Abstract
Banana (Musa sp var ‘Robusta’) fruits harvested at 75–80% maturity were dip treated with different concentrations of ethrel (250–1,000 ppm) solution for 5 min. Ethrel at 500 ppm induced uniform ripening without impairing taste and flavour of banana. Untreated control banana fruits remained shriveled, green and failed to ripen evenly even after 8 days of storage. Fruits treated with 500 ppm of ethrel ripened well in 6 days at 20 ± 1 °C. Changes in total soluble solids, acidity, total sugars and total carotenoids showed increasing trends up to 6 days during ripening whereas fruit shear force values, pulp pH and total chlorophyll in peel showed decreasing trends. Sensory quality of ethrel treated banana fruits (fully ripe) were excellent with respect to external colour, taste, flavour and overall quality.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
India is the largest producer of banana (Musa sp) in the world with an annual production of 23.205 million MT from an area of 0.647 million ha (NHB 2009). Banana var. ‘Robusta’ is one of the important tropical fruits grown commercially in India and is produced in large quantities throughout the year. Banana is a climacteric fruit and after harvest it exhibits a respiratory peak during natural ripening at 20 °C. Most of the banana bunches are harvested at complete maturity while they are green and unripe and subsequently allowed to ripen at ambient conditions. To meet the regular domestic requirement, green banana bunches are artificially ripened. Smoke treatment is one of the most common methods adopted to induce ripening (Ram et al. 1979). In this method, banana bunches are exposed to smoke generated by burning kerosene stove inside the airtight chambers for 24 h. As a result, the temperature inside the chamber also increases besides evolving ethylene gas with traces of other gases like acetylene and carbon monoxide. The ethylene gas triggers the enzymatic reactions in banana fruits causing early and uniform ripening with development of yellow colour on the skin along with its flavour. However, this type of smoke treatment is crude and ineffective as controlled ethylene treatment and also evolution of carbon monoxide is hazardous to health. Improper smoke treatment leads to uneven ripening and also poor external colour (yellow) development since the optimum temperature and relative humidity (RH) are not maintained in the ripening chamber. Otherwise generally, bananas are artificially ripened in local markets by the use of banned chemical calcium carbide. The use of this chemical is prohibited due to health reasons (PFA 2003). Extensive work has been done to study the effect of chemical substances to induce ripening (Singh et al. 1977; Nicholas and Thompson 1987). Commercially ethylene gas is used to ripen banana. But ethylene gas may be prove explosive when it reaches higher concentration. Hence, it has to be used very carefully. Commercial liquid like ethrel releases ethylene. It has been reported that exogenous application of ethylene in the form of ethrel accelerates ripening, increases colour and eating quality with reduced spoilage in different varieties of mango (Saltveit 1999; Singh and Janes 2001). The present study was taken up to evaluate the effect of ethrel dip treatment on ripening and sensory quality characteristics banana var. ‘Robusta’ at room temperature (20 °C).
Materials and methods
Twelve banana (var. ‘Robusta’) bunches were harvested at optimum stage of maturity from an orchard near Mysore and were immediately transported to the laboratory. Banana hands were separated from each bunch and delatexed for about 2 h to drain latex from cut portion. Banana hands present in the top and bottom of each bunch were discarded as these were not suitable for ripening studies. Selected banana hands were washed thoroughly with potable water to remove dirt and foreign materials present.
Post-harvest treatments and ripening conditions
Banana hands were dipped in different concentrations of ethrel (2-chloro ethyl phosphonic acid) solution ranging from 250 to 1,000 ppm for 5 min. One group of banana hands was used as untreated control fruits. Treated fruits were air dried to remove surface moisture. Five hands of each treatment (5 replicate for each treatment) were packed in ventilated round shaped plastic baskets and covered with polyethylene sheet to create high RH (80%) and kept for ripening at 20 ± 1 °C (Saeed et al. 2001). At each time of sampling, 6 fruits from 2 hands from each treatment were used for recording physical and textural characteristics periodically on 2, 4 and 6 days of storage at 20 ± 1 °C.
Physical attributes
The changes in cumulative physiological loss in weight (PLW) in both treated and control fruits were recorded during storage. Pulp to peel ratio of fruits for each treatment was determined by dividing weight of pulp by peel weight. The fruit colour was measured using UV–visible spectrophotometer (Shimadzu UV 2100, Japan) at wave length of 360–800 nm and expressed as L, a, b Hunter colour values (Hunter 1975). Fruit firmness (shear) of whole fruit was determined by using Instron Instrument (Model 4301, High Wycombe, UK) and values are expressed as Newton required to shear the fruit into two parts with speed 50 mm/min (Rushing and Huber 1983).
Chemical composition
Five fruits were randomly selected from each treatment at each stage and peel was removed, pulp was homogenized in warring blender. Moisture content in peel and pulp of banana fruits at each stage was determined by drying in oven at 70 °C for 24 h. The total soluble solids (TSS) of fruit pulp was measured with the help of Erma hand refractometer, while titratable acidity (% malic acid), pH and total sugars, starch and chlorophyll content were analyzed as per Ranganna (1986) methods. Total carotenoids of fruit pulp was extracted (by repeated extraction) with petroleum ether and acetone (3:2 v/v ratio, 60–80 °C) according to the method of Roy (1973).
Sensory quality characteristics
Descriptive sensory quality of ripe fruits viz., colour and appearance, texture (finger feel), taste, flavour and overall quality were assessed by a panel of 10 judges.
Statistical analysis
All the determinations were made in three different batches. The data were subjected to analysis of variance using the method of Snedecor and Cochran (1967).
Results and discussion
Physiological loss in weight (PLW)
Data on the cumulative loss in weight due to transpiration and respiration processes indicated that banana fruits kept in open condition without ethrel dip treatment lost weight up to 6.7% on 8th day of storage (Table 1). Fruits treated with ethrel solution of different concentrations showed increasing PLW ranging from 2.1 to 8.8%. Results showed that increase in PLW was directly proportional to increase in ethrel concentration. Maximum PLW (8.8%) was recorded in fruits treated with 1,000 ppm of ethrel at the end of 6 days. This increase in PLW of ethrel treated banana fruits during ripening could be due to upsurge in respiration rate leading to faster and uniform ripening compared to untreated fruits. Similar results were reported by Singh et al. (1977) and Mahajan et al. (2008).
Pulp/peel ratio
In ethrel treated banana fruits, weight of the fruit pulp increased with decrease in peel weight during ripening. Fruits treated with 1,000 ppm ethrel attained maximum increase in pulp to peel ratio of 2.3 followed by 2.2 in 500 ppm of treated fruits at the full ripe stage. Little change in pulp/peel ratio was recorded in control during storage. The results indicated that pulp to peel ratio of ethrel treated banana fruits during ripening increased with the increase in the ethrel concentration.
Changes in fruit colour
The intensity of greenness in the peel of all ethrel treated fruits decreased with fruit ripening and attained least ‘a’ values of −2.2, −1.2 and −1.63 in 250, 500 and 1,000 ppm of ethrel treated fruits, respectively on 6 days of storage. Fruits treated with ethrel also attained improvement in yellow colour of fruit peel as indicated by their respective ‘b’ values at the end of storage at 20 ± 1 °C. Tandon and Kalra (1995) also observed uniform yellow colour development in mango fruits treated by dipping in ethrel solution after harvest. There was no definite trend in the change of ‘L’ values.
Fruit firmness
The rapid decline in fruit firmness (shear force) values were recorded from 79.1 N at 0 day to 25.0 and 26.5 N in 1,000 and 500 ppm ethrel treated fruits, respectively at full ripe stage of 6 days (Table 1). Texture of fruits decreased at faster rate in treated fruits with increase in ethrel concentration. The decrease in texture of treated fruits during ripening could be due to breakdown of insoluble protopectin into soluble pectin or by cellular disintegration leading to membrane permeability (Brinston et al. 1988). In control fruits, texture of fruits decreased slowly from 79.1 N (at harvest day) to 63.6 N and remained unripe and shriveled even after 6 days. Similar observations were reported in banana by Peleg (1977) and Thompson and Burden (1995).
Chemical changes
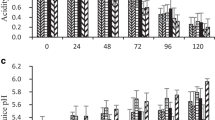
Moisture content in fruit pulp gradually increased during ripening while in peel there was a gradual decrease in all samples (Table 2). Ethrel treatment had mariginal effect on moisture content (Table 2). The increase in pulp moisture content during ripening is due to carbohydrate breakdown and osmotic transfer from the peel to pulp (John and Marchal 1995). The pH of pulp in all ethrel treated banana fruits decreased gradually during ripening whereas, in untreated samples, not much changes were observed. The decrease in pH of ethrel treated fruit pulp could be due to increase in total acidity during ripening. Banana fruits treated with 500 and 1,000 ppm of ethrel solution could reach optimum stage of ripening in 6 days of storage when pulp pH decreased to 4.8. Total acidity of ethrel treated fruits (500 ppm) increased gradually and reached a maximum of 0.54% at the optimum ripe stage. Control fruits developed total acidity of 0.34% on 6th day, as the fruits were still in unripe condition. TSS increased with increase in the concentration of ethrel during ripening. The TSS of pulp during ripening increased from 3.2° B (Brix) to 18.0° B in 250 ppm, 24.0° B in 500 ppm and 23.8° B in 1,000 ppm ethrel treated fruits at the end of 6 days storage. In untreated fruits, the change in TSS content in the pulp was slow and reached to a maximum of 7° Brix after 6 days of storage. The increase in TSS of fruit pulp could be due to the breakdown of starch into soluble sugars.
The total sugars in mature green fruits was 1.5% at harvest time. No appreciable difference was observed in the development of total sugars in control sample at the end of 6 days of ripening (2%). Total sugars accumulation was 17.6 and 17.4% in fruit pulp with 500 and 1,000 ppm of ethrel treated fruits, respectively at the end of 6 days of ripening. Starch contents were lower with higher levels of ethrel concentration and gradually decreased during ripening. The results indicated that the conversion of starch into sugars was rapid in ethrel treated fruits than in untreated fruits. This could be due to the rapid induction of pre-climacteric and climacteric phases and onset of climacteric peak in respiratory metabolic pathways in starch hydrolysis (Marriot 1980).
Chlorophyll content in peels of 500 and 1,000 ppm ethrel treated fruits decreased from 3.6 to 0.3 and to 0.2 mg/100 g, respectively after 6 days of ripening. Whereas in control fruits the total chlorophyll content decreased from 3.6 to 1.8 mg/100 g after 6 days of ripening. The results indicated that the degradation of chlorophyll pigments in fruit peel in ethrel treated fruits was more rapid than in untreated fruits. This could be due to accelerated rate of diffusion of exogenous ethylene in peel of ethrel treated fruits which triggered the degradation of chlorophyll pigments (Terai et al. 1973). In untreated fruits, there was less chlorophyll degradation in the peel tissues in the absence of exogenous ethylene treatment which probably depends on ethylene produced by pulp (Vendrell and McGlasson 1971). Similar results were reported by Srinivasan et al. (1973). The total carotenoids in ethrel treated fruits increased from 411 to 726 and 752 μg/100 g in 500 and 1,000 ppm of ethrel treated fruits, respectively after 6 days of ripening, whereas control fruits pulp developed 468 mg/100 g even after 6 days of ripening. The improper development of total carotenoid pigments in pulp of control samples could be due to delayed ethylene biogenesis.
Sensory quality characteristics
The descriptive sensory quality evaluation revealed that the fruits treated with ethrel solution of 500 and 1,000 ppm could reach optimum ripe stage on 6th day of storage with excellent overall organoleptic characteristics with respect to external colour, sweet taste, flavour and overall quality characteristics of ripe fruits. However, control fruits remained unripe and shriveled even after 6 days of storage.
Conclusion
It may be concluded that 500 ppm of ethrel solution was optimum for inducing uniform ripening of 75–80% mature banana fruits in 6 days at 20 ± 1 °C. The ethrel treated fruits ripened in 6 days at 20 ± 1 °C. At full ripe stage fruits had excellent sensory quality characteristics with respect to external yellow colour, sweetness, taste, flavour and overall quality. This method may be adapted by the small traders as replacement to calcium carbide treatment.
References
Brinston K, Dey PM, John MA, Pridhan JB (1988) Post-harvest changes in Mangifera indica mesocarp walls and cytoplasmic polysaccharides. Phytochem 27:719–723
Hunter S (1975) The measurement of appearance. Wiley, New York, pp 304–305
John P, Marchal J (1995) Ripening and biochemistry of the fruit. In: Gowen S (ed) Banana and plantains. Chapman and Hall, London, pp 434–467
Mahajan BVC, Singh G, Dhatt AS (2008) Studies on ripening behaviour and quality of winter guava with ethylene gas ethephon treatments. J Food Sci Technol 45:81–84
Marriot J (1980) Bananas—physiology and biochemistry of storage and ripening for optimum quality. CRC Crit Rev Food Sci Nutr 13:41–88
NHB (2009) Database National Horticulture Board. Ministry of Agriculture, Government of India, India
Nicholas JSS, Thompson AK (1987) The effects of temperature, concentration and exposure time to acetylene on initiation of banana ripening. J Sci Food Agric 40:43–50
Peleg M (1977) Textural changes in ripening of banana. J Texture Stud 7:457–463
PFA (2003) Prevention of Food Adulteration Act, 1954. Prevention of Food Adulteration Rules, 1955, 19th edn. International Law Book Co, Delhi, p 140
Ram HB, Singh SK, Singh RV, Surjeetsingh (1979) Effect of ethrel and smoking treatment on ripening and storage of banana cultivar. Himachal Prog Hort 11(3):69–75
Ranganna S (1986) Handbook of analysis and quality control for fruits and vegetables, 2nd edn. McGraw Hill, New Delhi
Roy SK (1973) A simple and rapid method for estimation of total carotenoids pigments in mango. J Food Sci Technol 10:45
Rushing JW, Huber DJ (1983) Colour and firmness of selected Florida glam tomato cultivars. Proc Fla Sta Hort Soc 96:107–109
Saeed A, Thompson AK, Ishfaq AH, Ali AA (2001) Effect of temperature on the ripening behavior and quality of banana fruit. Int J Agric Biol 3:224–227
Saltveit ME (1999) Effect of ethylene on quality of fresh fruits and vegetables. Postharvest Biol Technol 15:279–292
Singh Z, Janes J (2001) Effect of postharvest application of ethephon on fruit ripening, quality and shelf-life of mango under modified atmosphere packaging. Acta Hortic 553:599–601
Singh UR, Singh G, Khan A (1977) Studies on the artificial ripening of banana cultivars Basrai dwarf. Prog Hort 9(1):53–59
Snedecor GW, Cochran WG (1967) Statistical methods, 6th edn. Iowa State University Press, Ames
Srinivasan C, Subbaiah R, Shanmugam A (1973) Effect of ethephon on the chlorophyll fractions of banana during ripening. Hort Res 13:147–149
Tandon DK, Kalra SK (1995) Effect of postharvest ethrel treatments on ripening of early season mango fruits. Phala Samskarna-95, National Seminar on Post-harvest Technology of Fruits. Aug. 7–9, Bangalore, India, pp 249–253
Terai HY, Ueda Y, Ogata K (1973) Studies on the mechanism of ethylene action for fruit ripening. J Jpn Soc Hort Sci 42:75–80
Thompson AK, Burden OJ (1995) Harvesting and fruit care. In: Gowen S (ed) Banana and plantains. Chapmen and Hall, London, pp 403–433
Vendrell M, McGlasson WB (1971) Inhibition of ethylene production in banana fruit tissue by ethylene treatment. Aust J Biol Sci 24:885–895
Acknowledgement
Authors are grateful to Prakash V, Director, CFTRI, Mysore for his keen interest, constant encouragement and support during the course of investigation. Authors extend their sincere thanks to Rajarathnam S, Head, and Ramana KVR, Former Head, Department of Fruit and Vegetable Technology for their constant encouragement and guidance during the course of investigation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kulkarni, S.G., Kudachikar, V.B. & Keshava Prakash, M.N. Studies on physico-chemical changes during artificial ripening of banana (Musa sp) variety ‘Robusta’. J Food Sci Technol 48, 730–734 (2011). https://doi.org/10.1007/s13197-010-0133-y
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-010-0133-y