Abstract
Although calcium channel blockers, angiotensin II receptor blockers, and combination therapy are effective for hypertensive patients, the significant differences among them against stroke onset are undetermined. In this study, we investigated the significant beneficial effects of the combination therapy using amlodipine and irbesartan against stroke onset in hypertensive rats. The animals were fed an 8 % sodium diet and assigned to (1) vehicle, (2) amlodipine (2 mg/kg/day), (3) irbesartan (20 mg/kg/day), and (4) amlodipine + irbesartan groups. The drugs were given orally until 35 days, and incidences of stroke-related signs and mortality and blood pressure (BP) were monitored. Cerebral blood flow (CBF), brain water content, weight of the brain and left ventricle, and histological evaluations were conducted for the treated groups at 42 days after the start of the high-salt diet. Amlodipine and the combination therapy significantly reduced BP compared with the vehicle. Although the rates of stroke-related signs and mortality were high in the vehicle group, the rats in the treatment groups were mostly healthy until 35 days. After all drugs were discontinued, stroke onset was frequently seen in the monotherapy groups until 42 days, but no signs were observed in the combination therapy group. Although there were no significant differences in CBF or brain edema, the combination therapy reduced blood–brain barrier disruption, white matter injury, and reactive astrocytes compared with irbesartan, and the combination also inhibited left ventricular hypertrophy and preserved brain-derived neurotrophic factor (BDNF) expression on cerebral vessels compared to the monotherapies. These data suggest that the combination therapy had a persistent preventive effect on stroke onset in hypertensive rats, and the effects might be associated with BDNF preservation on cerebral vessels.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hypertension is known as an independent risk factor for stroke onset including cerebral infarction, intracerebral hematoma, subarachnoid hemorrhage, and hypertensive encephalopathy, all of which lead to high mortality and morbidity [1]. In addition, people who have a background of severe hypertension and excessive salt intake have increased blood pressure and have higher rates of stroke onset [2, 3]. Thus, it is very important to control blood pressure with suitable antihypertensive drugs for the prevention of stroke.
Calcium channel blockers (CCB) and angiotensin II receptor blockers (ARB) are recommended to be selected first for managing hypertension, and a combination therapy is recommended if a monotherapy is not sufficient for controlling blood pressure. Although clinical studies have shown that antihypertensive treatment using those drugs could inhibit stroke onset [4, 5], the significant differences between the monotherapies and the combination therapy for preventing stroke onset are still undetermined.
Brain-derived neurotrophic factor (BDNF) is known as a neurotropic factor and has an important role in adaptative brain plasticity after stroke [6]. BDNF is usually expressed in neurons, but brain microvascular endothelial cells also synthesize and secrete BDNF [7, 8]. Previously, it has been reported that BDNF was downregulated after a bilateral common carotid artery occlusion in hypertensive rats, and telmisartan, which is an ARB, improved hypertension-induced cognitive impairment via BDNF upregulation [9, 10]. Therefore, BDNF might have pivotal roles for prevention of hypertension-induced stroke onset.
In the current study, we hypothesized that the combination therapy including a CCB and an ARB would reduce the stroke onset more than each monotherapy. To address this hypothesis, we evaluated the effects of amlodipine and irbesartan in salt-loaded and spontaneously hypertensive stroke-prone (SHRSP) rats, which frequently show stroke onset after an 8 % sodium diet [11, 12]. Moreover, we sought the significant effects of the therapy combining amlodipine and irbesartan on blood–brain barrier disruption, white matter injury, and expressions of reactive astrocyte and BDNF, all of which can be modulated by hypertension-induced stroke.
Materials and Methods
Experimental Animals and Study Protocol
All experiments were approved by the institutional Animal Care and Use Committee of Kumamoto University. Forty male SHRSPs (Japan SLC, Shizuoka, Japan) were fed an 8 % sodium diet from 11 weeks of age and given either (1) vehicle [0.5 % carboxymethyl cellulose (CMC), n = 10], (2) amlodipine (2 mg/kg in 0.5 % CMC per day, n = 10), (3) irbesartan (20 mg/kg in 0.5 % CMC per day, n = 10), or (4) the combination of (2) and (3) (n = 10) every day by gastric gavage [11, 13]. At 28 days after the beginning of the high-salt diet, monitoring of the vehicle group was discontinued because the beneficial significance of the antihypertensive drugs was obvious. Thereafter, the animals in all groups were treated until 35 days, when all drugs were discontinued. Finally, it was decided that the endpoint of this study would be at 42 days after the rats were salt-loaded. Timeline of this study was summarized in Fig. 1a.
Schematic illustration of the timeline of the experiments (a) (Na, sodium diet; Veh, vehicle; AM, amlodipine; IR, irbesartan; BP, blood pressure; NF, neurological findings; CBF, cerebral blood flow; BW, weight of brain; BWC, brain water content; LVWI, left ventricle weight index). Blood pressure of SHRSP rats at 7 and 28 days (b) and body weight of the animals at each week (c) after high-salt diets in all groups. # P < 0.05 vs. vehicle, *P < 0.05 vs. AM + IR
Drugs
Amlodipine was purchased from LKT Laboratories, Inc. (Tokyo, Japan), and irbesartan was kindly supplied from Shionogi & CO., LTD. (Osaka, Japan).
Measurement of Blood Pressure and Body Weight
Blood pressure was measured in the conscious SHRSP rats via the tail cuff method (BP-98A; Softron Co, Tokyo, Japan) at 7 and 28 days, and the rats were weighed every week until 42 days after the start of the high-salt diet.
Monitoring of Stroke Onset and Death
The onset of stroke-related neurological deficits such as a paralytic gate, reduced motor activity, and sudden death were monitored every day for 42 days as previously described [14]. Stroke onset was defined as any of these signs. In addition, a 6-point scoring system (namely symptom scoring) with a modification of the method described by Nakayama et al.,[15] was assessed to all the rats in each week as follows; normal, 6; slight decrease in motor activity, 5; decrease in motor activity, 4; paralysis of hindlimbs, 3; slight movement, 2; cannot stand, 1; and death, 0.
Neurological Findings
Two neurological scoring tests were carried out at 28, 35, and 42 days after the start of the high-salt diet to emphasize the latent differences among the treatment groups. First, sensorimotor performance was scored from 0 to 4 using a beam walking apparatus described by Goldstein et al. [16], with a modification to measure for all animals (beam walking test). The rats’ performance was scored as follows: 0 or 1 points for rats that hung/stood off the beam, 2 points for animals that walked but fell down from the beam, and 3 or 4 for animals that could walk less than or at least 20 cm on the beam for 1 min [17]. Next, the rotarod test was performed to assess sensorimotor coordination and balance in each animal (MK-630B, Muromachi Kikai Co., LTD, Tokyo, Japan). Animals were placed on the rotating spindle at a constant race of 4 rotations per minute (RPM) for 1 min. Then, the rats were subjected to the trial on the accelerating spindle (4–40 RPM) for 5 min, and the time it took them to fall off the cylinder was recorded. The mean scores and times for three trials of beam walking and rotarod tests were used for each animal.
Measurement of Cerebral Blood Flow and Weight of Brain and Left Ventricle
At 42 days after the start of the salt-loaded diet, the animals’ cerebral blood flow (CBF) on the somatosensory cortex was recorded by a laser speckle blood flow imager (Omega Zone; Omegawave, Tokyo, Japan) under 2 % isoflurane [14]. Then, the animals were killed under deep anesthesia, and their brains were quickly removed and the whole brain was weighed (cut at the point of foramen magna). Next, the brains were divided at the point of bregma, and the rostral side of each brain was used to evaluate brain water content (BWC) as previously described [18]. Briefly, the brain weights were measured as soon as possible (wet weight), dried at 105 °C for 72 h, and weighed again to determine the dry weight of the brain. The following formula was calculated to measure the BWC: [(wet weight − dry weight) / wet weight] × 100 %. In addition, the left ventricle of each rat was weighted and divided by each rat’s body weight to evaluate the left ventricle weight index, which indicates left ventricular hypertrophy. On the other hand, the caudal side of each brain was immediately frozen in tissue-Tek OCT embedding medium (Sakura Finetek, Tokyo, Japan) and an 8-μm slice was made 1.5–2 mm caudally from the bregma for the following histological evaluations.
Assessment of Blood–Brain Barrier Disruption
Localizations of the immunoglobulin G (IgG) staining-positive region in each brain section were evaluated as previously described [14]. Briefly, two brain sections in each brain were randomly chosen and the sections were incubated with anti-IgG antibody (1: 500; Invitrogen Carlsbad, CA, USA) for 1 h, and then, the reaction product was visualized with diaminobenzidine (DAB). IgG stain lesions of those two slides were similar and were scored with one of the two sections as follows: IgG accumulation within the vessels (grade 0), IgG leakage around the vessel (grade 1), and extravasated IgG widespread to the interstitial space of one hemisphere (grade 2) or both hemispheres (grade 3).
Measurement of White Matter Injury
White matter lesions were evaluated using Klüver-Barrera staining in the corpus callosum as previously described [14]. Briefly, the brains were incubated overnight in Luxol fast blue solution at 60 °C and immersed in 100 % ethanol to remove excess stain. After adequate immersion with 0.05 % lithium carbonate and 70 % ethanol, the sections were immersed in 0.1 % cresyl violet solution for 5 min. The severity of the white matter lesions was graded on a 4-point scale: normal (grade 0), disarrangement of the nerve fibers (grade 1), formation of marked vacuoles (grade 2), and disappearance of myelinated fibers (grade 3) [19]. They were scored in four fields per rat at ×200 magnification and expressed as a mean score for each animal.
Immunohistochemistry
We immunostained the brain sections in each rat with anti-glial fibrillary acidic protein (GFAP; 1:300; Santa Cruz Biotechnology, Santa Cruz, CA, USA) and anti-BDNF (1:200; Santa Cruz Biotechnology) antibodies. Those samples were incubated overnight at 4 °C with the primary antibodies. After the slices were incubated with a secondary antibody (Santa Cruz Biotechnology), they were detected with DAB. Reactive astrocytes, which are highly immunoreactive to GFAP with large cell bodies and long and thick processes [20], were graded as follows: none or trace amounts (grade 0), a few (grade 1), moderate (grade 2), and marked or apparent glial scarring (grade 3). They were scored in the four fields of the somatosensory cortex on both sides for each rat at ×200 magnification and expressed as a mean score for each animal. On the other hand, BDNF-positive cerebral vessels, including cerebral arterioles and capillaries, were counted in four fields per rat at ×200 magnification and expressed as a mean number for each animal.
Statistical Analysis
The stroke onset and mortality of the animals were analyzed by a standard Kaplan-Meier analysis with a log rank test and Χ 2 analysis using GraphPad Prism version 5 for Windows (GraphPad Software). Parametric evaluations (blood pressure at 7 days, body weight at 42 days, cerebral blood flow) were analyzed by a one-way ANOVA with a Tukey-Kramer post hoc analysis. Other evaluations which did not show a normal distribution or an equal variance in each group were analyzed by a nonparametric test with a Kruskal-Wallis test followed by Dunn’s multiple comparison test. P < 0.05 was considered significant.
Results
Blood Pressure and Body Weight
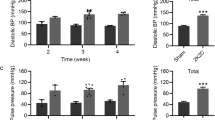
Although amlodipine significantly reduced blood pressure at 7 days after administration of the salt-loaded diet to the SHRSP rats, irbesartan did not show a statistically significant effect. On the other hand, amlodipine enhanced the blood pressure lowering effect of irbesartan at 7 and 28 days after the high-salt load (Fig. 1b). Body weight in the combination therapy group was significantly preserved compared with the weight in the vehicle group at 28 days and the weight in the irbesartan group at 42 days after the start of the salt-loaded diet (Fig. 1c).
Stroke Onset and Mortality
All of the antihypertensive treatments in this study prevented stroke onset, but amlodipine did not show the beneficial role against mortality at 28 days after administration of the salt-loaded diet (Fig. 2a, b). Although the rats in the monotherapy groups developed strokes and died gradually after the drugs were discontinued, there were no sign of stroke onset or death in the group that received the combination therapy. Finally, the percentage of stroke onset/death in each group was 60 %/30 % in the amlodipine group, 50 %/10 % in the irbesartan group, and 0 %/0 % in the combination group, which was a significantly different rate of stroke onset compared with the monotherapy groups. Although the symptom scoring was significantly higher in the combination group than in the amlodipine group, neurological findings including beam walking and rotarod tests at 28, 35, and 42 days after administration of the salt-loaded diet were not significantly different among the surviving rats (Fig. 2c and Table 1).
The rates of stroke onset (a), mortality (b), and symptom scoring (c) through the experiments after high-salt diets with vehicle (Veh), amlodipine (AM), irbesartan (IR), and AM + IR. The monitoring of the rats in vehicle group was stopped at 28 days after consumption of a high-salt diet. # P < 0.05 vs. vehicle, *P < 0.05 vs. AM + IR
Cerebral Blood Flow, Brain Edema, and Left Ventricular Hypertrophy
Next, we measured CBF and assessed the brain edema of the rats with their brain weight and BWC, which were moderately correlated, as demonstrated by the Spearman rank correlation coefficient (rs = 0.66, P < 0.05). Although the combination therapy reduced stroke onset as mentioned above, there were no significant changes in CBF or brain edema among the surviving rats at 42 days after administration of the salt-loaded diet (Fig. 3a–c). On the other hand, the combination therapy decreased the left ventricle weight index compared with the monotherapies (Fig. 3d).
Blood–Brain Barrier Disruption and White Matter Injury
As the extent of the IgG staining-positive area was markedly different in each rat, we defined the severity of the blood–brain barrier (BBB) disruption as the score of the IgG staining. According to the evaluation, the combination therapy significantly stabilized the BBB compared with irbesartan (Fig. 4a). Additionally, the white matter injury detected by Klüver-Barrera staining was significantly decreased in the combination group compared with that in the irbesartan group (Fig. 4b).
Evaluation of blood–brain barrier disruption (a), white matter injury (b), and reactive astrocyte (c) at 42 days after consumption of a high-salt diet with amlodipine (AM), irbesartan (IR), and AM + IR. The upper panels of the figures are representative pictures of each scoring. *P < 0.05 vs. AM + IR. Scale bar, 50 μm
Reactive Astrocyte and Brain-Derived Neurotrophic Factor Expressions on Cerebral Vessels in Somatosensory Cortex
There were infarct cortex detected by hematoxylin and eosin staining in one of the rats in the amlodipine group, two in the irbesartan group, and none in the combination groups (data not shown). Although upregulated reactive astrocytes were seen in almost all of the animals in all of the treatment groups, there were significantly fewer cells in the combination group than in the irbesartan group (Fig. 4c). Moreover, the number of BDNF expressions on cerebral vessels including the cerebral arterioles and capillaries was higher in the combination group than in the monotherapy groups (Fig. 5). Although BDNF was expressed in the neurons on the cortex, there were no changes among the groups.
Expression of brain-derived neurotrophic factor (BDNF) on cerebral vessels including arterioles and capillaries at 42 days after consumption of a high-salt diet with amlodipine (AM), irbesartan (IR), and AM + IR. The upper panels are representative pictures among the groups, and the arrows indicate BDNF-positive vessels. *P < 0.05 vs. AM + IR. Scale bar, 50 μm
Discussion
In the current study, we showed that monotherapies using either of the antihypertensive drugs amlodipine or irbesartan and the combination therapy with both drugs inhibited stroke onset in SHRSP rats at 28 days after administration of a salt-loaded diet. We also demonstrated that stroke onset was frequently seen in rats in the monotherapy groups after the drug was discontinued at 35 days, whereas the prevention of stroke onset persisted in the animal group treated with the combination therapy. Although there were no significant differences in hypertension-induced brain edema among the groups, the combination therapy reduced BBB disruption, white matter injury, and reactive astrocyte expressions, leading us to speculate that the persistence of the protective effects might come from BDNF preservation on cerebral vessels in the cortex.
The Pivotal Role of the Combination Therapy with Amlodipine and Irbesartan on Stroke Onset
Our main finding of this study was that the combination therapy with amlodipine and irbesartan persisted preventative effects on stroke onset. Increasing evidence from both clinical and basic research support the finding that the combination therapy with CCB and ARB is effective for improving hypertension and cardiovascular events [21, 22], and recent clinical guidelines in Japan also recommend the combination therapy for managing hypertension [23]. Based on stroke onset, we previously demonstrated that a combination therapy with amlodipine and valsartan prevented stroke onset compared with each monotherapy, and the effects came from the improvement in cerebral arteriolar remodeling and vascular endothelial dysfunction and reduction of oxidative stress, which independently lowered blood pressure [11]. In this study, there were no significant differences in sensorimotor function, CBF, or brain edema in the surviving hypertensive rats among the groups. Considering low frequency of cerebral infarction in surviving rats, we thought that this was because it is difficult to find significant differences in stroke outcome with mixed data including stroke-positive and stroke-negative brains. However, the combination therapy with amlodipine and irbesartan not only reduced “clinical and subclinical” stroke-related findings including BBB disruption, white matter injury, and reactive astrocyte expression compared with irbesartan but also inhibited stroke-related symptoms including death compared with amlodipine. The vulnerability of cerebral microvasculature and the microcirculatory disturbance induced by hypertension are associated with BBB disruption and white matter injury [24, 25], and they activate reactive astrocytes, which indicate brain injury such as brain edema and cerebral ischemia [26]. Thus, antihypertensive drugs have the possibility of inhibiting those brain injuries that are related to stroke onset. In this study, the combination therapy decreased blood pressure compared with irbesartan at 28 days after rats were salt-loaded, and the hypertension-induced left ventricular hypertrophy detected by the left ventricle weight index was significantly decreased in the combination group compared with the monotherapy groups. Although we did not measure blood pressure directly in the animals at 42 days after they were salt-loaded in order to avoid introducing extra stress to the weak rats, we speculate that the combination therapy should lower blood pressure significantly compared with the monotherapies. Therefore, we concluded that the combination therapy better controlled blood pressure through the study even after the drugs were discontinued and resulted in reductions of BBB instability and microcirculatory disturbance, which can induce white matter injury and reactive astrocytes.
The Implication of BDNF by the Combination Therapy on Cerebral Vessels Against Stroke Onset
Next, our novelty of this study was that BDNF expressions on cerebral vessels were higher in the combination therapy than in the monotherapies. BDNF is one of the neurotrophic factors, and its upregulation seems to have a neuroprotective effect against stroke [18, 27, 28]. In addition, BDNF has recently been found in the endothelial cells of cerebral arterioles after cerebral ischemia [29]. In hypertensive animals, BDNF was downregulated in the vulnerable hippocampal CA1 neurons after cerebral ischemia [9], and chronic hypertension lowered cerebral vascular reserves due to the thickening of the media of cerebral vessels [30]. We showed that BDNF expression on cerebral vessels, including cerebral arterioles and capillaries, was significantly higher in the combination therapy group, although CBF was similar among the treatment groups. We thought that the long-lasting hypertension throughout the study impaired cerebral autoregulation and induced cerebral ischemia detected by reactive astrocyte expression, resulting in the reduction of BDNF on cerebral vessels. Therefore, we concluded that the persistence of the beneficial effects in the combination therapy is associated with BDNF preservation on cerebral vessels, which might come from the reduction of hypertension-induced endothelial dysfunction.
Study Limitations
Although we showed significant roles of the combination therapy against stroke onset, there are still some limitations in this study. First, we did not assess the molecular pathways that were associated with the beneficial effect of the combination therapy. Previous reports have suggested that stroke onset induced by salt-loaded hypertension involved oxidative stress, and the combination therapy with CCB and ARB showed antioxidative stress [11]. Thus, we thought those mechanisms might be promising effects in this study. Second, although the combination therapy inhibited stroke onset and reduced BBB disruption, the therapy did not reveal significant beneficial effects in sensorimotor function as assessed by the beam walking and rotarod tests in CBF or in brain edema, which are deteriorated in stroke patients, suggesting that a longer endpoint might be more suitable for this study. In addition, further study is needed to clarify the severity of BBB disruption and changes of BDNF expressions in the sham group at 42 days after the beginning of the high-salt diet. Finally, we did not measure blood pressure at 42 days after rats were salt-loaded, and we substituted left ventricle weight index for the long-lasting strength of hyper blood pressure because measuring the blood pressure under waking conditions was stressful and could be a confound for the stroke-related outcome in weak rats. In view of the limitations in this study, we will clarify the molecular pathways and assess the sensorimotor function and blood pressure-lowering effect of those drugs over a longer period for the translation of these discoveries to clinical setting.
Conclusion
We showed that the combination therapy with amlodipine and irbesartan inhibited stroke onset even after those drugs were discontinued. In addition, we also demonstrated that the possible mechanism might be associated with BDNF expression on cerebral vessels. Because CCB and ARB are already used and are effective for lowering blood pressure in hypertensive patients, further study will provide the clinical significance of the combination treatment against stroke onset.
References
Lackland DT, Roccella EJ, Deutsch AF, Fornage M, George MG, Howard G, et al. Factors influencing the decline in stroke mortality: a statement from the American Heart Association/American Stroke Association. Stroke. 2014;45:315–53.
Ando K, Kawarazaki H, Miura K, Matsuura H, Watanabe Y, Yoshita K, et al. Report of the Salt Reduction Committee of the Japanese Society of Hypertension (1): role of salt in hypertension and cardiovascular diseases. Hypertens Res. 2013;36:1009–19.
Muiesan ML, Salvetti M, Rizzoni D, Paini A, Agabiti-Rosei C, Aggiusti C, et al. Resistant hypertension and target organ damage. Hypertens Res. 2013;36:485–91.
Neal B, MacMahon S, Chapman N. Effects of ACE inhibitors, calcium antagonists, and other blood-pressure-lowering drugs: results of prospectively designed overviews of randomised trials. Lancet. 2000;356:1955–64.
Reboldi G, Angeli F, Cavallini C, Gentile G, Mancia G, Verdecchia P. Comparison between angiotensin-converting enzyme inhibitors and angiotensin receptor blockers on the risk of myocardial infarction, stroke and death: a meta-analysis. J Hypertens. 2008;26:1282–9.
Lipsky RH, Marini AM. Brain-derived neurotrophic factor in neuronal survival and behavior-related plasticity. Ann N Y Acad Sci. 2007;1122:130–43.
Leventhal C, Rafii S, Rafii D, Shahar A, Goldman SA. Endothelial trophic support of neuronal production and recruitment from the adult mammalian subependyma. Mol Cell Neurosci. 1999;13:450–64.
Kim H, Li Q, Hempstead BL, Madri JA. Paracrine and autocrine functions of brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF) in brain-derived endothelial cells. J Biol Chem. 2004;279:33538–46.
Lee TH, Yang JT, Kato H, Wu JH. Hypertension downregulates the expression of brain-derived neurotrophic factor in the ischemia-vulnerable hippocampal CA1 and cortical areas after carotid artery occlusion. Brain Res. 2006;1116:31–8.
Kishi T, Hirooka Y, Sunagawa K. Telmisartan protects against cognitive decline via up-regulation of brain-derived neurotrophic factor/tropomyosin-related kinase B in hippocampus of hypertensive rats. J Cardiol. 2012;60:489–94.
Dong YF, Kataoka K, Tokutomi Y, Nako H, Nakamura T, Toyama K, et al. Beneficial effects of combination of valsartan and amlodipine on salt-induced brain injury in hypertensive rats. J Pharmacol Exp Ther. 2011;339:358–66.
Kim-Mitsuyama S, Yamamoto E, Tanaka T, Zhan Y, Izumi Y, Izumiya Y, et al. Critical role of angiotensin II in excess salt-induced brain oxidative stress of stroke-prone spontaneously hypertensive rats. Stroke. 2005;36:1083–8.
Nako H, Kataoka K, Koibuchi N, Dong YF, Toyama K, Yamamoto E, et al. Novel mechanism of angiotensin II-induced cardiac injury in hypertensive rats: the critical role of ASK1 and VEGF. Hypertens Res. 2012;35:194–200.
Nakagawa T, Hasegawa Y, Uekawa K, Ma M, Katayama T, Sueta D, et al. Renal denervation prevents stroke and brain injury via attenuation of oxidative stress in hypertensive rats. J Am Heart Assoc. 2013;2:e000375. doi:10.1161/JAHA.113.000375.
Nakayama T, Nagisa Y, Imamoto T, Nagai Y. Beneficial effects of TDN-345, a novel Ca2+ antagonist, on ischemic brain injury and cerebral glucose metabolism in experimental animal models with cerebrovascular lesions. Brain Res. 1997;762:203–10.
Goldstein LB, Davis JN. Beam-walking in rats: studies towards developing an animal model of functional recovery after brain injury. J Neurosci Methods. 1990;31:101–7.
Hasegawa Y, Suzuki H, Altay O, Rolland W, Zhang JH. Role of the sphingosine metabolism pathway on neurons against experimental cerebral ischemia in rats. Transl Stroke Res. 2013;4:524–32.
Hasegawa Y, Suzuki H, Altay O, Zhang JH. Preservation of tropomyosin-related kinase B (TrkB) signaling by sodium orthovanadate attenuates early brain injury after subarachnoid hemorrhage in rats. Stroke. 2011;42:477–83.
Wakita H, Tomimoto H, Akiguchi I, Lin JX, Ihara M, Ohtani R, et al. Ibudilast, a phosphodiesterase inhibitor, protects against white matter damage under chronic cerebral hypoperfusion in the rat. Brain Res. 2003;992:53–9.
Akin D, Ravizza T, Maroso M, Carcak N, Eryigit T, Vanzulli I, et al. IL-1β is induced in reactive astrocytes in the somatosensory cortex of rats with genetic absence epilepsy at the onset of spike-and-wave discharges, and contributes to their occurrence. Neurobiol Dis. 2011;44:259–69.
Ogawa H, Kim-Mitsuyama S, Matsui K, Jinnouchi T, Jinnouchi H, Arakawa K, et al. Angiotensin II receptor blocker-based therapy in Japanese elderly, high-risk, hypertensive patients. Am J Med. 2012;125:981–90.
Rafiq K, Sherajee SJ, Hitomi H, Nakano D, Kobori H, Ohmori K, et al. Calcium channel blocker enhances beneficial effects of an angiotensin II AT1 receptor blocker against cerebrovascular-renal injury in type 2 diabetic mice. PLoS ONE. 2013;8:e82082. doi:10.1371/journal.pone.0082082.
Shimamoto K, Ando K, Fujita T, Hasebe N, Higaki J, Horiuchi M, et al. The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2014). Hypertens Res. 2014;37:253–387.
Vital SA, Terao S, Nagai M, Granger DN. Mechanisms underlying the cerebral microvascular responses to angiotensin II-induced hypertension. Microcirculation. 2010;17:641–9.
Verhaaren BF, Vernooij MW, de Boer R, Hofman A, Niessen WJ, van der Lugt A, et al. High blood pressure and cerebral white matter lesion progression in the general population. Hypertension. 2013;61:1354–9.
Chen Y, Swanson RA. Astrocytes and brain injury. J Cereb Blood Flow Metab. 2003;23:137–49.
Han QQ, Jin W, Xiao ZF, Huang JC, Ni HB, Kong J, et al. The promotion of neurological recovery in an intracerebral hemorrhage model using fibrin-binding brain derived neurotrophic factor. Biomaterials. 2011;32:3244–52.
Ishrat T, Pillai B, Soliman S, Fouda AY, Kozak A, Johnson MH, Ergul A, Fagan SC. Low-dose candesartan enhances molecular mediators of neuroplasticity and subsequent functional recovery after ischemic stroke in rats. Mol Neurobiol. 2014.
Béjot Y, Prigent-Tessier A, Cachia C, Giroud M, Mossiat C, Bertrand N, et al. Time-dependent contribution of non neuronal cells to BDNF production after ischemic stroke in rats. Neurochem Int. 2011;58:102–11.
Clozel JP, Kuhn H, Hefti F. Effects of cilazapril on the cerebral circulation in spontaneously hypertensive rats. Hypertension. 1989;14:645–51.
Acknowledgments
This work was partially supported by a grant from Shionogi & CO., LTD. We thank Yuriko Shimamura, Miho Kataoka, Michie Uchikawa, Keiko Morozumi, Kazuko Noda, and Tomoko Moriyama for their support.
Disclosures
S. K-M received lecture fees and research grant from Astellas, AstraZeneca, Boehringer Ingelheim, Daiichi Sankyo, Novartis, Sionogi, Takeda, and Kyowa Hakko Kirin.
Conflict of Interest
The authors report no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hasegawa, Y., Nakagawa, T., Uekawa, K. et al. Therapy with the Combination of Amlodipine and Irbesartan Has Persistent Preventative Effects on Stroke Onset Associated with BDNF Preservation on Cerebral Vessels in Hypertensive Rats. Transl. Stroke Res. 7, 79–87 (2016). https://doi.org/10.1007/s12975-014-0383-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-014-0383-5