Abstract
Ubiquitin carboxyl-terminal hydrolase 37 (UCH37) is a member of deubiquitinating enzymes. It can suppress protein degradation through disassembling polyubiquitin from the distal subunit of the chain. The aim of this study was to assess the value of UCH37 in predicting tumor recurrence after curative resection in epithelial ovarian cancer (EOC) patients. In this study, the expression level of UCH37 in 5 paired EOC and normal tissue was tested by Western blot. And the association of UCH37 expression and prognostic value was analyzed in 100 tumor specimens from EOC patients, who underwent curative resection between 2003 and 2011. We found that UCH37 was up-regulated in most of the tumor tissue and high expression of UCH37 was an independent significant predictor associated with the poor outcome and recurrence of EOC (p = 0.0037 and p = 0.0042 in overall and disease-free survival, respectively), especially in the advanced stage of EOC (p = 0.0106 and p = 0.0115 in overall and disease-free survival, respectively), and may become a novel predictor for prognosis of EOC patients after curative resection. Our data suggest for the first time that UCH37 overexpression is associated with advanced tumor progression and poor clinical outcome of EOC patients and may help physicians make informed decisions regarding adjuvant treatment following curative resection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Ovarian cancer is one of the most common gynecologic malignancies worldwide [1]. It has a highly aggressive natural history and a highest mortality rate among all malignant cancers in female reproductive system [2]. Epithelial ovarian cancer (EOC) accounts for more than 80 % among all the pathological types of ovarian cancer. Although the survival rates of patients with early stage EOC are high, most patients are diagnosed at the advanced stages. The prognosis of patients with advanced EOC is still poor even though there has been great improvement on the treatment of EOC, such as surgery, radiotherapy, and chemotherapy [2, 3]. Molecular signatures which can define the prognosis of EOC are crucial and could allow appropriate therapeutic regimens applied in the earlier stage of the disease course.
The ubiquitin-proteasome system is a nonlysosomal route for intracellular protein degradation in eukaryotes [4, 5]. The proteasome recognizes substrate via the multiubiquitin chain and followed by the translocation and degradation of the substrate [6–8]. Ubiquitin carboxyl-terminal hydrolase 37 (UCH37) is a member of deubiquitinating enzymes (DUBs). It can suppress protein degradation through disassembling polyubiquitin from the distal subunit of the chain [9, 10]. It has reported that UCH37 assists in the mental development of mice [11]. Rolen et al. have found that the activity of the C-terminal hydrolases UCH37 is up-regulated in the majority of tumor tissues compared with the adjacent normal tissues [12]. Our previous research has also shown that UCH37 may play an important role on oncogenesis, tumor invasion, and migration in hepatocellular carcinoma and esophageal carcinoma [13–15]. All the evidences suggest that the up-regulation of UCH37 may play an important role in oncogenesis through promoting some proto-oncogenes’ expression and stem cell-like characteristics in the cell. However, the role UCH37 plays in ovarian cancer has not been reported.
In this study, the expression level of UCH37 in 5 paired EOC and normal tissue was tested by Western blot. And the association of UCH37 expression and prognostic value was analyzed in 100 tumor specimens from EOC patients to assess the value of UCH37 in predicting tumor recurrence after curative resection in EOC patients. Our results may help physicians make informed decisions regarding adjuvant treatment following curative resection.
Materials and methods
Patients and specimens
This study was reviewed and approved by the research ethics committee of Zhongshan Hospital, Fudan University. Informed consent was obtained from all of the patients. All specimens were handled in an anonymous way according to the ethical and legal standards. Paraffin blocks were obtained from the 100 patients with EOC who underwent curative resection at The Department of Obstetrics and Gynecology, Shanghai Zhongshan Hospital, Fudan University between 2003 and 2011, while 5 paired frozen tissues used in Western blot analysis were obtained in 2013. All the tissue samples were obtained from the Department of Pathology, Shanghai Zhongshan Hospital, Fudan University.
The inclusion and exclusion criteria of the patients include the following: (a) having a distinctive pathologic diagnosis of EOC, (b) having no anticancer treatment before ovarian resection, (c) having the curative ovarian resection, (d) having routinely formalin-fixed and paraffin-embedded specimens, and (e) the 5 specimens used in Western blot should have bilateral ovariectomy (including the tumor side and the normal side).
Tumor differentiation and staging was established according to the criteria of the 7th edition of FIGO staging system of American Joint Committee on Cancer (AJCC).
Follow-up and treatment for tumor recurrences
Patients had routine physical examination as a life-long follow-up in our hospital after the operation. The nearest follow-up time in this study was on August 20, 2013. The median follow-up was 44 months, ranging from 5 to 126 months. Most of the patients ended from oncological death, but two patients died from cerebral accident or heart failure. Typical imaging tests such as PET-CT and enhanced CT were the bases of the diagnosis of recurrence. Available clinical data, overall survival time, disease-free survival time, and the causes of death were linked in a blinded fashion to the immunohistochemistry (IHC) results ultimately. Patients received further treatment based on the specific condition when they were confirmed recurrence.
Western blot analysis
UCH37 expression in tissue specimens from 5 EOC patients was detected by Western blot. Frozen tissues were ground with liquid nitrogen. The tissues were lysed in lysis buffer and the overall protein extracts were clarified via centrifugation. The tissue-lysated proteins were separated on 12 % SDS-PAGE gel and then electrophoretically transferred to polyvinylidene fluoride membranes (Millipore). The membranes were then blocked with PBS with 0.05 % Tween-20 containing 5 % (w/v) nonfat dry milk at room temperature for 1 h, followed by incubation with the rat anti-UCH37 antibody (Santa Cruz, CA, USA) at 4 °C overnight. After incubating with appropriate secondary antibodies at 1:1,000 dilutions for 1 h, the blots were developed using an enhanced chemiluminescence kit (Beyotime, Beijing, China). Each experiment was repeated at least twice.
Immunohistochemistry
The specimens were fixed in 10 % neutral-buffered formalin and subsequently embedded with paraffin. The paraffin-embedded tissues were cut at 5 μm and then deparaffinized with xylene and rehydrated for further hematoxylin eosin (HE) or 3,3′-diaminobenzidine (DAB) IHC staining. The tissue sections were incubated for 30 min at 25 °C with rat anti-UCH37 antibody (dilution 1: 500, Santa Cruz, CA, USA) and secondary antibody after treating with 10 % normal goat serum for 45 min to block nonspecific binding sites. The specificity of the primary antibody has been validated by our previous studies [13–15]. After washing, peroxidase-labeled polymer and substrate-chromogen were then employed in order to visualize the staining of the interested proteins.
IHC staining was scored by two pathologists respectively without knowing patients’ characteristics. Any discrepancy was solved by consensus review. The score of immunoreactivity was performed by calculating the extent and intensity of cytoplasm staining positivity of cells in a semi-quantitative manner. Score interpretation was mentioned previously [13]. Interpretation scores were as follows: 0, ≤5 % cell positivity or negative staining; +1, 6~20 % cell positivity or mild staining; +2, 21~50 % cell positivity or moderate staining; and +3, ≥50 % cell positivity or intense staining. Total scores were the product of the two. And the final scores greater than 3.5 was considered high expression.
Statistical analyses
The software of SPSS version 19.0 for Windows (SPSS Inc., IL, USA) and GraphPad Prism 5 (GraphPad Software, Inc., CA, USA) were used for statistical analysis. Paired t test was used to compare the expression of UCH37 in five paired tumor and normal tissues in the Western blot result. When the data from different two groups were compared, the Student′s t test was used. The χ 2 test was used to assess the statistical significance of the association between UCH37 expression and clinicopathologic parameters. Kaplan-Meier curves were plotted to assess the effects of UCH37 expression on overall survival (OS) and disease-free survival (DFS). Survival curves were compared using the log-rank test. Cox proportional hazard regression models were used to assess the prognostic significance of UCH37 expression and several clinicopathologic parameters. It was considered statistically significant when p < 0.05.
Results
The high expression of UCH37 in tumor tissues by Western blot
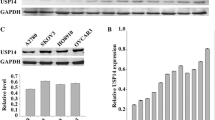
The differences of UCH37 expression in five paired EOC and non-tumorous tissues were performed by Western blot. Our result showed that the expression of UCH37 in tumor tissues is high (Fig. 1a). But because of the repairment of normal ovarian epithelium in women of childbearing age, high expression of UCH37 could also be found in the normal ovarian tissue (Fig. 1a). Although the expression of UCH37 was found higher in tumor tissues than the normal ones in most of tissue pairs, no statistic difference was found in UCH37 expression between the tumor tissue group and the normal tissue group (Fig. 1b).
Expression of UCH37 protein detected in EOC tissue by IHC
The expression and subcellular location of UCH37 was analyzed by IHC. UCH37 protein was detected in both nucleus and cytoplasm in most cancer samples (98 of 100, 98 %). Higher expression of UCH37 was also observed in the lower differentiation of ovarian cancer tissues (Fig. 2).
Expression of UCH37 protein in tissue by immunohistochemistry. a, b H&E staining. c, d Positive expression of UCH37 in epithelial ovarian cancer. e, f Weak staining of UCH37 in epithelial ovarian cancer tissues. g, h Negative and positive control (magnification, ×100 (a, c, e, g, and h) and × 200 (b, d, and f); positive control, esophageal squamous cell carcinoma)
Correlation between UCH37 expression and clinicopathological parameters
The association of UCH37 expression with various clinicopathological parameters of EOC tissues was summarized (Table 1). The expression of UCH37 in tumor was significantly associated with the tumor differentiation (p = 0.046). The EOC tissues with lower differentiation (III) more frequently showed higher UCH37 expression than those with higher differentiation (I~II). However, UCH37 expression was not correlated with age, tumor size, FIGO stage, CA125 expression in cancer tissue, ER expression in cancer tissue, and progesterone receptor (PR) expression in cancer tissue (all p > 0.05).
Survival analysis
The detailed clinical information of 100 EOC patients in high UCH37 expression and low UCH37 expression groups was reviewed to investigate the prognostic implications of UCH37 expression. The expression level of UCH37 in EOC was significantly correlated with the patients’ OS time and DFS time (p = 0.0037 and p = 0.0042, respectively), i.e., the higher level of UCH37 expression was correlated with shorter OS and DFS. The cumulative 3-year OS and DFS was 95.35 and 95.24 %, respectively, in the low UCH37 expression group, whereas it was only 71.23 and 73.65 % (p = 0.001 and 0.002, respectively) in high UCH37 expression group (Fig. 3).
Univariate analysis showed UCH37 expression, P53 expression, EP expression, and PR expression were associated with OS and DFS; while age, tumor differentiation, FIGO stage, and tumor size had no prognostic significance for OS and DFS (Table 2). Furthermore, the multivariate analyses identified the UCH37 expression (p = 0.008 and p = 0.007, respectively) and ER expression (both p = 0.001) as independent prognostic factors for OS and DFS (Table 3).
Interestingly, subgroup analyses according to clinical stage revealed that EOC patients with low clinical stage (I) had no significantly different prognosis between high and low UCH37 expression group (Fig. 4a, b), while patients with advanced stages (II~IV) in high UCH37 expression group demonstrated a significantly worse clinical outcome than those in low UCH37 expression group (Fig. 4c, d). The cumulative 3-year OS and DFS of EOC patients in advanced stages (II~IV) was 75.12 and 75.56 % in the low UCH37 expression group, whereas it was only 41.64 and 38.80 % in high UCH37 expression group (Fig. 4c, d).
Subgroup prognostic analyses according to clinical stage. EOC patients with low clinical stage (I) had no significantly different prognosis between high and low UCH37 expression group (a, b), while patients with advanced stages (II~IV) in high UCH37 expression group demonstrated a significantly worse clinical outcome than those in low UCH37 expression group (c, d)
Discussion
Despite the improvements in clinical treatment strategies, the prognosis of EOC still remains dismal [1, 3, 2]. Molecular signatures which can define the risk of recurrence and metastatic of EOC are crucial because they can allow appropriate therapeutic regimens to be applied earlier in the disease course.
Protein substrates covalently conjugated by ubiquitin are then targeted for the proteasome and degraded into small peptides. The ubiquitin-proteasome system is a nonlysosomal route for intracellular protein degradation in eukaryotes [4]. DUBs could remove Ub from the substrates and mediate degradation, and involve in numerous biological processes [16]. UCH37, a member of DUBs, can suppress protein degradation through disassembling polyubiquitin from the distal subunit of the chain. It has reported that UCH37 assists in the mental development of mice [11]. Our previous research has also shown that UCH37 may play an important role in oncogenesis, tumor invasion, and migration in hepatic carcinoma and esophageal squamous cell carcinoma [13–15].
Through the Western blot analysis, we found a high expression of UCH37 in the EOC tumor tissues. The expression of UCH37 in the tumor tissues was higher than most of the paired normal tissues, but there was no statistic difference between the two groups. The high expression of UCH37 could also be found in the normal ovarian tissue, mainly because of the repairment of normal ovarian epithelium in women of childbearing age. The ovulation, which causes repeated injury and repair process of the ovarian epithelium, upregulates the expression of oncoprotein, such as UCH37, and may be the risk factor of neoplasia. Whether the average level of UCH37 in the normal ovarian epithelium could predict the occurrence of EOC needs further investigations.
According to the IHC result, we found that UCH37 protein was detected in most of the tumor samples (98 of 100, 98 %), and located in both nuclei and cytoplasm, which confirmed our previous research [13, 15]. From the clinical data, we found that the expression of UCH37 was related to the tumor differentiation, which means higher expression of UCH37 that leads lower differentiation. But the expression of UCH37 was not related to the FIGO stage, perhaps because FIGO stage was not the best reflection of tumor malignancy.
Survival analysis showed that the expression level of UCH37 in EOC was significantly related with OS and DFS of the patients. And multivariate analysis showed UCH37 was an independent prognosticator for OS and DFS. All these results indicated that UCH37 was suitable to predict recurrence and prognosis in EOC after operation. Interestingly, subgroup analyses according to clinical stage revealed that EOC patients with advanced stages (II~IV) in high UCH37 expression group demonstrated a significantly worse clinical outcome than those in low UCH37 expression group, while no statistic significance was found between the two groups in the low clinical stage (I). Patients with low clinical stage (I) more easily received complete operation tumor resection without metastatic tumor residue than patients with advanced stages (II~IV). So, the molecular signatures may have less meaning to these kinds of patients. But to those patients with advanced stages (II~IV) of EOC, testing of UCH37 expression level can assess the prognosis and allow appropriate therapeutic regimens to be applied earlier in the disease course.
Although our study and previous study have confirmed that UCH37 does relate with the outcome and recurrence of the carcinoma, the mechanisms on the roles of UCH37 on tumorigenesis and metastasis is still unclear. Hence, the role of UCH37 on tumorigenesis and metastasis deserve further study.
Conclusion
In conclusion, our data suggest for the first time that UCH37 overexpression is associated with advanced tumor progression and poor clinical outcome of EOC patients. UCH37 may help physicians make informed decisions regarding adjuvant treatment following curative resection.
References
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. Cancer J Clin. 2014;64(1):9–29. doi:10.3322/caac.21208.
Wright JD, Shah M, Mathew L, Burke WM, Culhane J, Goldman N, et al. Fertility preservation in young women with epithelial ovarian cancer. Cancer. 2009;115(18):4118–26. doi:10.1002/cncr.24461.
Rota M, Pasquali E, Scotti L, Pelucchi C, Tramacere I, Islami F, et al. Alcohol drinking and epithelial ovarian cancer risk a systematic review and meta-analysis. Gynecol Oncol. 2012;125(3):758–63. doi:10.1016/j.ygyno.2012.03.031.
Hershko A, Ciechanover A. The ubiquitin system. Annu Rev Biochem. 1998;67:425–79. doi:10.1146/annurev.biochem.67.1.425.
Pickart CM. Mechanisms underlying ubiquitination. Annu Rev Biochem. 2001;70:503–33. doi:10.1146/annurev.biochem.70.1.503.
Glickman MH, Ciechanover A. The ubiquitin-proteasome proteolytic pathway: destruction for the sake of construction. Physiol Rev. 2002;82(2):373–428. doi:10.1152/physrev.00027.2001.
Wilkinson KD. Cell biology: unchaining the condemned. Nature. 2002;419(6905):351–3. doi:10.1038/419351a.
Goldberg AL. Protein degradation and protection against misfolded or damaged proteins. Nature. 2003;426(6968):895–9. doi:10.1038/nature02263.
Chung CH, Baek SH. Deubiquitinating enzymes: their diversity and emerging roles. Biochem Biophys Res Commun. 1999;266(3):633–40. doi:10.1006/bbrc.1999.1880.
Chen YJ, Ma YS, Fang Y, Wang Y, Fu D, Shen XZ. Power and promise of ubiquitin carboxyl-terminal hydrolase 37 as a target of cancer therapy. Asian Pacific journal of cancer prevention : APJCP. 2013;14(4):2173–9.
Al-Shami A, Jhaver KG, Vogel P, Wilkins C, Humphries J, Davis JJ, et al. Regulators of the proteasome pathway, Uch37 and Rpn13, play distinct roles in mouse development. PLoS One. 2010;5(10):e13654. doi:10.1371/journal.pone.0013654.
Rolen U, Kobzeva V, Gasparjan N, Ovaa H, Winberg G, Kisseljov F, et al. Activity profiling of deubiquitinating enzymes in cervical carcinoma biopsies and cell lines. Mol Carcinog. 2006;45(4):260–9. doi:10.1002/mc.20177.
Chen Y, Fu D, Xi J, Ji Z, Liu T, Ma Y, et al. Expression and clinical significance of UCH37 in human esophageal squamous cell carcinoma. Dig Dis Sci. 2012;57(9):2310–7. doi:10.1007/s10620-012-2181-9.
Fang Y, Mu J, Ma Y, Ma D, Fu D, Shen X. The interaction between ubiquitin C-terminal hydrolase 37 and glucose-regulated protein 78 in hepatocellular carcinoma. Mol Cell Biochem. 2012;359(1–2):59–66. doi:10.1007/s11010-011-0999-7.
Fang Y, Fu D, Tang W, Cai Y, Ma D, Wang H, et al. Ubiquitin C-terminal Hydrolase 37, a novel predictor for hepatocellular carcinoma recurrence, promotes cell migration and invasion via interacting and deubiquitinating PRP19. Biochim Biophys Acta. 2013;1833(3):559–72. doi:10.1016/j.bbamcr.2012.11.020.
Fang Y, Fu D, Shen XZ. The potential role of ubiquitin c-terminal hydrolases in oncogenesis. Biochim Biophys Acta. 2010;1806(1):1–6. doi:10.1016/j.bbcan.2010.03.001.
Wang L, Chen YJ, Hou J, Wang YY, Tang WQ, Shen XZ et al. Expression and clinical significance of BIRC6 in human epithelial ovarian cancer. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine. 2014. doi:10.1007/s13277-014-1641-6.
Acknowledgments
The authors would like to express gratitude to the staff of Prof. Xi-zhong Shen’s laboratory for their critical discussion and reading of the manuscript. The study was partly funded by the Youth Foundation of Zhongshan Hospital (ZSQN2012-36).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding authors
Additional information
Lin Wang and Yan-Jie Chen contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, L., Chen, YJ., Xu, K. et al. High expression of UCH37 is significantly associated with poor prognosis in human epithelial ovarian cancer. Tumor Biol. 35, 11427–11433 (2014). https://doi.org/10.1007/s13277-014-2446-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-2446-3