Abstract
The present study was aimed to evaluate the potential of petroleum ether and hydro-alcoholic extract of Linum usitatissimum (FPE and FHE) in STZ-nicotinamide induced diabetic nephropathy. GC–MS analysis of FPE revealed the presence of different fatty acids, heterocyclic compounds etc. Moreover, chromatography of FHE isolated Secoisolariciresinol diglycoside. After 30 days of STZ-administration, 100, 200 and 400 mg/kg of FPE and FHE were administered for 45 days. FPE and FHE produced significant attenuation in the glycemic status, renal parameter, lipid profile and level of antioxidant enzymes proving efficacy in diabetic nephropathy. Moreover, FPE and FHE produced significant reduction in the formation of AGEs in kidney. The results indicated that via amelioration oxidative stress and formation of AGEs, FPE and FHE produced significant nephroprotective effect in STZ- induced diabetic nephropathy in rats.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chronic hyperglycemia is the main culprit for the development of diabetic complications like diabetic nephropathy, diabetic neuropathy, diabetic retinopathy etc. Among these, diabetic nephropathy is the major cause of morbidity and mortality due to end stage renal disease (Pan et al. 2014). Diabetic nephropathy is a multistage condition characterized by decreased glomerular filtration rate, hypertrophy and increased renal blood flow (Thomas and Karalliedde 2015). Various metabolic hypotheses have been proposed for the development of diabetic complication among which polyol pathway is considered as a potential target for management of diabetic nephropathy. In polyol pathway, aldose reductase utilizes nicotinamide adenine dinucleotide phosphate (NADPH) to reduce glucose to sorbitol (Brownlee et al. 2008). In addition, two key mechanisms implicated in the development of diabetic nephropathy include advanced glycation and oxidative stress. Glycation of proteins leads to partial loss of protein activity and is one serious consequences of chronic hyperglycemia (Yamagishi and Matsui 2010; Tupe et al. 2015). Advanced glycation is the irreversible attachment of reducing sugars to amino groups of proteins to form advanced glycation end products (AGEs). The glycation reaction is subdivided into three main stages: early, intermediate and late (Baker et al. 1985). In the early stage, excessive glucose present in blood reacts with active amino groups of circulating proteins to yield chemically reversible glycosylation products (Schiff bases), fructosamines and Amadori products. In the intermediate stage, Amadori products degrade into a variety of carbonyl compounds (glyoxal, methylglyoxal, and deoxyglucosones). In the late stage, yellow–brown, often fluorescent, insoluble, irreversible advanced glycation end-products (AGEs) are formed. Furthermore, there is a growing body of evidence to indicate that glycated protein, AGEs and their signal-transducing receptor interactions induce the formation of reactive oxygen species, leading to concomitant oxidative stress and vascular inflammation, thereby playing a central role in the pathogenesis of various vascular complications in diabetes (Goh and Cooper 2008). Intracellular formation of AGEs can also cause generalized cellular dysfunction. Oxidative stress occurs as a result of the imbalance between ROS production and antioxidant defenses. Sources of ROS include the mitochondria, auto-oxidation of glucose, and enzymatic pathways including nicotinamide adenine dinucleotide phosphate reduced (NADPH) oxidase (Singh et al. 2013).
Normalization of glycemia, inhibition of aldose reductase, AGE formation inhibition and amelioration of oxidative stress reverses early changes of diabetic nephropathy (UK Prospective Diabetes Study (UKPDS) Group 1998). So, the management of hyperglycemia may pave a way to attenuation of diabetic nephropathy. But allopathic drugs used for the management of diabetes mellitus have plethora of side effects. So, the focus is shifted to herbal drug. But, herbal drugs which are considered safer than allopathic drugs are not purely devoid of side effects. So, food or food supplements have an upper edge in the management of diabetes and diabetic complications. Moreover, dietary measures are integral part of antidiabetic regimen. If dietary intervention could reduce the ensuing diabetic complications it can be a powerful adjunct to conventional antidiabetic treatment.
Linum usitatissimum L. (Linaceae), commonly known as Flaxseeds are used in daily food, preparation in many Asian countries. Various studies on the potential health benefits associated with consumption of flaxseed have increased in the past decade. These potential health benefits include anticancer effects, antiviral and bactericidal activity, anti-inflammatory activity, laxative and reduction of atherogenic risks (Cunnane et al. 1995; Thompson et al. 1996). Flaxseeds are a part of food in many parts of the world due to their dietary fiber content and other secondary plant metabolites viz, fixed oils, triterpenoids and polyphenolic compounds, lignans, α-linoleic acid etc. (Cunnane et al. 1995; Liggins et al. 2000). Flaxseeds inhibit glucosidase (Bhat et al. 2011) and porcine pancreatic α-amylase leading to the reduction in starch hydrolysis and eventually reduced glucose levels (Sudha et al. 2011). Moreover, the flaxseeds are reported to have renoprotective activity in ischemic reperfusion injury (Ghule et al. 2011). Flaxseed contains ω-3 fatty acids, lignans, and fiber that together may provide benefits to patients with cardiovascular disease. Circulating α-linolenic acid levels correlated with systolic and diastolic blood pressure, and lignan levels correlated with changes in diastolic blood pressure. Flaxseed showed one of the most potent antihypertensive effects achieved by a dietary intervention (Rodriguez-Leyva et al. 2013). Gok et al. 2016 investigated the effects of flaxseed intake on general metabolism, pentose phosphate pathway and glutathione-dependent enzymes in diabetic rats. The study concluded that Flaxseed has beneficial effects against diabetes-induced glucotoxicity by modulating glucose-6-phosphate dehydrogenase, 6-phosphogluconate dehydrogenase and glutathione reductase activities in tissues. Keeping these view points, present study was designed to evaluate the antihyperglycemic and renoprotective activity of petroleum ether and hydro-alcoholic (40%) extract of flaxseeds in diabetic nephropathy.
Materials and methods
Preparation of extract
Flaxseeds were procured from local supplier and authenticated by Dr. Sunita Garg, NISCAIR New Delhi. A voucher specimen (NISCAIR/RHMD/Consult/2013/2295/75) was deposited in the herbarium of NISCAIR, New Delhi for future reference. Seeds were powdered and extracted with Petroleum Ether 60–80 °C and hydro-alcoholic (40%) sequentially using soxhlet extraction method and extracts were stored at 4–8 °C until further use. The petroleum ether extract (FPE) was prepared in 0.25% w/v Tween-80 and hydroalcoholic extract (FHE) was redissolved in water.
Chemicals
Streptozotocin (STZ) was purchased from Sigma-Aldrich, Milwaukee, USA and Nicotinamide from Finar India Ltd. Diagnostic kits for the biochemical estimation were obtained from Reckon Diagnostics, India. All the other chemicals used were of analytical grade.
Quantitative analysis
Determination of total flavonoids
10 g of seed powder was extracted repeatedly with 100 ml of 80% aqueous methanol at room temperature. The whole solution was filtered through Whatman filter Paper No. 42. The filtrate was later transferred into a crucible and evaporated into dryness and weighed to a constant weight (Boham and Kocipal-Abyazan 1974).
Determination of total saponins
20 g of powdered seeds were taken in conical flask and 100 ml of 20% aq. ethanol was added. The mixture was heated over a hot water bath for 4 h with continuous stirring at about 55 °C. The mixture was filtered and the residue re-extracted with another 200 ml of 20% aq. ethanol. The combined extracts were reduced to 40 ml over water bath at about 90 °C. The concentrate was transferred into a 250 ml separating funnel and 20 ml of diethyl ether was added and shaken vigorously. The aqueous layer was recovered while the ether layer was discarded. The purification process was repeated. Then 60 ml n-butanol was added. The combined n-butanol extracts were washed twice with 10 ml of 5% aqueous sodium chloride. The remaining solution was heated in a water bath. After evaporation the sample was dried in the oven to a constant weight, the saponins content was calculated as percentage (Obadoni and Ochuko 2001).
Terpenoid extraction and TLC analysis
50 g of the powdered seeds were extracted with solvent combination of methanol and water (4:1) at room temperature for 24 h. The solution was filtered using Whatman filter Paper No. 1 and the filtrate was then evaporated to 1/10 volume at 40 °C. The remaining filtrate was acidified with 2 M sulphuric acid (pH 0.89) followed by chloroform extraction (three times the volume), stirred and allowed to stand in a separating funnel. Out of the two layers formed, the non-aqueous layer was taken and evaporated till dryness. The dried extract contained components like terpenoids which were further used for thin layer chromatography analysis. Glass plates (20 × 20 cm) were coated (0.5 mm) with silica gel (Qualigen fine chemicals) and samples (approximately 5 mg/ml) of each extract dissolved in methanol were applied. Chromatography was performed in 100% chloroform according to the method described by Harborne. The spots were visualized using concentrated sulphuric acid as a spray reagent followed by heating of plates at 100 °C for 10 min. The spots were identified based on the color, produced on reacting with a spray reagent (Harborne 1998).
Gas chromatography–mass spectrometry (GC–MS) analysis
FPE was subjected to GC–MS analysis. Samples were prepared by treating 10 mg of sample with 2 mL hexane followed by the addition of 0.2 mL of 2 M methanolic KOH. The tube was vortexed for 2 min at room temperature and after a light centrifugation aliquot of the hexane layer was collected for GC analysis. The column was held initially at 60 °C for 5 min after injection, then temperature was increased to 140 °C with 10 °C/min, heating ramp for 20 min and increased to 200 °C with 5 °C/min heating ramp for 20 min. Then temperature was then increased to 220 °C with 5 °C/min heating ramp for 20 min. Injector temperature 250 °C, detector temperature 275 °C, carrier gas H2: inlet pressure 45 psi linear, gas velocity 39 cm/s, column flow rate 2.4 mL/min; split ratio, 40:1 and injector volume 1μL.
Isolation of active compounds
The dried hydroalcoholic extract (10 g) was then subjected to column chromatography (silica gel packed column, Molychem 100–200 mesh, 160 g) by pre-adsorbing with silica gel. 5 g extract was eluted using chloroform (100%) and the mixture of chloroform and methanol up to 25%. The fractions (200 ml each) obtained from the column were collected and combined on monitoring TLC. At 20% (Fraction 80) yielded the compound.
In-vitro anti-glycation activity
In vitro antiglycation activity of FPE and FHE was evaluated by measuring their ability to inhibit the fluorescence of BSA (Matsuda et al. 2003). The reaction mixture of BSA (10 mg/ml), 1.1 M fructose in 0.1 M phosphate buffered-saline (PBS), pH 7.4 containing 0.02% sodium azide with or without extract (FPE and FHE; dissolved in PBS; 50–500 μg/ml) was incubated in darkness at 37 °C for 1, 2, 3, and 4 weeks. AGE formation was measured using Spectrofluorometer at an excitation wavelength 355 nm and emission wavelength 460 nm. Aminoguanidine (AG) was used as a positive control for this study. The percentage inhibition of AGE formation was determined by following formula:
Animals
Adult male Wistar rats weighing 250–300 g were housed in standard environmental conditions maintained at 23 ± 2 °C with 12 h light–dark cycle. Animals were fed with standard rodent diet and water ad libitum. Experimental protocol was approved by Institutional Animal Ethical Committee and the experiments were performed according to the guidelines of CPCSEA (MMCP/IAEC/13/11).
Induction of diabetic nephropathy
Diabetes was induced by a single i.p. of STZ (65 mg/kg) after 15 min of nicotinamide injection (230 mg/kg, i.p.). STZ was freshly prepared in 0.1 M citrate buffer (pH 4.5) and nicotinamide was prepared in normal saline. Diabetes mellitus was confirmed after 72 h of STZ injection and blood samples were collected via retro-orbital plexus. Rats with fasting blood glucose level more than 200 mg/dl were selected for the study. Doses of FPE and FHE (100, 200 and 400 mg/kg) were selected on the basis of oral acute toxicity studies reported in literature (Zanwar et al. 2011). After 30 days of STZ administration, different doses of FPE, FHE and Glimepride were continued for 45 days.
Rats were divided into nine groups and each group consists of six rats.
Group 1: normal control; Group 2: diabetic nephropathy (DN) control; Group 3: DN + 100 mg/kg FPE; Group 4: DN + 200 mg/kg FPE; Group 5: DN + 400 mg/kg FPE; Group 6: DN + 100 mg/kg FHE; Group 7: DN + 200 mg/kg FHE; Group 8: DN + 400 mg/kg FHE; Group 9: DN + 10 mg/kg Glimepride.
Rats were fasted overnight and blood samples were collected from retro-orbital plexus under light anesthesia. Biochemical estimation was done by measuring blood glucose level, serum insulin level, lipid profile (total cholesterol, triglyceride, HDL, LDL, VLDL levels), uric acid, urea, BUN and creatinine levels by using commercially available kits of Reckon Diagnostics Pvt. Ltd. Body weight was measured before the induction of diabetes and during the experimental period. Animals were sacrificed at the end of the study and tissues (liver, kidney and pancreas) were harvested and stored at −70 °C for histopathological studies.
Lipid peroxidation and activities of antioxidant enzymes in different tissues
Tissue (kidney, pancreas and liver) homogenate was used to estimate thiobarbituric acid reactive substances (TBARS) (Ohkawa et al. 1979) and level of antioxidant enzymes, viz. superoxide dismutase (SOD) and reduced glutathione (GSH) (Beutler et al. 1963).
AGEs estimation in kidneys
AGEs level in kidneys were determined by a method as previously described by Sensi et al. 1996. Briefly, perfused kidneys were homogenized in 2 ml of 0.25 M sucrose followed by centrifugation at 900 g at 5 °C and the supernatant was separated. The pellet was resuspended in 2 ml sucrose solution and centrifuged and the supernatant obtained was mixed with the previous one. The proteins present were precipitated by adding equal volume of trichloroacetic acid (TCA). After centrifugation at 4 °C with 900 g, the protein pellet obtained was mixed with 1 ml methanol twice to remove the lipid fraction. The insoluble protein, after washing with 10% cooled TCA was centrifuged and the residue was solubilized in 1 ml of 1 N NaOH and the protein concentration was estimated by measuring the absorbance at 280 nm against BSA standard curve. The AGEs content was then measured fluorometrically with an emission at 440 nm and excitation at 370 nm, and the results were expressed as relative fluorescence units (RFU)/mg protein.
Histopathology
Liver, kidney and pancreas were harvested from the animals and fixed in 10% neutral buffered formalin solution, dehydrated in ethanol and embedded in paraffin. Sections of 5 μm thickness were prepared using a rotary microtome and stained with hematoxylin and eosin (H & E) dye for histopathological examination.
Statistical analysis
Statistical analysis was performed using Graphpad Prism 6. Values were expressed as mean ± SEM and one way analysis of variance (ANOVA) was used for statistical analysis. ANOVA was followed by Tukey’s as post hoc multiple comparison test. The results were considered statistically significant if p ≤ 0.05.
Results
Quantitative analysis
The quantity of flavonoids, saponins and terpenoids was found in the following order 4.3% > 1.3% > 0.03% respectively. The terpenoidal extract when further subjected to TLC analysis using 100% chloroform showed 2 spots out of which spot 1 was brown and spot 2 was yellowish in color suggesting that spot 1 was limonene or α- pinene, and spot 2 of Pulgeone (Harborne 1984).
Gas chromatography mass spectroscopy analysis
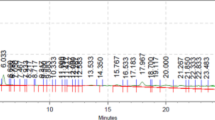
GC–MS analysis was conducted on the crude petroleum ether extract of Flaxseeds. The peaks in the chromatogram were integrated and compared with the database of spectra of known components (NIST-14) stored in the GC–MS library. Detailed tabulations of the GC–MS analysis of the extract is given in Table 1. Flaxseed extract revealed the presence of different fatty acids, heterocyclic compounds etc. The flaxseed extract showed 10 peaks in the GC–MS chromatogram indicating the presence of 10 phytochemical constituents.
Secoisolariciresinol diglycoside from L. usitatissimum (Flaxseed)
Chemical tests of phenolics were found positive and significant in hydroalcoholic extract of Linum usitatissimum. The hydroalcoholicic extract was subjected to column chromatography and its 20% (chloroform: methanol; 20: 80) fraction showed single spot on TLC and also have positive tests for phenolics. The UV λmax (265 & 358) and IR peaks (cm−1) [3391 (-OH), 2939 (C-H), 1637 (C = C), 1428 (C–C), 1346, 1279, 1240, 1208, 1130, 1067, 1052, 991, 941, 921, 867, 849, 682, 584, 472] showed presence of phenolics. The LC–MS Q-TOF micromass fragmentation pattern [704 (M++NH4); 687 (M++1), 524.3 (Secoisolariciresinol glycoside; 381.1, 441), 362.1 (Secoisolariciresinol; 325, 319, 281, 273, 247, 199, 183, 133, 73; Ricci and Piccolella 2012; Ghose et al. 2014] provides the confirmation of Secoisolariciresinol diglycoside (Fig. 1).
AGEs inhibition activity
In the present study, the formation of AGEs was monitored weekly by measuring fluorescence intensity of the BSA-fructose solutions for 4 weeks. A significant inhibition of AGEs formation (93.37%) was observed in fructose-induced glycated BSA plus aminoguanidine (500 μg/ml). In the 1st week, % inhibition of AGEs formation using FPE (50–500 μg/ml) was 23.54% whereas in the 4th week, the percentage inhibition was found to be 83.97%. Similarly, FHE % inhibition in the 1st week found to be 28.44% and in 4th week it was 91.74%.
Effect of FPE and FHE on body weight
Significant increase in reduced body weight was observed in DN control rats as compared to control animals. Whereas, administration of FPE and FHE (100, 200 and 400 mg/kg) in rats produced significantly (p < 0.001) reduced the weight loss as compared to DN control rats. Different doses of FHE (100, 200 and 400 mg/kg) also produced significant elevation in BW (6.55, 9.5 and 14.61% respectively) in comparison to DN control rats (Fig. 2a).
Effect of FPE and FHE on fasting blood glucose level
Administration of different doses of extracts was started after 30 days of STZ administration and continued for up to 75th day. Serum blood glucose level of animals was measured on 30th, 45th, 60th and 75th day of study. Fasting blood glucose level of control rats ranged from 93.68 to 96.03 mg/dl in 75 days. Whereas, in diabetic rats fasting glucose level was increased to 521.73 mg/dl from 325.82 mg/dl. Administration of different doses of FPE and FHE (100, 200 and 400 mg/kg) for 45 days, produced significant attenuation in blood glucose level and maximum attenuation was observed with 400 mg/kg of FHE as compared to DN control rats (Fig. 2b). Glimepride treatment also resulted in significant attenuation of blood glucose level in rats.
Effect of FPE and FHE on fasting insulin level
Administration of STZ produced a significant decrease in fasting insulin level (7.05 ± 0.209 μU/ml). Whereas administration of 100, 200 and 400 mg/kg of FPE and FHE (Fig. 2c) for 45 days produced significant increase in serum insulin level [FPE (8.20 ± 0.108, 9.45 ± 0.068, 9.45 ± 0.082 μU/ml respectively); FHE (9.23 ± 0.111, 10.33 ± 0.059, 12.92 ± 0.081 μU/ml respectively].
Effect of FPE and FHE on serum urea, uric acid, creatinine and BUN
Administration of STZ resulted in induction of diabetic nephropathy which can be assessed by the elevated level of serum urea, uric acid, creatinine and BUN as compared to normal control rats (Table 2). Whereas, administration of different doses of FPE, FHE (100, 200 and 400 mg/kg) significantly decreased the elevated level of serum urea, uric acid, creatinine and BUN. Moreover, attenuating effect was found to be dose dependent and 400 mg/kg of FPE and FHE produced maximum decrease in the level of markers of diabetic nephropathy. Glimepride also significantly attenuated the elevated level of these parameters.
Effect of FPE and FHE on serum lipid profile
Administration of STZ also produced dyslipidemia depicted in the form of elevated serum TC, TG, LDL, VLDL and reduced HDL level. 100, 200 and 400 mg/kg of FPE and FHE produced dose dependent attenuation in TC. Moreover, 400 mg/kg of FPE and FHE produced 56.94 and 64.40% respectively decrease in TC as compared to DN rats. Similarly, TG level was also found to be attenuated (120.08, 127.78, 107.24 mg/dl and 103.94, 90.86 and 79.70 mg/dl) by administration of different doses of (100, 200 and 400 mg/kg) of FPE and FHE respectively. Attenuation was also observed in level of LDL and VLDL at 100, 200 and 400 mg/kg of FPE, FHE. Serum HDL-c level was significantly reduced in diabetic nephropathy control rats whereas the level of HDL-c was found to be significantly elevated in FPE, FHE (100, 200 and 400 mg/kg) treated animals (Table 3).
Effect of FPE and FHE on antioxidant enzymes and TBARS
Oxidative stress ensues with the reduction of antioxidant enzymes and increased lipid peroxidation. Level of antioxidant enzymes (SOD and GSH) reduced significantly in pancreas, kidney and liver of diabetic nephropathy rats. Treatment with 100, 200 and 400 mg/kg of FPE and FHE significantly increased the level of antioxidant enzymes viz. SOD and GSH compared to DN rats. Level of TBARS was found to be elevated in DN rats as compared to normal control group whereas administration of FPE and FHE significantly reduced the level of TBARS in comparison to DN rats (Table 4).
Effect of FPE and FHE on AGEs in kidneys
Administration of STZ led to a significant increase in formation of AGEs in kidney of diabetic rats. FPE, FHE (100, 200 and 400 mg/kg) and Glimepride significantly inhibited the formation of AGEs when compared to diabetic control group (Table 4).
Histopathological studies of liver, kidney and pancreas
Kidney of normal control animals showed normal renal parenchyma with renal glomeruli as a glomerulus and Bowman’s capsule and surrounded by proximal and distal tubules. Kidney of DN rats showed mesangial expansion and thickening of glomerular capillaries. Glomeruli infiltrated by inflammation cells along with infiltration seen in cortex and medulla area. Atrophy of glomeruli was seen in STZ induced diabetic rats. In Glimepride treatment group, necrotic condition was reduced in convoluted tubules with reduced infiltration of inflammation cells in cortex and medulla. Flaxseed extracts treated group also showed protection viz, reduction in mesangial expansion, membrane thickness and atrophy (Fig. 3a).
Histopathological changes in a Kidney; b Liver; c Pancreatic islet of normal and treated rats (H&E × 100); “a” shows the structure of β-cells. (A) Normal, (B) diabetic nephropathy control, (C) standard, (D) FPE 100 mg/kg treated, (E) FPE 200 mg/kg, (F) FPE 400 mg/kg treated, (G) FHE 100 mg/kg treated, (H) FHE 200 mg/kg treated and (I) FHE 400 mg/kg treated
Normal central vein with radiating sinusoid cords were present in Liver of normal rats. There was no sinusoid congestion; swelling and necrotic cells. DN rats demonstrated perivenular inflammatory collection and hyperplasia of kupffer cell with condensed nuclei and fatty infiltration. These pathological changes were reversed by FPE and FHE extracts administered DN rats (Fig. 3b).
Pancreatic cells of normal control group showed normal architecture with normal acini and islets cells with no signs of edema and inflammation. In DN rats, inflammation, disorganization of the islets and steatosis were observed. Cell infiltration was seen in the acinar cells along with necrosis and shrinkage of islet cells. Treatment with FPE and FHE extracts and Glimepride, showed protective effect on islets of Langerhans and acinar cells as compared to diabetic rats and further reduction in edema, inflammation and shrinkage of islets (Fig. 3c).
Discussion
Hyperglycemia is the vital initiator of diabetic complications and instigates the activation of various metabolic pathways including polyol pathway and formation of AGEs. Excessive formation of AGEs under hyperglycemic condition plays a major role in the pathogenesis of diabetic nephropathy. Hyperglycemia leads to increased glucose level in tissues viz., kidneys which in turn leads to accumulation of AGEs. Accumulation of AGEs has been implicated in structural alterations in extracellular matrix leading to decreased glomerular filtration rate. Advanced glycation is the only pathway by which renal injury can be induced in diabetes (Forbes et al. 2003). The present study demonstrates that FPE and FHE attenuated the formation of AGEs in vitro which prompted us to evaluate the effect of FPE and FHE on diabetic nephropathy.
STZ has been widely used to induce diabetes in the experimental animals. It produces diabetes via its cytotoxic effects on pancreatic β-cells (Kim et al. 2003). The cytotoxic action of STZ in rats produces hyperglycemia, oxidative stress and renal injuries which closely reflect the natural progression and metabolic characteristics of diabetic nephropathy. Moreover, excessive production of reactive oxygen species ensues oxidative stress which plays a key role in the development of diabetic nephropathy (Singh et al. 2013). Along with hyperglycemia and oxidative stress, induction of diabetic nephropathy with STZ is associated with characteristic weight loss due to muscle wasting and catabolism of tissue proteins (Swanston-Fiatt et al. 1990).
The increased levels of fasting blood glucose in STZ-induced DN rats were lowered by the administration of FPE and FHE in dose and time dependent manner. The reduced glucose levels might be suggested by insulin like effect on peripheral tissues by either promoting glucose uptake metabolism by inhibiting hepatic gluconeogenesis, or by absorption of glucose into the muscle and adipose tissues, through the stimulation of a regeneration process and revitalization of the remaining beta cells (Kaur et al. 2016). Moreover, oral administration of FPE and FHE effectively reversed body weight loss by reversing muscle wasting and protein loss and also significantly attenuated hyperglycemia as compared to DN rats.
Hyperglycemia is responsible for diminution of antioxidant enzymes viz. SOD and GSH. LPO is another process which involves a source of secondary free radical; thus, it acts as a second messenger or can directly react with other biomolecules, further enhancing the biochemical lesions. Moreover, Administration of different doses of FPE and FHE ameliorated the oxidative stress via increasing level of antioxidant enzymes.
Phytochemical screening revealed that fatty acids, triterpenoid, phenolic compounds and saponins are present in FPE and FHE. Flaxseeds are grouped as “functional food and/or endocrine active food” by virtue of the presence of physiologically active food components that may provide health benefits beyond basic nutrition (Hasler et al. 2000). Trans fatty acids present in flaxseed oil offers an effective dietary strategy for the prevention of atherosclerotic cardiovascular disease (Bassett et al. 2011) and decrease postprandial glucose responses. Flaxseeds are richest source of plant lignans. The lignan SDG is present in greater quantity in the seed coat. In humans, ingested SDG is converted by bacteria in colon to biologically active lignans enterodiol and enterolactone (Hano et al. 2006). These SDG metabolites possess antioxidant activity and have been shown to effectively inhibit the development of type 1 and 2 diabetes (Prasad 2000). Effect of FHE on amelioration of DN can thus be attributed to the presence of SDG in the extract.
Diabetic Nephropathy is known by not only dysfunctional glucose metabolism but also dyslipidemia, especially in type 2 diabetes. Lipid abnormalities including increased level of TC, TG, LDL, VLDL and decreased HDL-c level predisposes diabetic patient to atherosclerosis and other cardiovascular complications like coronary heart disease (Thomas and Karalliedde 2015). Hyperglycemia, atherosclerosis and dyslipidemia are major causes of cardiovascular diseases and are linked to have interrelation with free radical generation (Singh et al. 2015). Similarly, in the present study, a positive correlation between hyperglycemia and dyslipidemia was observed. Serum TC, TG LDL and VLDL levels were found to be significantly elevated whereas HDL levels decreased. This dyslipidemia occurs due to increased breakdown of lipid and free fatty acids from peripheral stores. Moreover, hyperlipidemia with elevated levels of triglycerides, and free fatty acids results in oxidative stress and inflammation and may independently potentiate the adverse effects of hyperglycemia (O’Keefe and Bell 2007). Administration of FPE and FHE significantly reduced TC, TG, LDL and VLDL levels. Moreover, FPE and FHE effectively increased the level of serum HDL-c. Dyslipidemic effect of flaxseeds can be attributed to the presence of SDG (Prasad 2008).
DN is one of the most serious complications of diabetes and it is characterized by elevated the level of serum creatinine, blood urea nitrogen, creatinine clearance as well as kidney hypertrophy (Cowie et al. 1989). Hyperglycemia could enhance glomerulosclerosis and accelerate the progress of DN. These changes are a result of abnormal glucose regulation, hemodynamic changes within the kidney and increased oxidative stress (Aurell and Bjorck 1992). Free radicals attack important macromolecules leading to cell damage and homeostatic disruption, evidently leading to the degenerative diseases that afflict humanity (Tomás-Zapico and Coto-Montes 2007). DN is characterized by a series of renal structure abnormality including basement membrane thickening, mesangial expansion, glomerulosclerosis and tubulointerstitial fibrosis (Abe et al. 2011). These changes are attributed to persistent hyperglycemia and increased levels of BUN, creatinine, urea and uric acid. In the present study, histopathological observations of kidney showed glomeruli with mesangiocapillary proliferation along with amelioration of hemodynamic parameters of kidney. Liver showed perivenular inflammatory collection. Increased oxidative stress due to excess generation of free radicals, lipid peroxidation and decreased level of antioxidant enzymes causes deleterious structural changes in the liver of diabetic nephropathy rats (Polavarapu et al. 1998). The ultra structure of diabetic pancreas showed considerable reduction in the islet langerhans and depleted islets. Induction of hyperglycemia by STZ also produced similar structural alterations in liver, kidney and pancreas of experimental animals in the present study. Interaction between metabolic and hemodynamic factors adds to the deleterious effects of diabetes nephropathy (Cooper 2001). Good glycemic control ameliorates progression of diabetic nephropathy on both type 1 and 2 diabetes aided by slow development of kidney lesions (Thomas and Karalliedde 2015). Saponins isolated from medicinal plants are found to be renoprotective as they reduce fasting blood glucose and albuminuria, reverses the glomerular hyper-filtration state and ameliorates proliferative glomerular pathological changes during the early stages of diabetic nephropathy in rat models (Zhang et al. 2009). Treatment of DN rats with FPE and FHE effectively reduced the levels of urea, uric acid, creatinine and BUN indicating their increased clearance from kidney. Administration of FPE and FHE reversed these structural alterations in liver, kidney and pancreas of experimental animals. Moreover, administration of FPE and FHE also led to significant reduction in the level of AGEs in kidneys.
Conclusion
It can be concluded that FPE and FHE possess potential renoprotective activity. FPE and FHE ameliorated diabetic nephropathy by attenuating hyperglycemia and oxidative stress. Moreover, markers of diabetic nephropathy (uric acid, urea, creatinine and BUN level) and lipid profile was also improved after the administration of FPE and FHE in diabetic animals.
Abbreviations
- ANOVA:
-
Analysis of variance
- BUN:
-
Blood urea nitrogen
- FHE:
-
Flaxseed hydroalcoholic extract
- FPE:
-
Flaxseed petroleum ether extract
- GSH:
-
Glutathione
- HDL:
-
High density lipoproteins
- LDL:
-
Low density lipoproteins
- VLDL:
-
Very low density lipoproteins
- i.p. :
-
Intraperitoneal administration
- LPO:
-
Lipid peroxidation
- MAPKs:
-
Mitogen activated protein kinases
- SOD:
-
Superoxide dismutase
- SDG:
-
Secoisolariciresinol diglycoside
- STZ:
-
Streptozotocin
- TBARS:
-
Thiobarbituric acid reactive substances
- TNF-α:
-
Tumor necrosis factor-α
References
Abe H, Matsubara T, Arai H, Doi T (2011) Role of Smad1 in diabetic nephropathy: molecular mechanisms and implications as a diagnostic marker. Histol Histopathol 26:531–541
Aurell M, Bjorck S (1992) Determination of progressive renal disease in diabetes mellitus. Kidney Int 41:38–42
Baker JR, Metcalf PA, Johnson RN, Newman D, Rietz P (1985) Use of protein-based standards in automated colorimetric determinations of fructosamine in serum. Clin Chem 31:1550–1554
Bassett CM, McCullough RS, Edel AL, Patenaude A, LaVallee RK, Pierce GN (2011) The α-linolenic acid content of flaxseed can prevent the atherogenic effects of dietary trans fat. Am J Physiol Heart Circ Physiol 301(6):H2220–H2226
Beutler E, Duron O, Kelly BM (1963) Improved method for the determination of blood glutathione. J Lab Clin Med 61:882–890
Bhat M, Zinjarde SS, Bhargava SY, Kumar AR, Joshi BN (2011) Antidiabetic Indian plants: a good source of potent amylase inhibitors. Evid Based Complement Alternat Med 2011:810207
Boham BA, Kocipal-Abyazan R (1974) Flavonoids and condensed tannins from leaves of Hawaiian vaccinium vaticulatum and V. calycinum. Pac Sci 48:458–463
Brownlee M, Aiello LP, Cooper ME, Vinik AI, Nesto RW, Boulton AJM (2008) Com-plications of diabetes mellitus. In: Kronenberg HM, Melmed S, Polonsky KS, Larsen PR (eds) Williams textbook of endocrinology. Saun-ders Elsevier, Philadelphia, pp 1417–1501
Cooper ME (2001) Interaction of metabolic and haemodynamic factors in mediating experimental diabetic nephropathy. Diabetologia 44:1957–1972
Cowie CC, Port FK, Wolfe RA, Savage PJ, Moll PP, Hawthorne VM (1989) Disparities in incidence of diabetic end-stage renal disease according to race and type of diabetes. N Engl J Med 321:1074–1079
Cunnane SC, Hamadeh MJ, Liede AC, Thompson LU, Wolever TMS, Jenkins DJA (1995) Nutritional attributes of traditional flaxseed in healthy young adults. Am J Clin Nutr 61:62–68
Forbes JM, Cooper ME, Oldfield MD, Thomas MC (2003) Role of advanced glycation end products in diabetic nephropathy. J Am Soc Nephrol 14:S254–S258
Ghose K, Selvaraj K, McCallum J, Kirby CW, Sweeney-Nixon M, Cloutier SJ, Deyholos M, Datla R, Fofana B (2014) Identification and functional characterization of a flax UDP-glycosyltransferase glucosylating secoisolariciresinol (SECO) into secoisolariciresinol monoglucoside (SMG) and diglucoside (SDG). BMC Plant Biol 14(1):1
Ghule AE, Jadhav SS, Bodhankar SL (2011) Renoprotective effect of Linum usitatissimum seeds through haemodynamic changes and conservation of antioxidant enzymes in renal ischaemia-reperfusion injury in rats. Arab J Urol 9:215–221
Goh SY, Cooper ME (2008) The role of advanced glycation end products in progression and complications of diabetes. J Clin Endocrinol Metab 93:1143–11452
Gok M, Ulusu NN, Tarhan N, Tufan C, Ozansoy G, Arı N, Karasu C (2016) Flaxseed protects against diabetes-induced glucotoxicity by modulating pentose phosphate pathway and glutathione-dependent enzyme activities in rats. J Diet Suppl 13(3):339–351
Hano C, Martin I, Fliniaux O, Legrand B, Gutierrez L, Arroo RR et al (2006) Pinoresinol-lariciresinol reductase gene expression and secoisolariciresinol diglucoside accumulation in developing flax (Linum usitatissimum) seeds. Planta 224:1291–1301
Harborne JB (1984) Phytochemical methods: a guide to modern techniques of plant analysis, vol 3, 2nd edn. Chapman and Hall, New York, pp 100–117
Harborne JB (1998) Phytochemical methods: a guide to modern techniques of plant analysis. Chapman and Hall, London
Hasler CM, Kundrat S, Wool D (2000) Functional foods and cardiovascular disease. Curr Atheroscler Rep 2:467–475
Kaur N, Kishore L, Singh R (2016) Attenuating diabetes: What really works? Curr Diabetes Rev 12:259–78
Kim MJ, Ryu GR, Chung JS, Sim SS, Min Dos Rhie DJ, Yoon SH, Jo YM (2003) Protective effect of epicatechin against the toxic effects of STZ on rat pancreatic islets: in vivo and in vitro. Pancreas 26:292–299
Liggins J, Grimwood R, Bingham SA (2000) Extraction and quantification of lignan phytoestrogens in food and human samples. Anal Biochem 287:102–109
Matsuda H, Wang T, Managi H, Yoshikawa M (2003) Structural requirements of flavonoids for inhibition of protein glycation and radical scavenging activities. Bioorg Med Chem 11:5317–5323
O’Keefe JH, Bell DSH (2007) Postprandial hyperglycemia/hyperlipidemia (postprandial dysmetabolism): is a cardiovascular risk factor. Am J Cardiol 100:899–904
Obadoni BO, Ochuko PO (2001) Phytochemical studies and comparative efficacy of crude extracts of some homeostatic plants in Edo and Delta States of Nigeria. Glob J Pure Appl Sci 8:203–208
Ohkawa H, Ohishi N, Yagi K (1979) Assay of lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Pan D, Zhang D, Wu J, Chen C, Xu Z, Yang H, Zhou P (2014) A novel proteoglycan from Ganoderma lucidum fruiting bodies protects kidney function and ameliorates diabetic nephropathy via its antioxidant activity in C57BL/6 db/db mice. Food Chem Toxicol 63:111–118
Polavarapu R, Spitz DR, Sim JE, Follansbee MH, Oberley LW, Rahemtulla A, Nanji AA (1998) Increased lipid peroxidation and impaired antioxidant enzyme function is associated with pathological liver injury in experimental alcoholic liver disease in rats fed diets high in corn oil and fish oil. Hepatology 27:1317–1323
Prasad K (2000) Antioxidant activity of secoisolariciresinol diglucosidederived metabolites, secoisolariciresinol, enterodiol, and enterolactone. Int J Angiol 9:220–225
Prasad K (2008) Regression of hypercholesterolemic atherosclerosis in rabbits by secoisolariciresinol diglucoside isolated from flaxseed. Atherosclerosis 197:34–42
Ricci A, Piccolella S (2012) From the collisionally induced dissociation to the enzyme-mediated reactions: the electron flux within the lignan furanic ring. Tandem Mass Spectrom Appl Princ 26:619–634 (InTech, ISBN: 978-953-51-0141-3)
Rodriguez-Leyva D, Weighell W, Edel AL, LaVallee R, Dibrov E, Pinneker R, Maddaford TG, Ramjiawan B, Aliani M, Guzman R, Pierce GN (2013) Potent antihypertensive action of dietary flaxseed in hypertensive patients. Hypertension 62:1081–1089
Sensi M, Pricci F, Pugliese G, De Rossi MG, Petrucci AF, Cristina A et al (1996) Role of advanced glycation end-products (AGE) in late diabetic complications. Diabetes Res Clin Pract 28:9–17
Singh R, Kaur N, Kishore L, Gupta GK (2013) Management of diabetic complications: a chemical constituents based approach. J Ethnopharmacol 150:51–70
Singh R, Devi S, Gollen R (2015) Role of free radical in atherosclerosis, diabetes and dyslipidaemia: larger-than-life. Diabetes Metab Res Rev 31:113–126
Sudha P, Zinjarde SS, Bhargava SY, Kumar AR (2011) Potent α-amylase inhibitory activity of Indian Ayurvedic medicinal plants. BMC Complement Altern Med 11:5
Swanston-Fiatt SK, Day C, Bailey CJ, Flatt PR (1990) Traditional plant treatments for diabetes: studies in normal and streptozotocin diabetic mice. Diabetologia 33:462–464
Thomas S, Karalliedde J (2015) Diabetic nephropathy. Medicine 43:20–25
Thompson LU, Seidl MM, Rickard SE (1996) Antitumorigenic effect of a mammalian lignan precursor from flaxseed. Nutr Cancer 26:159–165
Tomás-Zapico C, Coto-Montes A (2007) Melatonin as antioxidant under pathological processes. Recent Pat Endocr Metab Immune Drug Discov 1:63–82
Tupe RS, Sankhe NM, Shaikh SA, Phatak DV, Parikh JU, Khaire AA, Kemse NG (2015) Aqueous extract of some indigenous medicinal plants inhibits glycation at multiple stages and protects erythrocytes from oxidative damage-an in vitro study. J Food Sci Technol 52(4):1911–1923
UK Prospective Diabetes Study (UKPDS) Group (1998) Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 352:837–853
Yamagishi S, Matsui T (2010) Advanced glycation end products, oxidative stress and diabetic nephropathy. Oxid Med Cell Longev 3:101–108
Zanwar AA, Hegde MV, Bodhankar SL (2011) Cardioprotective activity of flax lignan concentrate extracted from seed of Linum usitatissimum in isoprenalin induced myocardial necrosis in rats. Interdiscip Toxicol 4(2):90–97
Zhang J, Xie X, Li C, Fu P (2009) Systematic review of the renal protective effect of Astragalus membranaceus (root) on diabetic nephropathy in animal models. J Ethnopharmacol 126:189–196
Acknowledgements
Financial assistance (F. NO. SB/FT/LS-359/2012) from Department of Science and Technology, New Delhi, Government of India is highly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaur, N., Kishore, L. & Singh, R. Therapeutic effect of Linum usitatissimum L. in STZ-nicotinamide induced diabetic nephropathy via inhibition of AGE’s and oxidative stress. J Food Sci Technol 54, 408–421 (2017). https://doi.org/10.1007/s13197-016-2477-4
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-016-2477-4