glioma tissues (High grade, n = 45). ***P < 0.01 vs normal. d Kaplan–Meier curve of overall survival in 95 grade IV patients with low (blue curve) and high (green curve) Id2 expression. e Kaplan–Meier curve of overall survival in 64 grade IV patients receiving TMZ treatment with low (blue curve) and high (green curve) Id2 expression. All results are expressed as mean ± SD from at least three independent experiments
The expression change of Id2 mRNA was also studied using real-time PCR, and a similar upregulation pattern was observed in high-grade glioma (WHO III–IV) (n = 45) and low-grade glioma (WHO I–II) (n = 20) in comparison with normal brain tissues. Consistently, the increasing Id2 mRNA level was closely associated with tumor grades (normal tissue, 0.97 ± 0.37; low-grade, 2.81 ± 0.91; high-grade, 4.69 ± 1.23, respectively) (Fig. 2b), which is also true for Id2 protein level (normal tissue, 0.30 ± 0.19; low-grade, 1.09 ± 0.29; high-grade, 2.36 ± 0.88, respectively) (Fig. 2c).
Then, patient data were analyzed to determine the roles of Id2 in general prognosis, tumor responses to TMZ-treatment, and disease outcome. A total of 95 glioma patients of WHO IV grade who underwent glioma surgical resection from December 2007 to December 2011 were included in this study. As shown in Fig. 2d, patients with low protein expression level of Id2 based on immunohistochemistry had a prolonged overall survival time compared with those with high expression of Id2. Within this cohort, we further analyzed Id2 expression in 64 patients who postoperatively received TMZ treatment. We found that high Id2 expression was associated with a shorter overall survival time in patients receiving TMZ treatment, suggesting that high expression of Id2 could confer resistance to TMZ treatment in glioma cells (Fig. 2e).
Effects of Id2 downregulation on the chemosensitivity of GBM cells
In order to downregulate the expression of Id2, Id2-specific siRNA was utilized. Simultaneously, a scrambled siRNA was used as a negative control. To verify the silencing efficiency, Id2 protein levels were evaluated by Western blot. Compared with the untreated cells, Id2 siRNA significantly decreased the expression of Id2 protein in U87 cells (P < 0.05), while scrambled siRNA did not affect Id2 expression (P > 0.05), which confirmed the specificity of Id2 knockdown by siRNA (Fig. 3a, b). We then investigated whether the inhibition of Id2 by siRNA affected GBM cell lines’ sensitivity to anticancer drugs Me-CCNU, VM26, and TMZ. MTT assay revealed that the cell proliferation was significantly inhibited in the Id2 siRNA group in comparison with the scrambled siRNA group and U87 cells after treatment of anticancer drugs (Fig. 3c–e). The inhibition rates of Id2 siRNA were significantly higher than those of the control group treated with Me-CCNU, VM26, and TMZ, respectively (Fig. 3f).
Inhibition of Id2 by siRNA in human GBM cell line U87. a Western blotting showed that siRNA targeting Id2 downregulates Id2 protein expression significantly. 1. U87 group; 2. control group (si-ctl); 3. Id2 RNAi group (si-Id2). b Inhibition rate of Id2 by siRNA in human GBM cell line U87 (*P < 0.05). c The growth curve of U87 cells and U87 cells with Id2 silenced (si-Id2) and its control group (si-ctl) after treatment with Me-CCNU. d The growth curve of U87 cells and U87 cells with Id2 silenced (si-Id2) and its control group (si-ctl) after treatment with VM26. e The growth curve of U87 cells and U87 cells with Id2 silenced (si-Id2) and its control group (si-ctl) after treatment with TMZ. The x- and y-axes show time and cell number, respectively. MTT assay showed that the cell proliferation was significantly inhibited in Id2 siRNA group (triangle) in comparison with the control group (square) and U87 cells (diamond). f Inhibition rate of U87 cells with Id2 silenced (si-Id2) and its control group (si-ctl) after treatment with Me-CCNU, VM26, and TMZ, respectively (*P < 0.05)
Effects of Id2 downregulation on the apoptosis of GBM cells
To further explore the function of Id2 gene, we detected the apoptosis levels of U87 cells with Id2 knockdown. Compared with control cells, the results of annexin V–FITC apoptosis detection demonstrated that Id2 downregulation in U87 cells increased the percentage of cells in early-stage apoptosis from 7.86 ± 0.53 to 13.09 ± 1.22 % (P < 0.05) (Fig. 4a–d).
Apoptosis induced after Id2 RNAi in the U87 cell line. a Apoptosis of U87 cells, b apoptosis of control group cells (si-ctl), and c apoptosis of U87 cells after Id2 RNAi (si-Id2). The results of flow cytometry showed that compared with control group cells and U87 cells, the downregulation of the Id2 gene increased the apoptosis of U87 cells. d Apoptosis rate of U87 cells, control group, and Id2 siRNA group (*P < 0.05). e Expression of caspase 3 mRNA in the U87 group, control group, and si-Id2 group. f Expression of caspase 3 protein in the U87 group, control group, and Id2-knockdown group. The results of real-time PCR and Western blotting showed that the protein expression of caspase 3 had a negative correlation with Id2 expression
Effects of Id2 downregulation on the expression of caspase 3
Apoptosis is characterized by as many kinds of morphological changes and biochemical changes. Caspase family proteins like caspase 3, caspase 6, and caspase 7 are involved in this important biochemical change of apoptosis. Caspase activates catabolic hydrolases that degrade most of the macromolecules of the cell [6]. So we analyzed the effects of downregulation of Id2 on the expression of caspase 3 by real-time PCR and Western blot. Real-time PCR analysis showed that compared with the control group, the level of caspase 3 mRNA expression was significantly increased after Id2 knockdown (0.276 ± 0.610 vs 0.075 ± 0.024; P < 0.05) (Fig. 4e). And the protein level of caspase 3 was increased after Id2 knockdown by Id2 RNAi (0.94 ± 0.10 vs 0.56 ± 0.06; P < 0.05) (Fig. 4f).
Discussion
Id proteins belong to the HLH protein family, which were firstly identified by Benezra et al. in 1990 [5]. The Id2 gene is located in chromosome 2p25. The protein consists of 134 amino acids and lacks the basic DNA-binding domain. Id2 can function in a dominant-negative manner by binding and sequestering basic HLH (bHLH) transcription factors, thereby blocking the binding of bHLH proteins to DNA [6, 7]. Through binding bHLH proteins to inhibit DNA binding, Id2 regulates diverse cellular processes, including cell growth, differentiation, and apoptosis. And Id2 is frequently activated during tumorigenesis [8–10].
Accumulating evidence indicated that overexpression of Id2 promotes cell proliferation in multiple types of tumors, including ovarian cancer [11], pancreatic cancer [12], and neuroblastoma [13]. Several studies revealed that Id2 is a direct target of the N-myc transcription factor. The increased N-myc may stimulate the expression of Id2 in neuroblastoma, and abundant Id2 proteins combine unphosphorylated Rb, which inhibits cell differentiation and promotes cell proliferation and tumorigenesis [14].
The mean survival time of GBM patients is 1 year. Due to neovascularization, proliferation, and invasion, it is almost impossible to eliminate GBM cells completely by surgery. Adjuvant chemotherapy could increase 6 % survival rate in a year and prolong for 2 months in median survival time. One of the reasons for the limited improvement is the insensitivity or acquired resistance of glioma cells to anticancer drugs. We chose the anticancer drugs Me-CCNU, VM26, and TMZ for the reason that postsurgical patients in our department were treated with the combination of Me-CCNU and VM26 chemotherapy plan before 2009, and TMZ was included because of its increasing usage in the treatment of GBM in recent years [15].
In this study, we focused on the roles of Id2 in chemosensitivity of GBM cell lines. We found that Id2 protein expression was significantly elevated in malignant glioma cells compared with astrocyte cells and its expression level increases as tumor grade advances. These results strongly suggested that Id2 is a gene closely associated with glioma formation and progression. And we identified Id2 as a gene differentially expressed in glioma patients that was resistant to TMZ treatment. RNAi technology is a powerful approach to knock down gene expression for studies of gene function, and it has the potential for gene therapy [16]. Our in vitro experiments showed that after Id2 inhibition by siRNA, the chemosensitivity of U87 cells was strengthened.
Apoptosis inhibition is one of the important reasons not only for the growth of many malignant tumor cells but also for their resistance to chemotherapeutic agents [17]. A large number of studies have shown that many chemotherapeutics could induce cancer cells to undergo apoptosis by different pathways [18]. Cao et al. [19] reported knockdown of Id1 and Id2 gene expression induced apoptosis in RIE-1 cells, while overexpression of Id2 attenuated TGF-β-induced apoptosis. In the present experiment, annexin-V and PI staining revealed that apoptotic cells’ death rate increased in cells transfected with Id2 siRNA. Caspase proteins play an important role in apoptosis [20]. Using RT-PCR and Western blot, we found that caspase 3 activity was increased in U87 cells with Id2 downregulation, which suggested that Id2 might downregulate the expression of caspase 3 to inhibit the process of apoptosis. Therefore, downregulation of the Id2 gene by RNAi could enhance drug sensitivity through upregulation of the apoptotic pathway. The results will be tested in our further experiment in vivo. Furthermore, we believe that Id2 would be a good molecular target for glioblastoma gene therapy.
Abbreviations
- Id2:
-
Inhibitor of DNA binding 2, dominant negative helix–loop–helix protein
- GBM:
-
Glioblastoma
- RNAi:
-
RNA interference
- siRNA:
-
Small interfering RNA
- Me-CCNU, methyl-CCNU:
-
Semustine
- VM26:
-
Teniposide
- TMZ:
-
Temozolomide
- RT-PCR:
-
Reverse transcription-polymerase chain reaction
- HAc:
-
Human astrocytes-cerebellar
References
Zhao J, He H, Zhou K, Ren Y, Shi Z, Wu Z, et al. Neuronal transcription factors induce conversion of human glioma cells to neurons and inhibit tumorigenesis. PLoS One. 2012;7:e41506.
Tao BB, He H, Shi XH, Wang CL, Li WQ, Li B, et al. Upregulation of USP2a and FASN in gliomas correlates strongly with glioma grade. J Clin Neurosci. 2013;20:717–20.
Galanis E, Buckner J. Chemotherapy for high-grade gliomas. Br J Cancer. 2000;82:1371–80.
Zhao Z, Liu Y, He H, Chen X, Chen J, Lu YC. Candidate genes influencing sensitivity and resistance of human glioblastoma to semustine. Brain Res Bull. 2011;86:189–94.
Benezra R, Davis RL, Lockshon D, Turner DL, Weintraub H. The protein Id: a negative regulator of helix-loop-helix DNA binding proteins. Cell. 1990;61:49–59.
Mariño G, Niso-Santano M, Baehrecke EH, Kroemer G. Self-consumption: the interplay of autophagy and apoptosis. Nat Rev Mol Cell Biol. 2014;15:81–94.
Norton JD, Deed RW, Craggs G, Sablitzky F. Id helix-loop-helix proteins in cell growth and differentiation. Trends Cell Biol. 1998;8:58–65.
Yokota Y, Mori S. Role of Id family proteins in growth control. J Cell Physiol. 2002;190:21–8.
Trabosh VA, Divito KA, D Aguda B, Simbulan-Rosenthal CM, Rosenthal DS. Sequestration of E12/E47 and suppression of p27KIP1 play a role in Id2-induced proliferation and tumorigenesis. Carcinogenesis. 2009;30:1252–9.
Lasorella A, Rothschild G, Yokota Y, Russell RG, Iavarone A. Id2 mediates tumor initiation, proliferation, and angiogenesis in Rb mutant mice. Mol Cell Biol. 2005;25:3563–74.
Meng Y, Gu C, Wu Z, Zhao Y, Si Y, Fu X, et al. Id2 promotes the invasive growth of MCF-7 and SKOV-3 cells by a novel mechanism independent of dimerization to basic helix-loop-helix factors. BMC Cancer. 2009;9:75.
Coppe JP, Itahana Y, Moore DH, Bennington JL, Desprez PY. Id1 and Id2 proteins as molecular markers for human prostate cancer progression. Clin Cancer Res. 2004;10:2044–51.
Han W, Wu Z, Zhao Y, Meng Y, Si Y, Yang J, et al. FHL2 interacts with and acts as a functional repressor of Id2 in human neuroblastoma cells. Nucleic Acids Res. 2009;37:3996–4009.
Stallone G, Infante B, Pontrelli P, Ranieri E, Loverre A, Schena A, et al. ID2-VEGF-related pathways in the pathogenesis of Kaposi’s sarcoma: a link disrupted by rapamycin. Am J Transplant. 2009;9:558–66.
Spiegel BM, Esrailian E, Laine L, Chamberlain MC. Clinical impact of adjuvant chemotherapy in glioblastoma multiforme: a meta-analysis. CNS Drugs. 2007;21:775–87.
Wall NR, Shi Y. Small RNA: can RNA interference be exploited for therapy? Lancet. 2003;362:1401–3.
Pommier Y, Sordet O, Antony S, Hayward RL, Kohn KW. Apoptosis defects and chemotherapy resistance: molecular interaction maps and networks. Oncogene. 2004;23:2934–49.
Johnstone RW, Ruefli AA, Lowe SW. Apoptosis: a link between cancer genetics and chemotherapy. Cell. 2002;108:153–64.
Cao Y, Liu X, Zhang W, Deng X, Zhang H, Liu Y, et al. TGF-beta repression of Id2 induces apoptosis in gut epithelial cells. Oncogene. 2009;28:1089–98.
Cohen GM. Caspases: the executioners of apoptosis. Biochem J. 1997;326:1–16.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 81302187 and No. 30930094) and medical technological innovation funding of Nanjing Military Region in 2014 (No. ZD15).
Conflict of interest
None
Author information
Authors and Affiliations
Corresponding authors
Additional information
ZY. Zhao and H. Hua contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhao, Z., He, H., Wang, C. et al. Downregulation of Id2 increases chemosensitivity of glioma. Tumor Biol. 36, 4189–4196 (2015). https://doi.org/10.1007/s13277-015-3055-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3055-5