Abstract
Neuroendocrine tumours (NETs) belong to a relatively rare class of neoplasms. Nonetheless, their prevalence has increased significantly during the last decades. Peptide receptor radionuclide therapy (PRRT) is a relatively new treatment approach for inoperable or metastasised NETs. The therapeutic effect is based on the binding of radiolabelled somatostatin analogue peptides with NETs’ somatostatin receptors, resulting in internal irradiation of tumours. Pre-therapeutic patient-specific dosimetry is essential to ensure that a treatment course has high levels of safety and efficacy. This paper reviews the methods applied for PRRT dosimetry, as well as the dosimetric results presented in the literature. Focus is given on data concerning the therapeutic somatostatin analogue radiopeptides 111In-[DTPA0,D-Phe1]-octreotide (111In-DTPA-octreotide), 90Y-[DOTA0,Tyr3]-octreotide (90Y-DOTATOC) and 177Lu-[DOTA0,Tyr3,Thr8]-octreotide (177Lu-DOTATATE). Following the Medical Internal Radiation Dose (MIRD) Committee formalism, dosimetric analysis demonstrates large interpatient variability in tumour and organ uptake, with kidneys and bone marrow being the critical organs. The results are dependent on the image acquisition and processing protocol, as well as the dosimetric imaging radiopharmaceutical.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Neuroendocrine tumours (NETs) arise from cells of the diffuse endocrine system. They belong to a relatively rare class of neoplasms. Nevertheless, their prevalence has increased significantly during the last decades, mainly due to the improvement of the diagnostic procedures’ efficacy and to a better knowledge of this subject. According to Surveillance, Epidemiology, and End Results (SEER) data, the age-adjusted incidence of NETs rose from 1.9 to 5.25 cases per 100,000 people between 1973 and 2004 [1]. Some of the clinical and pathologic features of these tumours are characteristic of the organ of origin [2]. Excluding small-cell lung carcinomas, the most frequent NETs occur in the digestive tract (gastroenteropancreatic NETs—GEP NETs) (66 %), followed by the rest of the respiratory tract (31 %) [3]. NETs usually over-express specific somatostatin receptors (sstr) at their cell surfaces, sstr1–5, a characteristic which can be of great value in their localisation and treatment.
Treatment of NETs is typically multidisciplinary and should be individualised according to the tumour size, grade, location and secretory status [4]. Peptide receptor radionuclide therapy (PRRT) is a relatively new treatment approach for inoperable or metastasised NETs. It involves the systemic administration of a radiolabelled somatostatin analogue peptide, designed to target with high affinity and specificity somatostatin receptors over-expressed on tumours, and especially sstr2 [5].
Clinical trials have shown large interpatient variability in the biodistribution and tumour uptake of the radiopharmaceutical [6–8]. This fact stimulates the need for personalised therapy planning, rather than implementing a fixed therapeutic regimen approach. Pre-therapeutic patient-specific dosimetry is the tool for a treatment course with high levels of safety and efficacy. The main goal of dosimetry is the optimisation of the therapeutic outcome by maximising the dose delivered to the tumour, while keeping the dose delivered to critical organs at acceptable levels. In most cases, the critical organs are considered to be the kidneys and the bone marrow [9–11]. Patient-specific imaging is a prerequisite for the dosimetric estimations, so as to define the tracer biodistribution in each patient. Patient imaging data are acquired from planar, SPECT or PET images after injection of the imaging radiopharmaceutical. In some cases, the exact same radiopharmaceutical is used for both imaging (diagnostic and/or dosimetric) and therapeutic purposes, an approach referred in the bibliography as “theranostics” [8, 12–14]. Image quantification and absorbed dose estimations are based on the Medical Internal Radiation Dose (MIRD) Committee formalism [15]. Specific software programmes, such as MIRDOSE, OLINDA/EXM, LundADose, and STRATOS, are an essential tool when performing dosimetric estimations in a clinical environment [16–19].
This paper reviews the methods and results of PRRT dosimetry presented in the literature, mainly for the therapeutic somatostatin analogue radiopeptides 111In-[DTPA0,D-Phe1]-octreotide (111In-DTPA-octreotide), 90Y-[DOTA0,Tyr3]- octreotide (90Y-DOTATOC) and 177Lu-[DOTA0,Tyr3,Thr8]-octreotide (177Lu-DOTATATE).
Materials and methods
Studies inclusion criteria
This review includes studies that have performed dosimetry in adult patients for the therapeutic radiopharmaceuticals 111In-DTPA-octreotide, 90Y-DOTATOC and 177Lu-DOTATATE. Furthermore, dosimetric studies presented here are those published the last 15 years (2000–2014), with those of 177Lu-DOTATATE being published the last 5 years (2010–2014). An additional inclusion criterion was provision of dosimetric data for at least two regions of interest. Finally, this review includes data presented in the form of mGy/MBq, unless a fixed activity is administered to all patients, and in the form of a mean or a median value.
PRRT radiopharmaceuticals
Each nuclear medicine department implements its own PRRT protocol, according to which the total injected activity, the number of therapy cycles, the injected activity per cycle as well as the time interval between cycles are defined. In Table 1, some PRRT schemes, most commonly applied, are listed.
The therapeutic radiopharmaceutical consists of a radionuclide chelated to a somatostatin analogue peptide, for the purpose of delivering cytotoxic radioactivity to the tumour (Table 2). The salient features of the radiopharmaceutical which define its suitability as a PRRT radiopharmaceutical are:
-
The ligand. It consists of the chelating agent and the somatostatin analogue peptide. The chelating agent provides the stability of the radionuclide-ligand compound. The peptide is designed to target with high-affinity cell surface somatostatin receptors (sstr1–5), and especially sstr2 that is overexpressed on NETs.
-
The radionuclide. It is useful for its radioactive properties, namely adequate energy transfer to the tumour. The basic characteristics of radionuclides used in PRRT are presented in Table 3.
-
The activity. The injected activity has to be carefully selected so as to cause maximum damage to tumour cells, while keeping radiation exposure to normal tissues at safe levels.
Studies with 111In-DTPA-octreotide
Early studies, in the mid-to-late 1990s, investigated the safety and efficacy of using high activities of the imaging compound 111In-DTPA-octreotide as a therapeutic radiopharmaceutical [12, 20]. The idea of using this compound for therapy was based on the high linear energy transfer Auger electrons emitted by 111In. Despite some encouraging results in symptomatology, partial response (PR) was rarely observed (Table 4) [21]. The most common explanation for this was that the cytotoxic effect of the Auger electrons requires proximity of the 111In-labelled peptide to the nucleus, due to the very short particle range of Auger electrons [22]. Furthermore, serious side effects of leukaemia and myelodysplastic syndrome (MDS) were reported in three patients receiving the highest cumulative dose (90–100 GBq) (and estimated bone marrow radiation doses of more than 3 Gy) [23]. In Europe, therapy protocols with 111In-DTPA-octreotide were practically laid aside in favour of the more efficient β-emitters 90Y and 177Lu. Table 4 summarises the tumour response in treatment with 111In-DTPA-octreotide reported in various studies.
Studies with 90Y-DOTATOC
As 111In-DTPA-octreotide turned out to be ineffective for PRRT, researchers focused on the use of high-energy β-emitters, such as 90Y. 90Y is a nearly pure β-emitting radionuclide with a physical half-life of 64 h. The high-energy electrons (maximum energy 2.28 MeV) and maximum tissue penetration range of ~11 mm (Table 3), make 90Y more suitable for tumour cell killing, compared to 111In, also taking into consideration the cross-fire effect. Among other 90Y labelled peptides (such as 90Y-DOTATATE and 90Y-lanreotide), 90Y-DOTATOC is the most commonly used in PRRT [24–27]. The overall response rate (complete, partial and minor response—CR, PR and MR) reported in studies with 90Y-DOTATOC, fell in the range of 19–36 %, while the majority of patients showed disease stabilisation (5.2–69 %) (Table 4). In PRRT with 90Y-DOTATOC, kidneys are the dose-limiting organs, but also liver toxicity can be reported. In order to reduce the high kidney irradiation, positively charged amino acids were coinfused to competitively inhibit the proximal tubular reabsorption of the radiolabelled peptide. The coadministration of these amino acids led to a significant reduction in the renal absorbed dose. Specifically, in a study by de Jong et al., the renal absorbed dose was reduced by a mean of 27 % (range: 9–53 %) [28].
Studies with 177Lu-DOTATATE
177Lu emits both β- and γ- radiation, allowing post-treatment imaging and dosimetry assessments. Compared to 90Y, it has a lower tissue penetration range (Rmax ~ 2 mm), indicating that most of the energy is absorbed in small volumes. Some physical properties of 177Lu are listed on Table 3. The first treatment effects of 177Lu-DOTATATE were published in 2003 for 35 patients who had GEP NETs [29]. Since then, several centres worldwide conducted clinical trials with 177Lu-DOTATATE [30–32]. The reported overall response rates were higher when compared to 90Y-DOTATOC (Table 4). Serious side effects in patients treated with 177Lu-DOTATATE included renal toxicity (creatinine clearance loss of about 3.8 % per year), acute bone marrow toxicity (grade 3 and 4), mostly reversible (in 2–3 % of treatment cycles) and liver toxicity [5, 33].
Dosimetric imaging radiopharmaceuticals
Scintigraphy with radiolabelled somatostatin analogue peptides has become one of the most important imaging procedures for the diagnosis, staging and localisation of somatostatin receptor-positive tumours. It is also important for selecting patients with inoperable and/or metastatic tumours for subsequent PRRT. Furthermore, patient imaging is the prerequisite process for dosimetric assessment.
The imaging radiopharmaceutical may be the same or differ from the therapeutic one. Its selection is based on the following criteria:
-
The ligand. Ideally, the ligand of both imaging and therapeutic radiopharmaceutical should be the same. If this is not feasible, they should be chemically analogous, in order to eliminate the differences in the radiopharmaceutical biodistribution.
-
The radionuclide. The type of radiation emitted should enable imaging and its physical half-life should permit assessment of the radiopharmaceutical biokinetics in a short period of time.
-
The activity. In case that different radiopharmaceuticals are used the injected activity of the imaging radiopharmaceutical should be considerably lower than the therapeutic one, in order to keep patient radiation exposure to low levels.
In the dosimetric imaging procedure the renal protection regimen applied should be the same as the therapeutic one, since the amino acid infusion affects the degree of activity reabsorption in the proximal tubules. If dosimetric imaging is performed without amino acid coinfusion, the renal radiation doses are overestimated [34]. This overestimation is independent of the radionuclide used; however, attention should be paid to the imaging ligand, which should retain the chemical structure of the therapeutic one, so that the dosimetric procedure simulates the therapeutic one effectively.
Table 2 summarises the NETs imaging radiopharmaceuticals most often used, as well as the imaging technique used with each one.
Imaging with 111In
111In-DTPA-octreotide has been used for almost 20 years for the diagnosis and staging of somatostatin receptor-positive tumours and became the gold standard for the detection of NETs. 111In facilitates dosimetry assessments due to its γ-ray emission. Thereafter, it has been used as a surrogate for 90Y-DOTATOC, as the quantification of 90Y bremsstrahlung images was rather difficult. However, it was not the first choice for simulating 90Y-DOTATOC biokinetics because of the difference in the chemical nature of the ligands. In order to maintain similar chemical and kinetic behaviour, the use of 111In-DOTATOC has been investigated in several studies [35–37].
Compared to 111In-DTPA-octreotide, 111In-DOTATOC exhibited similar distribution and excretion patterns. However, the uptake in somatostatin receptor-positive organs and most tumours was higher for 111In –DOTATOC.
Imaging with 86Y
Another alternative for 90Y-DOTATOC image quantification, but a far more demanding solution from a technical and financial point of view, was to use DOTATOC labelled with the positron emitter 86Y. The major advantage of 86Y-DOTATOC is that it totally preserves the chemical structure of 90Y-DOTATOC. Therefore, it is generally considered as the gold standard for simulating 90Y-DOTATOC biokinetics. Additionally, PET offers improved accuracy and spatial resolution. Nevertheless, the low positron abundance (33 %), the emission of multiple, high-energy γ-rays, the high production cost and the low availability of 86Y were inhibiting factors in the routine utilisation of this compound [38]. In a comparison of 86Y-DOTATOC to 111In-DTPA-octreotide, the reported results indicated that the biokinetics of these two compounds were relatively similar in the organs but differed in the uptake pattern of the metastases. Particularly, 86Y-DOTATOC showed much higher uptake and slower washout in tumours [39].
Imaging with 177Lu
If PRRT is performed with 177Lu, its γ-ray emission enables dosimetry and therapy with the same compound. Therefore dosimetry is usually performed during PRRT, following the injection of 177Lu-DOTATATE. This approach eliminates the uncertainties occurring from the use of different compounds, due to the difference in biokinetics. Nevertheless, due to 177Lu’s long physical half-life of 6.7 days, a long time interval is required between the time of administration and acquiring the last dosimetric image acquisition. Ideally this should be more than 1 week [15]. Thus, patient discomfort could be pointed out as one of 177Lu imaging drawbacks.
Imaging with 68Ga
A further important step in the diagnosis of NETs was the introduction of somatostatin-based PET tracers labelled with the generator-produced radiometal 68Ga. The somatostatin analogue peptides clinically used are based on the three octapeptides [Tyr3]octreotide (TOC), [Tyr3,Thr8]octreotide (TATE) and [1-Nal3]octreotide (NOC), conjugated to DOTA. These somatostatin analogues showed high affinity for certain sstr. Additionally, since 68Ga is a generator product, rather than a cyclotron-produced product, a relatively simple labelling can be performed on an everyday basis [21]. In addition to these advantages, 68Ga peptides seem desirable for dosimetry, as they preserve the same chemical ligand structure with that of the therapeutic compound. Nonetheless, they are not suitable for accurate dosimetric calculations, mainly due to the very short physical half-life of 68Ga (68 min). Taking into account that the effective half-life is even shorter than the physical one, for accurate determination of time–activity curve, PET scans with 68Ga should last just a few minutes, in accordance to MIRD guidelines [15]. This would result in poor statistics and ineffective subsequent dosimetric evaluations.
Imaging with 99mTc
Clinical studies with 99mTc-somatostatin analogue peptides began in the late 1990s [40]. At present, 99mTc-ethylenediamine-N,N9-diacetic acid (EDDA)/HYNIC,Tyr3]octreotide (99mTc-HYNIC-TOC) is available in different European countries and its use in clinical practice for the diagnosis of NETs is gradually gaining acceptance. Among other advantages, its availability, its physical properties (γ-emitter, T 1/2 physical = 6 h) and the peptide –TOC, same as that with the therapeutic compound 90Y-DOTATOC, made it one of the principal choices for NETs imaging and dosimetry. 99mTc-HYNIC-TATE has also been used by some groups. Hubalewska et al. demonstrated superior image quality of 99mTc-HYNIC-TATE over 111In-DTPA-octreotide in 75 patients [41]. Cwikla et al. compared the 2 compounds 99mTc-HYNIC-TOC and 99mTc-HYNIC-TATE in the same 12 patients and concluded that both give excellent high-quality images. A similar number of metastatic sites could be seen with both agents, though 99mTc-HYNIC-TATE may have some advantage in lymph node and liver metastases [42].
Dosimetry
Patient-specific dosimetry can provide information on organs and NETs radiation absorbed doses and assess the risk of critical organ toxicity (kidneys, bone marrow). Different dosimetry methods can be applied, with various levels of complexity, depending on study objective, human and software resources, as well as workflow practical factors. All dosimetry methods require input data to be derived from patient-specific scintigraphic images and, when needed, from biological samples, such as blood and urine. Patient imaging can be performed with whole-body scans, or with SPECT. Planar and SPECT dosimetry gave comparable results in areas free of tumours. However, in tumour dosimetry, or in the case of overlapping organs, SPECT dosimetry was more accurate, although more time-demanding [11].
Since gamma camera systems provide the imaging data in counts or count rates, a mathematical model should be applied to convert counts to activity values [15]. For accurate image quantification several correction factors should be taken into account, including photon attenuation in the patient’s body, scatter, background activity, dead-time, source self-absorption, partial volume effects for small objects, collimator effects and proper calibration of the imaging system. Some of these factors are obtained from patient images (attenuation correction, scatter, background activity subtraction, source self-absorption) while others from experimental measurements. The determination of all these correction factors can be a painstaking process, and it is at the discretion of each dosimetry team which method of quantification should be applied.
Once the organ activity values are estimated, they are then converted to absorbed dose values. Dosimetric analysis is generally based on MIRD Committee schema [15]. The absorbed dose is estimated from the organ uptake and retention of administered radiopharmaceutical. In order to attain the therapeutic radiopharmaceutical biodistribution, simulation with an appropriate imaging radiopharmaceutical is being performed. The basic dosimetry equations, according to MIRD schema, are the following:
where \(\overline{D}_{target}\) is the mean target absorbed dose (Gy or rad), \(\tilde{A}_{source}\) is the source cumulated activity (Bq·sec or μCi·hr), \(S_{{target \leftarrow source}}\) is the mean absorbed dose per unit cumulated activity (Gy/Bq·sec or rad/μCi·hr), \(\tau_{source}\) is the source region residence time (sec or hr), \(A_{0}\) is the administered activity (Bq or μCi). From Eq. 1 and 3 it can be seen that the target absorbed dose depends on two main parameters:
-
(i)
Time-independent physical factors: the so-called S-factors, which include characteristics of type and energies of the radiations emitted, geometrical and composition aspects of the source, target and intervening media. The S-factors have been tabulated by MIRD in 1975 and 1978 [43, 44], and followed other relevant publications [45, 46].
-
(ii)
Time-dependent biokinetic factors: incorporated within à or τ.
Sequential imaging as a function of time postadministration of the radiopharmaceutical provides the time–activity curve for each source region. The area under the curve defines the cumulated activity, Ã. It should be noted that selection of adequate time points, appropriately time-distributed, for sampling radiopharmaceutical biokinetics has a significant effect on the resulting time–activity curve and conclusively on the precision of estimated target absorbed dose.
Data processing for absorbed dose estimations can be performed by computer software packages. There are several codes/programmes available now, varying from the most simple to the most complex; the latter being designed to apply patient-specific adjustments. Some examples of such programmes are MIRDOSE, OLINDA/EXM, LundADose, STRATOS [16–19]. The key features of these programmes differ significantly and the selection of the appropriate programme depends on the financial and computational cost as well as the dosimetric level accuracy desired. Some are based on standard mathematical phantoms and are simple and fast, while others are highly accurate, because they take the individualized patient anatomy into account, but they are time consuming.
Table 5 summarises the basic dosimetric protocols features for data acquisition and processing applied by several groups. It should be mentioned that in most cases of planar image acquisition, an additional SPECT scan was acquired for improved accuracy in organ depth determination.
Discussion
Results analysis
Due to large interpatient variability in tumour and organ uptake, accurate and reliable dosimetric estimations are needed to fully exploit the therapeutic potential of PRRT. In this way, the dose delivered to tumours is maximised, while the dose delivered to critical organs remains at acceptable levels. Nonetheless, and to the best of our knowledge, dosimetry is systematically implemented in a relatively limited number of nuclear medicine centres worldwide. This is mainly due to the time-consuming methods that are required for reliable dosimetry. Dosimetric procedures are demanding with respect to the patients’ discomfort, the level of staff commitment and available facilities. In most cases, after the patients are discharged, they should return to the Nuclear Medicine Department for the completion of image acquisition, which takes at least 2 days. Furthermore, reaching an adequate dosimetric outcome requires the collaboration of a multidisciplinary team, including medical physicists, nuclear medicine physicians, oncologists and technologists. If any link of this chain is reluctant to cooperate, dosimetric procedures become difficult to implement. Additionally, for accurate dosimetric estimations the department should be equipped with appropriate software programmes, imaging or/and counting systems and phantoms.
These difficulties raised objections as to whether dosimetry-based radionuclide therapy is worth the extra effort over fixed-dose therapies. The benefit of dosimetry-based radionuclide therapy over fixed dose therapy remains to be proven by randomised phase III clinical trials. Nevertheless, studies that provide initial evidence supporting further consideration of individualised therapy planning have been published. In a study of Stabin [47] the objections to dosimetry-based approach were addressed and the conclusion of the analysis was that careful use of patient-specific dosimetry should become routine practice. Furthermore, several investigators have shown that patient-specific dosimetry can produce strong correlations between absorbed dose, or biologically effective dose (BED), and observed effects in tumours and normal organs. Shen et al. [48] using a 90Y-antibody in radioimmunotherapy, found a strong correlation between bone marrow dose and observed marrow toxicity, whereas Pauwels et al. [34] correlated tumour absorbed dose with tumour reduction in their study with 90Y-DOTATOC. Taking radiobiological models into account, Barone [6] proved that BED is a reliable predictor of renal toxicity. The inclusion of the most advanced techniques in the dosimetric procedure, some of them being realistic body morphometry, radiobiological models, 3D voxel-by-voxel dosimetry, can produce accurate dose evaluations, which can subsequently be used for individual therapy planning.
In the Euratom Directives of 1997, it is stated that “for all medical exposures of individuals for radiotherapeutic purposes, exposures of target volumes shall be individually planned, taking into account that doses of non-target volumes and tissues shall be as low as reasonably achievable and consistent with the intended radiotherapeutic purpose of the exposure” [49]. These Directives form an additional stimulus for dosimetry. However, the fact that dosimetry is still not widely performed stresses the need for sufficient education of personnel and further development of dosimetric conscience.
Four parameters that significantly affect the dosimetric outcome are the selected dosimetric imaging radiopharmaceutical, the image acquisition method, the accuracy of activity quantification, as well as the temporal sampling.
For dosimetry of 90Y-DOTATOC, the chemically analogous 86Y-DOTATOC is generally considered as the gold standard. However, its already mentioned disadvantages in combination with the acceptable dosimetric results of 111In-DTPA-octreotide have laid the use of the PET tracer aside. As depicted by Table 6, both studies comparing the biokinetics of 111In-DTPA-octreotide versus 86Y-DOTATOC in the same patients have yielded similar organ uptake, while tumour uptake seems to be underestimated if 111In-DTPA-octreotide is selected, according to the studies of Förster et al. [39, 50]. In the case of 177Lu-DOTATATE the exact same radiopharmaceutical is used for both dosimetric imaging and therapeutic purposes.
As far as the image acquisition method is concerned, large differences can be obtained in the estimated absorbed dose values, depending on whether planar or SPECT dosimetry is used. Studies revealed that they provide comparable results in areas free of tumours, but planar dosimetry highly overestimated the absorbed dose in organs with tumours or in overlapping organs (Table 6) [11, 51]. Despite the improved accuracy of SPECT dosimetry, many dosimetry approaches are still based on 2D scintigraphic images. This is due to the much higher demands of SPECT in terms of imaging time and data post-processing. Another application of whole-body imaging by planar scintigraphy is bone marrow dosimetry. The activity in the whole-body from serial whole-body scans should be assessed to determine the cross-absorbed dose to the bone marrow, according to EANM Dosimetry Committee guidelines [52].
The fact that imaging systems measure counts and not activity is directly related to the need for absolute quantification, equipment calibration and inclusion of necessary corrections. The correction factors, mentioned in the dosimetry section, should be included in the dosimetric protocol as patient image acquisition differs by far from the ideal imaging conditions, due to broad-beam geometry conditions. The dosimetry team can face a number of difficulties in determining the correction factors, the most important of them being the time and skills needed, as well as the equipment availability for the experimental set-ups. Although being a time- and labour-intensive process, attention to determining these factors influences the accuracy of image quantification and hence the accuracy of the dosimetric results.
Furthermore, selection of image acquisition time points can have a significant effect on dosimetric estimation accuracy. For an appropriate temporal sampling, a minimum of three sampling points, near the effective half-life T eff , ~3 T eff and ~5 T eff , should be chosen for the uptake and washout phases, according to MIRD guidelines [15]. Inappropriate temporal sampling and data interpolation methods could lead to errors in absorbed dose estimation even greater than 20 %. For DOTATOC and DOTATATE peptides, whose time–activity curve generally comprises two kinetic phases, uptake and washout, at least six time points are needed. As each experimental point requires time-consuming acquisitions, the whole process would be demanding for patients, clinicians and personnel. In this respect, finding a compromise that balances accuracy and feasibility seems to be an optimal approach for clinical studies. Generally, radionuclides with relatively short physical half-lives, i.e., of a few hours, such as 99mTc (T 1/2 physical = 6 h), should be preferred for dosimetric imaging rather than long- lived, i.e., T 1/2 physical ~ days, such as 111In (2.8 days), or very short-lived radionuclides, i.e., T 1/2 physical ~ minutes, such as 68Ga (68 min). Dosimetric imaging with long-lived radionuclides is impractical for reasons of patients inconvenience, since the last scan should be acquired at ~5T eff , and radiation safety issues. Additionally, very short-lived radionuclides are impractical for dosimetric imaging, since the rapid activity falloff requires a very short scan duration, resulting in poor statistics.
Dosimetry software programmes can facilitate the dosimetric procedure in a busy clinical environment. Until the early 2000s the main dosimetric tool available was MIRDOSE3 software [16]. Using tabulated specific absorbed fractions (SAFs), radionuclide decay data and specific anthropomorphic models, the main function of this programme is to provide estimates of the mean radiation dose per unit administered activity, given the source organ residence times [53]. The software code OLINDA/EXM was designed as an update to MIRDOSE3 [17]. The updates include decay data for more than 800 radionuclides, including selected α-emitters, new absorbed fractions in the sphere model of MIRDOSE3.1 and the ability to modify organ masses to patient-specific mass values. The limitations of MIRDOSE and OLINDA/EXM, namely calculation only of self-dose for tumours, using a sphere model of tumours with a uniform activity distribution and the use of standard anatomic models, led to the development of more sophisticated programmes. Sjögreen and Ljungberg developed LundADose, which is used for dosimetry in clinical studies of 177Lu-DOTATATE and 90Y/111In labelled Ibritumomab Tiuxetan [18, 54]. For SPECT/CT dosimetry, a voxel-based Monte Carlo programme is used, while in planar dosimetry corrections for attenuation, scatter, and overlapping organs are applied. Furthermore, the programme offers the possibility calculating the BED. Further research in that field led to the release of more advanced software packages, like STRATOS, by Philips [19]. The STRATOS Dosimetry Solution performs 3D voxelised dose calculations using SPECT/CT and also PET/CT data, or even using planar images as input. It performs patient-specific dose calculations based on the dose point kernel (DPK) method. The key features of the STRATOS software include registration, segmentation and visualisation of 3D images, more supported therapy radionuclides, calculation of residence-time maps, energy–dose distributions and dose–volume–histograms per region. This gradual development of more realistic programmes, which are easily adopted in everyday clinical routine and are financially affordable, is expected to contribute to the inclusion of dosimetry in radionuclide therapies, in accordance with external beam radiation therapy practice.
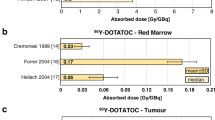
In PRRT, the kidneys together with the bone marrow are the dose-limiting organs at the usual administered activities. Proper kidney protection by the coinfusion of amino acid solutions enlarges the safety margin for administrating higher activities, enabling higher tumour radiation doses to be attained safely. The dosimetric results of Table 6 demonstrate that 90Y-DOTATOC kidney absorbed doses are higher than these obtained with 177Lu-DOTATATE. Consequently, 177Lu-labelled somatostatin analogues are likely to have higher renal toxicity thresholds than 90Y. Indeed, the PRRT schemes listed on Table 3 apply higher 177Lu injected activities than with 90Y. Thus, 177Lu-labelled peptides are considered a safer option in patients with reduced renal function or when designing a retreatment regimen.
The dosimetric results of the Barone 2008 study, presented in Table 6, pointed out the higher tumour uptake of the β-emitter, 90Y compared to 111In [55]. These data in combination with the low tumour response (<20 %, Table 4) proved the inappropriateness of 111In-labelled peptides for therapy, despite some encouraging preliminary results. Additionally, when comparing the radioisotopes used today in PRRT, 90Y and 177Lu, from data listed in Table 4, it can be seen that 177Lu led to higher corresponding tumour responses. Concluding, the optimal treatment regimen applied, namely administration of 90Y- or 177Lu-labelled peptides, or even a combination of them, is dependent on many parameters. The most important of them include the tumour size and location, the extent of disease and the patient’s clinical condition.
Improvements under development
The establishment of the optimal therapeutic and dosimetric protocol for PRRT is still a matter of research. Future steps for the improvement of PRRT dosimetry focus on three main aspects: development of efficient Neuroendocrine tumour imaging radiopharmaceuticals, proper selection of therapeutic schemes and better understanding of dosimetric procedures’ mathematical formalism and technical implementation.
Concerning Neuroendocrine tumour imaging, the relatively new compound 99mTc-HYNIC-TOC is a promising radiopharmaceutical. It binds to somatostatin receptors with high affinity (particularly subtype two and, to a lesser extent, subtypes three and five) and its use in clinical practice for the diagnosis of NETs is gradually gaining acceptance [42, 56]. In addition to its diagnostic value, 99mTc-HYNIC-TOC seems to be ideal for dosimetry assessments. Its physical properties (γ-emitter with a relatively short half-life of 6 h), allow the patient dosimetric imaging to be performed within no longer than 2 days. This is of great importance when considering the daily clinical schedule, the personnel burden and the patients’ comfort. The compound availability should also be included in its advantages. Furthermore, when dosimetric evaluations for the therapeutic radiopharmaceutical 90Y-DOTATOC are acquired, both the therapeutic and the imaging compound have the same peptide (-TOC), allowing similar biodistribution. Dosimetric data of 99mTc-HYNIC-TOC have been published by some groups [57–59].
In the search of methods for improving PRRT efficacy, studies lean towards combination therapy protocols. PRRT is commonly performed with a single radiopharmaceutical administration. The currently used therapeutic radionuclides 90Y and 177Lu differ markedly in their physical properties, including half-life, range and energy of emitted β-particles. Potential advantages of 177Lu for PRRT include a longer half-life and β-particle ranges suitable for small tumours. By contrast, 90Y emits β-particles with longer range and higher energies and therefore may be preferable for patients with bulk disease. These different characteristics led to the first clinical study of combination therapy in animals bearing tumours of various sizes [60]. Reports of clinical applications in humans, incorporating both 90Y- and 177Lu- labelled peptides, were published recently and indicated longer overall survival [61, 62]. Nonetheless, larger randomised trials are essential in order to confirm these results and optimise the combination treatment protocol.
In order to improve the accuracy of absorbed dose estimates, efforts have been directed towards more sophisticated dosimetry methods. While planar images are useful to derive biokinetics over time, 3D dosimetry based on quantitative SPECT/CT has potential advantages over planar approaches. Although requiring more time to acquire and more complex algorithms for activity quantification, SPECT/CT fused images provide insight into organ-specific 3D activity distribution, which can then be used as input data for voxel dosimetry. Berker et al. presented a hybrid 2D/3D activity quantification technique [63]. The proposed method of integrating 2D image information into 3D image-based dosimetry can speed up SPECT/CT-based 3D dosimetry without losing accuracy.
Several factors are involved in renal toxicity during or after PRRT, among which absorbed dose to kidneys plays a key role. In fractionated external radiation therapy the absorbed dose limit for causing 5 % probability of attaining radiation nephropathy within 5 years has been found to be 23 Gy, rising to 28 Gy with 50 % probability of nephropathy [64]. This dose is typically given at a high dose rate (1–10 Gy/min) in fractions of 1.5–2 Gy. However, in PRRT, where the maximum dose rate is much lower (~3 mGy/min), decaying exponentially with the radiopharmaceutical effective half-life and the organ dose distribution is inhomogeneous, the critical dose is expected to be higher and has not yet been determined [10]. In order to transfer the external radiation therapy dose limit to a corresponding limit in radionuclide therapy, the linear-quadratic (LQ) model can be applied. BED, obtained by the LQ model, has been proved to correlate strongly with renal toxicity, in contrast to kidney absorbed doses estimated by conventional dosimetry [6]. Conclusively, BED can be used to establish an accurate dose–response relationship. Even though BED values have not yet been established, studies report a mean kidney BED value of 40 Gy for patients without risk factors [3, 6].
As has been already mentioned, the imaging radiopharmaceutical should ideally be the same with the therapeutic one. However, for 90Y-labelled radiopharmaceutical imaging, quantification of the uptake and biodistribution is challenging, due to its nearly pure β-emission. In the absence of a photopeak, imaging of 90Y is dependent on bremsstrahlung radiation. A number of studies investigated energy windows, collimators and scatter correction techniques for 90Y bremsstrahlung imaging [13, 65, 66]. Some groups used Monte Carlo simulations in conjunction with phantom studies to optimise the accuracy of quantitative imaging, by enabling corrections to be made for attenuation, scatter and collimator-detector response [67]. These correction methods for quantification of 90Y imaging allowed the recent development of bremsstrahlung dosimetry studies, offering the opportunity for direct assessment of absorbed dose delivered in 90Y PRRT [68]. In a recent study of three patients, quantitative pretherapy 111In- ibritumomab SPECT/CT and planar whole-body (WB) imaging and therapy 90Y-ibritumomab SPECT/CT and WB imaging, resulted in differences between absorbed-dose estimates within 25 %, except for the lungs [69]. Recently, the presence of internal positron–electron pair production in 90Y was determined and the branching ratio was found to be (31.86 ± 0.47) × 10−6 [70]. Thus, PET imaging of 90Y might be a new method for dosimetric assessments. However, the low probability of positron emission requires large amounts of administered activity. 90Y PET-based dosimetry has been so far implemented in selective internal radiation therapy of the liver, where the 90Y micro-spheres are selectively located in the hepatic tumours and there is no biological removal [71].
Finally, improvements in the dosimetric process include the adoption of kidney multiregional dosimetric models. The administered dose in PRRT is limited by the radiation burden of critical organs. Studies proved inhomogeneous radioactivity distribution of the radiolabelled peptides in the kidneys [10, 72]. Ex vivo autoradiography of healthy kidney tissue, after injection of 111In-DTPA-octreotide, showed that the radioactivity is localised predominantly in the proximal tubule in the cortex [73]. More accurate radiation dose estimates may be determined either by adopting multiregional models or by assuming entire uptake by the cortex. In the first case, regional dosimetric estimations are performed in different substructures of the kidney (cortex, medulla, pelvis, papillae), while in the second activity is assumed to be distributed only in the cortex, which is 70 % of the total kidney mass [73, 74].
Conclusions
The dosimetric results demonstrated large interpatient variability in tumour and organ uptake, stressing the need for personalised therapy planning. Patient-specific dosimetry in PRRT is therefore needed, in order for the fixed dose regimens to be replaced with patient-tailored therapy planning. Comparing 86Y-DOTATOC to 111In-DTPA-octreotide for the dosimetry of 90Y-DOTATOC, the results demonstrated similar organ doses, while the tumour dose seemed to be underestimated if 111In-DTPA-octreotide is used. SPECT and planar dosimetry achieved comparable results in organ dose estimation, but SPECT provided a more accurate estimate of tumour dosimetry.
Kidneys are the dose-limiting organs due to both high absorbed doses and increased radiosensitivity. Additionally, kidney absorbed doses were higher in PRRT with 90Y-DOTATOC, allowing for higher 177Lu activities to be administered in 177Lu- DOTATATE therapies.
When comparing total tumour responses in 90Y- and 177Lu- PRRT schemes, 90Y led to overall response (CR, PR and MR) in up to 36 % of patients, while 177Lu achieved higher rates of 56 %.
Attention should be paid to the image quantification process, as it is can be a large source of error in dosimetry. In order for the dosimetry techniques to be systematically implemented in a busy clinical environment, the dosimetry software programmes can be an essential tool, as they help in making the procedures less time-intensive.
References
Yao JC, Hassan M, Phan A, Dagohoy C, Leary C, Mares JE, Abdalla EK, Fleming JB, Vauthey JN, Rashid A et al (2008) One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol 26:3063–3072. doi:10.1200/JCO.2007.15.4377
Klimstra DS, Modlin IR, Coppola D, Lloyd RV, Suster S (2010) The pathologic classification of neuroendocrine tumors. A review of nomenclature, grading, and staging systems. Pancreas 39:707–712. doi:10.1097/MPA.0b013e3181ec124e
Bodei L (2008) Peptide receptor radionuclide therapy with radiolabelled somatostatin analogues. Dissertation, University Medical Center Groningen
Novartis Oncology (2014) Multifaceted cancer that benefits from multidisciplinary care. http://www.neuroendocrinetumor.com/health-care-professional/managing-nets.jsp. Accessed on 29 March 2014
Zaknun JJ, Bodei L, Mueller-Brand J, Pavel ME, Baum RP, Hörsch D, O’Dorisio MS, O’Dorisiol TM, Howe JR, Cremonesi M et al (2013) The joint IAEA, EANM, and SNMMI practical guidance on peptide receptor radionuclide therapy (PRRNT) in neuroendocrine tumours. Eur J Nucl Med Mol Imaging 40:800–816. doi:10.1007/s00259-012-2330-6
Barone R, Borson-Chazot F, Valkema R, Walrand S, Chauvin F, Gogou L, Kvols LK, Krenning EP, Jamar F, Pauwels S (2005) Patient-specific dosimetry in predicting renal toxicity with (90)Y-DOTATOC: relevance of kidney volume and dose rate in finding a dose–effect relationship. J Nucl Med 46:99S–106S
Jamar F, Barone R, Mathieu I, Walrand S, Labar D, Carlier P, de Camps J, Schran H, Chen T, Smith MC et al (2003) 86Y-DOTA0-D-Phe1-Tyr3-octreotide (SMT487): a phase 1 clinical study: pharmacokinetics, biodistribution and renal protective effect of different regimens of amino acid co-infusion. Eur J Nucl Med Mol Imaging 30:510–518. doi:10.1007/s00259-003-1117-1
Forrer F, Krenning EP, Kooij PP, Bernard BF, Konijnenberg M, Bakker WH, Teunissen JJ, de Jong M, van Lom K, de Herder WW et al (2009) Bone marrow dosimetry in peptide receptor radionuclide therapy with [177Lu-DOTA0, Tyr3]octreotate. Eur J Nucl Med Mol Imaging 36:1138–1146. doi:10.1007/s00259-009-1072-6
Boerman OC, Oyen WJ, Corstens FH (2001) Between the Scylla and Charybdis of peptide radionuclide therapy: hitting the tumor and saving the kidney. Eur J Nucl Med 28:1447–1449
Konijnenberg MW (2003) Is the renal dosimetry for [90Y-DOTA0, Tyr3]Octreotide accurate enough to predict thresholds for individual patients? Cancer Biother Radiopharm 18:619–625
Sandström M, Garske U, Granberg D, Sundin A, Lundqvist HU (2010) Individualized dosimetry in patients undergoing therapy with (177)Lu- DOTA-D-Phe(1)-Tyr(3)-octreotate. Eur J Nucl Med Mol Imaging 37:212–225. doi:10.1007/s00259-009-1216-8
Krenning EP, de Jong M, Kooij PP, Breeman WA, Bakker WH, de Herder WW, van Eijck CH, Kwekkeboom DJ, Jamar F, Pauwels S et al (1999) Radiolabelled somatostatin analogue(s) for peptide receptor scintigraphy and radionuclide therapy. Ann Oncol 10:S23–S29
Shen S, DeNardo GL, DeNardo SJ (1994) Quantitative bremsstrahlung imaging of yttrium-90 using a Wiener filter. Med Phys 21:1409–1417
Öberg K (2012) Molecular imaging and radiotherapy: theranostics for personalized patient management of neuroendocrine tumors (NETs). Theranostics 2:448–458
Siegel JA, Thomas SR, Stubbs JB, Stabin MG, Hays MT, Koral KF, Robertson JS, Howell RW, Wessels BW, Fisher DR et al (1999) MIRD pamphlet no. 16: techniques for quantitative radiopharmaceutical biodistribution data acquisition and analysis for use in human radiation dose estimates. J Nucl Med 40:37S–61S
Stabin MG (1996) MIRDOSE: personal computer software for internal dose assessment in nuclear medicine. J Nucl Med 37:538–546
Stabin MG, Sparks RB, Crowe E (2005) OLINDA/EXM: the second-generation personal computer software for internal dose assessment in nuclear medicine. J Nucl Med 46:1023–1027
Sjögreen K, Ljungberg M, Wingårdh K, Minarik D, Strand SE (2005) The LundADose method for planar image activity quantification and absorbed-dose assessment in radionuclide therapy. Cancer Biother Radiopharm 20:92–97
Philips imalytics (2014) Research Dosimetry Solution- STRATOS and STRATOS+. http://www.imalytics.philips.com/sites/philipsimalytics/products/dosimetry/dosimetry.page. Accessed on 10 March 2014
de Jong M, Breeman WA, Bakker WH, Kooij PP, Bernard BF, Hofland LJ, Visser TJ, Srinivasan A, Schmidt MA, Erion JL et al (1998) Comparison of 111In-labeled somatostatin analogues for tumor scintigraphy and radionuclide therapy. Cancer Res 58:437–441
Kwekkeboom DJ, Kam BL, van Essen M, Teunissen JJ, van Eijck CH, Valkema R, de Jong M, de Herder WW, Krenning EP (2010) Somatostatin receptor-based imaging and therapy of gastroenteropancreatic neuroendocrine tumors. Endocr Relat Cancer 17:R53–R73. doi:10.1677/ERC-09-0078
Cremonesi M, Ferrari M, Bodei L, Tosi G, Paganelli GM (2006) Dosimetry in peptide radionuclide receptor therapy: a review. J Nucl Med 47:1467–1475
Valkema R, De Jong M, Bakker WH, Breeman WA, Kooij PP, Lugtenburg PJ, De Jong FH, Christiansen A, Kam BL, De Herder WW et al (2002) Phase I study of peptide receptor radionuclide therapy with [In-DTPA]-octreotide: the Rotterdam experience. Semin Nucl Med 32:110–122
Waldherr C, Pless M, Maecke HR, Haldemann A, Mueller-Brand JP (2001) The clinical value of [90Y-DOTA]-D-Phe1-Tyr3-octreotide (90Y-DOTATOC) in the treatment of neuroendocrine tumours: a clinical phase II study. Ann Oncol 12:941–945
Waldherr C, Pless M, Maecke HR, Schumacher T, Crazzolara A, Nitzsche EU, Haldemann A, Mueller-Brand J (2002) Tumor response and clinical benefit in neuroendocrine tumors after 7.4 GBq 90Y-DOTATOC. J Nucl Med 43:610–616
Bodei L, Cremonesi M, Zoboli S, Grana C, Bartolomei M, Rocca P, Caracciolo M, Mäcke HR, Chinol M, Paganelli G (2003) Receptor-mediated radionuclide therapy with 90Y-DOTATOC in association with amino acid infusion: a phase I study. Eur J Nucl Med Mol Imaging 3:207–216. doi:10.1007/s00259-002-1023-y
Valkema R, Pauwels S, Kvols L, Jamar F, Barone R, Bakker WH, Lasher J, Krenning EP (2003) Long-term follow-up of a phase 1 study of peptide receptor radionuclide therapy (PRRT) with [90Y-DOTA0, Tyr3]-octreotide in patients with somatostatin receptor positive tumours. [abstract]. Eur J Nucl Med Mol Imaging 30:S232
de Jong M, Krenning EP (2002) New advances in peptide receptor radionuclide therapy. J Nucl Med 43:617–620
Kwekkeboom DJ, Bakker WH, Kam BL, Teunissen JJM, Kooij PPM, Herder WW, Feelders RA, Eijck CHJ, Jong M, Srinivasan A et al (2003) Treatment of patients with gastro-entero-pancreatic (GEP) tumours with the novel radiolabelled somatostatin analogue [177Lu-DOTA(0), Tyr3]octreotate. Eur J Nucl Med Mol Imaging 30:417–422. doi:10.1007/s00259-002-1050-8
Bodei L, Cremonesi M, Grana CM, Fazio N, Iodice S, Baio SM, Bartolomei M, Lombardo D, Ferrari ME, Sansovini M et al (2011) Peptide receptor radionuclide therapy with 177Lu-DOTATATE: the IEO phase I-II study. Eur J Nucl Med Mol Imaging 38:2125–2135. doi:10.1007/s00259-011-1902-1
Claringbold PG, Brayshaw PA, Price RA, Turner H (2011) Phase II study of radiopeptide 177Lu-octreotate and capecitabine therapy of progressive disseminated neuroendocrine tumours. Eur J Nucl Med Mol Imaging 38:302–311. doi:10.1007/s00259-010-1631-x
Sansovini M, Severi S, Ambrosetti A, Monti M, Nanni O, Sarnelli A, Bodei L, Garaboldi L, Bartolomei M, Paganelli G (2013) Treatment with the radiolabelled somatostatin analog 177Lu-DOTATATE for advanced pancreatic neuroendocrine tumors. Neuroendocrinology 97:347–354. doi:10.1159/000348394
Valkema R, Pauwels SA, Kvols LK, Kwekkeboom DJ, Jamar F, de Jong M, Barone R, Walrand S, Kooij PP, Bakker WH et al (2005) Long-term follow-up of renal function after peptide receptor radiation therapy with 90Y-DOTA0, Tyr3-octreotide and 177Lu-DOTA0, Tyr3-octreotate. J Nucl Med 46:83S–91S
Pauwels S, Barone R, Walrand S, Borson-Chazot F, Valkema R, Kvols LK, Krenning EP, Jamar F (2005) Practical dosimetry of peptide receptor radionuclide therapy with 90Y-labeled somatostatin analogs. J Nucl Med 46:92S–98S
Kwekkeboom DJ, Kooij PP, Bakker WH, Mäcke HR, Krenning EP (1999) Comparison of 111In-DOTA-Tyr3-octreotide and 111In-DTPA-octreotide in the same patients: biodistribution, kinetics, organ and tumor uptake. J Nucl Med 40:762–767
Cremonesi M, Ferrari M, Zoboli S, Chinol M, Stabin MG, Orsi F, Maecke HR, Jermann E, Robertson C, Fiorenza M et al (1999) Biokinetics and dosimetry in patients administered with 111In-DOTA-Tyr3-octreotide: implications for internal radiotherapy with 90Y-DOTATOC. Eur J Nucl Med 26:877–886
Forrer F, Uusijärvi H, Waldherr C, Cremonesi M, Bernhardt P, Mueller-Brand J, Maecke HR (2004) A comparison of 111In-DOTATOC and 111In-DOTATATE: biodistribution and dosimetry in the same patients with metastatic neuroendocrine tumours. Eur J Nucl Med Mol Imaging 31:1257–1262. doi:10.1007/s00259-004-1553-6
Cremonesi M, Botta F, Di Dia A, Ferrari M, Bodei L, De Cicco C, Rossi A, Bartolomei M, Mei R, Severi SF et al (2010) Dosimetry for treatment with radiolabelled somatostatin analogues. A review. J Nucl Med Mol Imaging 54:37–51
Förster GJ, Engelbach MJ, Brockmann JJ, Reber HJ, Buchholz HG, Mäcke HR, Rösch FR, Herzog HR, Bartenstein PR (2001) Preliminary data on biodistribution and dosimetry for therapy planning of somatostatin receptor positive tumours: comparison of 86Y-DOTATOC and 111In-DTPA-octreotide. Eur J Nucl Med 28:1743–1750. doi:10.1007/s002590100628
Decristoforo C, Mather SJ, Cholewinski W, Donnemiller E, Riccabona G, Moncayo R (2000) 99mTc-EDDA/HYNIC-TOC: a new 99mTc-labelled radiopharmaceutical for imaging somatostatin receptor-positive tumours: first clinical results and intra-patient comparison with 111In-labelled octreotide derivatives. Eur J Nucl Med 27:1318–1325. doi:10.1007/s002590000289
Hubalewska-Dydejczyk A, Fröss-Baron K, Mikołajczak R, Maecke HR, Huszno B, Pach D, Sowa-Staszczak A, Janota B, Szybiński P, Kulig J (2006) 99mTc-EDDA/HYNIC-octreotate scintigraphy, an efficient method for the detection and staging of carcinoid tumours: results of 3 years’ experience. Eur J Nucl Med Mol Imaging 33:1123–1133. doi:10.1007/s00259-006-0113-7
Cwikla JB, Mikolajczak R, Pawlak D, Buscombe JR, Nasierowska-Guttmejer A, Bator A, Maecke HR, Walecki J (2008) Initial direct comparison of 99mTc-TOC and 99mTc-TATE in identifying sites of disease in patients with proven GEP NETs. J Nucl Med 49:1060–1065. doi:10.2967/jnumed.107.046961
Snyder WS, Ford MR, Warner GG, Watson SB (1975) S” absorbed dose per unit cumulated activity for selected radionuclides and organs. Mird pamphlet no.11. Society of Nuclear Medicine, New York
Snyder WS, Ford, Warner GG (1978) Estimates of specific absorbed fractions for photon sources uniformly distributed in various organs of a heterogeneous phantom. Mird PAMPHLET No. 5 Revised. Society of Nuclear Medicine, New York
Stabin MG, Siegel JA (2003) Physical models and dose factors for use in internal dose assessment. Health Phys 85:294–310
Stabin MG, Emmons MA, Segars WP, Fernald MJ (2009) The Vanderbilt University reference adult and pediatric phantom series. In: Xu XG (ed) Handbook of anatomical models for radiation dosimetry. Taylor & Francis, Philadelphia, pp 337–346
Stabin MG (2008) Update: the case for patient-specific dosimetry in radionuclide therapy. Cancer Biother Radiopharm 23:273–284. doi:10.1089/cbr.2007.0445
Shen S, Meredith RF, Duan J, Macey DJ, Khazaeli MB, Robert F, LoBuglio AF (2002) Improved prediction of myelotoxicity using a patient-specific imaging dose estimate for non-marrow-targeting (90)Y-antibody therapy. J Nucl Med 43:1245–1253
Council Directive 97/43/EURATOM (1997)
Helisch A, Förster GJ, Reber H, Buchholz HG, Arnold R, Göke B, Weber MM, Wiedenmann B, Pauwels S, Haus U et al (2004) Pre-therapeutic dosimetry and biodistribution of 86Y-DOTA-Phe1-Tyr3-octreotide versus 111In-pentetreotide in patients with advanced neuroendocrine tumours. Eur J Nucl Med Mol Imaging 31:1386–1392. doi:10.1007/s00259-004-1561-6
Garkavij M, Nickel M, Sjögreen-Gleisner K, Ljungberg M, Ohlsson T, Wingårdh K, Strand SE, Tennvall J (2010) 177Lu-[DOTA0, Tyr3] octreotate therapy in patients with disseminated neuroendocrine tumors: analysis of dosimetry with impact on future therapeutic strategy. Cancer 116:1084–1092. doi:10.1002/cncr.24796
Hindorf C, Glatting G, Chiesa C, Lindén O, Flux G (2010) EANM dosimetry committee guidelines for bone marrow and whole-body dosimetry. Eur J Nucl Med Mol Imaging 37:1238–1250. doi:10.1007/s00259-010-1422-4
Clairand I, Ricard M, Gouriou J, Di Paola M, Aubert B (1999) DOSE3D: EGS4 Monte Carlo code-based software for internal radionuclide dosimetry. J Nucl Med 40:1517–1523
Medicinsk strålningsfysik, Lund (2014) LundaDose program. http://www.msf.lu.se/forskning/nuclear-medicine-group/lundadose-program. Accessed on 19 Sept 2014
Barone R, Walrand S, Konijnenberg M, Valkema R, Kvols LK, Krenning EP, Pauwels S, Jamar F (2008) Therapy using labelled somatostatin analogues: comparison of the absorbed doses with 111In-DTPA-D-Phe1-octreotide and yttrium-labelled DOTA-D-Phe1-Tyr3-octreotide. Nucl Med Commun 29:283–290
Gabriel M, Decristoforo C, Donnemiller E, Ulmer H, Watfah Rychlinski C, Mather SJ, Moncayo R (2003) An intrapatient comparison of 99mTc-EDDA/HYNIC-TOC with 111In-DTPA-octreotide for diagnosis of somatostatin receptor-expressing tumors. J Nucl Med 44:708–716
González-Vázquez A, Ferro-Flores G, Arteaga de Murphy C, Gutiérrez-García Z (2006) Biokinetics and dosimetry in patients of 99mTc-EDDA/HYNIC-Tyr3-octreotide prepared from lyophilized kits. Appl Radiat Isot 64:792–797
Grimes J, Celler A, Birkenfeld B, Shcherbinin S, Listewnik MH, Piwowarska-Bilska H, Mikolajczak R, Zorga P (2011) Patient-specific radiation dosimetry of 99mTc-HYNIC-Tyr3-octreotide in neuroendocrine tumors. J Nucl Med 52:1474–1481. doi:10.2967/jnumed.111.088203
Chalkia MT, Stefanoyiannis AP, Prentakis A, Chatziioannou SN, Armeniakos I, Geronikola-Trapali X, Liotsou T, Efstathopoulos EP (2013) Patient-specific dosimetry of 99mTc-HYNIC-Tyr3-Octreotide in patients with neuroendocrine tumours. Annual Congress of the European Association of Nuclear Medicine, Lyon. http://www.eanm13.eanm.org/abstracts/abstract_search_result.php?navId=25)
de Jong M, Breeman W, Valkema R, Bernard BF, Krenning EP (2005) Combination radionuclide therapy using 177Lu and 90Y-labeled somatostatin analogs. J Nucl Med 46:13S–17S
Villard L, Romer A, Marincek N, Brunner P, Koller MT, Schindler C, Ng QK, Mäcke H, Müller-Brand J, Rochlitz C et al (2012) Cohort study of somatostatin-based radiopeptide therapy with [90Y-DOTA]-TOC versus [90Y-DOTA]-TOC plus [177Lu-DOTA]-TOC in neuroendocrine cancers. J Clin Oncol 30:1100–1106. doi:10.1200/JCO.2011.37.2151
Kunikowska J, Królicki L, Hubalewska-Dydejczyk A, Mikołajczak R, Sowa-Staszczak A, Pawlak D (2011) Clinical results of radionuclide therapy of neuroendocrine tumours with 90Y-DOTATATE and tandem 90Y/177Lu-DOTATATE: which is a better therapy option? Eur J Nucl Med Mol Imaging 38:1788–1797. doi:10.1007/s00259-011-1833-x
Berker Y, Goedicke A, Kemerink GJ, Aach T, Schweizer B (2011) Activity quantification combining conjugate-view planar scintigraphies and SPECT/CT data for patient-specific 3-D dosimetry in radionuclide therapy. Eur J Nucl Med Mol Imaging 38:2173–2185. doi:10.1007/s00259-011-1889-7
Emami B, Lyman J, Brown A, Coia L, Goitein M, Munzenrider JE, Shank B, Solin LJ, Wesson M (1991) Tolerance of normal tissue to therapeutic irradiation. Int J Radiat Oncol Biol Phys 21:109–122
Shen S, DeNardo GL, Yuan A, DeNardo DA, DeNardo SJ (1994) Planar gamma camera imaging and quantitation of yttrium-90 bremsstrahlung. J Nucl Med 35:1381–1389
Ito S, Kurosawa H, Kasahara H, Teraoka S, Ariga E, Deji S, Hirota M, Saze T, Minamizawa T, Nishizawa K (2009) (90)Y bremsstrahlung emission computed tomography using gamma cameras. Ann Nucl Med 23:257–267. doi:10.1007/s12149-009-0233-9
Minarik D, Sjögreen Gleisner K, Ljungberg M (2008) Evaluation of quantitative (90)Y SPECT based on experimental phantom studies. Phys Med Biol 53:5689–5703. doi:10.2967/jnumed.110.079897
Walrand S, Jamar F, van Elmbt L, Lhommel R, Bekonde EB, Pauwels S (2010) 4-Step renal dosimetry dependent on cortex geometry applied to 90Y peptide receptor radiotherapy: evaluation using a fillable kidney phantom imaged by 90Y PET. J Nucl Med 51:1969–1973. doi:10.2967/jnumed.110.080093
Minarik D, Sjögreen-Gleisner K, Linden O, Wingårdh K, Tennvall J, Strand SE, Ljungberg M (2010) 90Y bremsstrahlung imaging for absorbed-dose assessment in high-dose radioimmunotherapy. J Nucl Med 51:1974–1978. doi:10.2967/jnumed.110.079897
Selwyn RG, Nickles RJ, Thomadsen BR, DeWerd LA, Micka JA (2007) A new internal pair production branching ratio of 90Y: the development of a non-destructive assay for 90Y and 90Sr. Appl Radiat Isot 65:318–327
Lhommel R, van Elmbt L, Goffette P, Van den Eynde M, Jamar F, Pauwels S, Walrand S (2010) Feasibility of 90Y TOF PET-based dosimetry in liver metastasis therapy using SIR-Spheres. Eur J Nucl Med Mol Imaging 37:1654–1662. doi:10.1007/s00259-010-1470-9
de Jong M, Valkema R, van Gameren A, van Boven H, Bex A, van de Weyer EP, Burggraaf JD, Körner M, Reubi JC, Krenning EP (2004) Inhomogeneous localization of radioactivity in the human kidney after injection of [111In-DTPA]octreotide. J Nucl Med 45:1168–1171
Bouchet LG, Bolch WE, Blanco HP, Wessels BW, Siegel JA, Rajon DA, Clairand I, Sgouros G (2003) MIRD pamphlet No. 19: absorbed fractions and radionuclide S values for six age-dependent multiregion models of the kidney. J Nucl Med 44:1113–1147
Siegel JA, Stabin MG, Sharkey RM (2010) Renal dosimetry in peptide radionuclide receptor therapy. Cancer Biother Radiopharm 25:581–588. doi:10.1089/cbr.2010.0805
Valkema R, Pauwels S, Kvols LK, Barone R, Jamar F, Bakker WH, Kwekkeboom DJ, Bouterfa H, Krenning EP (2006) Survival and response after peptide receptor radionuclide therapy with [90Y-DOTA0, Tyr3]Octreotide in patients with advanced gastroenteropancreatic neuroendocrine tumors. Semin Nucl Med 36:147–156. doi:10.1053/j.semnuclmed.2006.01.001
Imhof A, Brunner P, Marincek N, Briel M, Schindler C, Rasch H, Mäcke HR, Rochlitz C, Müller-Brand J, Walter MA (2011) Response, survival, and long-term toxicity after therapy with the radiolabeled somatostatin analogue [90Y-DOTA]-TOC in metastasized neuroendocrine cancers. J Clin Oncol 29:2416–2423. doi:10.1200/JCO.2010.33.7873
Kontogeorgakos DK, Dimitriou PA, Limouris GS, Vlahos LJ (2006) Patient-specific dosimetry calculations using mathematic models of different anatomic sizes during therapy with 111In-DTPA-D-Phe1- octreotide infusions after catheterization of the hepatic artery. J Nucl Med 47:1476–1482
Chinol M, Bodei L, Cremonesi M, Paganelli G (2002) Receptor-mediated radiotherapy with 90Y-DOTA-DPhe1-Tyr3-octreotide: the experience of the European institute of oncology group. Semin Nucl Med 32:141–147. doi:10.1053/snuc.2002.31563
Rodrigues M, Traub-Weidinger T, Li S, Ibi B, Virgolini I (2006) Comparison of 111In-DOTA-DPhe1-Tyr3-octreotide and 111In-DOTA-lanreotide scintigraphy and dosimetry in patients with neuroendocrine tumours. Eur J Nucl Med Mol Imaging 33:532–540. doi:10.1007/s00259-005-0020-3
Hindorf C, Chittenden S, Causer L, Lewington VJ, Mäcke HR, Flux GD (2007) Dosimetry for 90Y-DOTATOC therapies in patients with neuroendocrine tumors. Cancer Biother Radiopharm 22:130–135. doi:10.1089/cbr.2007.306
Grassi E, Fioroni F, Sghedoni R, Sarti MA, Asti M, Fraternali A, Versari A, Salvo D, Borasi G (2008) Retrospective evaluation of 111In-DOTATOC dosimetry in patients enrolled for 90Y-DOTATOC PRRT. J Nucl Med 49:322P
Sandström M, Garske-Román U, Granberg D, Johansson S, Widström C, Eriksson B, Sundin A, Lundqvist H, Lubberink M (2013) Individualized dosimetry of kidney and bone marrow in patients undergoing 177Lu- DOTA-Octreotate treatment. J Nucl Med 54:33–41. doi:10.2967/jnumed.112.107524
Gupta SK, Singla S, Thakral P, Bal CS (2013) Dosimetric analyses of kidneys, liver, spleen, pituitary gland, and neuroendocrine tumors of patients treated with 177Lu-DOTATATE. Clin Nucl Med 38:188–194. doi:10.1097/RLU.0b013e3182814ac1
Schuchardt C, Kulkarni HR, Prasad V, Zachert C, Müller D, Baum RP (2013) The Bad Berka dose protocol: comparative results of dosimetry in peptide receptor radionuclide therapy using 177Lu-DOTATATE, 177Lu-DOTANOC, and 177Lu-DOTATOC. Recent Results Cancer Res 194:519–536. doi:10.1007/978-3-642-27994-2_30
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chalkia, M.T., Stefanoyiannis, A.P., Chatziioannou, S.N. et al. Patient-specific dosimetry in peptide receptor radionuclide therapy: a clinical review. Australas Phys Eng Sci Med 38, 7–22 (2015). https://doi.org/10.1007/s13246-014-0312-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13246-014-0312-7