Abstract
The Afrotropical tachinid “genus” Agaedioxenis Villeneuve is taken here as an example of the challenges faced by dipterists in classifying one of the most diverse and species rich families of organisms on Earth. Our study has revealed “two tribes hidden in one genus”, with one lineage representing a genus belonging to the tribe Goniini (Agaedioxenis) and the other representing a genus belonging to Eryciini (Eugaedioxenis gen. nov.). The two genera have been revised through an integrative approach of morphology and genetics (COI barcode sequences). The genus name Agaedioxenis replaces that of Gaedioxenis Townsend as a valid genus name. Agaedioxenis is recognized from five species consisting of two previously described species (Agaedioxenis setifrons (Villeneuve) and Agaedioxenis brevicornis (Villeneuve) both comb. nov.) and three new species (Agaedioxenis kirkspriggsi sp. nov., Agaedioxenis succulentus sp. nov., and Agaedioxenis timidus sp. nov.). Agaedioxenis propinqua (Villeneuve) is recognized as a subjective synonym of A. brevicornis (Villeneuve), syn. nov., and by First Reviser action, the latter is chosen as the senior of the two names. Eugaedioxenis gen. nov. is recognized based on two species, Eugaedioxenis haematodes (Villeneuve), type species and comb. nov., and Eugaedioxenis horridus sp. nov. All new species of both genera are described from South Africa. We further discuss how genetics, morphology, and natural history have contributed to revise the generic circumscription of Agaedioxenis, bringing about both the description of Eugaedioxenis and the revision of the suprageneric classification for these two taxa.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
An international effort is currently underway to prepare and publish the first Manual of Afrotropical Diptera (Kirk-Spriggs et al., http://afrotropicalmanual.org/index.html). A chapter will be devoted to each of the 108 families of flies known from the region, and an illustrated key to the genera of each family will be an intregral part of each family’s chapter. For parasitic flies of the family Tachinidae, there are several challenges associated with preparing a key to genera. First, there are significantly more genera of Tachinidae in the region than there are genera of any other family of Diptera (229 at last count, O’Hara 2014). Second, the classification of Afrotropical Tachinidae has received little attention since the catalogue and keys of Crosskey (1980, 1984). Third, even the classification of 30 years ago was, for practical reasons, seriously flawed phylogenetically for certain tribes of the Exoristinae (then known as the Goniinae).
Crosskey (1984: 202) was aware that the “externally visible adult morphology in Tachinidae frequently does not correlate very precisely with phyletic grouping”. This was particularly evident in the so-called Goniini-Carceliini-Sturmiini-Eryciini complex that Crosskey first discussed in his Australian conspectus (Crosskey 1973). Crosskey was familiar with the studies of the female reproductive system by Herting (1957, 1960) that revealed a lineage within this complex characterized by the production of a derived type of egg (termed “microtype”). He understood that a phylogenetic classification should take this into account, but noted that “it is impossible to adopt such a system as yet for the Australian fauna, in which the reproductive habit of most of the genera remains unstudied” (Crosskey 1973: 77). Crosskey (1973) thus followed a more traditional classificatory scheme that emphasized adult morphology and recognized four tribes in the complex: Carceliini, Eryciini, Goniini, and Sturmiini. Shortly thereafter, Mesnil (1975) formalized the results of Herting’s discoveries of the female reproductive system into a two-tribe system—the Goniini with microtype eggs and the Eryciini with macrotype eggs—but Crosskey (1976) maintained his four tribes for his Oriental conspectus and later for his Afrotropical catalogue and keys (Crosskey 1980, 1984).
An international team was formed in 2012 to investigate the phylogeny of the Tachinidae using morphological and molecular data (Stireman et al. 2013). It is anticipated that this study will result in a well-supported phylogeny that will aid in the development of a world classification of the family to replace the current regional classifications. This study, coinciding as it does with the Manual of Afrotropical Diptera project, has brought the Afrotropical Tachinidae under special scrutiny. We are endeavoring to modernize the supra-specific classification for the 1000-odd species, including reassigning the genera of Crosskey’s “Goniini-Carceliini-Sturmiini-Eryciini complex” along phylogenetic lines.
The “genus” Agaedioxenis Villeneuve is a prime example of the challenges faced by tachinidologists in classifying tachinids in general and resolving the Goniini-Carceliini-Sturmiini-Eryciini complex in particular. Villeneuve (1937) erected the genus, under the name Gaedioxenis, for two new species and later (Villeneuve 1939) added two more new species and placed one of them in a new subgenus, Agaedioxenis. Crosskey (1980) followed this classification and assigned the genus to the Eryciini. Crosskey (1984) keyed out the species of Gaedioxenis in two places in his key to Afrotropical eryciines, retaining the four species in one genus but noting that a revision was needed. Our study of Gaedioxenis sensu Villeneuve and Crosskey has revealed two problems, one nomenclatural and the other phylogenetic. We review the nomenclatural history of the genus name and show that its valid name is Agaedioxenis Villeneuve, 1939, stat. nov. We further demonstrate that the “genus” is in reality “two tribes hidden in one genus”, with one lineage representing a genus with microtype eggs and belonging to Goniini (Agaedioxenis) and the other representing a genus with macrotype eggs and belonging to Eryciini (Eugaedioxenis gen. nov.). The two genera are revised below, including the description of three new species of Agaedioxenis, the reassignment of Gaedioxenis haematodes Villeneuve to Eugaedioxenis (comb. nov.), and the description of one new species of the latter genus.
Materials and methods
Specimens
Male terminalia were dissected and prepared for examination following the methods described by O’Hara (2002). After examination, the terminalia were rehydrated and preserved in glycerol in a plastic microvial pinned below the specimen. Composite focus-stacking images were produced from multiple images captured using a Nikon DS-L1 digital camera (Nikon, Tokyo, Japan) mounted on a Leica MZ12.5 (Leica, Wetzlar, Germany) stereomicroscope (habitus, head, wing, abdomen) or on a Leica DMLS (Leica, Wetzlar, Germany) compound microscope, and processed with CombineZM 1.0 (http://combinezm.software.informer.com/). Environmental scanning electron microscope (ESEM) digital images were taken with a Hitachi TM1000 (Hitachi, Tokyo, Japan).
Label data of primary types are given verbatim using the following symbols:
-
/ End of a line and beginning of the next
-
// End of a label and beginning of the next (from top to bottom on the same pin)
Repositories of specimens are given in square brackets using the following acronyms:
- BMNH:
-
Natural History Museum [formerly British Museum (Natural History)], London, UK
- CNC:
-
Canadian National Collection of Insects, Ottawa, Canada
- JOSC:
-
J.O. Stireman collection at Wright State University, Dayton, Ohio, USA
- MZUR:
-
Zoological Museum, ‘Sapienza’ University of Rome, Rome, Italy
- NMDA:
-
Department of Arthropoda, Natal Museum, Pietermaritzburg, South Africa
- NMB:
-
National Museum, Bloemfontein, South Africa
Terminology
Morphological terminology essentially follows McAlpine (1981) with a few exceptions as described and discussed by Cerretti et al. (2014b [see in particular Supplementary File S2]). Sexual patches on the male abdominal tergites of Tachinidae were reviewed and discussed by Cerretti et al. (2014a). The term “Code” is used for the International Code of Zoological Nomenclature published in 1999 by the International Commission on Zoological Nomenclature (hereafter as ICZN 1999).
Genetic analysis
DNA was extracted from ethanol-preserved legs of the holotype (MZUR) and one female paratype (JOSC) of Agaedioxenis succulentus and of the holotype of Eugaedioxenis horridus (MZUR) that were collected in the Western Cape province of South Africa in 2012 (Table 1). Similarly, DNA was extracted from 10 additional exoristine tachinid taxa previously collected by JOS in the USA (Table 1). Extractions were performed using the Puregene Tissue Kit (Qiagen Inc.), slightly modifying the manufacturer’s protocol. PCR amplifications (30 μL) of COI mtDNA were composed of 13.35 μL deionized H20, 3 μL of 10x PCR buffer (TaKaRa), 3 μL of 10 mM dNTPs, 4.5 μL of 25 mM MgCl2, 0.15 μL of Taq polymerase (ExTaq, TaKaRa), 1.5 μL of each primer (5 pmol/μl), and 1 μL of DNA solution. The standard “barcode” sequencing primers LepF1 and LepR1 were used to amplify an approximately 700-bp fragment (e.g. Smith et al. 2006). After visualization on an agarose gel, samples were sent to the University of Arizona Genetics Core (uagc.arl.arizona.edu) for PCR product cleanup, quantification, sequencing reactions, and sequencing. In addition, 21 COI sequences of Exoristinae in the CNC were obtained from the Barcode of Life Data System (BOLD, www.boldsystems.org) (Ratnasingham and Hebert 2007), and 19 additional sequences were obtained from Genbank (primarily from Smith et al. 2007) (Table 1).
Sequencing output files were assembled and edited using CodonCode Aligner (CodonCode Corp.) and aligned using ClustalW and by eye in MEGA 6.0 (Tamura et al. 2013). A maximum parsimony analysis was conducted using MEGA 6.0 (Tamura et al. 2013), employing the SPR method with 10 initial random addition trees, retaining a maximum of 100 trees, and performing 500 bootstrap replicates. Maximum likelihood (ML) analysis was conducted using RAxML 8.0.24 (Stamatakis 2014) via the CIPRES Science Gateway (https://www.phylo.org). A GTRCAT substitutional model was used with threads = 6, Nruns = autoMRE, algorithm = “rapid bootstrap analysis and search for the best tree in one run”, and Winthemia sp. as the outgroup. A parallel Bayesian analysis was conducted using MrBayes 3.2.2 (Ronquist and Huelsenbeck 2003) with the following parameters: Nst=6, Rates=Invgamma, Ratepr=variable, all other priors=default, Nruns=2, ngen=100000, samplefreq=100, nchains=4, Burninfrac=0.25, contype=allcompat. Convergence was assessed using Tracer (Rambaut et al. 2014), and, despite wide variance in LnL, approximate stationarity was achieved within the first 25 % of generations. Trees were visualized in FigTree v.1.4.1 (Rambaut 2014). In addition, we estimated distances (uncorrected and ML) using MEGA 6.0 with variances estimated with 500 bootstraps.
Results
Genetics
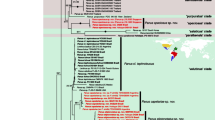
Phylogenetic relationships of the tribes Goniini and Eryciini were not well resolved in our parsimony, maximum likelihood or Bayesian analyses of COI sequences, and neither tribe was recovered as monophyletic in any analysis (Figs. 9 and 10). Bootstrap (bp.) and posterior probability (pp.) values were generally low for internal nodes above the level of genus, although erycines and goniines together form a somewhat well-supported clade (88 % pp., 36 % MP bp.), sister to the two blondeliine taxa included (100 % pp., 49 % MP bp.).
Regardless of the analysis, representatives of the focal taxa of this study, A. succulentus and E. horridus, were reconstructed in highly divergent positions. The former taxon was recovered as sister to the goniine genus Belvosia Robineau-Desvoidy in all analyses, but support for this relationship was very weak (27 % pp., 13 % ML bp., 10 % MP bp.; the shortest MP tree supported a Belvosia (Gonia Meigen + Agaedioxenis) relationship, but bootstrap support was marginally higher for Belvosia+Agaedioxenis). Eugaedioxenis was recovered in a distant clade, grouping with the eryciine genera Madremyia Townsend and Tsugaea Hall in both Bayesian and MP analyses, but this relationship was only moderately supported (66 % pp., 41 % bp in the MP consensus tree) and was not recovered in the ML analysis. Still, Eugaedioxenis was recovered far from Agaedioxenis in the ML analysis (not shown) as sister to an eryciine, the long-branched Ametadoria Townsend. Overall genetic distance between Agaedioxenis and Eugaedioxenis was 11.5 % (raw “p” distance), with ML estimated distances ranging from 12.8 % (uniform rates) to 24.7 % (gamma distributed rates, γ = 0.2). Mean (raw) distance between all (nominal) eryciine and goniine taxa was 10.5 % ± 0.6 (S.E.), and mean composite ML distances ranged from 11.5 % ± 0.8 (uniform rates) to 19.2 % ± 2.2 % (gamma distributed rates).
Morphology
Key to identify Agaedioxenis Villeneuve and Eugaedioxenis gen. nov. among Afrotropical Tachinidae
-
1.
Simultaneously: Parafacial covered with setulae; facial ridge with decumbent setae on lower 1/4–1/2; lower facial margin at least slightly visible in lateral view; ocellar setae well developed and proclinate; lower 1/2 of occiput and postgena covered with a majority of pale, hair-like setulae; prementum long and slender, 0.55–0.80 times as long as height of head; prosternum setose; proepisternal depression bare; the 3, strongest, basal postpronotal setae arranged in a line; 3 presutural acrostichal setae; 3 presutural and 3 or 4 postsutural (i.e., 3 + 3–4) dorsocentral setae; 1 presutural and 3 postsutural intra-alar setae; 3 postsutural supra-alar setae; first postsutural supra-alar seta longer than notopleural setae; 3–4 katepisternal setae; katepimeron and anepimeron bare; posterior lappet of metathoracic spiracle large, operculum-like; preapical anterodorsal setae of fore tibia at least as long and strong as preapical dorsal seta; mid tibia with 3–5, strong anterodorsal setae; costal spine well developed, at least as long as crossvein r-m; cs4 about as long as cs6; section of M between r-m and dm-cu longer than section between dm-cu and bend of M; hind tibia with 3–4 dorsal preapical setae ..................................................................................................2
-
-
Other combination of characters ................... [other Afrotropical Tachinidae]
-
2.
Scutum with 3 postsutural dorsocentral setae. Compound eye covered with scattered, long ommatrichia (longest ommatrichia at least as long as three eye facets). Hind tibia with 4 strong dorsal preapical setae. Basicosta yellow. Mid-dorsal depression on abdominal syntergite 1 + 2 not extended back to hind margin of syntergite. Scutellum without erect preapical setae. Three katepisternal setae. Tibiae yellow. Male: 2 strong proclinate orbital setae (Figs. 1a, c; 2e, g). Female: tergite 5 long, conical, 1.1–1.3 times as long as tergite 4 (Fig. 4a–b); egg macrotype, membranous, fully embryonated ................................................................. Eugaedioxenis Cerretti, O’Hara and Stireman, gen. nov.
-
-
Scutum with 4 postsutural dorsocentral setae. Compound eye bare. Hind tibia with 3 dorsal preapical setae. Basicosta black or blackish brown. Mid-dorsal depression on abdominal syntergite 1 + 2 extended back to hind margin of that segment. Scutellum with at least 1 pair of long, erect preapical scutellar setae. Usually 4 katepisternal setae. Tibiae dark. Male: proclinate orbital setae absent (Figs. 2a–d; 3a, c, e, g, i). Female: tergite 5 0.8–0.9 times as long as tergite 4 (Fig. 4c); egg microtype, planoconvex, fully embryonated and brownish black in dorsal view (Fig. 8a–d) ..................................................... Agaedioxenis Villeneuve
Fig. 1 a–b Eugaedioxenis gen. nov. haematodes (Villeneuve) habitus in lateral view: a male (holotype); b female. c–d Eugaedioxenis horridus gen. et sp. nov. habitus in lateral view: c male (holotype); d female (paratype). e–f Agaedioxenis setifrons (Villeneuve), habitus in lateral view: e male; f female (holotype)
Fig. 2 a Agaedioxenis brevicornis (Villeneuve), male, habitus. b Agaedioxenis kirkspriggsi sp. nov., male (holotype) habitus. c Agaedioxenis succulentus sp. nov., male (holotype) habitus. d Agaedioxenis timidus sp. nov., male (holotype) habitus. e–f Eugaedioxenis gen. nov. haematodes (Villeneuve) head in lateral view: e male; f female. g–h Eugaedioxenis horridus gen. et sp. nov. head in lateral view: g male (holotype), h female (paratype)
Fig. 3 a–b Agaedioxenis setifrons (Villeneuve) head in lateral view: a male; b female. c–d Agaedioxenis brevicornis (Villeneuve) head in lateral view: c male; d female. e–f Agaedioxenis kirkspriggsi sp. nov. head in lateral view: e male (holotype); f female. g–h Agaedioxenis succulentus sp. nov. head in lateral view: g male (holotype); h female. i Agaedioxenis timidus sp. nov., male (holotype) head in lateral view. j–k Male left hind tibia in posterodorsal view: j Agaedioxenis kirkspriggsi sp. nov. male (holotype); k Agaedioxenis brevicornis (Villeneuve)
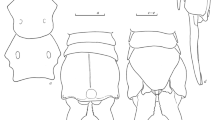
Fig. 4 a–c Female abdomen: a Eugaedioxenis gen. nov. haematodes (Villeneuve); b Eugaedioxenis horridus gen. et sp. nov. c Agaedioxenis succulentus sp. nov. d–e Male abdomen: d Agaedioxenis kirkspriggsi sp. nov. (holotype); e Agaedioxenis timidus sp. nov. (holotype). f–g Male abdomen in lateral view showing the sexual patches: f Eugaedioxenis gen. nov. haematodes (Villeneuve); g Agaedioxenis brevicornis (Villeneuve)
Tribe Eryciini
Eugaedioxenis Cerretti, O’Hara and Stireman, gen. nov.
(Figs. 1a–d; 2e–h; 4a–b, f; 5a–b; 6f; 7a–d)
a–b Eugaedioxenis gen. nov. haematodes (Villeneuve), epandrial complex in posterior (a) and lateral (b) view. c–d Agaedioxenis setifrons Villeneuve, epandrial complex in posterior (c) and lateral (d) view. e–f Agaedioxenis brevicornis (Villeneuve), epandrial complex in posterior (e) and lateral (f) view. Scale bar 0.2 mm. [red arrow indicates postero-proximal lobe of surstylus. Colour coding: green = cerci; yellow = surstylus]
a–b Agaedioxenis succulentus sp. nov. (holotype), epandrial complex in posterior (a) and lateral (b) view. c–d Agaedioxenis kirkspriggsi sp. nov. (holotype), epandrial complex in posterior (c) and lateral (d) view. Scale bar 0.2 mm. e Agaedioxenis brevicornis (Villeneuve), male terminalia in ventral view. f Eugaedioxenis gen. nov. haematodes (Villeneuve), male terminalia in ventral view. g Agaedioxenis kirkspriggsi sp. nov. (holotype), phallus, phallapodeme and ejaculatory apodeme in lateral view. [Arrow indicates median, proximal apophysis of surstylus. Colour coding (a–d): green = cerci; yellow = surstylus. Abbreviations (e–g): bp basiphallus, bs bacilliform sclerite, c cerci, dp distiphallus, e epandrium, edsd extension of dorsal sclerite of distiphallus, ep epiphallus, es ejaculatory apodeme, hy hypandrium, lv lateroventral region of distiphallus, ph phallus, pha phallapodeme, s surstylus.]
http://zoobank.org/urn:lsid:zoobank.org:act:FE7A5AB9-74D6-4627-8696-39D2398432E2
Type species. Gaedioxenis haematodes Villeneuve, 1937, by present designation.
Diagnosis. Medium to large size, strongly bristly, flies characterized by the following combination of character states: Compound eye covered with scattered, long ommatrichia; parafacial covered with fine setulae; facial ridge with decumbent setae on lower 1/4–1/2; lower facial margin at least slightly visible in lateral view; prementum long and slender; 5 postpronotal setae, the 3, strongest, basal setae arranged in a line; scutum with 3 postsutural dorsocentral setae; katepimeron bare; first postsutural supra-alar seta longer than notopleural setae; preapical anterodorsal setae of fore tibia at least as long and strong as preapical dorsal seta; hind tibia with 4 strong preapical setae; basicosta yellow; mid-dorsal depression on abdominal syntergite 1 + 2 not extended back to hind margin of syntergite; male with 2 strong proclinate orbital setae; female with tergite 5 long, conical, 1.1–1.3 times as long as tergite 4; oviscapt slightly elongated and telescopically retracted within segment 5 (Fig. 7); egg macrotype, membranous, fully embryonated. No remarkable sexual dimorphism has been observed between male and female in head chaetotaxy, frons width, compound eye size or postpedicel length.
Etymology. The generic name is a composite word formed from eu, meaning true, and the tachinid genus-group name Gaedioxenis Townsend.
Hosts. Unknown.
Distribution. South Africa.
Eugaedioxenis haematodes (Villeneuve, 1937), comb. nov.
(Figs. 1a–b, 2e–f, 4a, f, 5a–b, 6f)
Gaedioxenis haematodes Villeneuve, 1937: 207. Holotype male (CNC). Type locality: South Africa, Western Cape, between Somerset West and Strand [according to label data; published as “Colonie du Cap” (i.e., former Cape Province)].
Gaedioxenis haematodes: Crosskey (1980: 878), Afrotropical catalogue; Crosskey (1984: 288, 291), in key to genera of tropical and southern Africa.
Type material examined. Holotype ♂: ♂ // Somerset / W. Strand / Dr. Brauns. / Kapland [Cape of Good Hope] // Gaedioxenis / haematodes / Typ. Villen [label handwritten by Villeneuve] // Gaedioxenis / haematodes Vill. [handwritten by Mesnil] / L.P. Mesnil det., 1969 // TYPE [red label] // EX / L.-P. MESNIL / COLLECTION 1970 [CNC, in good condition except missing right fore leg]. Other material examined. 1♂, “Capland” [South Africa, Western Cape], Stellenbosch, 11.x.1925, Dr. H. Brauns (NMDA). 1♀, same data except xi.1924 (NMDA). 3♂♂, 1♀, [South Africa, Northern Cape], Calvinia District, Brandkop area, 14.x.1964, B. & P. Stuckenberg (NMDA).
Description.
Body length: 8–12 mm.
Male.
Colouration (Figs. 1a–b; 4a–b, f): Head ground colour black except genal groove, face, facial ridge and frontal vitta which are red. Head covered with whitish-grey reflecting microtomentum. Scape and pedicel reddish yellow; postpedicel and arista black. Palpus yellow. Thorax (except scutellum) black with light grey reflecting microtomentum; scutellum varying from red on apical 2/3 to almost entirely red. Presutural area of scutum with 4, well outlined, dark vittae (lateral pair subtriangular, median pair straight and narrow); postsutural area of scutum, when viewed from behind, with 5 dark vittae, 3 extending length of postsutural area and 2 on anterior portion only and continuous with median pair on presutural area. Femora and tarsi black, tibiae yellow. Tegula black; basicosta yellow. Wing membrane mostly hyaline, shading into yellowish antero-proximally. Abdomen mostly red with a median longitudinal dark vitta usually extending to tip of tergite 5 (median vitta tapers posteriorly, ending before posterior margin of tergite 5 in two examined specimens). Weak, grey reflecting micromentum present dorsally from syntergite 1 + 2 to tergite 5, without forming a distinct basal band.
Head (Figs. 1a; 2e): Compound eye covered with scattered ommatrichia, irregular in length (longest ommatrichia are at most as long as 4 eye facets). Frons 1.3–1.4 times as wide as compound eye in dorsal view. Inner and outer vertical setae long and robust in both sexes (outer vertical seta lateroclinate). Ocellar seta strong, proclinate. Fronto-orbital plate with only fine setulae lateral to frontal setae. Two upper reclinate orbital setae (only 1 in one specimen). Two strong proclinate and 2 strong lateroclinate orbital setae. Parafacial flat, at its narrowest point 1.1–1.3 times as wide as postpedicel; parafacial covered with fine, long, black setulae. Facial ridge usually slightly concave, with robust decumbent setae on lower 1/4–1/3. Vibrissa strong, arising slightly above level of lower facial margin. Face flat. Lower facial margin slightly visible in lateral view. Postpedicel 1.7–2.0 times as long as pedicel. Arista apparently bare, thickened on basal 2/5. First aristomere shorter than wide; second aristomere about 1.0–1.5 times as long as wide. Genal dilation well developed. Gena in profile 0.27–0.33 times as high as compound eye. Occiput convex; posterodorsal portion of occiput with 1–2 irregular rows of black setulae behind postocular row. Rest of occiput and postgena almost entirely covered with fine, pale setae. Palpus long and narrow, sub-cylindrical. Prementum slender, 0.7–0.8 times as long as height of head.
Thorax: Four or 5 postpronotal setae, 3 strong, basal setae arranged in a straight line; 1 strong anterior seta arising in front of mid basal one; 1 additional postpronotal seta usually present between and slightly posteriorly to the line of outer basal and mid basal postpronotal setae. Scutum with 3 + 3 acrostichal setae; 3 + 3 dorsocentral setae; 1 + 3 intra-alar setae; 1 or 2 posthumeral setae; 1 + 3 supra-alar setae (first postsutural supra-alar seta longer than notopleural setae); notopleuron with 2 strong setae, equal in length and thickness; postalar callus with 2 or 3 setae [when 3, then 1 is weaker than notopleural setae]. Anatergite bare. Prosternum with several long setulae on lateral margin. Proepisternal depression bare. Katepimeron bare or nearly so. Usually 3 katepisternal setae (nearly in line) [holotype with 4 setae on right side, fourth seta below middle seta]. Anterior and posterior lappets of metathoracic spiracle unequal in size (posterior lappet larger, operculum-like). Scutellum with 1 pair of crossed apical setae (standing slightly erect), varying from 1/2 (holotype) to 2/3 as long as subapical setae; 1 pair of subapical setae, 1 pair of lateral setae, and 1 pair of basal setae; lateral and subapical setae strong and subequal in size; 1 pair of widely separated discal setae; preapical setae absent. Postmetacoxal area membranous.
Legs: Fore tibia with 2–3 strong posterior setae. Preapical anterodorsal seta of fore tibia well developed, about 2/3 as long as preapical dorsal seta. Fore claws about as long as fifth tarsomere (sometimes slightly longer). Mid tibia with 3–5 anterodorsal setae. Submedian ventral seta of mid tibia present. Hind tibia with several anterodorsal setae, irregular in size (i.e., not forming a comb-like row). Preapical posteroventral seta of hind tibia moderately well developed, varying from 1/2 to 2/3 as long as preapical anteroventral seta. Hind tibia with 4 dorsal preapical setae.
Wing: Costal spine well developed, about 1.5 times as long as crossvein r-m. Vein R4+5 with 2–3 setulae at base. Bend of vein M nearly right-angled. Costal sector cs2 ventrally bare. Costal sector cs4 about as long as cs6. Section of M between crossveins r-m and dm-cu clearly longer than section between dm-cu and bend of M. Section of M between dm-cu and bend of M shorter than postangular section of M. Cell r4+5 closed at wing margin or short petiolate. Wing membrane uniformly covered with microscopic setulae.
Abdomen (Fig. 4f): Ventral edges of syntergite 1 + 2 and tergites 3 and 4 entirely overlapping the corresponding sternites. Mid-dorsal depression of syntergite 1 + 2 not extending posteriorly to hind margin of syntergite (i.e., confined to approximately anterior 4/5). Syntergite 1 + 2 with 2 pairs of median marginal setae (inner median pair less than half as long as outer median pair, and only barely noticeable in holotype), and 1 pair of lateral marginal setae; tergite 3 with 3 pairs of median marginal setae (inner median pair 1/3–2/3 as long as outer median pairs), and 2 pairs of lateral marginal setae; tergite 4 with a complete row of regular marginal setae; tergite 5 covered with erect setae, not arranged in rows. Tergites 3 and 4 with several median discal setae about as long and robust as inner median marginal setae. Tergites 4 and 5 ventrally with sexual patches (i.e., patches of appressed fine setulae with underlying glandular organs) (Fig. 4f; cf. Cerretti et al. 2014a) (barely visible patches sometimes also on tergite 3 [incl. holotype]). Tergite 5 about 0.9–1.0 times as long as tergite 4.
Male terminalia: Tergite 6 more or less plate-like but indented anteriorly at mid point, not fused with segment 7. Syntergosternite 7 + 8 relatively narrow. Sternite 6 asymmetrical and right side connection to segment 7 narrowly membranous. Sternite 5 with anterior margin almost straight; posterior margin with a deep median U-shaped cleft. Membranous transverse band on sternite 5 well developed. Epandrium short and convex. Lateral epandrial lobe not developed. Cerci in posterior view long and narrow, distally only slightly separated (Fig. 5a). Surstylus well developed, only slightly shorter than cerci, more or less lobe-like in lateral view (Fig. 5b), not fused with epandrium; with several short setulae on lateral and medial surfaces. Bacilliform sclerite rod-shaped and narrowly fused to surstylus anterobasally; surstylus with a long and narrow median, proximal apophysis (Fig. 6f). Hypandrial arms not fused posteromedially. Pregonite well developed, sub-triangular, moderately hook-shaped distally and provided with fine setulae along posterior margin. Postgonite narrow, distally rounded and gently bent anteriorly. Intermedium well developed. Ejaculatory apodeme present, small. Basal processes of basiphallus present. Epiphallus well developed and arising dorsally on basiphallus, in sub-basal position. Ventral wall of distiphallus concave. Lateroventral region of distiphallus sclerotized. Medioventral ridge of distiphallus not developed. Extension of dorsal sclerite of distiphallus well developed (Fig. 6g).
Oviscapt slightly elongated and telescopically retracted within segment 5 (Fig. 7d); egg membranous, macrotype. Eggs are stored fully embryonated within a long and spiralling common oviduct.
For other characters, female differs from male as follows:
Thorax: Fore claws 0.5–0.7 times as long as fifth tarsomere. Abdomen: Tergite 5 sub-conical, 1.1–1.3 times as long as tergite 4 (Fig. 4a).
Distribution. Afrotropical: South Africa (Northern Cape, Western Cape).
Eugaedioxenis horridus Cerretti, O’Hara and Stireman, sp. nov.
http://zoobank.org/urn:lsid:zoobank.org:act:221425B1-CA8A-4BC7-875F-5FD23373FBF1
Type material. Holotype ♂: South Africa: Western Cape / Anysberg Nature Reserve at: / 33°26′37.76″S 20°47′29.25″E / 14.X.2012, 840 m (hilltop) / P. Cerretti, J. Stireman, J. O’Hara, / I. Winkler & A.H. Kirk-Spriggs leg // SA020 [MZUR]. Paratype ♀: Sth Africa Cape Prov / 10 km E Garies [Northern Cape] / 3017DB 3.ix.1981 / J. Londt, L. Schoeman / and B. Stuckenberg. / Succulent Karoo [NMDA].
Etymology. The species epithet derives from the Latin adjective horridus meaning bristly.
Recognition. Eugaedioxenis horridus shares many characters with congener E. haematodes in regard to habitus, wing venation and chaetotaxy, but is distinguishable from it in its darker colouration of abdomen (Fig. 1a–d) and remarkably different morphometric ratios of the head (Fig. 2e–h) (see key below).
Description.
Body length: 11–12 mm.
Male.
Colouration: Head ground colour black except genal groove and facial ridge which are red, and frontal vitta which is brown. Head covered with grey reflecting microtomentum. Scape and pedicel reddish yellow; postpedicel and arista black. Palpus yellow. Thorax (except scutellum) black; scutellum mostly black with red tip. Presutural area of scutum with 4 well outlined dark vittae (lateral pair subtriangular, median pair straight and narrow); postsutural area of scutum, when viewed from behind, with 5 dark vittae, 3 extending length of postsutural area and 2 on anterior portion only and continuous with median pair on presutural area. Femora and tarsi black, tibiae yellow. Tegula black; basicosta yellow. Wing membrane mostly hyaline, shading into yellowish antero-proximally. Syntergite 1 + 2 black dorsally, red lateroventrally, dorsally covered with weak, grey microtomentum. Tergites 3 and 4 mostly black dorsally, largely red lateroventrally; grey reflecting microtomentum present on both tergites but dense only on tergite 4. Tergite 5 entirely black and covered with weak brownish microtomentum.
Head (Figs. 1c, 2g): Compound eye covered with scattered ommatrichia, irregular in length (longest ommatrichia are at most as long as 4 eye facets). Frons 1.7–1.9 times as wide as compound eye in dorsal view. Inner and outer vertical setae long and robust in both sexes (outer vertical seta lateroclinate). Ocellar seta strong, proclinate. Fronto-orbital plate with only short setulae lateral to frontal row. Two upper reclinate orbital setae. Two strong proclinate and 2 strong lateroclinate orbital setae. Parafacial convex, at its narrowest point 2.0–2.3 times as wide as postpedicel; parafacial covered with fine, long, black setae. Facial ridge straight to slightly concave just above vibrissa, with robust decumbent setae on lower 1/2. Vibrissa strong, arising slightly above lower facial margin. Face flat. Lower facial margin slightly visible in lateral view. Postpedicel 1.7–2.0 times as long as pedicel. Arista bare, thickened on basal 1/2. First aristomere shorter than wide; second aristomere about 1.5 times as long as wide. Genal dilation well developed, narrow. Gena in profile about 0.4–0.5 times as high as compound eye. Occiput convex; posterodorsal portion of occiput with 1–2 irregular rows of black setulae behind postocular row. Rest of occiput and postgena almost entirely covered with fine, pale setae. Palpus long and narrow, sub-cylindrical. Prementum slender, 0.60–0.65 times as long as height of head.
Thorax: Five postpronotal setae, 3 strong, basal setae arranged in a straight line; 1 strong anterior seta arising in front of mid basal one; 1 additional postpronotal seta present between and slightly posteriorly to the line of outer basal and mid basal postpronotal setae. Scutum with 3 + 3 acrostichal setae; 3 + 3 dorsocentral setae; 1 + 3 intra-alar setae; 3 posthumeral setae; 1 + 3 supra-alar setae (first postsutural supra-alar seta longer than notopleural setae); notopleuron with 2 strong setae, equal in length and thickness; postalar callus with 3 setae. Anatergite bare. Prosternum with several long setulae on lateral margin. Proepisternal depression bare. Katepimeron bare. Three katepisternal setae (nearly in line). Anterior and posterior lappets of metathoracic spiracle unequal in size (posterior lappet larger, operculum-like). Scutellum with 1 pair of crossed apical setae (standing slightly erect) about 2/3 as long as subapical setae; 1 pair of subapical, 1 pair of lateral setae, and 1 pair of basal setae; lateral and subapical setae subequal in size; 1 pair of widely separated discal setae; preapical setae absent. Postmetacoxal area membranous.
Legs: Fore tibia with 2 strong posterior setae. Preapical anterodorsal seta of fore tibia about as long and robust as preapical dorsal seta. Fore claws about as long as fifth tarsomere. Mid tibia with 5 anterodorsal setae. Submedian ventral seta on mid tibia present. Hind tibia with several anterodorsal setae, irregular in size. Preapical posteroventral seta of hind tibia well developed, about 2/3 as long as preapical anteroventral seta. Hind tibia with 4 dorsal preapical setae.
Wing: Costal spine well developed, about 1.5 times as long as crossvein r-m. Vein R4+5 with 2–3 setulae at base. Bend of vein M nearly right-angled. Costal sector cs2 ventrally bare. Costal sector cs4 about as long as cs6. Section of M between crossveins r-m and dm-cu clearly longer than section between dm-cu and bend of M. Section of M between dm-cu and bend of M shorter than postangular section of M. Cell r4+5 closed at wing margin. Wing membrane uniformly covered with microscopic setulae.
Abdomen (Fig. 1c): Ventral edges of syntergite 1 + 2 and tergites 3 and 4 entirely overlapping the corresponding sternites. Mid-dorsal depression of syntergite 1 + 2 not extending posteriorly to hind margin of syntergite. Syntergite 1 + 2 with 2 pairs of median marginal setae (inner median pair less than 1/2 as long as outer median pair), and 1 pair of lateral marginal setae; tergites 3 with 3 pairs of median marginal setae (inner median pair about 1/3 as long as outer median pairs) and 2 pairs of lateral marginal setae; tergite 4 with a complete row of marginal setae; tergite 5 covered with erect setae not arranged in rows. Tergites 3 and 4 with several median discal setae about as long and robust as inner median marginal setae. Tergites 3 to 5 ventrally with sexual patches. Tergite 5 about 0.9–1.0 times as long as tergite 4.
Male terminalia: not examined.
Female (Figs. 1d, 2h, 4b) differs from male as follows:
Colouration: Scutellum almost entirely red. Abdomen entirely black in ground colour (Figs. 1d, 4b). Legs: Fore claws about 0.7 times as long as fifth tarsomere. Abdomen: Tergite 5 sub-conical, 1.2 times as long as tergite 4 (Fig. 4b).
Distribution. Afrotropical: South Africa (Northern Cape, Western Cape).
Key to species of Eugaedioxenis gen. nov.
-
1.
Frons at its narrowest point 1.3–1.4 times as wide as compound eye in dorsal view. Parafacial at its narrowest point 1.1–1.3 times as wide as width of postpedicel (Figs. 1a–b, 2e–f). Abdomen mainly reddish with a narrow median black vitta on syntergite 1 + 2 to tergite 5 (Fig. 4a) ................................................................................................... E. haematodes (Villeneuve)
- Frons at its narrowest point 1.7–1.9 times as wide as compound eye in dorsal view. Parafacial at its narrowest point 2.0–2.3 times as wide as width of postpedicel (Figs. 1c–d; 2g–h). Abdomen mainly black with dark red sides (Fig. 4b) in male, entirely black in female ....... E. horridus Cerretti, O’Hara and Stireman, sp. nov.
Tribe Goniini
Agaedioxenis Villeneuve, 1939, stat. nov.
(Figs. 1e–f, 2a–d, 3a–k, 4c–e, g, 5c–f, 6a–e, g, 8a–d)
Gaedioxenis Villeneuve, 1937: 206. Nomen nudum (proposed after 1930 without designation of type species from two included species).
Gaedioxenis Villeneuve, 1939: 1. Nomen nudum (proposed after 1930 without designation of type species from two included species).
Agaedioxenis Villeneuve, 1939: 2 (as subgenus of Gaedioxenis Villeneuve, 1937 [nomen nudum]). Type species: Gaedioxenis (Agaedioxenis) brevicornis Villeneuve, 1939, by monotypy.
Gaedioxenis Townsend, 1943: 335. Type species: Gaedioxenis setifrons Villeneuve, 1937, by original designation.
Review of genus-group names. Villeneuve (1937) proposed the new genus Gaedioxenis for two new species, G. setifrons and G. haematodes. Later, Villeneuve (1939) added two new species to this genus, Gaedioxenis propinqua and G. brevicornis, placing the latter in a new subgenus, Agaedioxenis. The type species of the genus-group name Agaedioxenis was thus fixed by monotypy as G. (A.) brevicornis. In neither of Villeneuve’s papers was a type species fixed for Gaedioxenis from among the two species described in 1937 or the two species available for type species fixation in 1939 (G. setifrons and G. propinqua; G. haematodes was not mentioned in this work and G. brevicornis was fixed as type species of Agaedioxenis). Townsend (1943: 335) attempted to remedy this situation by designating G. setifrons as the type species of Gaedioxenis (by “designation herein”). Crosskey (1980: 878) followed this interpretation, attributing authorship of Gaedioxenis to Villeneuve, 1937 and the type species designation to Townsend (1943) [as 1941, treating the “Addenda and corrigenda” to Part XI of Townsend’s (1941) Manual of Myiology as published at the same time as the rest of the volume].
Evenhuis et al. (2008) discovered that earlier interpretations of Gaedioxenis were contrary to the Code that required the description of a new genus after 1930 to “be accompanied by the fixation of a type species in the original publication” (Article 13.3 of ICZN 1999). Thus, Gaedioxenis was unavailable from Villeneuve (1937, 1939) and took authorship and date from Townsend (1943). This interpretation was discussed in more detail by Evenhuis et al. (2015).
We agree with Evenhuis et al. (2008) and Evenhuis et al. (2015) that Gaedioxenis is correctly attributed to Townsend (1943), but we do not treat this name as valid. The genus-group name Agaedioxenis Villeneuve, 1939 has priority over Gaedioxenis Townsend, 1943 and is the valid name of the genus when these names are treated as subjective synonyms, as they are herein.
Diagnosis. Medium to large size, moderately bristly, exoristine characterized by the following combination of character states: Compound eye bare; parafacial covered with fine setulae; facial ridge with decumbent setae approximately on lower 1/4; lower facial margin protruded and clearly visible in lateral view; prementum long and slender; 4 or 5 postpronotal setae, the 3, strongest, basal setae arranged in a line; scutum with 4 postsutural dorsocentral setae; katepimeron bare or at most with 1–2 hair-like setulae on anterior 1/4; first postsutural supra-alar seta longer than notopleural setae; preapical anterodorsal setae of fore tibia at least as long and strong as preapical dorsal seta; hind tibia with 3 strong dorsal preapical setae; basicosta brownish black; mid-dorsal depression on abdominal syntergite 1 + 2 extended back to hind margin of that segment; male without proclinate orbital setae; female with tergite 5 normally developed, sub-conical, 0.8–0.9 times as long as tergite 4; egg microtype, planoconvex, fully embryonated; convex side hard-shelled, blackish brown, with 1 subcircular aeropilar area attached to anterior margin. Sexual dimorphism involves head chaetotaxy and morphometric ratios of head; specifically females have proclinate and lateroclinate orbital setae, a wider frons and in some cases also a shorter postpedicel.
Hosts. Unknown.
Distribution. South Africa, Zimbabwe.
Agaedioxenis setifrons (Villeneuve, 1937), comb. nov.
Gaedioxenis setifrons Villeneuve, 1937: 207. Holotype female (CNC). Type locality: South Africa, Western Cape, Stellenbosch.
Gaedioxenis setifrons: Villeneuve (1939: 1); Townsend (1943: 335), type species designation; Crosskey (1980: 878), Afrotropical catalogue; Crosskey (1984: 291), note in key to genera of tropical and southern Africa; Evenhuis et al. (2008: 14), newly recognized as type species of Gaedioxenis Townsend; Evenhuis et al. (2015), type species of Gaedioxenis in list of Townsend genus-group names.
Type material. Holotype ♀: [blank small red square label] // Capland / Stellenbosch / Nov-10 1925 / Dr. H. Brauns. // Gaedioxenis / setifrons / Villen. / Typ. [label handwritten by Villeneuve] // Gaedioxenis / setifrons Vill. [handwritten by Mesnil] / L.P. Mesnil det., 1969 // TYPE [red label] // EX / L.-P. MESNIL / COLLECTION 1970 [CNC, in good condition except missing both front legs]. Other material examined. 1♂, South Africa, Western Cape, West Coast National Park, 33° 07′ 31″ S 18° 03′ 38″ E 30 m, J.G.L. & A. Londt, 13.ix.2001, Seeberg area, lagoon bush; 1♀, South Africa, Cape Prov., 10 km E Garies, 3017DB, 3.ix.1981, J. Londt, L. Schoeman and B. Stuckenberg, Succulent Karoo (both in NMDA).
Description.
Body length: 10.5–11.0 mm.
Male.
Colouration: Head ground colour black except genal groove and facial ridge which are dark red, and frontal vitta which is dark brown. Head covered with whitish-grey reflecting microtomentum. Scape and pedicel reddish yellow; postpedicel and arista black. Palpus blackish or brownish (reddish brown in holotype). Thorax (except scutellum) black; scutellum anteriorly and anterolaterally black turning to red on posteromedian 1/2. Presutural area of scutum with 4 dark vittae (lateral pair subtriangular, median pair straight and narrow); postsutural area of scutum, when viewed from behind, with 5 dark vittae, 3 extending length of postsutural area and 2 on anterior portion only and continuous with median pair on presutural area. Legs black. Tegula and basicosta black. Wing membrane hyaline. Abdomen almost entirely black in ground colour, only faintly reddish laterally on tergite 4 or on both tergites 3 and 4. Tergites 3 and 4 with a band of whitish reflecting microtomentum on anterior 1/2; tergite 5 with a narrow band of whitish and brownish reflecting microtomentum on anterior 1/4–1/3.
Head (Figs. 1e, 3a): Compound eye bare. Frons 1.33 times as wide as compound eye in dorsal view. Inner and outer vertical setae long and robust (outer vertical seta lateroclinate). Ocellar seta strong, proclinate. Fronto-orbital plate with 2 irregular rows of several strong medioclinate setae lateral to lower frontal setae. Four to 6 upper reclinate orbital seta (2 of them lateroclinate). Proclinate orbital setae absent. Parafacial flat, at its narrowest point about 0.8 times as wide as postpedicel; parafacial covered with fine, long, black setae. Facial ridge concave, with decumbent setae on lower 1/4–1/5. Vibrissa strong, arising slightly above or at level of lower facial margin. Face slightly raised and usually visible in lateral view. Lower facial margin protruded and clearly visible in lateral view. Postpedicel about 4 times as long as pedicel. Arista bare, thickened on basal 4/5 or more. First aristomere at most as long as wide; second aristomere about 3–5 times as long as wide. Genal dilation well developed. Gena in profile ca. 0.4 times as high as compound eye. Occiput slightly convex; posterodorsal portion of occiput with 2 irregular rows of black setulae behind postocular row. Rest of occiput and postgena almost entirely covered with fine, pale setae. Palpus long and narrow, sub-cylindrical. Prementum slender, 0.60–0.75 times as long as height of head.
Thorax: Four postpronotal setae, the 3 strongest basal setae arranged in a straight line; 1 strong anterior seta arising in front between mid basal and inner basal setae. Scutum with 3 + 3 acrostichal setae; 3 + 4 dorsocentral setae; 1 + 3 intra-alar setae; 3 posthumeral setae; 1 + 3 supra-alar setae (first postsutural supra-alar seta longer than notopleural setae); notopleuron with 2 strong setae, equal in length and thickness; postalar callus with 2 strong setae, 1 additional, smaller seta may be present. Anatergite bare. Prosternum with several long setulae on lateral margin. Proepisternal depression bare. Katepimeron bare or with 1 fine setula anteriorly. Four katepisternal setae. Anterior and posterior lappets of metathoracic spiracle unequal in size (posterior lappet larger, operculum-like). Apical scutellar setae strong, crossed and varying from almost horizontal to slightly erect. One pair of subapical scutellar setae, 1 pair of lateral setae, and 1 pair of basal setae; lateral scutellar setae about 4/5 as long as subapical setae; 1 pair of widely separated discal setae and 1–2 pairs of strong, erect preapical setae, irregular in length. Postmetacoxal area membranous.
Legs: Fore tibia with 2 posterior setae. Preapical anterodorsal seta of fore tibia distinctly longer than preapical dorsal seta. Fore claws shorter than fifth tarsomere. Mid tibia with 3–5 anterodorsal setae. Submedian ventral seta on mid tibia present. Row of anterodorsal setae of hind tibia very irregular in size (i.e., not forming a comb-like row). Preapical posteroventral seta of hind tibia distinctly shorter than preapical anteroventral seta. Hind tibia with 3 dorsal preapical setae (mid-dorsal one shorter than preapical anterodorsal and preapical posterodorsal).
Wing: Costal spine well developed, about twice as long as crossvein r-m. Vein R4+5 with 2–3 setulae at base. Bend of vein M nearly right-angled. Costa sector cs2 ventrally bare. Costal sector cs4 longer than cs6. Section of M between crossveins r-m and dm-cu clearly longer than section between dm-cu and bend of M. Section of M between dm-cu and bend of M shorter than postangular section of M. Cell r4+5 closed at wing margin. Wing membrane uniformly covered with microscopic setulae.
Abdomen: Ventral edges of syntergite 1 + 2 and tergites 3 and 4 entirely overlapping corresponding sternites. Mid-dorsal depression of syntergite 1 + 2 extending posteriorly to hind margin of that segment. Syntergite 1 + 2 with 2 pairs of equally strong median marginal setae, and 2–3 pairs of lateral marginal setae; tergite 3 with 2 pairs of median marginal setae and 4 pairs of lateral marginal setae; tergite 4 with a complete row of regular marginal setae; tergite 5 with erect marginal and discal setae not arranged in rows. Tergites 3 and 4 with irregular median discal setae barely distinguishable from strong erect general setulae (median discal setae more developed on tergite 4). Tergites 3 to 5 without sexual patches. Tergite 5 about 0.9–1.0 times as long as tergite 4.
Male terminalia (Fig. 5c–d): Tergite 6 divided into 2 large hemitergites, not fused with segment 7. Syntergosternite 7 + 8 relatively broad. Sternite 6 asymmetrical and right side connection to segment 7 narrowly membranous. Sternite 5 with anterior margin almost straight; posterior margin with a deep median U-shaped cleft. Membranous transverse band on sternite 5 well developed. Epandrium short and convex. Lateral epandrial lobe not developed. Cerci in posterior view long and sub-triangular, distally only slightly separated. Surstylus laterally compressed, shorter than cerci; distal 2/3 rounded in lateral view and proximally not fused with epandrium; with several short setulae on both lateral and medial surfaces. Bacilliform sclerite rod-shaped and fused to surstylus antero-basally; surstylus with a stout median, proximal apophysis, visible in lateral view in form of a postero-proximal lobe (Fig. 5d, red arrow). Hypandrial arms strongly approximated, not fused posteromedially. Pregonite well developed, sub-triangular, hook-shaped distally. Postgonite narrow, distally pointed and gently bent anteriorly. Intermedium well developed. Ejaculatory apodeme present, small. Basal processes of basiphallus present. Epiphallus very short and membranous (barely distinguishable), arising dorsally in distal position (i.e., close to junction between basiphallus and distiphallus) (as in Fig. 6g). Ventral wall of distiphallus concave. Lateroventral region of distiphallus sclerotized. Medioventral ridge of distiphallus not developed. Extension of dorsal sclerite of distiphallus well developed (as in Fig. 6g).
Female differs from male as follows:
Head (Figs. 1f, 3b): Frons 1.40–1.51 times as wide as compound eye in dorsal view. Fronto-orbital plate with 1 irregular row of several strong medioclinate setae lateral to lower frontal setae. Two or 3 upper reclinate orbital setae, 2 strong proclinate orbital setae, and 2 lateroclinate orbital setae. Postpedicel about 2.5 times as long as pedicel. Thorax: Postalar callus with 2 setae. Legs: Hind tibia with 3 dorsal preapical setae subequal in size. Abdomen: Syntergite 1 + 2 with 2 pairs of median marginal setae, inner pair weak. Tergite 3 with 1 strong and 1 weak pair of median marginal setae and 2 or 3 pairs of lateral marginal setae; tergite 5 sub-conical, 0.9 times as long as tergite 4.
Distribution. Afrotropical: South Africa (Western Cape).
Remarks. Agaedioxenis setifrons is morphologically divergent and is separable from congeners by having a different shape of the antennae and head and by having apical scutellar setae well developed and crossed. Moreover, males of A. setifrons do not have sexual patches on abdominal tergites, which otherwise characterize the remaining species; i.e., A. brevicornis, A. kirkspriggsi, A. succulentus and A. timidus. Sexual patches on tergites 3 and 4, although present also in several other Goniini and Eryciini (see Cerretti et al. 2014a), may represent a homoplasious autapomorphy supporting monophyly of this group of species.
Agaedioxenis brevicornis (Villeneuve, 1939), comb. nov.
(Figs. 2a, 3c–d, k; 4g, 5e–f; 6e, 8a–d)
Gaedioxenis (Agaedioxenis) brevicornis Villeneuve, 1939: 1. Holotype male (BMNH). Type locality: Zimbabwe, Mutare [as “Umtali”] District.
Gaedioxenis brevicornis: Crosskey (1980: 878), Afrotropical catalogue.
Gaedioxenis propinqua Villeneuve, 1939: 2. Holotype female (not located). Type locality: South Africa, KwaZulu-Natal [as “Natal”]. Syn. nov.
Gaedioxenis propinqua: Crosskey (1980: 878), Afrotropical catalogue.
Note about synonymy. Gaedioxenis brevicornis and Gaedioxenis propinqua were described in the same paper by Villeneuve (1939). We consider these names to be subjective synonyms, with the former based on a male and the latter on a female of the same species. Acting as the First Reviser, we select G. brevicornis as the senior synonym (Article 24.2.2 of the Code, ICZN 1999).
Note about the holotype of Gaedioxenis brevicornis Villeneuve. The holotype is pinned sideways on a micropin (minuten) and double-mounted on a rectangular card. The head is detached and glued, face up, to the same card. The left fore leg and right hind leg are missing. The specimen is otherwise in good condition but generally rather dirty (D. Whitmore, pers. comm. April 2014).
Material examined. 1♂, “Capland” [South Africa, Western Cape], Stellenbosch, 10.ix.1926, Dr. H. Brauns (NMDA). 1♂, same data except 13.ix.1926 (NMDA). 1♀, same data except 15.x.1926 (NMDA). 1♀, same data except 21.x.1926 (NMDA). 1♂, same data except 25.x.1926 (NMDA). 1♂, Kapstadt [South Africa, Western Cape, Cape Town], 13.x–7.xi.1958, Lindner (CNC). 1♀, “Natal” [South Africa, KwaZulu-Natal], Mooi River, Willow Grande, Well Brook, 18.x.1913, R.C. Wroughton (CNC, abdomen missing). 2♂♂, “S. Rhodesia” [Zimbabwe], “Umtali Dist.” [Mutare District], Vumba Mountains, iii.1938, A. Cuthbertson (CNC, labelled by Villeneuve as a paratype of Gaedioxenis brevicornis but the species was described from a single male).
Description.
Body length: 9–12 mm.
Male.
Colouration: Head ground colour black except genal groove and facial ridge which are dark red. Head covered with grey reflecting microtomentum. Scape and pedicel yellow to reddish brown; postpedicel and arista black. Palpus mainly yellow and usually with blackish tip. Thorax (except scutellum) black; scutellum anteriorly black turning into red on posterior 2/3 or more. Presutural area of scutum with 4 well-outlined dark vittae (lateral pair subtriangular, median pair straight and narrow); postsutural area of scutum, when viewed from behind, with 5 dark vittae, all extending length of postsutural area though ending shortly anterior to transverse suture. Legs black. Tegula and basicosta black. Wing membrane hyaline. Abdomen almost entirely black in ground colour, sometimes faintly reddish laterally on tergite 4. Tergites 3 and 4 with a band of whitish reflecting microtomentum on anterior 1/3 or less, tergite 5 with at most a narrow band of whitish or brownish reflecting microtomentum.
Head (Figs. 2a, 3c): Compound eye bare. Frons 0.70–0.85 times as wide as compound eye in dorsal view. Inner vertical seta long and robust. Outer vertical seta not or only slightly [often varying within single specimens] differentiated from postocular row. Ocellar seta strong, proclinate. Fronto-orbital plate with an irregular row of strong medioclinate setae lateral to frontal setae running most of length of frontal setae but decreasing in size on upper portion, sometimes with several additional strong setae on lower portion. Two to 4 upper reclinate orbital setae. Proclinate and lateroclinate orbital setae absent. Parafacial flat, at its narrowest point 1.30–1.45 times as wide as postpedicel; parafacial covered with fine, long, black setae (sometimes confined to lower 1/2 of parafacial). Facial ridge concave, with decumbent setae on lower 1/4–1/5. Vibrissa strong, arising at or slightly above level of lower facial margin. Face slightly raised and usually barely visible in lateral view. Lower facial margin protruded and clearly visible in lateral view. Postpedicel 1.1–1.2 times as long as pedicel. Arista apparently bare, thickened on basal 1/2–3/4. First aristomere at most as long as wide; second aristomere about 1.0–1.5 times as long as wide. Genal dilation well developed. Gena in profile 0.28–0.43 times as high as compound eye. Occiput flat or slightly convex; posterodorsal portion of occiput with 1–2 irregular rows of black setulae behind postocular row. Rest of occiput and postgena almost entirely covered with fine, pale setae. Palpus long and narrow, sub-cylindrical. Prementum slender, 0.55–0.65 times as long as height of head.
Thorax: Four postpronotal setae, the 3 strongest basal setae arranged in a straight line; 1 strong anterior seta arising between mid basal and inner basal setae. Scutum with 3 + 3 acrostichal setae; 3 + 4 dorsocentral setae; 1 + 3 intra-alar setae; 1 to 3 posthumeral setae; 1 + 3 supra-alar setae (first postsutural supra-alar seta longer than notopleural setae); notopleuron with 2 strong setae, equal in length and thickness; postalar callus with 2–3 setae. Anatergite bare. Prosternum with several long setulae on lateral margin. Proepisternal depression bare. Katepimeron bare. Four katepisternal setae. Anterior and posterior lappets of metathoracic spiracle unequal in size (posterior lappet larger, operculum-like). Apical scutellar setae varied from absent to long and thin, if present then divergent. One pair of subapical scutellar setae, 1 pair of lateral, and 1 pair of basal setae; lateral and subapical setae subequal in size or lateral pair slightly shorter; 1 pair of widely separated discal setae and 1–2 pairs of strong, erect preapical setae. Postmetacoxal area membranous.
Legs: Fore tibia with 2 posterior setae. Preapical anterodorsal seta of fore tibia about as long and robust as preapical dorsal seta. Fore claws longer than fifth tarsomere. Mid tibia with 3–5 anterodorsal setae. Submedian ventral seta on mid tibia present. Row of anterodorsal setae of hind tibia very irregular in size (i.e., not forming a comb-like row) (Fig. 3k). Preapical posteroventral seta of hind tibia distinctly shorter than preapical anteroventral seta. Hind tibia with 3 dorsal preapical setae (mid-dorsal one usually shorter than preapical anterodorsal and preapical posterodorsal setae).
Wing: Costal spine well developed, about 1.2 times as long as crossvein r-m. Vein R4+5 with 2–3 setulae only at base. Bend of M vein obtuse. Costa sector cs2 ventrally bare. Costal sector cs4 about as long as cs6. Section of M between crossveins r-m and dm-cu clearly longer than section between dm-cu and bend of M. Section of M between dm-cu and bend of M shorter than postangular section of M. Cell r4+5 closed at wing margin or short petiolate. Wing membrane uniformly covered with microscopic setulae.
Abdomen (Fig. 4g): Ventral edges of syntergite 1 + 2 and tergites 3 and 4 entirely overlapping corresponding sternites. Mid-dorsal depression of syntergite 1 + 2 extending posteriorly to hind margin of syntergite. Syntergite 1 + 2 with 1 pair of median marginal setae, and 1–3 pairs of lateral marginal setae; tergite 3 with 1 pair of median marginal setae, and 1–2 pairs of lateral marginal setae (the longest median marginal setae 0.9–1.3 times as long as corresponding tergite measured at its maximum median length); tergite 4 with a complete row of marginal setae; tergite 5 with erect marginal and discal setae not arranged in rows. Tergites 3 and 4 usually without median discal setae; short median discal setae occasionally present only on tergite 4. Tergites 3 and 4 ventrally with a pair of sexual patches (Fig. 4g). Tergite 5 about 0.9–1.0 times as long as tergite 4.
Male terminalia (Figs. 5e–f, 6e): Tergite 6 divided into 2 hemitergites, not fused with segment 7. Syntergosternite 7 + 8 relatively narrow. Sternite 6 asymmetrical and right side connection to segment 7 narrowly membranous. Sternite 5 with anterior margin almost straight; posterior margin with a deep median U-shaped cleft. Membranous transverse band on sternite 5 well developed. Epandrium short and convex. Lateral epandrial lobe not developed. Cerci in posterior view long and sub-triangular, distally only slightly separated. Surstylus laterally compressed, shorter than cerci; distal 2/3 rounded in lateral view and proximally not fused with epandrium; with several short setulae on both outer and inner surfaces. Bacilliform sclerite rod-shaped and fused to surstylus antero-basally; surstylus with a stout median, proximal apophysis (Fig. 6e, red arrow), visible in lateral view in form of a postero-proximal lobe (Fig. 5f, red arrow). Hypandrial arms strongly approximated, fused or not fused posteromedially. Pregonite well developed, sub-triangular, hook-shaped distally. Postgonite narrow, distally pointed and gently bent anteriorly. Intermedium well developed. Ejaculatory apodeme present, small. Basal processes of basiphallus present. Epiphallus very short and membranous, arising dorsally in distal position (i.e., close to junction between basiphallus and distiphallus) (as in Fig. 6g). Ventral wall of distiphallus concave. Lateroventral region of distiphallus sclerotized. Medioventral ridge of distiphallus not developed. Extension of dorsal sclerite of distiphallus well developed (as in Fig. 6g).
Female differs from male as follows:
Head: Frons 1.20–1.33 times as wide as compound eye in dorsal view. Two or 3 upper reclinate orbital setae, 2–3 strong proclinate orbital setae and 2 lateroclinate orbital setae. Outer vertical seta well developed, lateroclinate. Thorax: Fore claws 0.7–0.9 times as long as fifth tarsomere.
Egg (Fig. 8a–d): Length: 217–232 μm; width: 117–132 μm. Shape: plano-convex. Microsculpture of thick, dorsal chorion: smooth with a barely visible polygonal microsculpture; one subcircular aeropilar area is present on anterior edge of egg. Colour: dorsal side and lateral margins blackish brown, mid-ventral, flat side whitish to pale yellow.
Distribution. Afrotropical: South Africa (KwaZulu-Natal, Western Cape), Zimbabwe.
Agaedioxenis kirkspriggsi Cerretti, O’Hara and Stireman, sp. nov.
(Figs. 2b, 3e–f, j, 4d, 6c–d, g)
http://zoobank.org/urn:lsid:zoobank.org:act:4A4A9BA3-FD22-4ACD-9132-893DAB95AE17
Type material. Holotype ♂: Malaise trap, Leucosedea[error for Leucosidea]-dominated scrub // RSA, Free State, Harrismith / Mooihekkop / 28° 10′ 50.0″ S 29° 10′ 51.1″ E / 14-16.ix.2009, ca. 1800 m / A.H. Kirk-Spriggs // Entomology Dept. / National Museum / P.O. Box 266 / Bloemfontein 9300 / South Africa // BMSA (D) 10351 [NMB]. Paratypes: 3♂♂, 1♀: same data as holotype [1♂ 1♀ in NMB, 1♂ in MZUR, 1♂ in CNC].
Etymology. Dedicated to our colleague Dr Ashley Kirk-Spriggs (NMB), who collected the type specimens.
Description.
Body length: 7–11 mm.
Male differs from A. brevicornis as follows:
Colouration: Abdominal tergite 3 with a band of whitish reflecting microtomentum on anterior half (Fig. 4d). Head (Figs. 2b, 3e): Frons 0.75–0.85 times as wide as compound eye in dorsal view. Parafacial flat, at its narrowest point 1.25–1.62 times as wide as postpedicel. Postpedicel 1.13–1.50 times as long as pedicel. Gena in profile 0.24–0.29 times as high as compound eye. Prementum 0.66–0.75 times as long as height of head. Legs. Hind tibia with a comb-like row of 15–20 anterodorsal setae with only one larger seta in submedian position (Fig. 3j). Male terminalia: As in A. brevicornis except for shape of cerci in posterior view (Fig. 6c) and shape of surstylus in lateral view (Fig. 6d); phallus as in Fig. 6g.
Female. Differs from male as follows:
Head (Fig. 3f): Frons 1.06 times as wide as compound eye in dorsal view. Two or 3 upper reclinate orbital setae, 3 strong proclinate orbital setae and 2 lateroclinate orbital setae. Outer vertical seta well developed, lateroclinate. Thorax: Fore claws about as long as fifth tarsomere.
Distribution. Afrotropical: South Africa (Free State).
Agaedioxenis succulentus Cerretti, O’Hara and Stireman, sp. nov.
http://zoobank.org/urn:lsid:zoobank.org:act:A63F53B9-30CB-4544-B437-042827FD983D
Gaedioxenis brevicornis: Cerretti et al. (2013: 23), misidentification.
Type material. Holotype ♂: SA007 // South Africa, Western Cape / Ceres Bergfynbos Reserve at: / 33° 23′ 1.91″ S 19° 17′ 20.16″ E / 12.X.2012, 459 m, P. Cerretti, J. Stireman, J. O’Hara / I. Winkler & A.H. Kirk-Spriggs leg. [MZUR]. Paratypes: 1♀: SA006 // [same data as holotype] [MZUR]; 1♀ [same data as holotype] [JOSC].
Etymology. The specific epithet derives from the habitat in which the type specimens were collected, the Succulent Karoo, and should be treated as a Latin adjective.
Description.
Body length: 10.0–11.4 mm.
Male differs from A. brevicornis as follows:
Head (Figs. 2c, 3g): Frons 0.65 times as wide as compound eye in dorsal view. Parafacial flat, at its narrowest point 1.18–1.54 times as wide as postpedicel. Postpedicel 1.12–1.35 times as long as pedicel. Gena in profile 0.21–0.38 times as high as compound eye. Prementum 0.60–0.65 times as long as height of head. Abdomen: Abdominal tergite 3 with 2 pairs of median marginal setae (outer pair slightly shorter). Male terminalia: As in A. brevicornis except for shape of cerci in posterior view (Fig. 6a) and shape of surstylus in lateral view (Fig. 6b).
Female differs from male as follows:
Head (Fig. 3h): Frons 1.15–1.25 times as wide as compound eye in dorsal view. Two or 3 upper reclinate orbital setae, 2–4 strong proclinate orbital setae and 2 lateroclinate orbital setae. Outer vertical seta well developed, lateroclinate. Thorax: Fore claws about 0.9 times as long as fifth tarsomere. Abdomen: Abdominal tergite 3 with a complete row of median marginal setae (Fig. 4c).
Distribution. Afrotropical: South Africa (Western Cape).
Agaedioxenis timidus Cerretti, O’Hara and Stireman, sp. nov.
http://zoobank.org/urn:lsid:zoobank.org:act:42D3708E-CF9D-40B2-ABB9-158CB696F15A
Type material. Holotype ♂: Kapstadt [Cape Town] / 13.X–7.XI.1958 / Lindner leg. // Agaedioxenis / brevicornis / L.P. Mesnil det, 1969 // EX / L.-P. MESNIL / COLLECTION 1970 [CNC].
Etymology. The species epithet is derived from the Latin adjective timidus, meaning shy, alluding to the fact that this species has been rarely collected.
Description.
Body length: 7.0 mm.
Male differs from A. brevicornis as follows:
Colouration: Scutellum black, shading into dark brown apically. Abdomen entirely black with wide bands of weak and barely visible greyish microtomentum on anterior 4/5 or more (tergite 3) or anterior 1/2 (tergite 4) (Fig. 4e). Head (Figs. 2d, 3i): Frons 0.83 times as wide as compound eye in dorsal view. Parafacial at its narrowest point 1.66 times as wide as postpedicel. Postpedicel 1.08 times as long as pedicel. Second aristomere about 2.6 times as long as wide. Gena in profile 0.43 times as high as compound eye. Prementum slender, about 0.75 times as long as height of head. Abdomen: The longest median marginal setae of tergite 3, at least twice as long as corresponding tergite measured at its maximum median length. Male terminalia: not examined.
Female unknown.
Distribution. Afrotropical: South Africa, Western Cape (known only from the type locality of Cape Town).
Key to species of Agaedioxenis Villeneuve
-
1.
Postpedicel 4 (♂), 1.9–2.4 (♀) times as long as pedicel (Figs. 1e–f; 3a–b). Second aristomere 3–5 times as long as its diameter at midlength. Arista thickened on about its basal 4/5. Scutellum with crossed apical setae. Female: frons about 1.4–1.5 times as wide as compound eye in dorsal view; 2 upper reclinate orbital setae, 2 strong proclinate orbitals, 2 strong lateroclinate orbital setae. Male: frons 1.33 times as wide as compound eye in dorsal view; abdomen without sexual patches .................................................. A. setifrons (Villeneuve)
-
-
Postpedicel 0.9–1.5 times as long as pedicel (Figs. 2a–d; 3c–h). Second aristomere at most 2.6 times as long as its diameter at midlength. Arista thickened on its basal 1/2–2/3. Apical scutellar setae varying from diverging to sub-parallel, sometimes reduced or absent. Female: frons about 1.00–1.35 times as wide as compound eye in dorsal view; 2–4 strong proclinate orbital setae and 1–2 lateroclinate orbital setae. Male: frons 0.65–0.90 times as wide as compound eye in dorsal view; tergite 3 and 4 with sexual patches ventrally (Fig. 4g) ......................................................................... 2
-
2.
Abdominal syntergite 1 + 2 (♂) and tergite 3 (♂♀) with 4 or more median marginal setae. Male: Frons about 0.65 times as wide as compound eye in dorsal view; cerci in posterior view as in Fig. 6a; surstylus in lateral view as in Fig. 6b ................................................................................ .. A. succulentus Cerretti, O’Hara and Stireman, sp. nov.
-
-
Abdominal syntergite 1 + 2 and tergite 3 with 2 median marginal setae. Male: Frons 0.75–0.85 times as wide as compound eye in dorsal view; cerci and surstylus not as above [male terminalia of A. timidus not examined] ................................................................ 3
-
3.
Longest median marginal setae of tergite 3 at least twice as long as tergite 3 measured at its maximum median length (Fig. 2d). Second aristomere about 2.5 times as long as wide. Scutellum almost entirely black, only dark brown apically. Abdominal microtomentum weak and not forming distinct basal bands (Fig. 4e). [female unknown] ............ A. timidus Cerretti, O’Hara and Stireman, sp. nov.
-
-
Longest median marginal setae of tergite 3 0.9–1.3 times as long as same tergite measured at its maximum median length. Second aristomere at most 2 times as long as wide. Scutellum reddish yellow at least on apical 1/3. Abdominal microtomentum in form of distinct basal bands ............................................................................... 4
-
4.
Hind tibia with a comb-like row of 15–25 anterodorsal setae with only one larger seta in submedian position (Fig. 3j). Basal band of microtomentum on abdominal tergite 3 confined on anterior half (Fig. 4d). Male: terminalia as in Fig. 6c–d .................................................................................. A. kirkspriggsi Cerretti, O’Hara and Stireman, sp. nov.
-
-
Anterodorsal setae on hind tibia not arranged in a regular comb-like row (i.e., with at least three setae longer and stronger than others, Fig. 3k). Basal band of microtomentum on abdominal tergite 3 confined on anterior 1/3. Male: terminalia as in Fig. 5e–f ................................................. A. brevicornis (Villeneuve)
Discussion
If it is through a rigorous implementation of cladistic principles that systematics plays a fundamental role in evolutionary biology, then it is the task of taxonomists to improve the classification of living organisms by identifying monophyletic assemblages, removing inconsistencies, and instituting taxonomic changes to better reflect evolutionary history.
It is apparent from our integrative study of morphology and genetics that Agaedioxenis sensu Crosskey (1980, 1984, as Gaedioxenis) represented a polyphyletic assemblage of species that were combined under one genus due to a misleading combination of external traits. Although all examined females share a long and coiled common oviduct (uterus) capable of retaining large quantities of eggs, dissection of the female genitalia has revealed two different reproductive strategies.
We did not observe females in the act of laying their eggs, but there are several morphological traits from which we can infer a lot about their oviposition behaviour. Eugaedioxenis haematodes and E. horridus produce macrotype, membranous eggs, which are laid ready to hatch. Also, females of these species have a relatively long, conical tergite 5 and an oviscapt that is slightly elongated and telescopically retracted within segment 5. These traits are characteristic of several eryciine genera that deposit their eggs directly on hosts (e.g. some species of Phryxe Robineau-Desvoidy and Erycia Robineau-Desvoidy). Females of Agaedioxenis have an unmodified tergite 5, short oviscapt and produce microtype, plano-convex, hard-shelled eggs. These eggs are of the type that are laid ready to hatch on the food plants of their hosts, and must be swallowed by feeding hosts before they can hatch and the first instars can penetrate into the host’s haemocoel through the gut wall (see Herting 1960; Wood 1987; Mellini 1991). As mentioned in the “Introduction” section, tachinids sharing this reproductive, strategy are grouped within the tribe Goniini (cf. O’Hara 2013). Other than egg morphology there are no known characters that unambiguously separate the Eryciini from the Goniini (see Tschorsnig 1985; Cerretti and Barraclough 2007), and it is therefore necessary to examine the female genitalia and eggs of a species in this group to determine to which tribe it belongs.
Monophyly of the Eryciini has been questioned by several authors due to the lack of synapomorphies defining this tribe, in contrast to the Goniini, which are widely accepted as a monophyletic group (Herting 1960; Wood 1987), well supported by at least three morphological autapomorphies (Cerretti et al. 2014b). Using molecular data, the hypothesis of Goniini monophyly was rejected by Stireman (2002), while more recently, it was upheld by Tachi and Shima (2010). In particular, Stireman (2002), using two genes (EF1α and 28S rDNA), found taxa of Eryciini and Goniini interspersed within a single clade, i.e., not forming two distinct clades. On the other hand, Tachi and Shima (2010), using four genes (16S, 18S, 28S rDNA, and white), reconstructed the Eryciini as a paraphyletic grade from which a monophyletic Goniini arose. These results are consistent with the assumption that increasing the number of loci should lead to an increase in phylogenetic resolution (e.g. Rokas and Carroll 2005).
Our analyses of “barcode” sequences yielded contrasting topologies with very low branch support, but consistently recovered both the Eryciini and Goniini as nonmonophyletic and Agaedioxenis and Eugaedioxenis in highly divergent positions (see the “Results” section and Figs. 9 and 10). Also interesting is that all analyses yielded Eryciini + Goniini as a well-supported monophyletic assemblage (Figs. 9 and 10). To comment further on the monophyly of the Eryciini and Goniini is beyond our present scope, because COI sequences are clearly not informative enough to robustly resolve relationships at this level. However, the overall genetic distance between Agaedioxenis and Eugaedioxenis is higher than the mean genetic distance between all the eryciines and goniines we tested. Furthermore, Agaedioxenis was always reconstructed as sister to a member of the Goniini and Eugaedioxenis was always reconstructed as sister to an eryciine, although the identity of that sister taxon varied across analyses. Therefore, despite instability in the trees, genetic data clearly indicate that these two taxa are not closely related, corroborating the morphological evidence of their phylogenetic disparity.
The most parsimonious tree recovered from maximum parsimony analysis of representative eryciine (red) and goniine (yellow) taxa based on COI sequence data (treelength 1721). Numbers above bars indicate bootstrap probabilities of branches (500 replicates). Only probabilities >10 % are reported. The focal taxa in the current study, Agaedioxenis and Eugaedioxenis, are indicated in bold
References
Cerretti, P., & Barraclough, D. A. (2007). Anomalostomyia namibica, a new genus and species of Afrotropical Tachinidae (Diptera). Italian Journal of Zoology, 74, 101–106.
Cerretti, P., O’Hara, J. E., Stireman, J. O., III, Winkler, I. S., & Kirk-Spriggs, A. H. (2013). To ‘Die Hel’ and back. Expeditions of the phylogeny of World Tachinidae Project. Part I: Western Cape, South Africa. The Tachinid Times, 26, 20–29.
Cerretti, P., Di Giulio, A., Romani, R., Inclan, D. J., Whitmore, D., Di Giovanni, F., Scalici, M., & Minelli, A. (2014a). First report of exocrine epithelial glands in oestroid flies: the tachinid sexual patches (Diptera: Calyptratae: Oestroidea: Tachinidae). Acta Zoologica. doi:10.1111/azo.12085.
Cerretti, P., O’Hara, J. E., Wood, D. M., Shima, H., Inclan, D. J., & Stireman, J. O., III. (2014b). Signal through the noise? Phylogeny of the Tachinidae (Diptera) as inferred from morphological evidence. Systematic Entomology, 39, 335–353.
Crosskey, R. W. (1973). A conspectus of the Tachinidae (Diptera) of Australia, including keys to the supraspecific taxa and taxonomic and host catalogues. Bulletin of the British Museum (Natural History). Entomology. Supplement, 21, 221 pp.
Crosskey, R. W. (1976). A taxonomic conspectus of the Tachinidae (Diptera) of the Oriental Region. Bulletin of the British Museum (Natural History). Entomology. Supplement, 26, 357 pp.
Crosskey, R. W. (1980). Family Tachinidae. In R. W. Crosskey (Ed.), Catalogue of the Diptera of the Afrotropical Region (pp. 822–882). London: British Museum (Natural History).
Crosskey, R. W. (1984). Annotated keys to the genera of Tachinidae (Diptera) found in tropical and southern Africa. Annals of the Natal Museum, 26, 189–237.
Evenhuis, N. L., Pape, T., & Pont, A. C. (2008). The problems of subsequent typification in genus-group names and use of the Zoological Record: a study of selected post-1930 Diptera genus-group names without type species designations. Zootaxa, 1912, 1–44.
Evenhuis, N. L., Pont, A. C., & Whitmore, D. (2015). Nomenclatural studies toward a world list of Diptera genus-group names. Part IV: Charles Henry Tyler Townsend. Zootaxa.
Herting, B. (1957). Das weibliche Postabdomen des calyptraten Fliegen (Diptera) und sein Merkmalswert für die Systematik der Gruppe. Zeitschrift für Morphologie und Ökologie der Tiere, 45, 429–461.
Herting, B. (1960). Biologie der westpaläarktischen Raupenfliegen. Dipt., Tachinidae. Monographien zur angewandten Entomologie, 16, 188 pp.
International Commission on Zoological Nomenclature. (1999). International Code of Zoological Nomenclature. Fourth edition adopted by the International Union of Biological Sciences. London: International Trust for Zoological Nomenclature.
McAlpine, J. F. (1981). Morphology and terminology — adults. In McAlpine, J. F. Peterson, B. V. Shewell, G. E. Teskey, H. J. Vockeroth, J. R. & Wood, D. M. (Eds), Manual of Nearctic Diptera. Vol. 1. Research Branch, Agriculture Canada Monograph, 27, 9–63.
Mellini, E. (1991). Sinossi di biologia dei Ditteri Larvevoridi. Bollettino dell’Istituto di Entomologia “Guido Grandi” dell’Università di Bologna, 45(1990), 1–38.
Mesnil, L. P. (1975). 64g. Larvaevorinae (Tachininae). Die Fliegender Palaearktischen Region, 10 (Lieferung 309), 1305–1384.
O’Hara, J. E. (2002). Revision of the Polideini (Tachinidae) of America north of Mexico. Studia Dipterologica. Supplement, 10, 170 pp.
O’Hara, J. E. (2013). History of tachinid classification (Diptera, Tachinidae). ZooKeys, 316, 1–34.
O’Hara, J. E. (2014). World genera of the Tachinidae (Diptera) and their regional occurrence. Version 8.0. PDF document, 87 pp. http://www.nadsdiptera.org/Tach/WorldTachs/Genera [verified November 2014].
Rambaut, A. (2014). FigTree 1.4.1. Tree Figure Drawing Tool (http://tree.bio.ed.ac.uk/).
Rambaut, A., Suchard, M. A., Xie, D., & Drummond, A. J. (2014). Tracer v1.6, Available from http://beast.bio.ed.ac.uk/Tracer.
Ratnasingham, S., & Hebert, P. D. N. (2007). BOLD: The Barcode of Life Data System (http://www.barcodinglife.org). Molecular Ecology Notes, 7, 355–364.
Rokas, A., & Carroll, S. B. (2005). More genes or more taxa? The relative contribution of gene number and taxon number to phylogenetic accuracy. Molecular Biology and Evolution, 22, 1337–1344.
Ronquist, F., & Huelsenbeck, J. P. (2003). MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics, 19, 1572–1574.
Smith, M. A., Woodley, N. E., Janzen, D. H., Hallwachs, W., & Hebert, P. D. N. (2006). DNA barcodes reveal cryptic host-specificity within the presumed polyphagous members of a genus of parasitoid flies (Diptera: Tachinidae). Proceedings of the National Academy of Sciences USA, 103, 3657–3662.
Smith, M. A., Wood, D. M., Janzen, D. H., Hallwachs, W., & Hebert, P. D. N. (2007). DNA barcodes affirm that 16 species of apparently generalist tropical parasitoid flies (Diptera: Tachinidae) are not all generalists. Proceedings of the National Academy of Sciences USA, 104, 4967–4972.
Stamatakis, A. (2014). RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics. doi:10.1093/bioinformatics/btu033.
Stireman, J. O., III. (2002). Phylogenetic relationships of tachinid flies in subfamily Exoristinae (Tachinidae: Diptera) based on 28S rDNA and elongation factor-1α. Systematic Entomology, 27, 409–435.
Stireman, J. O., III, O’Hara, J. E., Moulton, K., Cerretti, P., & Winkler, I. S. (2013). Progress towards a phylogeny of world Tachinidae. Year 1. The Tachinid Times, 26, 4–9.
Tachi, T., & Shima, H. (2010). Molecular phylogeny of the subfamily Exoristinae (Diptera, Tachinidae), with discussions on the evolutionary history of female oviposition strategy. Systematic Entomology, 35, 148–163.
Tamura, K., Stecher, G., Peterson, D., Filipski, A., & Kumar, S. (2013). MEGA6: molecular evolutionary genetics analysis version 6.0. Molecular Biology and Evolution, 30, 2725–2729.
Townsend, C. H. T. (1943) Addenda and corrigenda [to Townsend’s Manual of myiology in twelve parts, Part XI, 1941]. Privately published, Itaquaquecetuba, São Paulo. Pp. 331–342.
Tschorsnig, H.-P. (1985). Taxonomie forstlich wichtiger Parasiten: Untersuchungen zur Struktur des männlichen Postabdomens der Raupenfliegen (Diptera, Tachinidae). Stuttgarter Beiträge zur Naturkunde Serie A (Biologie), 383, 1–137.
Villeneuve, J. (1937). Descriptions de myodaires supérieurs. Revue de Zoologie et de Botanique Africaines, 29, 205–212.
Villeneuve, J. (1939). Myodaires supérieurs africains (descriptions et observations). Bulletin du Musée Royal d’Histoire Naturelle de Belgique, 15(48), 1–10.
Wood, D. M. (1987) Tachinidae. In McAlpine, J. F. Peterson, B. V. Shewell, G. E. Teskey, H. J. Vockeroth, J. R., & Wood, D. M. (Eds), Manual of Nearctic Diptera. Vol. 2. Research Branch, Agriculture Canada Monograph, 28, 1193–1269.
Acknowledgments
We would like to thank Ashley Kirk-Spriggs (NMB), Burgert Muller (NMDA), and Nigel Wyatt (BMNH) who enabled our studies of the valuable material preserved in their institutions. Thanks to Daniel Whitmore (BMNH) for information about the holotype of A. brevicornis and to Mara Tisato (Centro Nazionale Biodiversità Forestale, Verona, Italy) for help composing plates. Thomas Pape (Natural History Museum of Denmark, Copenhagen) provided the nomenclatural advice on the interpretation of Villeneuve’s genus-group names. Finally, we thank Knut Rognes (University of Stavanger, Stavanger, Norway) and an anonymous reviewer for their constructive comments on earlier drafts of this manuscript.
Funding
This work was supported by U.S. National Science Foundation DEB-1146269 to principal investigators J.O.S. and J.E.O. and to major collaborator P.C.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work has been registered in ZooBank, http://zoobank.org/urn:lsid:zoobank.org:pub:0C53D317-5C05-45D7-A9E4-FB6E6D6F28DE
Rights and permissions
About this article
Cite this article
Cerretti, P., O’Hara, J.E., Winkler, I.S. et al. Two tribes hidden in one genus: the case of Agaedioxenis Villeneuve (Diptera: Tachinidae: Exoristinae). Org Divers Evol 15, 489–512 (2015). https://doi.org/10.1007/s13127-015-0211-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13127-015-0211-0