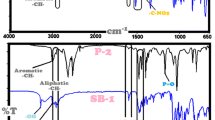
Abstract
In this paper, a series of Schiff bases and their polyazomethine derivatives were synthesized via condensation and oxidative polycondensation reactions of 4-aminophenol with 4-isopropylbenzaldehyde or 4-t-butylbenzaldehyde. Polymerization reaction conditions for polymerization yield of polyazomethines were investigated by changing some experimental parameters such as time and temperature using different oxidants like alkaline NaOCl (30%) and air. The structural characterizations of Schiff bases and polymers were performed using FT-IR, 1H NMR, 13C NMR and SEC techniques. Photophysical properties were studied UV-vis and photoluminescence spectroscopies. Electrochemical properties were also investigated cyclic voltammetry (CV) and electrochemical band gaps determined in the range 1.73 to 2.63 eV. Thermal properties of the compounds were investigated using TG-DTA and DSC techniques and char residue at 1000 oC determined between 3-6% and 39-34% for Schiff bases and polyazomethines, respectively. Moreover, conductivity measurements were done using four-point probe technique at both doped with iodine vapor and un-doped state.

Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
D. Sek, A. Iwan, B. Jarzabek, B. Kaczmarczyk, J. Kasperczyk, Z. Mazurak, M. Domanski, K. Karon, and M. Lapkowski, Macromolecules, 41, 6653 (2008).
S. Barik, S. Bishop, and W. G. Skene, Mater. Chem. Phys., 129, 529 (2011).
I. Kaya, M. Yildirim, and A. Avci, Synth. Met., 160, 911 (2010).
H. J. Kim, J. H. Lee, M. Lee, and T. S. Lee, React. Funct. Polym., 68, 1696 (2008).
M. F. Zaltariov, M. Cazacu, C. Racles, V. Musteata, A. Vlad, and A. Airine, J. Appl. Polym. Sci., 132, 41361 (2015).
M. F. Zaltariov, M. Cazacu, S. Shova, C. D. Varganici, L. Vacareanu, V. Musteata, and A. Airinei, Des. Monomers Polym., 17, 668 (2014).
M. Kamaci and I. Kaya, J. Taiwan Inst. Chem. Eng., 59, 536 (2016).
L. Sicard, D. Navarathne, T. Skalski, and W. G. Skene, Adv. Funct. Mater., 23, 3549 (2013).
A. Iwan, B. Boharewicz, I. Tazbir, M. Malinowski, M. Filapek, T. Kla, B. Luszczynska, I. Glowacki, K. P. Korona, M. Kaminska, J. Wojtkiewicz, M. Lewandowska, and A. Hreniak, Sol. Energy, 117, 246 (2015).
H. Bednarski, J. Gisiorowski, M. Domaski, B. Hajduk, J. Jurusik, B. Jarzibek, and J. Weszka, Acta Phys. Polym. A, 122, 1083 (2012).
H. Ma, H. L. Yip, F. Huang, and A. K. Y. Jen, Adv. Funct. Mater., 20, 1371 (2010).
C. Mallet, M. L Borgne, M. Starck, and W. G. Skene, Polym. Chem., 4, 250 (2013).
S. A. P. Guarín, M. Bourgeaux, S. Dufresne, and W. G. Skene, J. Org. Chem., 72, 2631 (2007).
S. Barik and W. G. Skene, Polym. Chem., 2, 1091 (2011).
S. A. P. Guarìn and W. G. Skene, Mater. Lett., 61, 5102 (2007).
S. Dineshkumar, A Muthusamy, and J. Chandrasekaran, J. Mol. Struct., 1128, 730 (2017).
D. Senol, F. Kolcu, and I. Kaya, J. Fluoresc., 26, 1579 (2016).
I. Kaya and M. Yildirim, Eur. Polym. J., 43, 127 (2007).
N. Y. Baran, M. Karakisla, H. O. Demir, and M. Saçak, J. Mol. Struct., 1123, 153 (2016).
I. Kaya, A. Bilici, and M. Sacak, Synth. Met., 159, 1414 (2009).
S. Dineshkumar, A. Muthusamy, P. Chitra, and S. Anand, J. Adhes. Sci. Technol., 29, 2605 (2015).
B. Jarzabek, B. Kaczmarczyk, J. Jurusik, M. Siwy, and J. Weszka, J. Non-Cryst. Solids, 375, 13 (2013).
M. Grigoras and C. O. Catanescu, J. Macromol. Sci., Part C: Polym. Rev., 44, 131 (2004).
A. Iwan and D. Sek, Prog. Polym. Sci., 33, 289 (2008).
S. Dufresne and W. G. Skene, J. Phys. Org. Chem., 25, 211 (2012).
S. G. Ciechanowicz, K. P. Korona, A. Wolos, A. Drabinska, A. Iwan, I. Tazbir, J. Wojtkiewicz, and M. Kaminska, J. Phys. Chem. C, 120, 11415 (2016).
S. Bronnikov, S. Kostromin, V. Mustea, and V. Cozan, Liq. Cryst., 42, 1102 (2015).
S. Culhaoglu and I. Kaya, Polym. Korea, 39, 225 (2015).
M. Kamaci and I. Kaya, J. Inorg. Organomet. Polym., 24, 803 (2014).
I. Kaya, M. Yildirim and M. Kamaci, Eur. Polym. J., 45, 1586 (2009).
A. Avci, M. Kamaci, I. Kaya, and M. Yildirim, Mater. Chem. Phys., 163, 301 (2015).
M. Kamaci, A. Avci, and I. Kaya, Prog. Org. Coat., 88, 325 (2015).
A. M. Navarchian, Z. Hasanzadeh, and M. Joulazadeh, Adv. Polym. Technol., 32, 21356 (2013).
D. V. Matyushov, R. Schmid, and B. M. Ladanyi, J. Phys. Chem. B, 101, 1035 (1997).
C. D. Hein, X. M. Liu, and D. Wang, Pharm. Res., 25, 2216 (2008).
M. Kamaci and I. Kaya, Polym. Eng. Sci., 54, 1664 (2014).
I. Kaya, M. Yildirim, and A. Avci, Synth. Met., 160, 911 (2010).
M. Yildirim and I. Kaya, Polym. Bull., 71, 3067 (2014).
M. Kamaci and I. Kaya, J. Fluoresc., 25, 1339 (2015).
M. Kamaci and I. Kaya, J. Inorg. Organomet. Polym., 25, 1250 (2015).
S. Afzal, A. Gul, and Z. Akhter, J. Inorg. Organomet. Polym., 24, 321 (2014).
D. Şenol, F. Kolcu, and İ. Kaya, J. Fluoresc., 26, 1579 (2016).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaya, İ., Gökpınar, M. & Kamacı, M. Reaction conditions, photophysical, electrochemical, conductivity, and thermal properties of polyazomethines. Macromol. Res. 25, 739–748 (2017). https://doi.org/10.1007/s13233-017-5072-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-017-5072-2